Abstract
The expression of ATP-sensitive K+ (KATP) channels by magnocellular cholinergic basal forebrain (BF) neurones was investigated in thin brain slice and dissociated cell culture preparations using a combination of whole-cell, perforated-patch and single-channel recording techniques. Greater than 95% of BF neurones expressed functional KATP channels whose activation resulted in membrane hyperpolarization and a profound fall in excitability. The whole-cell KATP conductance was 14.0 ± 1.5 nS and had a reversal potential of −91.4 ± 0.9 mV that shifted by 59.6 mV with a tenfold increase in [K+]o. IKATP was inhibited reversibly by tolbutamide (IC50 of 34.1 μm) and irreversibly by glibenclamide (0.3–3 nm) and had a low affinity for [ATP]i (67% reduction with 6 mm[MgATP]i). Using perforated-patch recording, a small proportion of the conductance was found to be tonically active. This was weakly potentiated by diazoxide (0.1 mm extracellular glucose) but insensitive to pinacidil (≤500 μm). Single-channel KATP currents recorded in symmetrical 140 mm K+-containing solutions exhibited weak inward rectification with a mean conductance of 66.2 ± 1.9 pS. Channel activity was inhibited by MgATP (>50 μm) and activated by MgADP (200 μm). The K+ channels opener diazoxide (200–500 μm) increased channel opening probability (NPo) by 486 ± 120% whereas pinacidil (500 μm) had no effect. In conclusion, the characteristics of the KATP channels expressed by BF neurones are very similar to channels composed of SUR1 and Kir6.2 subunits. In the native cell, their affinity for ATP is close to the resting [ATP]i, potentially allowing them to be modulated by physiologically relevant changes in [ATP]i. The effect of these channels on the level of ascending cholinergic excitation of the cortex and hippocampus is discussed.
K+ channels that are tonically active at subthreshold potentials play a pivotal role in regulating the excitability of many neurones (Brown & Selyanko, 1985; Benson et al. 1988; Millar et al. 2000). Small changes in the activity of this type of channel can produce profound changes in cell excitability. One example of subthreshold K+ channels are the so-called ATP-sensitive K+ channels which open in response to a fall in the intracellular ATP/ADP ratio (Ashcroft & Gribble, 1998). Structurally KATP channels are hetero-octamers made up from a combination of four pore forming subunits (either Kir6.1 or 6.2) and four sulphonylurea receptors (SUR 1, 2A, 2B and 2C) (Ashcroft & Gribble, 1998; Babenko et al. 1998a). Both the pore forming and receptor subunits must be expressed and co-assemble in order to form a functional channel (Clement et al. 1997). KATP channel subunits are widely distributed throughout the nervous system including the brain (Treherne & Ashford, 1991; Dunn-Meynell et al. 1998). Within basal forebrain nuclei, large numbers of high affinity binding sites for the sulphonylurea receptor antagonist glibenclamide have been described (Mourre et al. 1990). In situ hybridization studies have revealed overlapping moderate to high mRNA levels for the channel-forming Kir6.2 and sulphonylurea receptor SUR1 subunits in medial septal and diagonal band regions of the basal forebrain (Karschin et al. 1997). In addition, 3 days after selective lesioning of the nucleus basalis Kir6.2 mRNA levels in the cortex have been shown to fall by 47% (Xu et al. 2002). Together these findings indicate that a significant population of the cells in these regions may express functional somatic as well as presynaptic KATP channels.
However, basal forebrain nuclei contain a heterogeneous population of neurones, and the identity of the cells expressing KATP channels is unknown. In this study, the functional expression of KATP channels by individually identified cholinergic basal forebrain neurones has been examined. These large neurones provide the principal ascending cholinergic input to both the hippocampus and cerebral cortex and the integrity of their projections is vital for various learning and memory processes and also for maintaining normal levels of cortical arousal and attentiveness (Jones, 1993; Everitt & Robbins, 1997). A preliminary report of these findings has appeared in abstract form (Allen, 2003).
Methods
Preparation of dissociated basal forebrain cultures
Sprague Dawley rat pups (12–14 days old) were anaesthetized by chloroform inhalation prior to decapitation in accordance with UK legislation. The brain was rapidly removed and placed in ice-cold Gey's balanced salt solution supplemented with 8 mm Mg2+ and 0.6%d-glucose. The brain was hemisected and 450 μm thick coronal sections cut using a McIllwain tissue chopper. Basal forebrain areas, namely the medial septum (MS), diagonal band of Broca (DBB) and substantia innominata (SI) were isolated and placed in Hanks' balanced salt solution (HBSS) containing 10 mm Hepes and 1.25 mgl−1 trypsin and incubated for 60 min at 37°C. The tissue fragments were subsequently washed in HBSS containing 10% fetal bovine serum (FBS) and 8 mm Mg2+ before being gently dissociated using a flamed Pasteur pipette. The resulting cell suspension was centrifuged at 600 r.p.m. for 6 min, the supernatant discarded and the cell pellet re-suspended in Neurobasal medium containing B27 supplement (Gibco, UK), 0.01 mg l−1 nerve growth factor and 10% FBS. Cells were plated onto 13 mm diameter poly-d-lysine-coated glass coverslips (1 per 35 mm Petri dish) and left to settle for 10 min before the entire dish was gently flooded with medium. The medium was replaced after 4 h. Subsequent re-feeding took place after 18 h and thereafter weekly with medium of the same composition minus FBS. Cells were maintained in a 5% CO2 incubator at 37°C for periods of up to 8 weeks prior to use.
Brain slice preparation
Sprague Dawley rat pups (12–14 days old) were anaesthetized as above before decapitation and removal of the brain to ice-cold (4°C) Krebs solution containing (mm): NaCl 118; KCl; 3; Hepes 5; NaHCO3 25; glucose 11; CaCl2 0.5; MgCl2 6; kynurenic acid 1, adjusted to pH 7.3 (where necessary) and gassed with 95% O2–5% CO2. The brain was hemisected and 200–250 μm thick coronal sections containing the different basal forebrain regions (as detailed above) were cut using a Vibratome (model 1000 plus). Sections were incubated for 1 h at room temperature in oxygenated Krebs solution (composition as above) prior to the start of electrophysiological recording. Brain slices were transferred to a recording chamber and gently held in place by a flattened C-shaped platinum wire bridged by fine nylon fibres. Cells were visualized using water immersion objectives on an upright microscope (Olympus BX50WI) equipped with differential interference contrast (DIC) optics.
The preparation was continuously superfused (non-recirculating) with oxygenated Krebs (see below for composition), containing 1 mm kynurenic acid, 10 μm 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) and 30 μm bicuculline methochloride. All experiments were carried out at room temperature using the whole-cell configuration of the patch-clamp technique (see below).
Electrophysiology
Whole-cell and perforated-patch recording from cell cultures
Coverslips bearing the cell culture were transferred into a recording bath mounted on the microscope stage (Olympus BX50WI). Unless otherwise stated, cells were superfused at a rate of 5–8 ml min−1 at room temperature (22–26°C) with Krebs solution of composition (mm): NaCl, 122; KCl, 3; Hepes, 5; NaHCO3, 15; glucose, 11; CaCl2, 2; MgCl2, 1; adjusted to pH 7.3 and gassed with 95% O2–5% CO2. Recordings were carried out using either the tight-seal whole-cell configuration of the patch-clamp technique or the amphotericin-B perforated-patch method (Rae et al. 1991), using an Axoclamp 2 amplifier coupled to a Digidata 1200 (Axon Instruments) interface and pCLAMP 8 (Axon Instruments) acquisition software. Data was acquired at a sampling rate of 10–40 kHz and filtered at 1–3 kHz (voltage clamp) or 20 kHz (bridge voltage recording) prior to acquisition. When necessary, additional filtering was carried out off-line. Patch pipettes were pulled from 1.5 mm o.d. × 1.17 mm i.d. borosilicate glass (Harvard Apparatus) coated to within 100 μm of the tip with Sylgard (Dow Corning). Electrode resistance ranged between 5 and 9 MΩ for whole-cell recording and between 3 and 5 MΩ for perforated-patch recording. The composition of the pipette solution (unless otherwise stated) was (mm): potassium acetate, 108; KCl, 11; Hepes, 40; NaOH, 17; EGTA, 3; CaCl2, 0.52, MgCl2, 1.2 (pH 7.3). Voltage-clamp recordings were carried out using the discontinuous single-electrode voltage-clamp technique. During perforated-patch recordings, series resistance (Rs) ranged between 7 and 28 MΩ.
Whole-cell brain slice recording
All brain slice recordings were carried out at room temperature using the whole-cell variant of the patch-clamp technique (see above).
Cell-attached and inside-out patch recording
Recordings were carried out using an Axopatch 200 A (Axon Instruments) patch-clamp amplifier (interface and acquisition software as above). Patch electrodes were filled with a high K+-containing solution of composition (mm): potassium acetate 140; Hepes 20; EGTA 0.5 mm; MgCl2 1; and the pH was adjusted to 7.3 with KOH. For inside-out patch recording the bathing and pipette solutions were of the same composition (see above). Additional drugs were directly added to the bathing solution.
Assessment of changes in excitability
Measurement of excitability changes were carried out using standard bridge voltage recording mode. Excitability was quantified in terms of the minimum current (100–500 ms stimuli) required to reach spike threshold. Measurements were made following activation or inhibition of the KATP conductance both before and after nulling any associated changes in resting potential. As alterations in firing frequency were only observed to occur as a secondary consequence of membrane potential changes resulting in the activation or inhibition of other conductances and were extremely variable from cell to cell, this form of measurement was not considered to be a very reliable or direct measure of KATP channel-induced excitability changes.
Choline acetyltransferase (ChAT) single-cell RT-PCR
When harvesting cell contents for single-cell RT-PCR, electrodes were filled with 3.75 μl of filling solution of composition (mm): potassium acetate 108; KCl 10.6; Hepes 40; MgCl2 1.2; EGTA 3; NaOH 12 (pH 7.3). After whole-cell patch clamping the cell, a steady negative pressure was applied to the pipette until as much of the cytoplasm as possible was aspirated, taking care not rupture the membrane seal. Having removed the cytoplasmic contents, negative pressure was released, and the electrode was quickly removed from the bath and its contents aspirated into a sterile Eppendorf tube containing 6.25 μl of a solution containing 10 U RNase inhibitor, 100 U M-MLV RNase H− point mutant reverse transcriptase (Promega), oligo(dT)15 (final concentration 5 μm), 0.1 μg μl−1 acetylated BSA and the four deoxyribonucleoside triphosphates (final concentration 0.5 mm).
The tubes containing approximately 10 μl were then incubated at 37°C for 1 h to synthesize single-stranded complimentary DNA (cDNA) followed by a further 15 min incubation at 72°C for enzyme inactivation. The samples were then frozen at –25°C prior to carrying out PCR. PCR was used to amplify mRNA transcripts using ChAT subunit specific pair of primers. ChAT mRNA (Brice et al. 1989) was identified using a pair of primers flanking a splicing site near the 3′ terminus of the coding region. The upper primer was 5′-ATGGCCATTGACAACCATCTTCTG (nucleotides 1729–1752) located on exon 14. The lower primer was 5′-CCTTGAACTGCAGAGGTCTCTCAT (nucleotides 2052–2029) located on exon 15, and the fragment size was 323 bp (Yan & Surmeier, 1996). Taq DNA polymerase (1 μl; 10 × Titanium Taq DNA polymerase, Clontech, UK), in a buffer containing 3.5 mm Mg2+, 0.2 mm dNTPs and the primer pair (5 pmol each), was added to 10 μl of RT product to give a final volume of 50 μl. After 3 min at 94°C, 35 cycles (94°C 30 s, 55°C 30 s, 72°C 40 s) were performed, followed by an elongation period of 10 min at 72°C. The amplification product was then purified using High Pure microspin tubes (Roche Diagnostics GmbH). The purified product (5 μl) was used as a template for the second round of PCR amplification: 40 cycles (94°C 30 s, 55°C 30 s, 72°C 40 s) with a 10 min final elongation period at 72°C.
Data acquisition and analysis
Data was analysed using a combination of the pCLAMP 8 suite of software (Axon Instruments), Origin 5 (Microcal Software Inc., Northampton, MA, USA) and Coreldraw 8 (Corel graphics, Corel Corporation, Ontario, Canada). Unless otherwise stated all values are means ± s.e.m. Significance values were calculated using Student's paired t test.
Drugs
All drugs were applied either via the superfusing solution or by inclusion in the pipette filling solution. 4-Aminopyridine, Na2ADP, MgADP, apamin, Na2ATP, MgATP, cAMP, charybdotoxin, chloroform, diazoxide, GDP-β-S, glibenclamide, GTP, halothane, paclitaxel, phalloidin, pinacidil, spermine, tetraethylammonium chloride and tolbutamide were from Sigma. Tetrodotoxin was from Alomone Laboratories. Bicuculline methochloride and 6-cyano-7-nitroquinoxaline-2,3-dione disodium (CNQX) were from Tocris.
Results
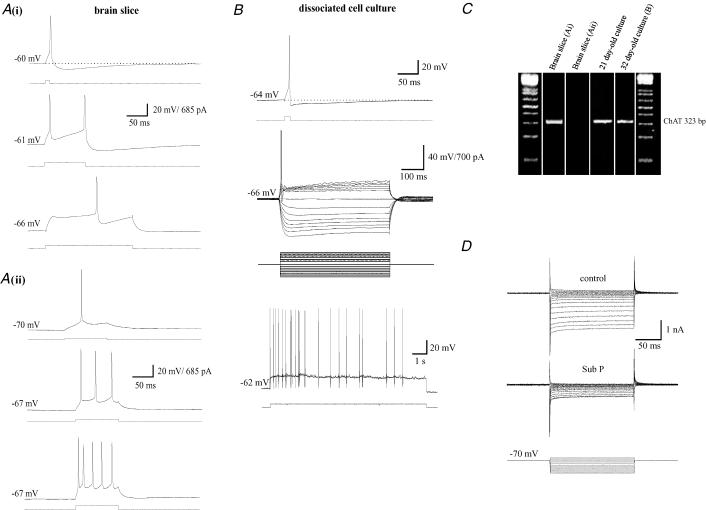
Whole-cell patch-clamp recordings were made from 139 magnocellular cholinergic basal forebrain (BF) neurones in thin brain slice preparations. A further 194 whole-cell, perforated-patch or excised-patch recordings were made from magnocellular BF neurones in mixed cell cultures. Cells were selected on the basis of their size (diameters 20–38 μm), location (see Methods), firing characteristics (see Fig. 1A and B) and current–voltage relationships (see Fig. 1D). The cholinergic nature of the cells meeting these criteria was also confirmed by carrying out single-cell RT-PCR for ChAT on 23 cells (15 of which were cultured cells with the other 8 from acute slices) of which 21 gave a positive product (see Fig. 1C). No consistent differences were observed in the electrophysiological characteristics of the neurones from the different preparations, and therefore data were pooled.
Figure 1. Characteristics of basal forebrain (BF) neurones from brain slice and dissociated cell culture preparations.
A, recording from BF neurones in brain slice preparations (13-day-old rats). Ai illustrates the spike after-hyperpolarization (AHP) (upper panels), slow firing rate and pronounced A-type current-induced delay of spike initiation (lower panel) that are typical features of cholinergic basal forebrain neurones. The cholinergic nature of this cell was confirmed using single-cell RT-PCR for ChAT (see panel C, gel lane 1). Aii shows typical firing characteristics (high frequency discharge with no spike AHP) associated with non-cholinergic BF neurones. Again the non-cholinergic nature of the cell was confirmed using RT-PCR (see panel C, lane 2). B illustrates that the firing characteristics of cholinergic BF neurones are still maintained after prolonged periods in culture. The cell shown was from a 32-day-old culture and the recording was carried out using perforated patch recording. Upper, middle and lower panels, respectively, show the spike AHP, I–V relationship and slow rate of firing characteristic of cholinergic neurones in intact preparations. Again the cholinergic nature of the cells was confirmed by RT-PCR (see panel C, lane 4). C, composite gel showing the presence and absence of ChAT (323 bp) reaction product in 4 different BF neurones. Cholinergic BF neurones also express a Substance P-sensitive inward rectifier current (Yamaguchi et al. 1990). D shows that this current is also maintained when the cells are maintained in culture (17-day-old culture). From a holding potential of –70 mV voltage steps from –125 to –55 mV (160 ms duration; 5 mV increments) were imposed to generate an I–V relationship under control conditions (0.5 μm TTX present throughout) and in the presence of 600 nm substance P.
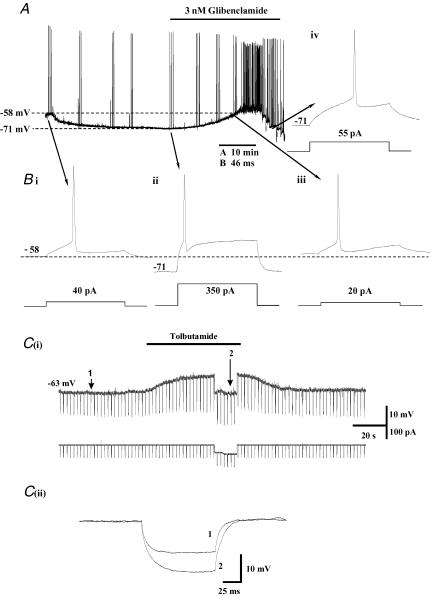
Immediately after breaking through to the whole-cell recording configuration (0 mm[ATP]i), the average resting potential of basal forebrain neurones was –60.3 ± 1.13 mV (n = 69); as cell dialysis proceeded membrane potential slowly hyperpolarized over a period of 10–15 min, reaching a maximum resting potential of −70.6 ± 1.14 mV (n = 60). Figure 2 shows a typical example of this run-up phenomenon recorded from a cholinergic basal forebrain neurone in a 250 μm thick brain slice of the rat diagonal band region. As the cell hyperpolarized, its excitability declined markedly and much more current needed to be injected in order to elicit action potential discharge. Typically, the current required to evoke firing at the peak of the hyperpolarization (range 0.35–1.56 nA; mean 812.5 ± 89 pA; n = 17) was significantly greater (P > 0.0001; paired t test) than what was initially required at the start of the recording (range 0.02–0.28 nA; mean 158 ± 24 pA; n = 17). The fall in excitability resulted from a combination of the membrane hyperpolarization, which also had the additional inhibitory effect of removing inactivation from A-type K+ channels, and the shunting effect on the membrane due to the increase in resting conductance (see Fig. 2Bii and iv). The hyperpolarization and conductance increase were unaffected by the calcium-activated K+ channel blockers apamin (30 nm; n = 4) or charybdotoxin (100 nm; n = 3) and were not potentiated by the twin pore K+ channel modulators, chloroform (1 mm; n = 4) or halothane (3 mm; n = 4). However, both the membrane hyperpolarization and the resulting fall in excitability could largely be prevented by application of the sulphonylurea receptor antagonists glibenclamide (1–10 nm) or tolbutamide (30–200 μm), indicating the involvement of KATP channels. In a few cells, excitability in the presence of these antagonists was slightly higher than at the start of the recording, indicating that a proportion of the KATP channels might be tonically activated under resting conditions. In order to investigate this possibility under more physiological conditions, experiments were carried out using perforated-patch recording to minimize any disturbance to the normal intracellular ATP/ADP ratio in the presence of 2 mm extracellular glucose. Under these conditions, application of tolbutamide (100 μm) induced a small reversible depolarization (mean 7.2 ± 1.6 mV; n = 9) and increase in input resistance (mean increase 52.6 ± 16.2 MΩ; n = 7) in 9 out of 11 cells tested (Fig. 2C).
Figure 2.
A, whole-cell recording of a basal forebrain (BF) neurone from the diagonal band region of a thin (250 μm) brain slice. Immediately upon breaking through to whole-cell (1 mm[MgATP]i), resting potential (Vm) was –58 mV. Within less than a minute of cell dialysis commencing, Vm slowly began to hyperpolarize, reaching a maximum of –71 mV after approximately 15 min. During this period, excitability declined markedly (see Bi and ii). Subsequent application of glibenclamide (3 nm) reversed both the hyperpolarization and fall in excitability (Biii). In this particular cell, excitability in the presence of glibenclamide was slightly higher than under control conditions. At the end of the record shown in A (glibenclamide present), current was directly injected through the electrode in order to restore the membrane potential to that observed at the peak of the hyperpolarization. Biv shows that the increase in excitability observed in the presence of glibenclamide (Biii) was not simply the result of the change in Vm (compare Bii and Biv). Ci, perforated-patch recording from a BF neurone maintained in culture for 13 days (2 mm extracellular glucose). Under control conditions, Vm was –63 mV. Hyperpolarizing current steps (100 pA (100 ms)−1) were applied at 0.5 Hz to monitor changes membrane resistance (Rin). Application of tolbutamide (100 μm) caused a membrane depolarization associated with a fall in Rin. Cii illustrates the direct effect of tolbutamide on Rin after the associated depolarization had been nulled by current injection.
During prolonged (>30 min) whole-cell recording, the activity of the underlying KATP channel conductance was frequently observed to slowly decline and excitability returned to levels similar to those observed at the start of the recording (data not shown).
Ionic basis of the run-up current
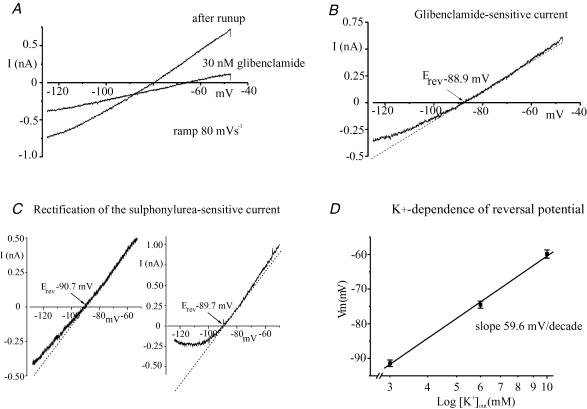
The mechanism underlying the membrane hyperpolarization and increase in conductance was examined under voltage clamp. Figure 3A shows membrane current in response to ramping membrane potential from –125 to –47 mV (ramp rate 80 mV s−1) following full current run-up and after subsequent application of glibenclamide (30 nm). Panel B shows the current–voltage (I–V) relationship of the glibenclamide-sensitive component of the current. Typically, the sulphonylurea-sensitive current between –130 and –50 mV exhibited weak outward rectification which was most pronounced at strongly hyperpolarized potentials. The degree of rectification varied considerably between cells (see Fig. 3C). In cells exhibiting relatively marked rectification, the I–V curve of the sulphonylurea-sensitive current occasionally exhibited a region of negative slope conductance at strongly negative potentials (see right-hand panel of Fig. 3C and (Sim & Allen, 1998). A similar phenomenon has been observed in other Kir channels and has been attributed to voltage-dependent block of the channels by extracellular Na+ ions (Standen & Stanfield, 1979).
Figure 3. Voltage and K+ dependence of the sulphonylurea-sensitive run-up current.
A, shows the whole-cell membrane current in response to ramping the membrane potential (Vm) from –126 to –47 mV (ramp 80 mV s−1) following current run-up and after subsequent application of glibenclamide (30 nm). B, I–V relationship of the glibenclamide-sensitive component of the current shown in A. C, examples of the extremes of rectification observed in the I–V relation of the sulphonylurea-sensitive from different cells. D, the reversal potentials of the sulphonylurea-sensitive current measured in 3, 6 and 10 mm[K+]o were 91.4 ± 0.94, –74.5 ± 1.01 and –59.9 ± 1.17 mV (n = 5), respectively, slope 59.6 mV for a 10-fold change in [K+]o.
In Fig. 3B, the reversal potential (Erev) of the sulphonylurea-sensitive current in 3 mm[K+]out was –88.9 mV, a value close to the calculated potassium equilibrium potential (EK). The K+ selectivity of the underlying channels was examined further by measuring the shift in the reversal potential of the tolbutamide-sensitive run-up current in response to changing the extracellular [K+]. Mean Erev values for the sulphonylurea-sensitive current in 3, 6 and 10 mm[K+]o were –91.4 ± 0.94, –74.5 ± 1.01 and –59.9 ± 1.17 mV (n = 5), respectively, yielding a predicted shift of 59.6 mV for a 10-fold change in [K+]o, a value consistent with what would be predicted if the underlying channels were highly selective for K+ ions (see Fig. 3D).
Kinetics of the run-up current
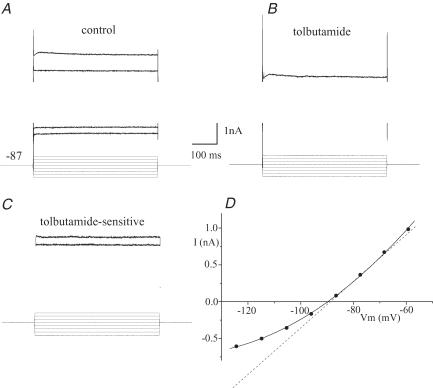
The activation and inactivation kinetics of the sulphonylurea-sensitive run-up current were examined. Figure 4A and B shows membrane currents evoked between –125 and –60 mV from a holding potential of –87 mV following current run-up and in the presence of tolbutamide (100 μm). Over this range of potentials the sulphonylurea-sensitive component of the current exhibited no observable time dependence of either its activation or inactivation (see Fig. 4C). However, switch-clamp recordings have a limited frequency response and any kinetic components with a time constant of less than 1–2 ms would be unresolved. The kinetics of the current were not examined at potentials more depolarized than –60 mV as the native transient outward K+ currents in BF cells were too large to clamp sufficiently well to be able to obtain accurate subtraction currents.
Figure 4. Kinetics of the whole-cell KATP channel current.
I–V relationship (–127 to –57 mV in 10 mV increments) in the presence of (TTX, 0.5 μm) under (A) control conditions (Vh–87 mV) and (B) after addition of tolbutamide (100 μm). C, the tolbutamide-sensitive component of current exhibits no fast kinetic activation or inactivation. D, the I–V curve of the steady state (end of pulse) tolbutamide-sensitive current shown in B.
Activation of the run-up current
Dialysis of the cell contents during whole-cell recording invariably triggered current run-up; it did not occur when disruption of cytoplasmic contents was minimized by employing the perforated patch recording technique, indicating that it was triggered by loss of a specific cytoplasmic factor. Under whole-cell recording conditions, neither the rate nor amplitude of the run-up current were affected by inclusion in the pipette solution of the microtubule or cytoskeletal stabilizing agents paclitaxol (300 nm) and phalloidin (10 μm); the mean run-up conductances (Grun-up) in paclitaxol and phalloidin were 12.3 ± 2.7 nS (n = 5) and 16.7 ± 3.2 nS (n = 5), respectively, compared to a value of 14.0 ± 1.5 nS (n = 18) under control conditions.
Basal forebrain neurones also express other Kir channels whose rectification at depolarized potentials results in large part from channel block by intracellular polyamines (Bajic et al. 2002). During prolonged whole-cell recordings endogeneous polyamines may be dialysed away, resulting in loss of rectification due to unblock of the channels, which could contribute to the observed run-up current at potentials positive to EK. However, inclusion of spermine (1 μm) in the pipette filling solution did not significantly reduce the amplitude of the run-up conductance. Mean conductance (positive to EK) in the presence spermine was 13.9 ± 2.2 nS (n = 4), compared to a value 14.0 ± 1.5 nS (n = 18) under control conditions.
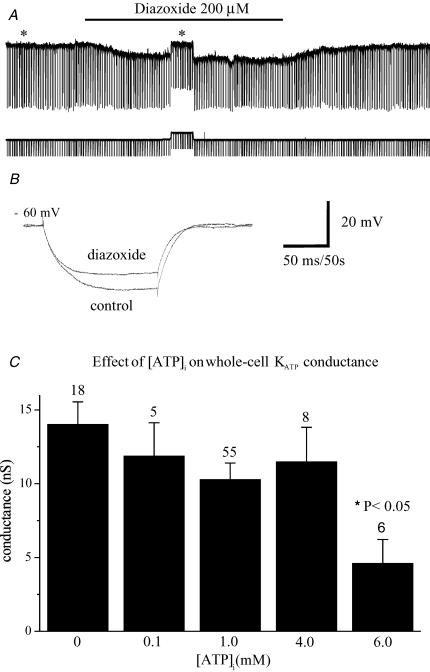
Depending upon their subunit composition, KATP channels exhibit varying sensitivities to the different K+ channel activators (KCOs) (Babenko et al. 1998a; Schwanstecher et al. 1998). As run-up was activated under whole-cell conditions even in the presence of high intracellular ATP concentration (see later section), it was not possible to test these compounds in this recording configuration. Thus the ability of these compounds to activate the whole-cell KATP current in basal forebrain neurones was investigated using the perforated-patch recording technique. Under these conditions, in the presence of normal to high extracellular glucose concentrations (2–10 mm), the ATP-sensitive K+ channel activators diazoxide and pinacidil (100–500 μm) both failed to activate the K+ run-up current (n = 12). This lack of effect was surprising, as almost all subtypes of KATP channel are sensitive to at least one of the compounds. However, previous studies have shown that ATP competes with diazoxide/pinacidil for control of channel opening and that their effect can be completely suppressed if the intracellular ATP concentration exceeds 2–5 mm (Dunne et al. 1987; Fan et al. 1990). Thus if, as has been reported in other cells (Ashcroft, 1988), basal forebrain neurones have a high resting submembrane [ATP]i, then this could explain the observed lack of effect of these compounds. In order to test this possibility, the effects of KCOs were tested on cells bathed with Krebs solution containing low (0.1 mm) extracellular glucose. This should result in a reduction in the intracellular ATP concentration and allow the KCOs access to the active site on the SUR subunit. Under these conditions, diazoxide (200 μm; see Figs 5A and B), but not pinacidil (200–500 μm), was then observed to hyperpolarize the resting cell membrane potential by 3.3 ± 1.15 mV and decrease input resistance by 41.3 ± 17.3 MΩ in 3 out of 8 cells tested.
Figure 5.
A, activation of the KATP conductance by diazoxide (200 μm) in a perforated-patched cell superfused with low extracellular glucose (0.1 mm) containing Krebs solution. hyperpolarizing current steps (100 pA (100 ms)−1) were applied at 0.5 Hz to monitor changes membrane resistance (Rin). B illustrates the direct effect of diazoxide on Rin (*traces) after the associated depolarization had been nulled by current injection. C, histogram showing the sensitivity of whole-cell KATP run-up conductance to intracellular [ATP]. In each case, the KATP conductance was calculated by measuring the amplitude of the tolbutamide-sensitive current activated in response to stepping from –80 to –40 mV following full current run-up. A significant reduction in the amplitude of the run-up conductance was only observed with [ATP]i of 6 mm.
Modulation by [ATP]i and other high energy phosphates
The dependence of the run-up current on the intracellular ATP concentration was examined. The histogram in Fig. 5C shows the amplitude of the sulphonylurea-sensitive run-up conductance in the presence of different intracellular ATP concentrations. In the absence of ATP, the average conductance increase due to the activation of KATP channels was 14.0 ± 1.5 nS (n = 18). Increasing [ATP]i had no effect upon the amplitude of the conductance until its concentration exceeded 4 mm. With 6 mm[ATP]i, the mean run-up conductance was reduced by approximately 67% to 4.6 ± 1.6 nS (n = 6). In contrast, inclusion of physiologically relevant intracellular concentrations of GTP (0.5 mm), GDP-βS (1 mm), ADP (1 mm) or cAMP (0.5–1 mm) in the intracellular solution failed to prevent run-up, the mean sulphonylurea-sensitive conductances under these conditions being 14.3 ± 3.5 nS (n = 4), 13.0 ± 1.2 nS (n = 3), 17.0 ± 2.8 nS (n = 8) and 16.6 ± 5.1 nS (n = 4), respectively.
Pharmacology of the KATP current
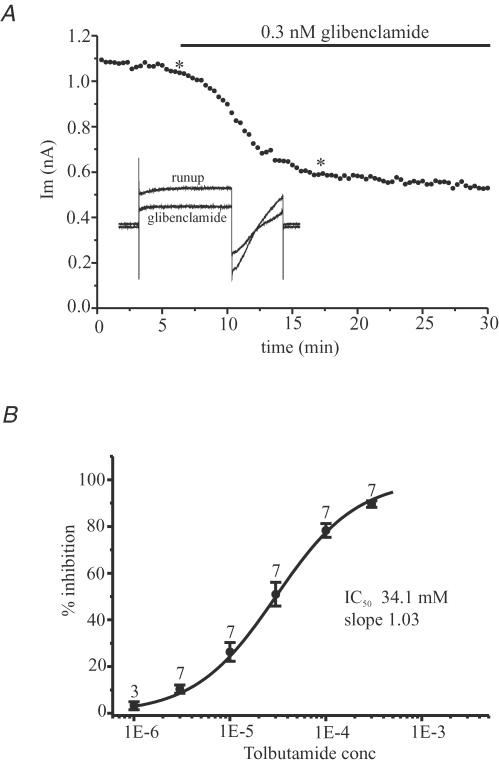
KATP channels can be assembled from a variety of different subunits. To date, five different channel subtypes have been identified, each displaying distinctive pharmacological and biophysical characteristics. Their sensitivity to inhibition by sulphonylurea compounds is one indicator of individual channel subunit composition. Channels composed of SUR1–Kir 6.2 or SUR2B–Kir6.1 subunits are both blocked by nanomolar concentrations of glibenclamide (Dunne et al. 1987; Gribble et al. 1998). In channels assembled from SUR1–Kir 6.2 subunits the block is irreversible, whereas in channels composed of SUR2B–Kir6.1 subunits the block is reversible (Gribble & Ashcroft, 2000). By contrast, tolbutamide reversibly inhibits all KATP channels but displays its highest affinity (Ki∼ 2–32 μm) for channels composed of SUR1–Kir 6.2 subunits (Inagaki et al. 1995; Gribble et al. 1997, 1998). In basal forebrain neurones, glibenclamide potently inhibited the KATP current at concentrations as low as 0.1–3 nm (see Fig. 6A). Inhibition by glibenclamide was found to be essentially irreversible with no recovery being observed during the time course of recordings (=45 min). The irreversible nature of the block precluded direct measurement of the IC50 value. By contrast tolbutamide reversibly inhibited the current with a mean IC50 of 34.1 ± 6.5 μm (n = 7; see Fig. 6B).
Figure 6.
A, inhibition of run-up current by glibenclamide. The KATP conductance was allowed to maximally activate under whole-cell recording conditions (0 mm[ATP]i). Ordinate is membrane current (Im) evoked by stepping from –80 to –40 mV (see inset) after run-up and in the presence of 0.3 nm glibenclamide. Inset shows membrane current in response to a 2 s step to –40 mV from Vh–80 mV followed by a 0.1 s step to –120 mV and ramp change in Vm to –40 mV (ramp 80 mV s−1) before stepping back to Vh (protocol repeated every 20 s) at the points recording indicated by the asterisk. B, dose–response curve for the KATP current to tolbutamide. The curve was constructed from the mean IC50 and slope values obtained from the individual cells. IC50 and Hill slope values were 34.1 μm and 1.03, respectively. All points are mean ± s.e.m.
KATP channels also display relatively low affinities for the K+ channel blockers tetraethylammonium (Fatherazi & Cook, 1991) and 4-aminopyridine (Koh et al. 1998). In cholinergic basal forebrain neurones, TEA and 4-AP (5 mm) blocked only 27.4 ± 4.2% (n = 6) and 2.75 ± 2.75% (n = 4), respectively, of tolbutamide-sensitive (200 μm) KATP current.
Single-channel properties
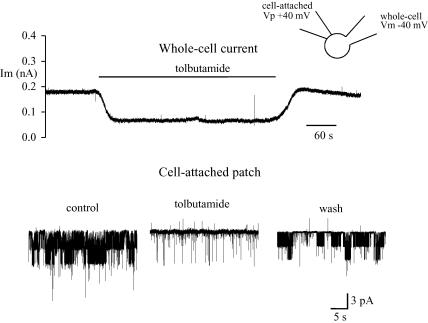
Information about the subunit composition of the channels can be also obtained from their single-channel characteristics. In order to correlate the whole-cell KATP current with the underlying single channels, an initial series of experiments was carried out to record single-channel activity using simultaneous cell-attached patch and macroscopic whole-cell current recording from the same cell. Figure 7 shows an example of a dual recording from a cholinergic basal forebrain neurone. In this case, the cell had been obtained from a 14-day-old animal and maintained in culture for 3 weeks. The basic configuration for the recording is shown in the top right panel. The whole-cell voltage-clamp recording was carried out as previously described. In addition, a second electrode containing the same filling solution as the whole-cell electrode was used to obtain a cell-attached patch recording from the same cell. A holding potential of +40 mV was applied to the patch pipette, whilst the cell was voltage clamped at –40 mV. This produced a driving force for K+ ion movement across the patch of 80 mV and approximately 50 mV for the whole-cell current. Following run-up of the whole-cell current, channel activity in the form of bursts of openings with many brief closures was observed in the patch (see lower panel in Fig. 7). Application of 100 μm tolbutamide reduced both the standing whole-cell current and inhibited channel activity in the patch. Both the whole-cell current and associated channel activity recovered upon washing.
Figure 7. Simultaneous recording of single-channel and whole-cell KATP currents from a cholinergic basal forebrain neurone maintained in culture.
Upper trace shows the whole cell current recorded from the cell after full activation of the KATP current. The cell was voltage clamped at a depolarized potential (–40 mV). The lower traces show single-channel activity from a cell-attached patch on the same cell (Vp+40 mV). Both the whole-cell and patch-electrodes were filled with 140 mm K+-containing solutions, whilst the bathing Krebs solution contained 3 mm K+. Under control conditions, an outward current was recorded by the whole-cell electrode and a high level of KATP channel activity was observed in the patch. Application of tolbutamide (100 μm) to the bathing solution reduced both the standing membrane current and single-channel activity. On washout the KATP current and channel activity both recovered. Note the occasional brief upward deflections from the zero current level are recording artefacts not channel openings.
Inside-out patches
In order to directly compare the properties of these KATP channels with those from cells in other studies, recordings were also carried out using inside-out patches excised cholinergic BF neurones maintained in culture. Patches were excised into high 140 mm K+ containing solution. On excision into solutions containing 0 mm ATP, initial KATP channel activity was usually quite high but generally declined to a much lower value within a few seconds. The extent of this initial loss of activity varied greatly between patches and in many cases channel activity was almost totally lost in less than a minute. Furthermore, most patches also contained other constitutively active inwardly rectifying K+ channels (Bajic et al. 2002), making it relatively difficult to study KATP channel activity in isolation. In patches containing only KATP channels, their activity was characterized by bursts of openings containing many brief closures (see Figs 8A and 9Ai), with individual bursts of channel openings being interspersed by prolonged closed states.
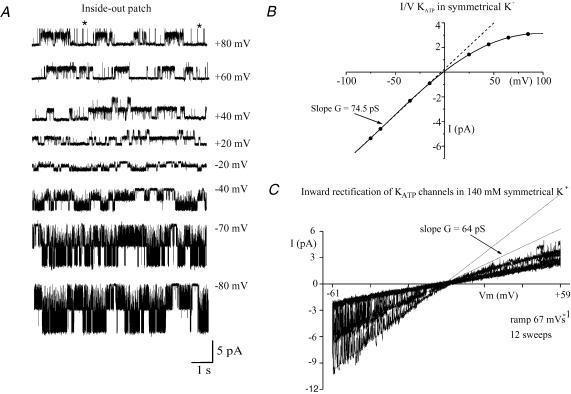
Figure 8. Voltage-dependent rectification by KATP channels in the inside-out recording configuration.
A, steady-state channel activity recorded in symmetrical 140 mm K+-containing solution for membrane potentials ranging between –80 and +80 mV (Note: the larger brief channel openings marked with an asterisk on the +80 mV record are clipped openings of a much larger unidentified channel). B, I–V curve for the single-channel currents shown in A. (Note: channel slope conductance was measured over the relatively linear region of the curve between –25 and –80 mV). C, rectification of single-channel currents as revealed by slowly ramping patch potential between –61 and +59 mV (ramp 67 mV s−1). The patch contained at least two KATP channels.
Figure 9. KATP channel pharmacology (inside-out configuration in symmetrical 140 mm K+).
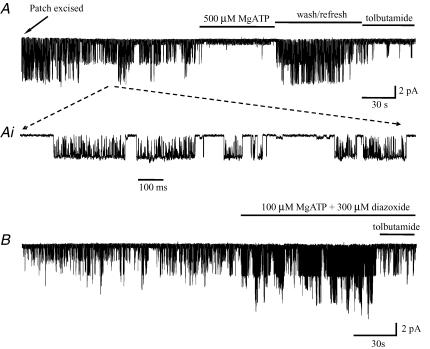
A, on excision there was an initial high level of channel activity which characteristically declined to a lower level (Vp+50 mV). Application of 0.5 mm MgATP inhibited all KATP channel activity (remaining small unidentified channel openings remain). On washout channel activity returned to a level similar to that observed immediately after excision but higher than that immediately prior to adding ATP (channel refreshment). Application of tolbutamide (100 μm) greatly reduced the frequency of channel openings. B, a second patch displaying increased channel opening in the presence of diazoxide (300 μm; Vp+80 mV). Again channel activity was greatly reduced by application of tolbutamide.
Single-channel conductance
Figure 8A shows typical channel activity over a range of membrane potentials between –80 and +80 mV. Under these conditions the channels displayed inward rectification at all potentials more positive than –20 mV. A plot of single-channel current versus membrane potential for the same patch is shown in panel B. Figure 8C shows channel rectification in a second patch in response to ramping the patch potential from –60 to +60 mV. In the example shown, the patch contained at least two channels and the dashed lines indicate the predicted single-channel currents in the absence of any rectification. Single-channel slope conductance was measured over the relatively linear region of the I–V curve between –80 and –30 mV. Under these conditions channel conductance ranged between 60 and 74.5 pS (mean 66.2 ± 1.9 pS, n = 7).
ATP sensitivity
Application of MgATP at concentrations =20 μm was observed to stimulate KATP channel activity. At higher concentrations (=50 μm) MgATP reduced channel activity and at concentrations (=500 μm) it virtually suppressed all KATP channel activity (Fig. 9A). Prolonged exposure to MgATP (0.2–1 mm) resulted, on washout, in transient refreshment of channel activity, restoring it to levels similar to those observed immediately following patch excision and significantly higher than that immediately prior to ATP application (n = 6; see Fig. 9A). In the presence of blocking concentrations of MgATP (100–500 μm), application of MgADP (200–500 μm) increased channel activity in 4 out of 5 patches tested.
Effects of K+ channel openers (KCOs) on single-channel currents
The sensitivity of KATP channels to KCOs can also be indicative of the sulphonylurea subunit composition. Diazoxide strongly activates channels with subunit composition SUR1–Kir6.2, but is largely ineffective on channels of composition SUR2A–Kir 6.2. In BF neurones, application of diazoxide (200–500 μm) in the presence of 100 μm MgATP strongly activated the KATP channels (mean increase in NPo486 ± 120%; n = 5; see Fig. 9B). By contrast pinacidil (200–500 μm), which most potently activates SUR2A/Kir 6.2 and has only a very weak effect upon SUR2B–Kir6.2- or SUR1–Kir6.2-containing channels, had no discernable effect (n = 5).
Glucose sensing
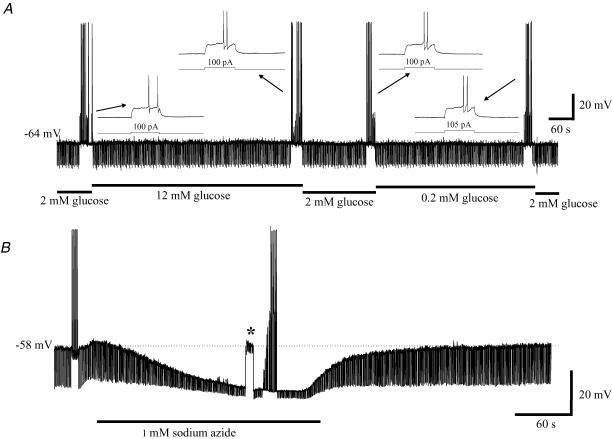
Within the brain a subpopulation of the neurones expressing KATP channels are also glucose responsive, i.e. regulate their firing rate by modulating KATP channel activity in response to changes in the extracellular glucose concentration (Levin et al. 2001). The possibility that some basal forebrain neurones might be glucose responsive/sensing was examined using perforated-patch recording. In vitro, BF neurones rarely exhibit any spontaneous action potential discharge. Therefore the effect of acute changes in extracellular glucose on resting membrane potential (Vm), input resistance (Rin) and excitability were assessed in response to direct current stimulation on switching from normal (2 mm) to low (0.1–0.2 mm; n = 11) or high (10–12 mm; n = 6) extracellular glucose for a period of 5 min (bath temperature 34–36°C; see Fig. 10A). Under these conditions, no consistent changes in Vm, Rin, the amplitude of the stimulating current required to evoke an action potential or the pattern of spike discharge in response to 100–500 ms depolarizing current stimuli were observed. By contrast metabolic inhibition in response to exposure to NaN3 (1 mm) for =5 min consistently activated KATP channels and reduced excitability (n = 5; see Fig. 10B).
Figure 10.
A, examination of the effect of acute changes in extracellular glucose levels on the excitability of cholinergic BF neurones. The illustrated recording was made from a cell maintained in culture for 6 days using the perforated-patch recording technique. Downward deflections are membrane voltage responses to current pulses delivered at 0.5 Hz (100 pA, 200 ms) used to monitor changes in input resistance. The threshold current required to evoke spike discharge was determined just prior to changing the extracellular glucose concentration (upward deflections). Insets show firing (200 ms duration pulses) and the threshold current under each condition. Note, prolonged current stimulation (0.5–1 s evoked a sustained slow rate of firing). B, the effect upon excitability of exposure to sodium azide (1 mm). Downward deflections are membrane voltage responses to 50 pA, 200 ms duration current pulses delivered at 1 Hz. On exposure to sodium azide membrane potential typically depolarized transiently before slowly hyperpolarizing. The hyperpolarization was associated with a fall in input resistance (Rin) and inhibited by tolbutamide confirming the involvement of KATP channels (data not shown). In the cell shown, Rin fell from 340 to 60 mΩ whilst the current required to evoke an action potential increased from 130 to 850 pA. Asterisk marks the region of the voltage trace where the change in membrane potential was nulled by direct current injection. From this it can be see that the majority of the fall in input resistance was the result of sodium azide-induced channel opening rather than as a secondary consequence of membrane hyperpolarization.
Discussion
The results of the present study indicate that virtually all magnocellular cholinergic basal forebrain neurones express functional KATP channels and that when activated they exert a profound inhibitory effect upon cell excitability. The single-channel conductance of basal forebrain KATP channels measured in symmetrical high K+-containing solutions ranged between 60 and 74.5 pS (mean 66.2 ± 1.9 pS). These values are similar to that which have been reported in several other central neurones (Ohno-Shosaku & Yamamoto, 1992; Schwanstecher & Panten, 1993) and consistent with the reported unitary conductance values for channels composed of SUR1–Kir6.2 (pancreatic β cell type) subunits (50–75 pS; Findlay et al. 1985; Ashcroft et al. 1988; Williams et al. 1993). By contrast SUR2A–Kir6.2 (cardiac ventricular type) containing channels have conductances of 67–90 pS (Nakayama et al. 1991; Babenko et al. 1998b; Kono et al. 2000) whilst SUR2B–Kir6.2 (smooth muscle type) channels range between 27 and 59 pS (Koh et al. 1998; Lee et al. 1999; Teramoto et al. 2000).
Pharmacologically the high affinity for glibenclamide coupled with the irreversible nature of the block is also consistent with BF KATP channels cells being composed of SUR1–Kir6.2 subunits (Gribble & Ashcroft, 2000). The affinity of the channels for tolbutamide (IC50 of 34.1 ± 6.5 μm) was somewhat lower than has previously been reported for SUR1–Kir6.2-containing channels (4–7 μm; Trube et al. 1986; Sakura et al. 1995; Gribble et al. 1998). This could indicate that these cells express multiple KATP channels isoforms. However, in all of the cells studied, the dose–response curves could be well fitted to a single binding equation.
Studies with KCOs were rather equivocal. Pinacidil, which strongly stimulates cardiac channels with almost no effect on pancreatic β-cell-type channels (Babenko et al. 1998b), was ineffective when applied either to excised patches or under whole-cell conditions. Diazoxide, which has a broader spectrum of activity, clearly activated KATP channels in excised membrane patches in the presence of low (0.5 mm) MgATP. However, under whole-cell perforated-patch recording conditions in the presence of normal extracellular glucose it was ineffective. This lack of effect of the KCOs could be due to the presence of a high intracellular ATP concentration which would compete with KCOs for the active site on the SUR subunit (Dunne et al. 1987; Fan et al. 1990). Consistent with this idea, reducing the intracellular ATP concentration by placing the cells in low glucose-containing solutions did increase the number of cells responding to diazoxide. A further contributory factor to the weak effect of diazoxide could be the presence of high levels of phosphatidlyinositol 4,5-bisphosphate (PIP2) within the cell (see below), which is also known to reduce the effectiveness of diazoxide (Baukrowitz et al. 1998).
Data from the present study, in particular, the observation that a large proportion of the KATP channels activated even when the recording electrode contained a high concentration of MgATP (4–6 mm), suggest that in intact cholinergic basal forebrain neurones the KATP channels exhibit a low affinity for ATP. In intact cells, reported values for the intracellular ATP concentration range from about 1.5 mm to 5 mm (Ashcroft & Ashcroft, 1990; Gribble & Ashcroft, 2000). Thus, if the ATP affinity of the KATP channels in intact BF cells is in the millimolar range then one might expect to observe some tonic activation, and indeed, using perforated-patch recordings which cause minimal disruption to the cell cytoplasm, a small proportion of the KATP channels were found to be tonically activated in a significant proportion of the cells tested.
In excised membrane patches the ATP affinity of the channels is in the micromolar range. Whilst a number of factors can subtly modulate the ATP affinity of KATP channels, the most likely explanation for the much lower affinity of the macroscopic current is the presence of high levels of PIP2, which has been reported to be capable of reducing the ATP affinity of the channels by several orders of magnitude (Baukrowitz et al. 1998). If PIP2 is involved, then this also raises the possibility that activation of receptor pathways resulting in changes in membrane phosphoinositide levels could indirectly modulate the excitability of BF cells by shifting the KATP channel activation threshold by altering channel affinity for ATP.
What is the functional role of the KATP channels expressed by cholinergic basal forebrain neurones? Within the CNS, a small population of so-called, glucose-responsive cells use KATP channels to directly regulate their firing in response to changes in ambient glucose levels (Miki et al. 2001). Short-term exposure (=5 min) of BF neurones to low or high extracellular glucose was found to have no discernable effect upon excitability. This apparent lack of a direct glucose-sensing role is similar to that which has been reported in the vast majority of other CNS neurones expressing KATP channels. The difference between the cells that can sense glucose and those that can't is believed to be the result of differential expression of the genes involved in glucose metabolism such that only the glucose-sensing neurones express the low affinity glucose transporter GLUT2 and the glucose phosphorylating enzyme glucokinase (Miki et al. 2001). Whilst basal forebrain neurones do not appear to be glucose responsive, their KATP channels could be activated in response to a reduction in intracellular ATP levels following metabolic inhibition with sodium azide. The most common role ascribed to KATP channels in non-glucose-sensing neurones is neuroprotection against excessive transmitter release and excitotoxicity or depolarization leading to excessive cell discharge as a result of hypoxia or ischaemia (Fellows et al. 1993). Under these conditions, the KATP channels open and hyperpolarize the cell membrane to protect them. The protective role of KATP channels may not be restricted to short-term metabolic insults. Chronic metabolic stress is also believed to play a role in various neurodegenerative disorders, including Alzheimer's disease (Mattson et al. 2001). Glucose administration has been shown to improve memory in Alzheimer's patients (Manning et al. 1993), whilst in rats glucose-induced improvements in memory have been shown to be attenuated by glibenclamide (Rashidy-Pour, 2001). Thus KATP channels could play a role in neuropathological conditions involving prolonged metabolic dysfunction.
All of the roles for the KATP channels discussed so far are pathophysiological in nature. However, the KATP channels may play an important physiological role. KATP channels have been implicated in several types of memory processes (Stefani et al. 1999; Stefani & Gold, 2001). Substantial evidence indicates that glucose may enhance memory by augmenting cholinergic functions (Durkin et al. 1992; Ragozzino et al. 1996). Intraseptal injections of glibenclamide or glucose to block KATP channels enhances spatial working memory (Stefani et al. 1999), whilst intrahippocampal infusion of glibenclamide increases ACh output and spatial memory (Stefani & Gold, 2001). KATP channel activators such as lemakalim have the converse effect (Stefani & Gold, 2001).
A high level of ascending cholinergic excitation from the basal forebrain is vital for maintaining cortical activation (Jones, 1993; Everitt & Robbins, 1997). However, during prolonged periods of wakefulness, there is a slow decline in glucose and ATP levels and a rise in the extracellular adenosine concentration (Netchiporouk et al. 2001; Van den Noort & Brine, 1970). The magnitude of the adenosine rise and thus by association the level of energy depletion is not homogeneous throughout the brain; rather, it is site specific, being significantly higher in the cholinergic region of the basal forebrain and, to a lesser extent the cortex, than in other brain regions (Porkka-Heiskanen et al. 1997, 2000). The adenosine activates inhibitory A1 receptors, resulting in a reduction in acetylcholine release and cortical excitation (Fredholm et al. 1990; Mogul et al. 1993). This scheme places adenosine as the prime candidate for the induction of sleep (Porkka-Heiskanen, 1999). One source for the extracellular adenosine is the hydrolysis of [ATP]i and its subsequent transport to the extracellular space by nucleoside transporters (Porkka-Heiskanen et al. 1997). As a consequence, there will also be a parallel fall in the intracellular ATP/ADP ratio, potentially leading to activation of KATP channels. The present study shows that if activated in this manner they could exert an additional, very powerful inhibitory effect on the excitability of cholinergic BF neurones that could also contribute, along with adenosine, to the induction, or maintenance of the duration or depth of sleep.
In conclusion, cholinergic basal forebrain neurones express KATP channels with properties very similar to those of channels composed of SUR1–Kir6.2 subunits. These channels exhibit a low affinity for [ATP]i and in many cells a small proportion of the channels are tonically activated and may thus play a modulatory role in controlling resting excitability. Further activation of these channels as a result of metabolic stress has a profound effect upon their excitability which may have important consequences for the maintenance of normal levels of cortical arousal and certain mneumonic processes.
Acknowledgments
The authors would like to thank Dr M. Mistry and Dr F. Abogadie for their help with the single-cell RT-PCR. This work was supported by the Medical Research Council.
References
- Allen TGJ. Regulation of excitability in cholinergic basal forebrain neurones by KATP channels. J Physiol. 2003;547.P:C23. doi: 10.1113/jphysiol.2003.055889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashcroft FM. Adenosine 5′-triphosphate-sensitive potassium channels. Annu Rev Neurosci. 1988;11:97–118. doi: 10.1146/annurev.ne.11.030188.000525. [DOI] [PubMed] [Google Scholar]
- Ashcroft FM, Ashcroft SJ, Harrison DE. Properties of single potassium channels modulated by glucose in rat pancreatic β-cells. J Physiol. 1988;400:501–527. doi: 10.1113/jphysiol.1988.sp017134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashcroft FM, Gribble FM. Correlating structure and function in ATP-sensitive K+ channels. Trends Neurosci. 1998;21:288–294. doi: 10.1016/s0166-2236(98)01225-9. [DOI] [PubMed] [Google Scholar]
- Ashcroft SJ, Ashcroft FM. Properties and functions of ATP-sensitive K-channels. Cell Signal. 1990;2:197–214. doi: 10.1016/0898-6568(90)90048-f. [DOI] [PubMed] [Google Scholar]
- Babenko AP, Aguilar-Bryan L, Bryan J. A view of sur/KIR6.X, KATP channels. Annu Rev Physiol. 1998a;60:667–687. doi: 10.1146/annurev.physiol.60.1.667. [DOI] [PubMed] [Google Scholar]
- Babenko AP, Gonzalez G, Aguilar-Bryan L, Bryan J. Reconstituted human cardiac KATP channels: functional identity with the native channels from the sarcolemma of human ventricular cells. Circ Res. 1998b;83:1132–1143. doi: 10.1161/01.res.83.11.1132. [DOI] [PubMed] [Google Scholar]
- Bajic D, Koike M, Albsoul-Younes AM, Nakajima S, Nakajima Y. Two different inward rectifier K+ channels are effectors for transmitter-induced slow excitation in brain neurons. Proc Natl Acad Sci U S A. 2002;99:14494–14499. doi: 10.1073/pnas.222379999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baukrowitz T, Schulte U, Oliver D, Herlitze S, Krauter T, Tucker SJ, Ruppersberg JP, Fakler B. PIP2 and PIP as determinants for ATP inhibition of KATP channels. Science. 1998;282:1141–1144. doi: 10.1126/science.282.5391.1141. [DOI] [PubMed] [Google Scholar]
- Benson DM, Blitzer RD, Landau EM. An analysis of the depolarization produced in guinea-pig hippocampus by cholinergic receptor stimulation. J Physiol. 1988;404:479–496. doi: 10.1113/jphysiol.1988.sp017301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brice A, Berrard S, Raynaud B, Ansieau S, Coppola T, Weber MJ, Mallet J. Complete sequence of a cDNA encoding an active rat choline acetyltransferase: a tool to investigate the plasticity of cholinergic phenotype expression. J Neurosci Res. 1989;23:266–273. doi: 10.1002/jnr.490230304. [DOI] [PubMed] [Google Scholar]
- Brown DA, Selyanko AA. Membrane currents underlying the cholinergic slow excitatory post-synaptic potential in the rat sympathetic ganglion. J Physiol. 1985;365:365–387. doi: 10.1113/jphysiol.1985.sp015777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clement J, Pt Kunjilwar K, Gonzalez G, Schwanstecher M, Panten U, Aguilar-Bryan L, Bryan J. Association and stoichiometry of K(ATP) channel subunits. Neuron. 1997;18:827–838. doi: 10.1016/s0896-6273(00)80321-9. [DOI] [PubMed] [Google Scholar]
- Dunne MJ, Illot MC, Peterson OH. Interaction of diazoxide, tolbutamide and ATP4− on nucleotide-dependent K+ channels in an insulin-secreting cell line. J Membr Biol. 1987;99:215–224. doi: 10.1007/BF01995702. [DOI] [PubMed] [Google Scholar]
- Dunn-Meynell AA, Rawson NE, Levin BE. Distribution and phenotype of neurons containing the ATP-sensitive K+ channel in rat brain. Brain Res. 1998;814:41–54. doi: 10.1016/s0006-8993(98)00956-1. [DOI] [PubMed] [Google Scholar]
- Durkin TP, Messier C, de Boer P, Westerink BH. Raised glucose levels enhance scopolamine-induced acetylcholine overflow from the hippocampus: an in vivo microdialysis study in the rat. Behav Brain Res. 1992;49:181–188. doi: 10.1016/s0166-4328(05)80163-9. [DOI] [PubMed] [Google Scholar]
- Everitt BJ, Robbins TW. Central cholinergic systems and cognition. Annu Rev Psychol. 1997;48:649–684. doi: 10.1146/annurev.psych.48.1.649. [DOI] [PubMed] [Google Scholar]
- Fan Z, Nakayama K, Hiraoka M. Pinacidil activates the ATP-sensitive K+ channel in inside-out and cell-attached patch membranes of guinea-pig ventricular myocytes. Pflugers Arch. 1990;415:387–394. doi: 10.1007/BF00373613. [DOI] [PubMed] [Google Scholar]
- Fatherazi S, Cook DL. Specificity of tetraethylammonium and quinine for three K channels in insulin-secreting cells. J Membr Biol. 1991;120:105–114. doi: 10.1007/BF01872393. [DOI] [PubMed] [Google Scholar]
- Fellows LK, Boutelle MG, Fillenz M. ATP-sensitive potassium channels and local energy demands in the rat hippocampus: an in vivo study. J Neurochem. 1993;61:949–954. doi: 10.1111/j.1471-4159.1993.tb03607.x. [DOI] [PubMed] [Google Scholar]
- Findlay I, Dunne MJ, Petersen OH. ATP-sensitive inward rectifier and voltage- and calcium-activated K+ channels in cultured pancreatic islet cells. J Membr Biol. 1985;88:165–172. doi: 10.1007/BF01868430. [DOI] [PubMed] [Google Scholar]
- Fredholm BB, Duner-Engstrom M, Fastbom J, Hu PS, van der Ploeg I. Role of G proteins, cyclic AMP, and ion channels in the inhibition of transmitter release by adenosine. Ann N Y Acad Sci. 1990;604:276–288. doi: 10.1111/j.1749-6632.1990.tb32000.x. [DOI] [PubMed] [Google Scholar]
- Gribble FM, Ashcroft FM. Sulfonylurea sensitivity of adenosine triphosphate-sensitive potassium channels from beta cells and extrapancreatic tissues. Metabolism. 2000;49:3–6. [PubMed] [Google Scholar]
- Gribble FM, Tucker SJ, Ashcroft FM. The interaction of nucleotides with the tolbutamide block of cloned ATP- sensitive K+ channel currents expressed in Xenopus oocytes: a reinterpretation. J Physiol. 1997;504:35–45. doi: 10.1111/j.1469-7793.1997.00035.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gribble FM, Tucker SJ, Seino S, Ashcroft FM. Tissue specificity of sulfonylureas: studies on cloned cardiac and beta-cell K(ATP) channels. Diabetes. 1998;47:1412–1418. doi: 10.2337/diabetes.47.9.1412. [DOI] [PubMed] [Google Scholar]
- Inagaki N, Gonoi T, Clement JP, Namba N, Inazawa J, Gonzalez G, Aguilar-Bryan L, Seino S, Bryan J. Reconstitution of IKATP: an inward rectifier subunit plus the sulfonylurea receptor. Science. 1995;270:1166–1170. doi: 10.1126/science.270.5239.1166. [DOI] [PubMed] [Google Scholar]
- Jones BE. The organization of central cholinergic systems and their functional importance in sleep-waking states. Prog Brain Res. 1993;98:61–71. doi: 10.1016/s0079-6123(08)62381-x. [DOI] [PubMed] [Google Scholar]
- Karschin C, Ecke C, Ashcroft FM, Karschin A. Overlapping distribution of K (ATP) channel-forming Kir6.2 subunit and the sulfonylurea receptor SUR1 in rodent brain. FEBS Lett. 1997;401:59–64. doi: 10.1016/s0014-5793(96)01438-x. [DOI] [PubMed] [Google Scholar]
- Koh SD, Bradley KK, Rae MG, Keef KD, Horowitz B, Sanders KM. Basal activation of ATP-sensitive potassium channels in murine colonic smooth muscle cell. Biophys J. 1998;75:1793–1800. doi: 10.1016/S0006-3495(98)77621-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kono Y, Horie M, Takano M, Otani H, Xie LH, Akao M, Tsuji K, Sasayama S. The properties of the Kir6.1–6.2 tandem channel co-expressed with SUR2A. Pflugers Arch. 2000;440:692–698. doi: 10.1007/s004240000315. [DOI] [PubMed] [Google Scholar]
- Lee K, Dixon AK, Richardson PJ, Pinnock RD. Glucose-receptive neurones in the rat ventromedial hypothalamus express KATP channels composed of Kir6.1 and SUR1 subunits. J Physiol. 1999;515:439–452. doi: 10.1111/j.1469-7793.1999.439ac.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin BE, Dunn-Meynell AA, Routh VH. Brain glucosensing and the K(ATP) channel. Nat Neurosci. 2001;4:459–460. doi: 10.1038/87405. [DOI] [PubMed] [Google Scholar]
- Manning CA, Ragozzino ME, Gold PE. Glucose enhancement of memory in patients with probable senile dementia of the Alzheimer's type. Neurobiol Aging. 1993;14:523–528. doi: 10.1016/0197-4580(93)90034-9. [DOI] [PubMed] [Google Scholar]
- Mattson MP, Duan W, Pedersen WA, Culmsee C. Neurodegenerative disorders and ischemic brain diseases. Apoptosis. 2001;6:69–81. doi: 10.1023/a:1009676112184. [DOI] [PubMed] [Google Scholar]
- Miki T, Liss B, Minami K, Shiuchi T, Saraya A, Kashima Y, Horiuchi M, Ashcroft F, Minokoshi Y, Roeper J, Seino S. ATP-sensitive K+ channels in the hypothalamus are essential for the maintenance of glucose homeostasis. Nat Neurosci. 2001;4:507–512. doi: 10.1038/87455. [DOI] [PubMed] [Google Scholar]
- Millar JA, Barratt L, Southan AP, Page KM, Fyffe RE, Robertson B, Mathie A. A functional role for the two-pore domain potassium channel TASK-1 in cerebellar granule neurons. Proc Natl Acad Sci U S A. 2000;97:3614–3618. doi: 10.1073/pnas.050012597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mogul DJ, Adams ME, Fox AP. Differential activation of adenosine receptors decreases N-type but potentiates P-type Ca2+ current in hippocampal CA3 neurons. Neuron. 1993;10:327–334. doi: 10.1016/0896-6273(93)90322-i. [DOI] [PubMed] [Google Scholar]
- Mourre C, Widmann C, Lazdunski M. Sulfonylurea binding sites associated with ATP-regulated K+ channels in the central nervous system: autoradiographic analysis of their distribution and ontogenesis, and of their localization in mutant mice cerebellum. Brain Res. 1990;519:29–43. doi: 10.1016/0006-8993(90)90057-i. [DOI] [PubMed] [Google Scholar]
- Nakayama K, Fan Z, Marumo F, Sawanobori T, Hiraoka M. Action of nicorandil on ATP-sensitive K+ channel in guinea-pig ventricular myocytes. Br J Pharmacol. 1991;103:1641–1648. doi: 10.1111/j.1476-5381.1991.tb09841.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netchiporouk L, Shram N, Salvert D, Cespuglio R. Brain extracellular glucose assessed by voltammetry throughout the rat sleep-wake cycle. Eur J Neurosci. 2001;13:1429–1434. doi: 10.1046/j.0953-816x.2001.01503.x. [DOI] [PubMed] [Google Scholar]
- Ohno-Shosaku T, Yamamoto C. Identification of an ATP-sensitive K+ channel in rat cultured cortical neurons. Pflugers Arch. 1992;422:260–266. doi: 10.1007/BF00376211. [DOI] [PubMed] [Google Scholar]
- Porkka-Heiskanen T. Adenosine in sleep and wakefulness. Ann Med. 1999;31:125–129. doi: 10.3109/07853899908998788. [DOI] [PubMed] [Google Scholar]
- Porkka-Heiskanen T, Strecker RE, McCarley RW. Brain site-specificity of extracellular adenosine concentration changes during sleep deprivation and spontaneous sleep: an in vivo microdialysis study. Neuroscience. 2000;99:507–517. doi: 10.1016/s0306-4522(00)00220-7. [DOI] [PubMed] [Google Scholar]
- Porkka-Heiskanen T, Strecker RE, Thakkar M, Bjorkum AA, Greene RW, McCarley RW. Adenosine: a mediator of the sleep-inducing effects of prolonged wakefulness. Science. 1997;276:1265–1268. doi: 10.1126/science.276.5316.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rae J, Cooper K, Gates P, Watsky M. Low access resistance perforated patch recordings using amphotericin B. J Neurosci Meth. 1991;37:15–26. doi: 10.1016/0165-0270(91)90017-t. [DOI] [PubMed] [Google Scholar]
- Ragozzino ME, Unick KE, Gold PE. Hippocampal acetylcholine release during memory testing in rats: augmentation by glucose. Proc Natl Acad Sci U S A. 1996;93:4693–4698. doi: 10.1073/pnas.93.10.4693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rashidy-Pour A. ATP-sensitive potassium channels mediate the effects of a peripheral injection of glucose on memory storage in an inhibitory avoidance task. Behav Brain Res. 2001;126:43–48. doi: 10.1016/s0166-4328(01)00242-x. [DOI] [PubMed] [Google Scholar]
- Sakura H, Ammala C, Smith PA, Gribble FM, Ashcroft FM. Cloning and functional expression of the cDNA encoding a novel ATP-sensitive potassium channel subunit expressed in pancreatic beta-cells, brain, heart and skeletal muscle. FEBS Lett. 1995;377:338–344. doi: 10.1016/0014-5793(95)01369-5. [DOI] [PubMed] [Google Scholar]
- Schwanstecher C, Panten U. Tolbutamide- and diazoxide-sensitive K+ channel in neurons of substantia nigra pars reticulata. Naunyn Schmiedebergs Arch Pharmacol. 1993;348:113–117. doi: 10.1007/BF00168546. [DOI] [PubMed] [Google Scholar]
- Schwanstecher M, Sieverding C, Dorschner H, Gross I, Aguilar-Bryan L, Schwanstecher C, Bryan J. Potassium channel openers require ATP to bind to and act through sulfonylurea receptors. EMBO J. 1998;17:5529–5535. doi: 10.1093/emboj/17.19.5529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sim JA, Allen TG. Morphological and membrane properties of rat magnocellular basal forebrain neurons maintained in culture. J Neurophysiol. 1998;80:1653–1669. doi: 10.1152/jn.1998.80.4.1653. [DOI] [PubMed] [Google Scholar]
- Standen NB, Stanfield PR. Potassium depletion and sodium block of potassium currents under hyperpolarization in frog sartorius muscle. J Physiol. 1979;294:497–520. doi: 10.1113/jphysiol.1979.sp012943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefani MR, Gold PE. Intrahippocampal infusions of K-ATP channel modulators influence spontaneous alternation performance: relationships to acetylcholine release in the hippocampus. J Neurosci. 2001;21:609–614. doi: 10.1523/JNEUROSCI.21-02-00609.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefani MR, Nicholson GM, Gold PE. ATP-sensitive potassium channel blockade enhances spontaneous alternation performance in the rat: a potential mechanism for glucose-mediated memory enhancement. Neuroscience. 1999;93:557–563. doi: 10.1016/s0306-4522(99)00128-1. [DOI] [PubMed] [Google Scholar]
- Teramoto N, Brading AF, Ito Y. Possible underestimation of the channel conductance underlying pinacidil-induced K+ currents using noise analysis in pig urethral myocytes. J Pharm Pharmacol. 2000;52:1395–1403. doi: 10.1211/0022357001777397. [DOI] [PubMed] [Google Scholar]
- Treherne JM, Ashford ML. The regional distribution of sulphonylurea binding sites in rat brain. Neuroscience. 1991;40:523–531. doi: 10.1016/0306-4522(91)90138-e. [DOI] [PubMed] [Google Scholar]
- Trube G, Rorsman P, Ohno-Shosaku T. Opposite effects of tolbutamide and diazoxide on the ATP-dependent K+ channel in mouse pancreatic beta-cells. Pflugers Arch. 1986;407:493–499. doi: 10.1007/BF00657506. [DOI] [PubMed] [Google Scholar]
- Van den Noort S, Brine K. Effect of sleep on brain labile phosphates and metabolic rate. Am J Physiol. 1970;218:1434–1439. doi: 10.1152/ajplegacy.1970.218.5.1434. [DOI] [PubMed] [Google Scholar]
- Williams BA, Smith PA, Leow K, Shimizu S, Gray DW, Ashcroft FM. Two types of potassium channel regulated by ATP in pancreatic B cells isolated from a type-2 diabetic human. Pflugers Arch. 1993;423:265–273. doi: 10.1007/BF00374405. [DOI] [PubMed] [Google Scholar]
- Xu XH, Pan YP, Wang XL. mRNA expression alterations of inward rectifier potassium channels in rat brain with cholinergic impairment. Neurosci Lett. 2002;322:25–28. doi: 10.1016/s0304-3940(02)00071-x. [DOI] [PubMed] [Google Scholar]
- Yamaguchi K, Nakajima Y, Nakajima S, Stanfield PR. Modulation of inwardly rectifying channels by substance P in cholinergic neurones from rat brain in culture. J Physiol. 1990;426:499–520. doi: 10.1113/jphysiol.1990.sp018151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Z, Surmeier DJ. Muscarinic (m2/m4) receptors reduce N- and P-type Ca2+ currents in rat neostriatal cholinergic interneurons through a fast, membrane-delimited, G-protein pathway. J Neurosci. 1996;16:2592–2604. doi: 10.1523/JNEUROSCI.16-08-02592.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]