Abstract
We hypothesized that ultrafiltrate crossing the luminal endothelial glycocalyx through infrequent discontinuities (gaps) in the tight junction (TJ) strand of endothelial clefts reduces albumin diffusive flux from tissue into the ‘protected region’ of the cleft on the luminal side of the TJ. Thus, the effective oncotic pressure difference (σΔπ) opposing filtration is greater than that measured between lumen and interstitial fluid. To test this we measured σΔπ across rat mesenteric microvessels perfused with albumin (50 mg ml−1) with and without interstitial albumin at the same concentration within a few micrometres of the endothelium as demonstrated by confocal microscopy. We found σΔπ was near 70% of luminal oncotic pressure when the tissue concentration equalled that in the lumen. We determined size and frequency of TJ strand gaps in endothelial clefts using serial section electron microscopy. We found nine gaps in the reconstructed clefts having mean spacing of 3.59 μm and mean length of 315 nm. The mean depth of the TJ strand near gaps was 67 nm and the mean cleft path length from lumen to interstitium was 411 nm. With these parameters our three-dimensional hydrodynamic model confirmed that fluid velocity was high at gaps in the TJ strand so that even at relatively low hydraulic pressures the albumin concentration on the tissue side of the glycocalyx was significantly lower than in the interstitium. The results conform to the hypothesis that colloid osmotic forces opposing filtration across non-fenestrated continuous capillaries are developed across the endothelial glycocalyx and that the oncotic pressure of interstitial fluid does not directly determine fluid balance across microvascular endothelium.
The classic Starling equation describing the balance of hydrostatic and colloid osmotic (oncotic) forces which determine filtration and reabsorption across the capillary wall includes four forces:
where Pc and Pt are the hydrostatic pressures in the capillary lumen and tissue, respectively, and πc and πt are the corresponding lumen and tissue oncotic pressures. Jv/A is the filtration rate per unit area, σ is the reflection coefficient to the plasma proteins and Lp is the hydraulic conductivity of the vessel wall. This relation has been tested by changing Pc and πc (only rarely by changing Pt or πt) and confirmed many times in both whole organ and isolated microvessels, but usually under conditions where the tissue protein concentration was low, due to wash-down of the tissue proteins after rapid filtration, or washout of tissue protein in exposed superfused tissue (Pappenheimer & Soto-Rivera, 1948; Michel et al. 1974; Granger & Taylor, 1980). Thus the precise contribution of the interstitial Starling forces warrants further study. For example, Reed and colleagues have demonstrated that large negative pressures can contribute to oedema formation under inflammatory conditions (Reed et al. 1992). With respect to interstitial oncotic pressure, filtration of normally low protein concentration fluid into interstitium tends to diminish πt by dilution of the interstitial fluid. This change opposes further increase in fluid filtration, providing a buffering or moderating influence on fluid filtration. The latter idea, incorporated into the concepts of safety factors against oedema, resulted from direct measurements of interstitial pressure and interstitial fluid composition (Guyton, 1963; Aukland & Reed, 1993).
Over recent years evidence has accumulated that the tissue oncotic pressure may not contribute to the balance of osmotic and hydrostatic forces to the same extent as the other three forces. Specifically, Levick pointed out that, if the most accurate values for tissue oncotic pressure are substituted into the Starling equation, then most tissues are not in fluid balance, and would filter far more fluid than can be accounted for by measured lymph flows (Levick, 1991). Further, using direct measurements Levick demonstrated in the synovium that tissue albumin had much less effect on fluid exchange than the same concentration administered intravascularly (McDonald & Levick, 1993).
Michel (1997) and Weinbaum (1998) proposed that the effective protein osmotic barrier is not the whole capillary wall, but the luminal glycocalyx which acts as the primary molecular filter. If this is the case, the plasma protein concentration on the tissue side of the glycocalyx in the cleft between adjacent endothelial cells may be much less than it is in the tissue because the back-diffusion of the plasma protein from the tissue into the cleft will be reduced by the tissue-directed convective flow. In particular, the degree of reduction depends on the velocity of the ultrafiltrate in the cleft distal to the glycocalyx. Thus the concentration difference of the plasma protein across the glycocalyx may be much larger than that estimated from the plasma-to-tissue concentration difference. The effectiveness of this mechanism depends on the geometry of the filtration path in the cleft, and on the gradients of tissue plasma proteins near the tissue side of the vascular wall.
A detailed model of the albumin concentration gradients across the glycocalyx and in the cleft between adjacent endothelial cells was developed by Hu and Weinbaum, based on the carefully measured geometry of the size and frequency of junction strand breaks in frog mesenteric microvessels (Adamson & Michel, 1993; Hu & Weinbaum, 1999). In our previous paper we directly compared the prediction of a three-dimensional model of filtration through a fibre-matrix-junctional break model after loading the tissue with serum albumin at a concentration of 50 mg ml−1, and perfusing a vessel with albumin at the same concentration. We found that there was no significant difference between the oncotic pressure exerted by the perfusate when there was no albumin in the tissue and when albumin was in the tissue at the same concentration as in the lumen.
The key observation is that albumin is filtered at the glycocalyx, and the ultrafiltrate with its low concentration of albumin is funnelled into infrequent breaks in the junction strand so that the water velocity at these breaks reduces the flux of albumin from tissue toward the glycocalyx, even when filtration rates are quite low. The aim of the present experiments was to test this hypothesis in detail in mammalian capillaries. Little information about the size and frequency of breaks in the junctional strand was available for mammalian capillaries when we began this investigation. In fact, the series of serial sections reported by Bundgaard, on rat heart microvessels, focused on very small breaks in the junction strand that might act as molecular filters and provided no detailed data on the few larger breaks that were noted in rat heart (Bundgaard, 1984).
Thus, the first of three key steps in evaluation of this new hypothesis has been to measure oncotic pressure across rat mesenteric microvessels perfused with albumin at a concentration of 50 mg ml−1, with and without albumin at the same concentration in the superfusate. The second step has been to carry out detailed reconstructions of the organization of the junction strands in the microvessels from serial sections. All reconstructions have been carried out on microvessels in which the hydraulic conductivity was also measured. A final important step was to directly measure the gradients of albumin in the tissue around the microvessels during both high and low filtration rates. The microvessels we studied have hydraulic conductivities similar to those of rat heart, and overlap the range of Lp values reported for mammalian skeletal muscle and skin (Renkin, 1977). Our results provide new evidence that the effective oncotic pressure opposing net filtration in mammalian continuous capillaries is developed across the endothelial glycocalyx and that the oncotic pressure of interstitial fluid does not directly determine fluid balance across microvascular endothelium.
Methods
Animal preparation
Experiments were carried out on rats (male, Sprague–Dawley, 350–450 g, Hilltop Laboratory Animals, Inc.) anaesthetized with pentobarbital (65 mg (kg body weight)−1 given subcutaneously). Anaesthesia, assessed by toe-pinch reflex, was maintained by giving additional pentobarbital (subcutaneous 3 mg per dose) as needed. At the end of experiments, animals were killed with a pentobarbital overdose. All animal protocols were approved by the Institutional Animal Care and Use Committee of the University of California, Davis. Each rat was anaesthetized as above and placed on a heating pad to maintain normal body temperature. A midline surgical incision (2–3 cm) was made in the abdominal wall, and the gut gently taken out from the abdominal cavity to spread the mesentery over a quartz pillar for transillumination and observation on the fixed stage of an inverted microscope. The upper surface of the mesentery was continuously superfused with Ringer solution maintained at 35–37°C during preparation and experimentation. Because our present techniques are not suited to cannulation and perfusion of vessels smaller than about 15 μm diameter we have not investigated the properties of true capillaries (diameter ca 6 μm). Therefore, experiments were performed on straight non-branched segments of venular microvessels typically 25–40 μm in diameter. Prior to cannulation, all vessels selected for experiments had brisk blood flow and were free of leucocytes sticking or rolling on the vessel wall.
Measurement of Lp and effective oncotic pressure
All measurements were based on the modified Landis technique, which measures the volume flux of water across the wall of a microvessel perfused via a glass micropipette following downstream occlusion of the vessel. The assumptions and limitations of the measurement have been evaluated in detail (Michel, 1980; Curry et al. 1983; Kendall & Michel, 1995; Adamson et al. 2002). The initial transcapillary water flow per unit area of the capillary wall (Jv/A) was measured at predetermined capillary pressures of 15–60 cmH2O. Microvessel Lp was calculated as the slope of the relation between Jv/A and applied hydraulic pressure.
Implicit in our calculations is the assumption that Pt (interstitial hydrostatic pressure) is negligible in our rat mesentery preparation. If it were negative we would underestimate the driving force for outward fluid filtration and if it were positive we would overestimate the force, leading us to over- or underestimate Lp, respectively. However, as has been demonstrated previously in frog mesentery, when both the perfusate and superfusate are protein free, the relationship between Jv/A and Pc is linear and extrapolates through the origin, a condition which would not be expected for non-zero Pt (Michel et al. 1974). Moreover, direct measurements using servo-null techniques in rat mesentery show that Pt is within 0.5 cmH2O of atmospheric pressure and does not vary under high or low filtration (Kajimura et al. 2001). Therefore, it is reasonable to assume that Pt is negligible in the rat mesentery preparation.
The primary set of experiments was designed to determine the effective oncotic pressure of albumin (50 mg ml−1) in the perfusate, first when there was no albumin in the extravascular space (control) and then when albumin was present in the tissue at the same concentration as in the perfusate. The latter condition was achieved by adding albumin (50 mg ml−1) to the superfusate. During control measurements the surface of the mesentery was continuously bathed in protein-free Ringer solution. Jv/A was estimated from the motion of marker erythrocytes using the modified Landis technique (Michel, 1980; Hu et al. 2000). Before each measurement the pressure in the vessel was held at 60 cmH2O for 30 s by partial downstream occlusion to establish a high filtration steady state in which the albumin concentration on the downstream side of the selectivity barrier was washed down to a minimal level, thus maximizing the effective oncotic pressure difference (Michel, 1984; Michel & Curry, 1999). Jv/A was measured by monitoring the motion of flow marker erythrocytes during brief (5–8 s) full occlusions. Measurements of Jv/A at a microvessel pressure of 15 cmH2O were made after setting up steady-state filtration at 60 cmH2O, occluding, and rapidly (< 0.5 s) switching the applied hydraulic pressure from 60 to 15 cmH2O. Reabsorptive water movement was seen as marker erythrocytes moved away from the occlusion site toward the cannulation site as fluid moved into the vessel. Three to five measurements were made at each pressure, yielding a control relationship between Jv/A and applied hydraulic pressure. The slope of the relationship is the hydraulic conductivity (Lp) and the effective oncotic pressure (σΔπ) of the serum albumin was equal to the intercept on the pressure axis. Under the condition of initially high Jv/A the value of σΔπ approximates to σ2πc (Michel, 1984). Using control measurements of Jv/A having protein-free Ringer as the superfusate and the known value of perfusate oncotic pressure we calculated σ of the vessel wall to albumin. Our test solution of bovine serum albumin (BSA; 50 mg ml−1) had an oncotic pressure of 27 cmH2O at 37°C (calculated from published equations: McDonald & Levick, 1993).
To investigate the effect of the presence of extravascular albumin on the effective oncotic pressure, measurements were repeated on each vessel using the same protocol as above except that serum albumin was added to the superfusate at the same concentration as in the perfusate (50 mg ml−1) and allowed to equilibrate for 20 min before starting measurements. If the high concentration of extravascular albumin were effective in balancing the oncotic pressure of the perfusate albumin, we would have expected to see no effective oncotic pressure and the extrapolated relationship between Jv/A and applied pressure should have intercepted the pressure axis at the origin.
As a second test of the effective oncotic pressure of extravascular serum albumin we measured filtration rates across the vessel wall, having set up steady-state filtration not only at 60 cmH2O, but also at lower pressures (10–40 cmH2O). Partial occlusions were used at each pressure to set up the steady-state flow for 60 s prior to measuring Jv/A during a full occlusion (Michel & Phillips, 1987; Hu et al. 2000).
The use of erythrocytes as flow markers overestimates transluminal volume flow due to the tendency of the erythrocytes to move with flow along the vessel centreline (Kendall & Michel, 1995). In rat mesentery venular microvessels this results in over-estimation of Jv/A by nearly a factor of two. To accurately predict volume flows in our model we needed to estimate the true value of Jv/A. We assumed that flow marker erythrocytes travelled with the centreline flow of the vessel. Mean fluid velocity in the vessel lumen (u) was calculated from marker cell velocity (uc) using the relation
where rc is the radius of the erythrocyte (3.5 μm) and R is the vessel radius (Michel et al. 1974; Kendall & Michel, 1995). All values of Jv/A and Lp were reported as true values.
Confocal microscopy to determine distribution of extravascular albumin
Methods for collecting images and analysing the distribution of fluorescently labelled albumin within mesenteric interstitium were similar to those previously published (Adamson et al. 1994; Hu et al. 2000). Briefly, the tray holding the rat had a glass coverslip through which to observe the mesentery using an inverted microscope with a laser confocal attachment (Zeiss LSM 510). Confocal images (256 intensity levels) were taken transversely to the vessel axis with a × 40 (1.3 NA) lens using 543 nm excitation wavelength and 560 nm long-pass emission detection. Acquisitions consisting of 35 line scans (x–z mode) were collected every 20 s with 1.3 μm optical section and 2 μm vertical step yielding images of tissue 230 μm by 35 μm centred on the perfused vessel. Sequences of images were collected for up to 20 min and examined for fluorescence intensity profiles (Zeiss LSM 510 and NIH Image J Software).
Confocal microscopy to determine cleft length per unit area of vessel wall
To estimate total cleft length per unit area of vessel wall (Fig. 1A) we used confocal microscopy techniques as previously published (He & Adamson, 1995; Curry et al. 2003). Briefly, after completion of Lp measurements, each vessel was cannulated and perfused via a pipette containing AgNO3 (1 mg ml−1) for 10–20 s. The pipette was removed and replaced with one containing Ringer solution plus BSA (10 mg ml−1), the vessel was occluded with pipette pressure set to 50 cmH2O, and fixative (2% glutaraldehyde, 4% formaldehyde, 0.2 m phosphate, 0.15 m NaCl) was dripped on the mesentery. After development of silver lines under a bright light, the vessel was imaged with the laser scanning confocal microscope in transmission mode (× 25 lens, 0.8 NA). The perimeters (pc) and areas (Ac) of individual cells were measured on the captured images. Cleft length per unit area (Lc) was calculated as (0.5 pcAc−1).
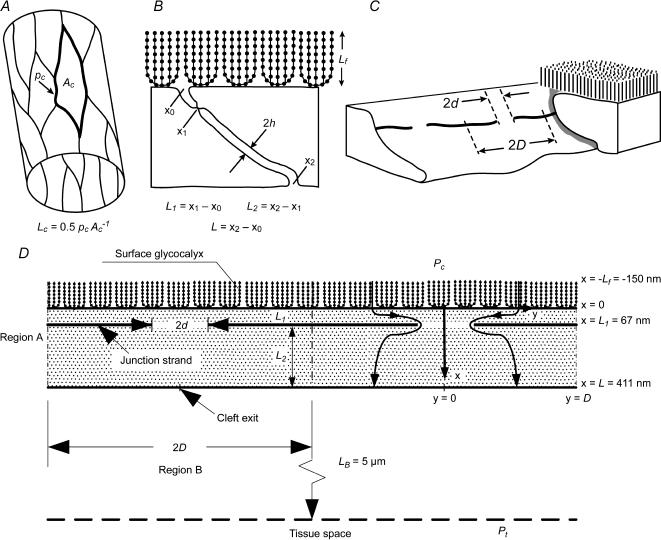
Figure 1. Measured parameters and geometric model of the endothelial cleft.
Diagrams illustrating measurement parameters used to construct mathematical model. A, area (Ac) and perimeter (pc) measurements of individual cells seen in silver-stained whole mounts of venular microvessels were used to calculate cleft length per unit area, Lc. B, from each electron microscopic image the cleft depth, L, was measured along the contour between facing cells from the luminal cleft opening (x0) to the abluminal cleft exit (x2). The total distance from lumen to interstitium through the cleft is the sum of L1, the distance to the tight junction strand (x1) from luminal cleft entrance, and L2, the distance from tight junction strand to the abluminal cleft exit. The cleft width is the distance between the outer leaflets of the cell membranes of the two facing cells. Lf, depth of the glycocalyx as determined by Squire et al. (2001). C, oblique view of cleft segment reconstructed from serial sections illustrates the length of tight junction strand gaps, 2d, and the mean distance between strand gap centres, equal to the functional unit length, 2D. D, the idealized diagram representing the mathematical model with values for the cleft and glycocalyx incorporating results of the present study and literature values as given in Table 1. Pc, hydraulic pressure in the vessel lumen; Pt, hydraulic pressure in the interstitium; LB, radius of the near-field region; dimension x corresponds to orientation of cleft depth, L; dimension y corresponds to orientation of cleft length, Lc; dimension z (perpendicular to plane of diagram) corresponds to orientation of cleft width, 2h; other parameters as above.
Ultrastructure
In the experiments to examine ultrastructural morphology, each venule was perfused with a solution containing BSA (Sigma-Aldrich A4378) at 10 mg ml−1 in Ringer solution. For these experiments, Jv/A was estimated from each occlusion at one hydraulic pressure with the assumption that the net effective pressure determining fluid flow was equal to the applied hydraulic pressure minus 3 cmH2O, the approximate oncotic pressure contributed by the BSA in the perfusates. Usually between 5 and 10 occlusions at 50 cmH2O over 10–20 min were used to estimate the Lp. After the final determination of Jv/A, fixation for electron microscopy was started by dripping ice-cold glutaraldehyde (3% v/v in 0.1 m cacodylate buffer) on the mesentery. This was done while vessels were still perfused and occluded to ensure that the final perfusion conditions and geometry were maintained during fixation. A sketch of the area enabled later identification of the perfused vessel segment. After 10 min in situ fixation, tissue was cut away from the animal and glutaraldehyde fixation continued on ice for a total of 1 h before changing to OsO4 (1% in 0.1 m cacodylate, pH 7.2) for 1 h. Tissue was treated with tannic acid (0.5%) in maleate buffer (0.05 m, pH 6) for 30 min to enhance membrane staining, and placed in uranyl acetate (2% in maleate buffer, pH 6) overnight. After dehydration in a graded series of acetone, the tissues were flat embedded in epoxy resin.
Ribbons of serial sections (from 20 to 40 sections per ribbon with 40 or 50 nm section thickness) from transversely sectioned vessels were collected on formvar-coated slots. Sections were stained with saturated uranyl acetate and lead citrate and examined on a Philips CM120 at 80 kV using a goniometric stage. Images were recorded at a nominal electronic magnification of × 27 500 using a 2K × 2K digital camera yielding a pixel size of about 1 nm by 1 nm (MegaScan, Gatan). The goniometric stage was tilted (up to ±50 deg) to resolve the presence or absence of tight junctions. We measured contour lengths of clefts from the luminal entrance to the abluminal exit, depth of tight junction strands and cleft widths (Fig. 1B). Measurement software (Digital Micrograph, Gatan) was calibrated using a replica grating (2160 lines mm−1) and 0.261 μm latex spheres. The position of the cleft entrance or exit was defined as the point at which the cleft width became abruptly larger than the nearby wide region (typically >18 nm). The width of the cleft away from tight junctions, i.e. the wide region, was determined from intensity plots drawn across the cleft in regions of images showing greatest membrane contrast and minimal cell membrane thickness to ensure that the cleft was orientated as nearly as possible perpendicularly to the electron beam. The cleft width was defined as the distance between the minimal intensities corresponding to the staining of the outer membrane leaflets of the facing cells.
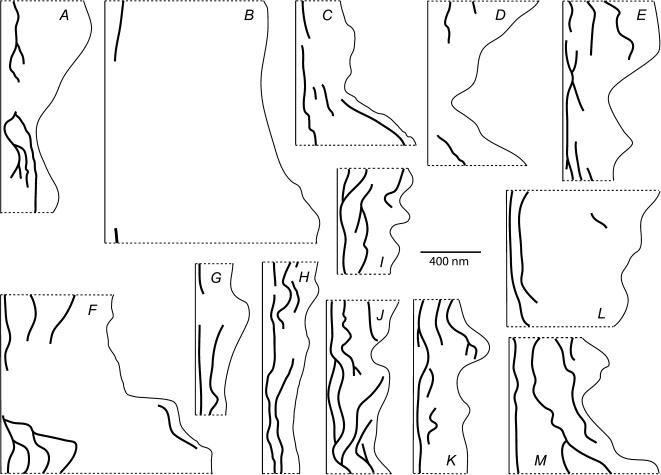
Continuity of tight junction strands was assessed by following the appearance of tight junctions from section to section. When present, tight junctions occluded the space between endothelial cells and the outer membrane leaflets appeared to touch and, at some locations, appeared to fuse. Where tight junction continuity ended, the distance between cells opened to equal the width of the wide part of the cleft, typically 18 nm. The length of tight junction strand gaps was determined by multiplying the number of sections in which a tight junction region was open by the nominal section thickness. The mean distance from one strand gap centre to the next was calculated as the total length of serially sectioned cleft divided by the number of gaps found (Fig. 1C).
We examined all images for cleft-spanning structures only after they had been collected as above to be used for tight junction analysis. Because the collections were not optimized for analysis of the crossbridges we did not attempt to quantify their frequency or structural details.
Solutions and reagents
Mammalian Ringer solution was composed of (mm): 132 NaCl, 4.6 KCl, 2 CaCl2, 1.2 MgSO4, 5.5 glucose, 5.0 NaHCO3, and 20 Hepes and Na-Hepes. The ratio of acid-Hepes to Na-Hepes was adjusted to achieve pH 7.40–7.45 for Ringer solutions. All perfusates were mammalian Ringer solution containing additional BSA at 10 or 50 mg ml−1 as indicated. BSA was labelled with the fluorophore Alexa Fluor 546 (Molecular Probes) as per manufacturer's instructions. A trace amount (0.1 mg ml−1) of Alexa Fluor 546–BSA was added to the superfusate during confocal imaging experiments performed to examine the equilibration of albumin between superfusate and tissue.
Statistics concerning in vivo experiments
Throughout, averaged values were reported as mean ± s.e.m. The indicated statistical tests were performed assuming significance for probability levels < 0.05.
Mathematical model of fluid flux through the cleft
Our theoretical model for the prediction of Lp and the steady-state concentration profiles in the cleft and tissue for rat mesentery venules is shown in Fig. 1D. This model is based on the morphometric measurements summarized in Table 1 and the mean values of Lp and σ for albumin found in the present in vivo measurements. In the current mathematical model, σ of the glycocalyx is assumed to equal the value found in our in vivo measurements. Key dimensions for the cleft geometry are shown in the figure. This basic model differs from that used previously for frog mesentery capillary in three important respects (Hu & Weinbaum, 1999; Hu et al. 2000). First, as described by Weinbaum and colleagues, the model for the surface glycocalyx has been modified to correspond to the ultrastructure suggested by the latest autocorrelation imaging techniques described by Squire and colleagues, where quasi-periodic structures were observed for both the scattering centres along and between the core proteins and the underlying actin scaffold (Squire et al. 2001; Weinbaum et al. 2003). The glycocalyx is composed of bush-like clusters of core proteins emanating from central foci that are spaced at 100 nm intervals in an ordered hexagonal array. The scattering centres along the core proteins are 10–12 nm in diameter and their spacing is roughly 20 nm both in the directions parallel and perpendicular to the plasma membrane. Squire and colleagues suggested the possibility that the observed dimensions and spacing of the scattering centres could correspond to the distribution of aggregated glycan side chains along a proteoglycan molecule. Second, our present morphological observations and the previous studies by Schulze & Firth (1992) on mammalian heart capillaries indicate that there is a periodic array of cleft-spanning structures that probably fill the entire cleft. These could be cadherins or an equivalent molecule that is responsible for the remarkably uniform gap height of the cleft outside of the junctional region. Based on the extensive measurements in Schulze & Firth (1992) we have assumed that the cleft-spanning molecules are spaced in a hexagonal array at 15 nm intervals and their diameter is taken as 2.5 nm, the diameter of cadherins (Shapiro et al. 1995; Boggon et al. 2002). Third, the tight junction strand has been located asymmetrically in the cleft, only 15% of the cleft depth or 67 nm from the apical margin of the cell. In our previous studies on frog mesentery we had for convenience placed the strand midway between apical and basal aspects of the cleft. Based on an earlier mathematical model in the appendix to Adamson & Michel (1993), where the endothelial glycocalyx was omitted, it was shown that the effect of the location of the junction strand had little effect on Lp. However, we have found that the location of the junction strand relative to the glycocalyx does have a significant effect on Lp since it can create a narrow water channel on the apical side of the junction strand through which fluid streamlines must pass if the strand is located close to the apical margin (Weinbaum et al. 2003).
Table 1.
Cleft, junction strand gap and glycocalyx parameters
| Gap length1 | 2d | 315 nm |
| Gap spacing1 | 2D | 3590 nm |
| Depth of strand1 | L1 | 67 nm |
| Total cleft depth1 | L | 411 nm |
| Width of cleft1 | 2h | 18 nm |
| Cleft length per area vessel wall1 | Lc | 0.10 μm−1 |
| Cleft linker spacing2 | Δc | 15 nm |
| Cleft linker diameter3 | 2rc | 2.5 nm |
| Glycocalyx fibre spacing4 | Δf | 8 nm |
| Glycocalyx fibre diameter4 | 2rf | 12 nm |
We have assumed 150 nm for the thickness of the glycocalyx for two reasons. First, previous work indicates that approximately 60% of the hydraulic resistance across the microvessel wall is contributed by the glycocalyx (Adamson, 1990). In our present model this condition is satisfied by a 150 nm thick glycocalyx (see Fig. 9 and Discussion). Second, imaging of rapidly frozen microvessels revealed a surface layer with a thickness of approximately 150 nm (Squire et al. 2001).
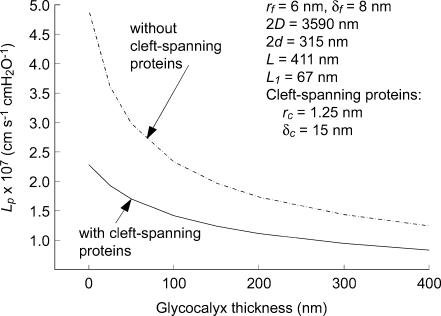
Figure 9. Model predictions of Lp with and without cleft-spanning structures.
Predicted Lp based on the measured parameters described in the text and listed in Table 1. The cleft-spanning structures contribute about 50% of the hydraulic resistance for all values of glycocalyx thickness.
We briefly summarize the important features of the new model as they apply to the present predictions. Since the flow crossing the glycocalyx travels nearly parallel to the core proteins in the outer regions of the glycocalyx layer (streamlines in Fig. 1D) and then basically transverse to the core proteins near the base of each bush-like structure, we have developed a model for the Darcy permeability of the glycocalyx layer which is based on an equal weighting of flow parallel and perpendicular to the core protein fibres. The latter are modelled as circular cylinders whose diameter, 12 nm, is taken as the observed diameter of the quasi-periodic scattering centres (Squire et al. 2001). The flow in the wide part of the cleft, where we assume a hexagonal array of cleft-spanning structures, is described using a Brinkman equation (Tsay & Weinbaum, 1991). Since the spacing of the structures, 15 nm, is of the same order as the measured gap height of the cleft, 18 nm, the fibre interaction layers near the cell membrane boundaries must be considered in determining the effective Darcy permeability Kp of the cleft. Once this effective Kp is determined, the governing equation for the flow in the cleft can be treated as a two-dimensional Darcy flow satisfying a potential flow equation for the pressure field. The junction strand itself is treated as an impermeable boundary for both water and solute. The treatment of flow in region B, the region surrounding the cleft exit, differs from that summarized previously in Hu & Weinbaum (1999) in that region B is now solved numerically using a full three-dimensional convection–diffusion equation with continuity of concentration and flux at both the cleft exit and the interface with the tissue space of the far field. The treatment of flow in the far field tissue space is the same as previously described (Hu & Weinbaum, 1999).
Results
Protein equilibration between superfusate and tissue
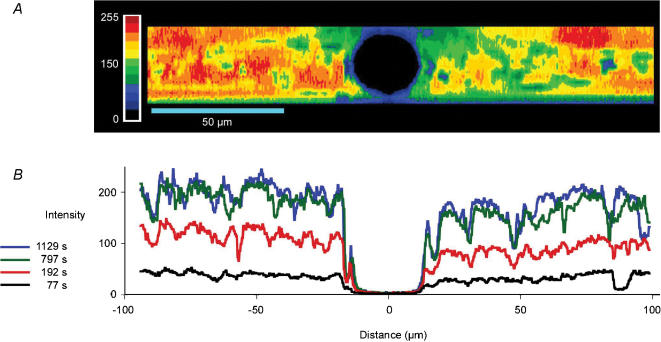
An important requirement of our experimental design to test the oncotic effect of extravascular serum protein is to access the tissue space immediately surrounding the vessel wall and set the local concentration of serum protein at a known value. To establish the time required for albumin to diffuse from the superfusate into the tissue, we collected confocal microscope images such as in Fig. 2. Figure 2A is a confocal image taken transverse to the longitudinal axis of a venular microvessel in rat mesentery showing the distribution of fluorescently labelled serum albumin within the extravascular space 15 min after adding the labelled albumin to the superfusate. Figure 2B shows the temporal evolution of the albumin distribution along a profile in the mid-plane of the tissue as it diffuses into the tissue from the superfusate over almost 20 min. The superfusate included unlabelled serum albumin (50 mg ml−1) and a trace amount of Alexa Fluor 546–BSA (0.1 mg ml−1). The vessel was cannulated (>500 μm distant) and perfused with solution containing unlabelled albumin (50 mg ml−1) only. To reduce the resistance of the mesothelium to albumin diffusion and facilitate solute diffusion between the tissue fluid space and superfusate, before applying the fluorescently labelled superfusate the mesothelium was stroked with a fine glass rod about 100 μm from either side of the venule. Figure 2B shows that by about 13 min the tissue concentration was near steady state. The superfusate fluorescent intensity on this scale was about 230 units and thus the tissue concentration reached an apparent concentration of 80–90% of the superfusate value. In several experiments we found that steady state was reached between 8 and 15 min. Therefore, in subsequent experiments to test the effects of extravascular albumin we allowed a minimum of 20 min superfusion with BSA-containing solutions before measuring Jv/A. In Fig. 2B the non-uniformities in the tissue concentration gradient are due to sites where isolated cells or dense collagen bundles exclude albumin.
Figure 2. Loading the interstitium with serum albumin.
A, fluorescence image of a microvessel oriented transversely to the vessel axis collected 912 s after adding fluorescent albumin to the superfusate (at top). The vessel (centre, dark) was perfused with non-fluorescent solution. Serum albumin was able to penetrate throughout the interstitial space. B, intensity profiles taken along the centreline of images such as in A at various times demonstrate that the concentration of albumin near the vessel wall reaches steady state by about 13 min.
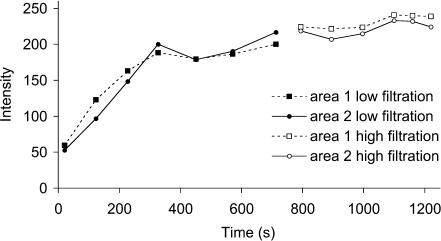
Figure 3 shows fluorescence intensity of labelled albumin as it develops over time in two areas within 5 μm of a microvessel wall taken from a time series of confocal images similar to that in Fig. 2A. The filled symbols in Fig. 3 represent data collected under free flow conditions (first ∼12 min), during which Pc was near 20 cmH2O and filtration rate was very low. The open symbols represent data collected after raising Pc to 60 cmH2O to maintain an increased filtration rate. There was no significant change in the albumin concentration near the microvessel after increasing filtration rate, showing that increased filtration did not significantly dilute the concentration of extravascular albumin near the microvessel wall when the tissue was exposed to albumin in the superfusate.
Figure 3. Interstitial albumin loading not diminished by high filtration from vessel.
Fluorescence intensity was recorded from two locations within 5 μm of a vessel wall and monitored over time during perfusion near 20 cmH2O. After raising the luminal hydraulic pressure to 60 cmH2O the intensity did not decrease indicating that interstitial albumin concentration was high during both low and high filtration states.
Effective oncotic pressure with and without extravascular protein
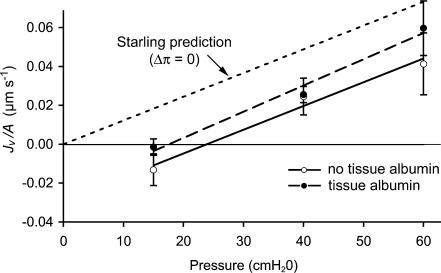
Figure 4 shows the results of a single experiment in which Jv/A was measured in a rat mesenteric venular microvessel perfused with solution containing BSA (50 mg ml−1) having πc equal to 27 cmH2O. Measurements were made first when the superfusate was protein-free Ringer solution. Under these conditions with no albumin in the superfusate and πt equal to 0 cmH2O, the intercept on the pressure axis measured the effective oncotic pressure (24 cmH2O) due to albumin in the perfusate and the Lp was 1.2 × 10−7 cm s−1 cmH2O−1 (continuous line). Then we changed the superfusate to one containing BSA (50 mg ml−1) so that there was no albumin concentration difference between lumen and interstitium and again measured Jv/A. Under these latter conditions (πt=πc), we predicted, according to the classic Starling equation, that the pressure intercept should go through the origin and the slope (Lp) would be unchanged (dotted line). While the Lp (1.3 × 10−7 cm s−1 cmH2O−1) was not different from expectation, we found the effective oncotic pressure was 17 cmH2O (dashed line).
Figure 4. Effective oncotic pressure remains high in the presence of high extravascular albumin.
Filtration flux, Jv/A, as a function of microvessel hydrostatic pressure during perfusion with BSA (50 mg ml−1) was measured in this vessel first with protein-free Ringer superfusate (○) and then with superfusate also containing BSA at the same concentration •). Intercept on the pressure axis indicates the effective oncotic pressure and shows that when interstitial albumin is present at the same concentration as in the perfusate the effective oncotic pressure is greatly different from the expected value of zero (line of Starling prediction).
These comparisons were made in six vessels. When the superfusate included albumin at the same concentration as the perfusate, the mean effective oncotic pressure was 17 ± 2 cmH2O. This is significantly different from the expected value of 0 cmH2O predicted from the Starling equation (P < 0.001, Student's t test). When the superfusate was protein-free Ringer solution the mean effective oncotic pressure was 24 ± 2 cmH2O. Mean Lp under both conditions was 1.0 ± 0.1 × 10−7 cm s−1 cmH2O−1. For two of the vessels we measured Jv/A with serum albumin in the superfusate before switching to Ringer superfusate and found that the order of superfusates had no effect on the outcome. Since we knew from the confocal experiments that the albumin concentration just outside the vessel wall was not significantly different from that in the bulk interstitium, the colloid osmotic force opposing filtration must have been exerted across a structure within the microvessel wall, which we propose to be the glycocalyx.
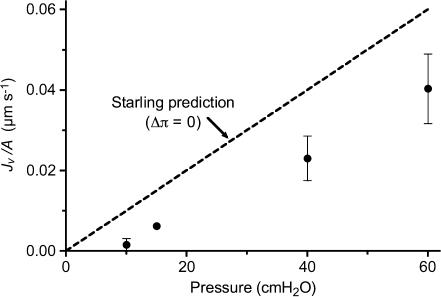
For four of the six vessels reported above we also succeeded in measuring filtration rates across the vessel wall having set up steady-state filtration not only at 60 cmH2O, but also at lower pressures. These measurements were made while the mesentery was bathed in superfusate containing serum albumin at the same concentration as in the perfusate (50 mg ml−1). Therefore, the expected Jv/A was the Starling equation prediction under the condition of no oncotic pressure difference between lumen and tissue (Δπ= 0) and having Lp being equal to the mean of the four vessels (1.3 × 10−7 cm s−1 cmH2O−1). The data of Fig. 5 show that for all pressures, the measured Jv/A was substantially less than the value expected, indicating that albumin oncotic pressure on the downstream side (tissue side) of the sieving structure in the vessel wall was substantially lower than the luminal value. Moreover, the slight filtration seen at low Pc conforms to the curvature of the predicted relationship (see also Fig. 11).
Figure 5. Steady state filtration at multiple pressures.
Filtration flux, Jv/A, as a function of microvessel hydrostatic pressure was measured after having established steady-state conditions at each indicated intraluminal pressure. The superfusate contained BSA at the same concentration as the perfusate (50 mg ml−1). Values shown are means ± s.e.m. for 4 vessels. The relationship expected based on the lack of a protein osmotic pressure difference between perfusate and interstitial fluid is also shown (Starling prediction).
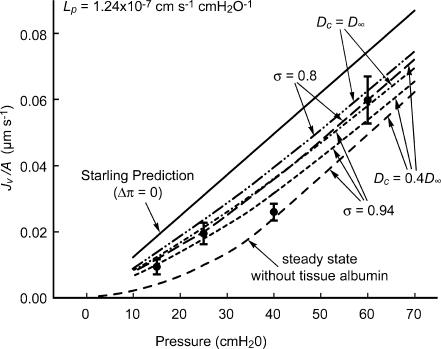
Figure 11. Model prediction of steady-state filtration relationship.
Jv/A is plotted as a function of microvessel hydrostatic pressure for steady-state filtration at each pressure. Model values are predicted from geometric parameters as in Fig. 10 and correspond to an Lp of 1.24 × 10−7 cm s−1 cmH2O−1. The classic Starling equation predicts no oncotic pressure difference when albumin concentration is 50 mg ml−1 in both lumen and tissue (continuous line). The absence of tissue albumin predicts maximal reduction of Jv/A (lowest dashed curve). Predictions of present model when albumin concentration is 50 mg ml−1 in both lumen and tissue are shown for effective albumin diffusion coefficient within the cleft (Dc) equal to the free solution diffusion coefficient (D∞) and 0.4D∞, and also for reflection coefficients for albumin (σ) of 0.8 and 0.94. Steady-state data are shown from a representative microvessel experiment which had Lp of 1.34 × 10−7 cm s−1 cmH2O−1 and σ equal to 0.93 (•; mean ± s.e.m. of 5–7 measurements at each pressure).
Morphology of the endothelial cleft
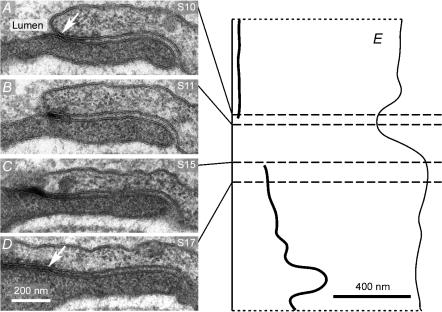
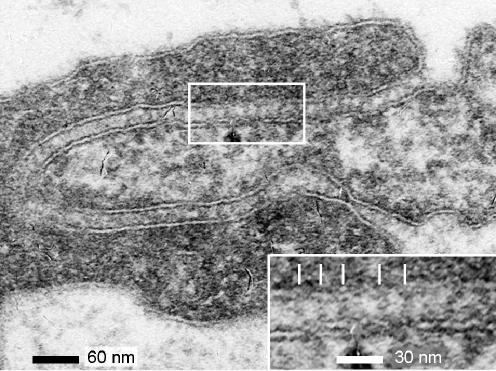
Five venular microvessels were examined to investigate the ultrastructural morphology of the endothelial cleft. They had a mean Lp of 0.8 ± 0.1 × 10−7 cm s−1 cmH2O−1, similar to the value measured in the previous section. From each vessel we examined at least two serial runs of sections and recorded images from several clefts. In serial runs, we examined 27 clefts totalling 32.3 μm of cleft length. Four electron micrographs are shown from a set of serial sections demonstrating a break in the tight junction strand between two endothelial cells of a mesenteric venule (Fig. 6). Figure 6A shows the tight junction intact; in Figs 6B and 6C there is a patent pathway from the lumen to the interstitium; Fig. 6D shows the tight junction intact on the other side of the strand discontinuity. In this case, the junction strand break was 250 nm long. A reconstruction of the 30 sections in the region of the strand gap is shown in Fig. 6E. The tight junction is characteristically close to the luminal opening of the cleft (about 60 nm in section 10) and the path from lumen to interstitium is about 700 nm.
Figure 6. Tight junction strand gap seen in electron micrographs from serial sections.
Four sections from a serial run of 30 sections are shown. A, all sections up to and including sections 10 (S10) included a continuous tight junction (arrow) occluding the cleft. B, section 11 (S11) had no tight junction. C, section 15 (S15) had no tight junction. D, section 17 (S17) and all subsequent sections had a tight junction (arrow) that could be traced from one section to the next. E, reconstruction of the tight junction strand illustrated in A–D. Heavy line is tight junction strand, light lines are luminal and abluminal cleft entrances. Dotted lines represent beginning and ending section edges. Section locations on reconstruction shown by dashed lines.
In addition to the gap shown in Fig. 6, we found eight more gaps, for a total of nine junction strand gaps in the 32.3 μm of reconstructed cleft (Fig. 7A–H). The mean length of gaps was 315 ± 114 nm (n = 9; ranging from 1 section to 22 sections, i.e. 50 nm to 1100 nm). This value was given to parameter 2d (Fig. 1 and Table 1). Therefore, the total proportion of open cleft was 0.088 or about 9%. This frequency of gaps (9 per 32.3 μm) yielded a mean gap spacing (centre to centre) of 3.59 μm, which is the value used for parameter 2D in our theoretical model. The mean depth of the nine strand gaps (distance from luminal opening to the edges of the strand ends) was 67 ± 7 nm and the mean depth of the cleft path from lumen to interstitium was 411 nm. These values were assigned to parameters L1 and L, respectively. Wissig suggested that cleft pathways through strand gaps might be found as overlapping but unconnected strands, and therefore not seen in single thin sections (Wissig & Williams, 1978). Only two of the strand gaps reconstructed from serial sections showed some overlap by deeper strands (Fig. 7E and H). To demonstrate further the variation of tight junction strand arrangements five reconstructions of cleft regions in which no strand gaps were found are also shown (Fig. 7I–M). A tight junction strand forming a continuous barrier was often very near the luminal entrance. Multiple strands were usually present. However, many of the strands towards the interstitium were segments either branching from the primary continuous strand or unconnected to other strands. We examined a total 602 micrographs of clefts to determine an average number of tight junctions without regard to the presence of strand breaks and found that on average there are 2.2 tight junctions per cleft in these venular microvessel clefts. The width of the wide part of the cleft was found to average 18 nm (range was 14–21 nm).
Figure 7. Reconstructions of tight junction strands.
A–H, eight additional reconstructions of clefts exhibiting discontinuous tight junction strands. I–M, reconstructions of clefts with no strand gaps for comparison. Luminal entrances are shown as straight light line at left of each reconstruction and abluminal exits are at right. Dotted lines represent beginning and ending section edges. Heavy lines represent tight junction strands.
The perimeter and area of individual endothelial cells were measured from confocal images of silver-stained endothelial cell clefts of venular microvessels. Mean cell perimeter was 139 ± 4 μm and mean cell area was 705 ± 24 μm2. For each cell the perimeter per unit area (Lc = 0.5 pcAc−1) was calculated and the mean value of Lc was 0.100 ± 0.002 μm−1(n = 32). The latter value, equivalent to an average circumferential distance between clefts of 10 μm, was used in our model of the vessel wall. These values correspond closely to those of rat trachea for which Lc was 0.105 μm−1 for 20 μm diameter venules (McDonald, 1994). Our typical vessel diameter was 32 μm, having 10 clefts per circumference.
In several endothelial clefts we found regularly spaced electron densities that spanned the space between the two endothelial cells (Fig. 8). They were usually seen only in limited regions within any one cleft image. However, they were found throughout the wide part of clefts both on the luminal and abluminal side of junction strands. The spacing from structure to structure ranged from about 10 nm to 20 nm, similar to that previously reported (Schulze & Firth, 1992).
Figure 8. Cross-bridging structures in the endothelial cleft.
Electron dense structures spanning the space between endothelial cells were seen in segments of several clefts. They are apparent in the abluminal part of this cleft (rectangle). Inset, same region at higher magnification with structures marked (arrows).
Model predictions for Lp
In Fig. 9 are the model predictions for the filtration coefficient Lp as a function of the glycocalyx thickness, Lf. All other geometric parameters in the model are obtained from the measured morphological data described in the preceding paragraphs and summarized in Table 1. Two solutions are presented, one without cleft-spanning structures and one assuming cleft-spanning structures are present throughout the wide part of all clefts and having the 15 nm spacing measured in rat heart capillaries (Schulze & Firth, 1992). A typical measured value of Lp was 1.0 × 10−7 cm s−1 cmH2O−1. One observes that for any glycocalyx thickness the value of Lp with cleft-spanning structures is about one-half the value for the same cleft without these space-spanning structures. This indicates that one-half of the total cleft resistance including the glycocalyx resides in the cleft-spanning structures. We have not directly investigated the thickness of the glycocalyx in the rat mesentery venules, but the thickness of this layer in frog mesentery microvessels has been estimated as 150 nm (Squire et al. 2001; Hu et al. 2000). For the range 150–250 nm the model predicts that Lp would be a factor of two too large if the cleft-spanning structures were not included. In contrast, with cleft-spanning structures the predicted Lp would lie between 1.0 and 1.25 × 10−7 cm s−1 cmH2O−1, well within the range of the average measured value. In subsequent analyses Lf is assumed to be 150 nm and, using the parameters of Table 1, for a typical microvessel the model predicts Lp equals 1.24 × 10−7 cm s−1 cmH2O−1.
Model predictions for concentration profiles
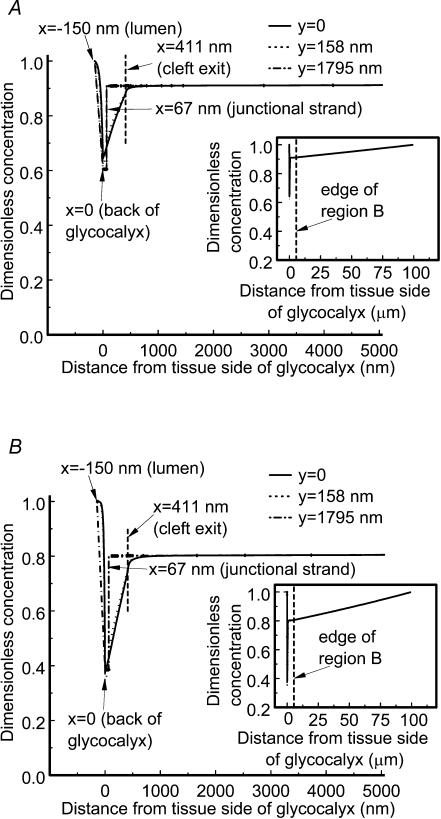
In Fig. 10 we have plotted the albumin concentration through the cleft and tissue along the centre line of a periodic junction strand break (y = 0), near the edge of the strand break (y = 158 nm) and at the edge of the periodic unit (y = 1795 nm). Figure 10B is for a high filtration state where the lumen pressure is 60 cmH2O in which a steady state has been achieved after the tissue has been equilibrated with albumin in the superfusate by damaging the mesothelium 100 μm from the microvessel and allowing albumin to diffuse into the tissue around the microvessel. Note that the predicted albumin concentration on the lumen side of the junction strand is only about 35% that in the superfusate or lumen. The convective flow through the break in the junction strand is sufficiently strong to greatly hinder diffusive flux of albumin from the tissue space into the protected space on the tissue side of the glycocalyx. The effective oncotic pressure driving the flow is approximately 65% of the total oncotic pressure in the lumen of the vessel. At a Pc of 30 cmH2O (Fig. 10A) the concentration in the tissue just outside the cleft exit is reduced by only 10% from the superfusate concentration, yet the concentration on the lumen side of the junction strand beneath the glycocalyx is reduced by nearly 40% from the lumen value. In both cases the effective oncotic difference, the difference across the glycocalyx, is much greater than the difference between lumen and tissue.
Figure 10. Model prediction of steady-state BSA concentration gradient during low and high filtration.
Dimensionless concentration profiles from the lumen into the tissue are shown for a path through the centre of a strand gap (y = 0, continuous line), the edge of a strand gap (y = 158 nm, dashed line) and across the tight junction equidistant between two strand gaps (y = 1795 nm, dashed–dotted line). Lf is assumed to be 150 nm, other values are as in Fig. 9 with cleft-spanning structures. A, with Pc 30 cmH2O, protein concentration at the abluminal side of the glycocalyx is reduced by 40% from its value in the lumen and the superfusate. Concentration on the tissue side of the cleft is low near the strand gap and rises toward the tissue. Inset, concentration just outside the cleft is only 10% reduced from the steady-state value in the lumen and the superfusate. B, at high pressure, with Pc= 60 cmH2O, the protein concentration on the abluminal side of the glycocalyx is reduced by 65% from the luminal value. Inset, even at high Pc the protein concentration just outside the cleft is reduced by only 20% from the value in superfusate.
Model prediction for Jv/A
In Fig. 11 we show our model predictions for steady-state filtration curves as a function of lumen pressure using parameter values as in Fig. 10. The curves with albumin loaded into the tissue lie between the prediction for the classic steady-state Starling equation, where the effective oncotic pressure measured between plasma and tissue is expected to be 0 cmH2O, and the result that would be predicted if there were no albumin in the tissue, i.e. the experiment performed by Michel & Phillips (1987), for which the luminal oncotic pressure is maximally effective at reducing Jv/A. Also in the figure are data from a representative rat mesentery microvessel experiment for which the measured Lp was 1.34 × 10−7 cm s−1 cmH2O−1, and which therefore provides a close test of the mathematical model (predicts Lp of 1.24 × 10−7 cm s−1 cmH2O−1). Our new model based on the hypothesis that the effective oncotic pressure is due to the local difference in oncotic pressure across the glycocalyx layer provides reasonable agreement with our experimental data for all pressures shown for the representative experiment. To explore the sensitivity of the model to the input data we show predictions for Jv/A for a reflection coefficient that varies between σ= 0.8 and σ= 0.94 and a diffusion coefficient in the cleft, Dc, that varies between the value in free solution, D∞, and 0.4D∞. The latter variation accounts for the presence of the crossbridging fibres in the estimation of Dc. It is clear from this sensitivity analysis that a reasonable variation of these key parameters has only a minor effect on the Jv/A curves.
Discussion
Both the experiments to measure the effective osmotic pressure of albumin across the wall of rat mesenteric microvessels, and the calculation of the albumin concentration gradients across the glycocalyx, at the breaks in the cleft and in the tissue, conform to the hypothesis that the hydrostatic and osmotic forces which determine fluid filtration across the microvessel wall are across the endothelial glycocalyx. Furthermore, our direct measurements of the albumin concentration on both sides of the microvessel wall during high and low filtration states demonstrate that the local albumin concentration behind the glycocalyx must differ from that in the tissue. These results support the growing evidence that tissue proteins contribute less to the balance of osmotic forces across the microvessel wall than expected from the usual form of the Starling equation. The unique feature of our experiment was the test of the relation under conditions where the tissue concentration is high and directly measured. As pointed out in the introduction, the diminished contribution of the tissue proteins was likely to have been missed in earlier evaluations of the Starling balance for fluid exchange because the tissue concentrations were usually low due to experimental conditions.
Our results confirm and extend, for rat mesenteric microvessels, the observations, previously described for frog mesenteric microvessels using a similar approach, that large protein osmotic pressures were developed across the microvessel wall even when albumin concentrations in the lumen and the tissue were equal (Hu et al. 2000). However, there are several important refinements in our experimental methods and the model used to explain the observations that we evaluate below. The most important is the measurement of the concentration of albumin in tissue close to the vessel wall under conditions where there is a high filtration rate of water across the vessel wall. There was no measurable difference in the concentration of albumin in the tissue within a few micrometres of the wall under conditions of high filtration rate compared with low filtration. This clearly demonstrates that filtration does not contribute significantly to the dilution of tissue albumin concentration and hence the tissue oncotic pressure when the latter is controlled by the superfusate. Under these conditions tissue albumin does not contribute strongly to the formation of a protein osmotic pressure difference.
Extravascular albumin gradients under the conditions of our experiments
The main reason that water leaving the cleft does not significantly modify the albumin concentration close to the wall is that water velocity falls to very low values as it spreads into the tissue. At about 2 μm from the cleft exit into the interstitial space the mean fluid velocity falls to less than 0.4% of the value at the cleft exit. Diffusive mechanisms then dominate the distribution of the albumin in the vicinity of the vessel wall (Hu et al. 2000). These observations re-emphasize that the primary mechanism for maintaining a low albumin concentration on the tissue side of the glycocalyx is the presence of small but infrequent gaps in the junctional strands that funnel the transendothelial water flows, such that the velocity of water at the gaps is a significant fraction (estimated values 30–100%) of the albumin diffusion velocity at the gaps. The result is that the diffusion of albumin from the tissue into the cleft beyond the strand gaps is reduced by convection, creating a steep gradient of albumin concentration in the distal region of the cleft as shown in Fig. 10. This helps to maintain the low albumin concentration in a ‘protected region’ just underneath the glycocalyx. One notes that in regions removed from the breaks (y = 1795 nm), where convection is low, the concentration in the distal part of the cleft nearly equals that at the cleft exit due to diffusion of albumin in the tissue.
We can further understand the effects of the protected region by examining the effective oncotic pressures in the experiments of Fig. 4. When the superfusate was protein-free Ringer solution and steady state was achieved by setting the perfusion pressure to a high value (60 cmH2O), the mean effective oncotic pressure was 24 cmH2O. Using simple one-dimensional analysis and the assumption that at high fluid flux the effective oncotic pressure (σΔπ) across the sieving matrix approaches σ2πc, we calculated that the σ for serum albumin was 0.94 ± 0.03. This mean value is the same as previously reported for rat mesenteric vessels using similar techniques (Kendall & Michel, 1995). When albumin was in the tissue the effective oncotic pressure was reduced to 17 cmH2O, but not reduced to 0 as would be expected if the entire microvessel wall was the sieving structure. Thus, with albumin in the tissue σΔπ was close to 70% of that measured when there was no albumin in the tissue. This indicates that some albumin may diffuse back into the protected space, but the amount is far less than would occur if the funnelling mechanism was not operating. Assuming σ for albumin equalled 0.94 and πc equalled 27 cmH2O, the oncotic pressure on the tissue side of the endothelial surface matrix is estimated to have been 9 cmH2O, corresponding to about 21 mg ml−1 serum albumin, close to 40% of the luminal concentration. This corresponds well with the prediction from our model seen in Fig. 10B for the concentration along the centreline of the gap at the back of the glycocalyx. A more complete model including prediction of reflection coefficients to macromolecules is needed for further analysis of the measured effective oncotic pressure.
Modelling cleft ultrastructure
Another important improvement in our experiments is that the ultrastructure of the endothelial cleft and continuity of junctional strands was determined by extensive serial sectioning on the same microvessels as used for the measurement of Lp. The gaps in the junctional strands were longer in rat than those in frog microvessels (315 nm versus 150 nm) described by Adamson & Michel (1993). The frequency of strand gaps observed in serial reconstructions was comparable (on average one every 3.6 μm in rat versus near 4.4 μm in frog). So it appears that the effective area for exchange of water between adjacent endothelial cells in the rat mesenteric microvessels at the level of the breaks in the junction strand was larger than in the frog (9% in rat compared with close to 4% in the frog) as the gap height of the openings (near 18 nm) was not significantly different in the two preparations. When this result is combined with the observation that the overall Lp of rat vessels is about one-half that in frog, we conclude that, at any value of microvessel pressure, the water velocity at the level of the break is likely to be lower in the rat than in the frog. Thus the local Peclet number at the strand gap (ratio of water velocity to albumin diffusion velocity) is expected to be smaller in the rat microvessels. Therefore we expect more back-diffusion of albumin from the tissue, even at high microvessel pressures, and a smaller albumin concentration difference across the glycocalyx at these pressures. These trends are consistent with the measured results. Although the measured albumin osmotic pressure approaches that of the luminal osmotic pressure, even when albumin is in the tissue, the values in rat microvessels are close to 70% of luminal pressure while the measured values in the frog approached 90% of the luminal concentration.
There are important uncertainties still to be resolved with respect to the ultrastructural data. We have shown previously that visualizing strand gaps only through morphological reconstruction (compared with the use of a tracer such as lanthanum nitrate) underestimates their frequency by about a factor of two (Adamson & Michel, 1993). Thus the above estimates of the proportion of open strand are minimum values for both rat and frog. Furthermore, using this proportion of open strand and assuming a glycocalyx thickness of between 100 nm and 400 nm, the Lp predicted from our model was about 2-fold higher than the measured range (dashed curve on Fig. 9). Thus there are likely to be additional resistances to water flow in the cleft. We noted that the primary junctional strand was consistently closer to the lumen than the midpoint of the cleft in rat. This contributed to some additional resistance to water movement since the flow was confined to a narrow channel between the underside of the glycocalyx and the tight junction strand as it moved toward the site of breaks in the junction strand. This additional resistance is not sufficient to account for the observed discrepancy between measured and predicted Lp in the rat vessels. We also found clear evidence for crossbridging structures within the wide part of the cleft, similar to those described in rat heart (Schulze & Firth, 1992). Our collection and analysis of sections was not optimized to obtain additional data on the distribution of these structures in the wide part of the cleft, but, when included as an array of posts 2.5 nm in diameter spaced in an array 15 nm apart, we estimated that they accounted for close to 50% of the resistance to water flow across the microvessel wall (continuous curve Fig. 9), bringing the predicted Lp well within the measured range (0.5–1.6 × 10−7 cm s−1 cmH2O−1).
The question of the contribution of these structures to selectivity has not been studied, and our data allow us to draw only tentative conclusions. We suggest that it is unlikely that these structures in the wide part of the cleft contribute significantly to sieving mechanisms as any colloid osmotic pressure developed across the wide part of the cleft distal to the glycocalyx would actually oppose the osmotic pressure across the glycocalyx, because the direction of the gradient is reversed! A structure with similar appearance in thin sections has been described from the adherens junction of retinal pigmented epithelium (RPE) of chickens (Miyaguchi, 2000). These crossbridging structures, estimated to have a density corresponding to an intercrossbridge distance of near 35 nm in the RPE, are thought to be cadherin molecules. In addition to VE-cadherin, other cell adhesion molecules are likely to be present in the endothelial cleft. The precise geometry and distribution of these molecules in both frog and rat microvessels necessary for modelling resistance to fluid and solute flux should be a subject for further investigation.
Modelling glycocalyx structure
Another refinement in our theoretical modelling is to describe the glycocalyx structure as quasi-periodic, as defined by the detailed analyses of glycocalyx structure using autocorrelation techniques (Squire et al. 2001). An analysis of this structure as a molecular sieve has been published elsewhere (Weinbaum et al. 2003), and the approach has been extended to obtain the results in Figs 9 and 10. The key observations are that the fibres which form the main part of the sieving matrix project from the endothelial surface as a series of bush-like structures with an effective diameter of the ‘branches’ of each of bush being 10–12 nm. In this model the sieving elements are pore-like channels, 7–8 nm diameter, between the core proteins forming the glycoproteins on the cell surface, aligned normal to the endothelial cell surface so that the direction of filtered fluid flow is mainly parallel to the core proteins, except close to the base of each bush. Although this structure is significantly different from the model we previously suggested where sieving occurs in the spacing between the much narrower side chains of the glycoproteins (0.6 nm radius), aligned predominantly transverse to the flow, the model predicts similar overall sieving properties because the spacing between the narrow fibres was also close to 7–8 nm in each model. The principal difference between the models is that in the present model the glycocalyx accounts for a larger fraction of the resistance to water flow than in the previous model, and this is in accord with experimental results previously described where enzymatic removal of the glycocalyx decreases the resistance to water flows by about 50% (Adamson, 1990).
Comparison with other investigations
Ultrastructure
The structure of the interendothelial cleft and the organization of the junction strand observed in rat heart capillaries by Bundgaard, (1984) was qualitatively similar to frog and rat mesentery microvessels except that the length of the breaks was much shorter, typically the width of a single transmission section, 40–50 nm. While Bundgaard stated that non-connected overlapping junction strands leading to tortuous pathways through the clefts such as proposed by Wissig & Williams (1978) were not considered, it is not clear if any such pathways were encountered in rat heart vessels. It is possible that such pathways were not included in the total. In the rat mesentery vessels we found limited evidence for patent pathways from lumen to tissue around overlapping junction strands, such that the pathways would not be seen in single thin sections (Figs 7E and 7H). However, the hydraulic resistance of such pathways would necessarily be much greater than that of the majority of strand gaps and, combined with the low frequency of these pathways, we can conclude that the hydraulic flow is dominated by pathways through the non-overlapping strand gaps. At present there are no serial section ultrastructural studies for mammalian microvessels equivalent to that for frog mesentery (Adamson & Michel, 1993). Our data are therefore the most detailed to date on mammalian tissue. However, our results do not rule out the possibility that the size and frequency of breaks may be different in other mammalian microvascular beds. This may be an area for future study.
Tissue oncotic pressure
Our observations that the oncotic pressure of the mixed interstitial fluid does not exert its full effect at the capillary wall and that it is the oncotic pressure just abluminal to the endothelial glycocalyx that is a primary determinant of the fluid flow may appear to conflict with observations by Smaje and colleagues. They varied the extravascular concentration of serum albumin while measuring fluid efflux from rat cremaster capillaries and concluded that the filtration rate was in direct proportion to the interstitial oncotic pressure (Smaje et al. 1970). To evaluate this result we used our model to estimate effective oncotic pressures in cremaster capillaries in which the intravascular hydrostatic pressure was near normal capillary pressure (between 20 and 30 cmH2O). When using this perfusion pressure we found that the oncotic pressure on the abluminal side of the glycocalyx varies roughly in proportion to the oncotic pressure in the interstitium, but is somewhat lower, with the local value dependent on the local fluid velocity. This reflects the lower fluid velocity through the strand gaps and an increasing diffusive flux into the cleft from the tissue at low filtration. Therefore, while the results of Smaje and colleagues appear at first to be in contrast to our results, we believe they are in line with our model at normal pressures. This analysis assumes a similar ultrastructure for the true capillaries of the cremaster and the venular microvessels of the mesentery. Further experiments which combine the two techniques may be able to resolve this matter more fully.
Similarly, elevation of interstitial colloid osmotic pressure increased fluid efflux from fenestrated synovial capillaries of rabbit knee, clearly demonstrating an effectiveness of interstitial colloid in determining fluid balance (McDonald & Levick, 1993; Sabaratnam et al. 2002). However, the interstitial colloid exerted only about half of its potential oncotic pressure at the abluminal capillary surface, confirming that fluid flux through the fenestrations was sufficient to ‘wash-down’ the peri-fenestral colloid concentration. Nonetheless, as pointed out by Levick, the porous exit from a fenestration is nearly in direct communication with the interstitium and thus is expected to be more responsive to the interstitial oncotic pressure at low flow than the subglycocalyx region of a continuous capillary which is protected by the throat effect of the strand gap (Sabaratnam et al. 2002). Most important is that, for both fluid flow pathways, fenestration or continuous capillary cleft, local fluid flows, determined by pore exit geometry, determine the effectiveness of interstitial colloid in fluid balance.
Implications for fluid balance
The conditions of our experiments which demonstrate that the oncotic effect of interstitial albumin is less than expected are more extreme than those found in normal tissue. In our experiments we set the concentration, and therefore the oncotic pressure, of extravascular albumin to be equal to that in the vessel lumen, in effect ‘clamping’ the extravascular concentration at a higher than normal level. In a variety of tissues interstitial oncotic pressure is estimated to be from 30 to 60% of the value in plasma (Levick, 1991). The source of the plasma protein in interstitium is the infrequent ‘large pore’ pathway usually thought to be vesicle shuttling or chains of vesicles across the endothelium (Michel & Curry, 1999). One of the ‘safety factors against oedema’ that have been described is the dilution of this albumin, which occurs as ultrafiltrate enters the interstitium. Our results indicate that, during high filtration, because of the fluid velocity through the junction strand gaps, the interstitial protein concentration has very little influence on the concentration of protein just beneath the glycocalyx. However, at low-to-normal fluid filtration there must be a balance between convection washing albumin away from the glycocalyx and diffusive flux of albumin from the tissue into the subglycocalyx region. Under these conditions the safety factor still exists, but it is magnified by the throat effect of the junction strand, which constricts fluid flow to a narrow region. Any increase in Pc, for example during exercise in muscle, not only results in a diminished interstitial protein concentration, but also raises the fluid velocity in the cleft to levels such that convective drag on plasma proteins should very rapidly wash down the subglycocalyx region and quickly establish a high oncotic pressure gradient. An important aspect of the mechanism is the small volume of the subglycocalyx region, which enables very rapid wash-down and predicts nearly moment-to-moment adjustment of the oncotic gradient.
While the mechanism to wash-down the protein concentration in the subglycocalyx region helps maintain the oncotic gradient under normal conditions, it also helps recovery from acute inflammatory oedema. The results of Fig. 10A (initially constructed to investigate the specific conditions of our experiments) can be thought of as similar to conditions in a filtering capillary during the early stages of recovery from a local inflammatory stimulus. Protein has accumulated in the interstitium to a high level and intravascular pressure is high. Once inflammatory gaps have closed and the tight junctions have reformed the vessel approaches the state seen in the figure. In this example the local πt is nearly 24 cmH2O while the value in the subglycocalyx has washed down to about 15 cmH2O, greatly diminishing the net force for outward filtration.
It is clear from model results that the concentration profile of larger solutes would be affected similarly to that of albumin. As solute size increases, the Peclet number increases and the throat effect of the junction strand gaps becomes greater. That is, the concentration in the subglycocalyx region of the cleft would be lower than in tissue for ferritin, immunoglobulins and other large solutes present in both plasma and lymph. While the molar concentration of these high molecular weight components is low relative to albumin they do contribute significantly to the net oncotic gradient and this mechanism serves to enhance their oncotic effect. Small solutes, with greater diffusivities in the tissue and cleft, and having minimal convective flux, would be distributed by diffusion.
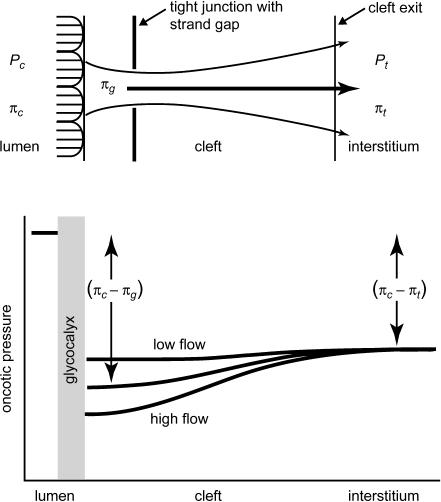
The concepts of our model are emphasized in an idealized diagram of the oncotic gradient from the lumen into the tissue immediately surrounding a fluid filtering microvessel under normal conditions (Fig. 12). Here the oncotic pressure of the mixed interstitial fluid, πt, is about 40% of the value in the vessel lumen, πc. In the classic Starling equation it is the difference (πc–πt), multiplied by σ, that offsets the hydrostatic pressure difference primarily responsible for outward filtration. However, in our model it is the difference between πc and the value in the subglycocalyx region, πg, that determines the effective oncotic pressure, σ(πc–πg). Everywhere along the filtering vessel (πc–πg) is greater than (πc–πt) and the predicted value of Jv/A is lower when (πc–πg) is recognized as the directly effective value. The value of πg is lowest at high fluid velocity through the cleft and therefore varies primarily as Pc varies along the vessel and with variations in vascular tone. Based on our current model we expect πg to be in the range 0.7–0.9πt under normal hydrostatic pressures, conferring several additional centimetres of water to the reabsorptive oncotic pressure difference.
Figure 12. Proposed model for the effective oncotic pressure in a filtering microvessel.
The effective oncotic pressure difference determining fluid balance in filtering microvessels is greater when calculated using the oncotic pressure in the subglycocalyx region rather than the mixed interstitial fluid. See Discussion for details.
The role of highly localized water flows on the downstream side of fenestrae has been examined both experimentally and theoretically by Levick and coworkers with reference to fluid exchange in the synovium (McDonald & Levick, 1993). In experiments in which the tissue concentration of albumin was increased, these investigators also showed that the effects of extravascular albumin on fluid exchange were much less than intravascular albumin. When these results are combined with our present results, they help to resolve the paradox, first pointed out by Levick in 1991, that when the most accurate values for tissue oncotic pressure are substituted into the Starling equation most tissues are not expected to be in fluid balance, and would filter far more fluid than can be accounted for by measured lymph flows. This paradox is resolved if the tissue albumin concentrations contribute far less to the balance of protein osmotic forces than the plasma concentration. This is because the plasma protein concentration difference across the glycocalyx and not the whole capillary wall is the primary determinant of the balance of Starling forces regulating transvascular fluid exchange.
Acknowledgments
This work was supported by NIH HL44485.
References
- Adamson RH. Permeability of frog mesenteric capillaries after partial pronase digestion of the endothelial glycocalyx. J Physiol. 1990;428:1–13. doi: 10.1113/jphysiol.1990.sp018197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adamson RH, Curry FE, Adamson G, Liu B, Jiang Y, Aktories K, Barth H, Daigeler A, Golenhofen N, Ness W, Drenckhahn D. Rho and rho kinase modulation of barrier properties: cultured endothelial cells and intact microvessels of rats and mice. J Physiol. 2002;539:295–308. doi: 10.1113/jphysiol.2001.013117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adamson RH, Lenz JF, Curry FE. Quantitative laser scanning confocal microscopy on single capillaries: permeability measurement. Microcirculation. 1994;1:251–265. doi: 10.3109/10739689409146752. [DOI] [PubMed] [Google Scholar]
- Adamson RH, Michel CC. Pathways through the intercellular clefts of frog mesenteric capillaries. J Physiol. 1993;466:303–327. [PMC free article] [PubMed] [Google Scholar]
- Aukland K, Reed RK. Interstitial-lymphatic mechanisms in the control of extracellular fluid volume. Physiol Rev. 1993;73:1–78. doi: 10.1152/physrev.1993.73.1.1. [DOI] [PubMed] [Google Scholar]
- Boggon TJ, Murray J, Chappuis-Flament S, Wong E, Gumbiner BM, Shapiro L. C-cadherin ectodomain structure and implications for cell adhesion mechanisms. Science. 2002;296:1308–1313. doi: 10.1126/science.1071559. [DOI] [PubMed] [Google Scholar]
- Bundgaard M. The three-dimensional organization of tight junctions in a capillary endothelium revealed by serial-section electron microscopy. J Ultrastruct Res. 1984;88:1–17. doi: 10.1016/s0022-5320(84)90177-1. [DOI] [PubMed] [Google Scholar]
- Curry FE, Huxley VH, Sarelius IH. Techniques in microcirculation: measurement of permeability, pressure and flow. In: Linden T, editor. Cardiovascular Physiology. P309/1. New York: Elsevier; 1983. pp. 1–34. [Google Scholar]
- Curry FE, Zeng M, Adamson RH. Thrombin increases permeability only in venules exposed to inflammatory conditions. Am J Physiol Heart Circ Physiol. 2003;285:H2446–H2453. doi: 10.1152/ajpheart.00262.2003. [DOI] [PubMed] [Google Scholar]
- Granger DN, Taylor AE. Permeability of intestinal capillaries to endogenous macromolecules. Am J Physiol. 1980;238:H457–H464. doi: 10.1152/ajpheart.1980.238.4.H457. [DOI] [PubMed] [Google Scholar]
- Guyton AC. A concept of negative interstitial pressure based on pressures in implanted perforated capsules. Circ Res. 1963;12:399–414. doi: 10.1161/01.res.12.4.399. [DOI] [PubMed] [Google Scholar]
- He P, Adamson RH. Visualization of endothelial clefts and nuclei in living microvessels with combined reflectance and fluorescence confocal microscopy. Microcirculation. 1995;2:267–276. doi: 10.3109/10739689509146772. [DOI] [PubMed] [Google Scholar]
- Hu X, Adamson RH, Liu B, Curry FE, Weinbaum S. Starling forces that oppose filtration after tissue oncotic pressure is increased. Am J Physiol Heart Circ Physiol. 2000;279:H1724–H1736. doi: 10.1152/ajpheart.2000.279.4.H1724. [DOI] [PubMed] [Google Scholar]
- Hu X, Weinbaum S. A new view of Starling's hypothesis at the microstructural level. Microvascular Res. 1999;58:281–304. doi: 10.1006/mvre.1999.2177. [DOI] [PubMed] [Google Scholar]
- Kajimura M, Wiig H, Reed RK, Michel CC. Interstitial fluid pressure surrounding rat mesenteric venules during changes in fluid filtration. Exp Physiol. 2001;86:33–38. doi: 10.1113/eph8602106. [DOI] [PubMed] [Google Scholar]
- Kendall S, Michel CC. The measurement of permeability in single rat venules using the red cell microperfusion technique. Exp Physiol. 1995;80:359–372. doi: 10.1113/expphysiol.1995.sp003853. [DOI] [PubMed] [Google Scholar]
- Levick JR. Capillary filtration-absorption balance reconsidered in light of dynamic extravascular factors [published erratum appears in Exp Physiol 1992 March; 77 (2): 403] Exp Physiol. 1991;76:825–857. doi: 10.1113/expphysiol.1991.sp003549. [DOI] [PubMed] [Google Scholar]
- McDonald DM. Endothelial gaps and permeability of venules in rat tracheas exposed to inflammatory stimuli. Am J Physiol. 1994;266:L61–L83. doi: 10.1152/ajplung.1994.266.1.L61. [DOI] [PubMed] [Google Scholar]
- McDonald JN, Levick JR. Effect of extravascular plasma protein on pressure-flow relations across synovium in anaesthetized rabbits. J Physiol. 1993;465:539–559. doi: 10.1113/jphysiol.1993.sp019692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel CC. Filtration coefficients and osmotic reflexion coefficients of the walls of single frog mesenteric capillaries. J Physiol. 1980;309:341–355. doi: 10.1113/jphysiol.1980.sp013512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel CC. Fluid movements through capillary walls. In: Renkin EM, Michel CC, editors. Handbook of Physiology, section 2, The Cardiovascular System, Microcirculation, part 1. IV. Bethesda: American Physiological Society; 1984. pp. 375–409. [Google Scholar]
- Michel CC. Starling: the formulation of his hypothesis of microvascular fluid exchange and its significance after 100 years. Exp Physiol. 1997;82:1–30. doi: 10.1113/expphysiol.1997.sp004000. [DOI] [PubMed] [Google Scholar]
- Michel CC, Curry FE. Microvascular permeability. Physiol Rev. 1999;79:703–761. doi: 10.1152/physrev.1999.79.3.703. [DOI] [PubMed] [Google Scholar]
- Michel CC, Mason JC, Curry FE, Tooke JE, Hunter PJ. A development of the Landis technique for measuring the filtration coefficient of individual capillaries in the frog mesentery. Q J Exp Physiol. 1974;59:283–309. doi: 10.1113/expphysiol.1974.sp002275. [DOI] [PubMed] [Google Scholar]
- Michel CC, Phillips ME. Steady-state fluid filtration at different capillary pressures in perfused frog mesenteric capillaries. J Physiol. 1987;388:421–435. doi: 10.1113/jphysiol.1987.sp016622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyaguchi K. Ultrastructure of the zonula adherens revealed by rapid-freeze deep-etching. J Struct Biol. 2000;132:169–178. doi: 10.1006/jsbi.2000.4244. [DOI] [PubMed] [Google Scholar]
- Pappenheimer JR, Soto-Rivera A. Effective osmotic pressure of the plasma proteins and other quantities associated with the capillary circulation in the hind-limbs of cats and dogs. Am J Physiol. 1948;152:471–491. doi: 10.1152/ajplegacy.1948.152.3.471. [DOI] [PubMed] [Google Scholar]
- Reed RK, Rubin K, Wiig H, Rodt SA. Blockade of beta 1-integrins in skin causes edema through lowering of interstitial fluid pressure. Circ Res. 1992;71:978–983. doi: 10.1161/01.res.71.4.978. [DOI] [PubMed] [Google Scholar]
- Renkin EM. Multiple pathways of capillary permeability. Circ Res. 1977;41:735–743. doi: 10.1161/01.res.41.6.735. [DOI] [PubMed] [Google Scholar]
- Sabaratnam S, Coleman PJ, Badrick E, Mason RM, Levick JR. Interactive effect of chondroitin sulphate C and hyaluronan on fluid movement across rabbit synovium. J Physiol. 2002;540:271–284. doi: 10.1113/jphysiol.2001.013468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulze C, Firth JA. The interendothelial junction in myocardial capillaries: evidence for the existence of regularly spaced, cleft-spanning structures. J Cell Sci. 1992;101:647–655. doi: 10.1242/jcs.101.3.647. [DOI] [PubMed] [Google Scholar]
- Shapiro L, Fannon AM, Kwong PD, Thompson A, Lehmann MS, Grubel G, Legrand JF, Als-Nielsen J, Colman DR, Hendrickson WA. Structural basis of cell-cell adhesion by cadherins. Nature. 1995;374:327–337. doi: 10.1038/374327a0. [DOI] [PubMed] [Google Scholar]
- Smaje L, Zweifach BW, Intaglietta M. Micropressures and capillary filtration coefficients in single vessels of the cremaster muscle of the rat. Microvascular Res. 1970;2:96–110. doi: 10.1016/0026-2862(70)90055-5. [DOI] [PubMed] [Google Scholar]
- Squire JM, Chew M, Nneji G, Neal C, Barry J, Michel C. Quasi-periodic substructure in the microvessel endothelial glycocalyx: a possible explanation for molecular filtering? J Struct Biol. 2001;136:239–255. doi: 10.1006/jsbi.2002.4441. [DOI] [PubMed] [Google Scholar]
- Tsay R, Weinbaum S. Viscous flow in a channel with periodic cross-bridging fibers of arbitrary aspect ratio and spacing. J Fluid Mech. 1991;226:125–148. [Google Scholar]
- Weinbaum S. 1997 Whitaker Distinguished Lecture: Models to solve mysteries in biomechanics at the cellular level; a new view of fiber matrix layers. Ann Biomed Eng. 1998;26:627–643. doi: 10.1114/1.134. [DOI] [PubMed] [Google Scholar]
- Weinbaum S, Zhang X, Han Y, Vink H, Cowin SC. Mechanotransduction and flow across the endothelial glycocalyx. Proc Natl Acad Sci USA. 2003;100:7988–7995. doi: 10.1073/pnas.1332808100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wissig SL, Williams MC. Permeability of muscle capillaries to microperoxidase. J Cell Biol. 1978;76:341–359. doi: 10.1083/jcb.76.2.341. [DOI] [PMC free article] [PubMed] [Google Scholar]