Abstract
Two-pore-domain potassium channels are a family of ion channels that are widely believed to play an important role in maintaining and regulating neuronal excitability. It has been shown that they can be modulated by an extraordinarily diverse range of endogenous and exogenous factors. One particular member of the family, TREK-1 (also known as KCNK2), is activated by increasing temperature, membrane stretch and internal acidosis, but is also sensitive to the presence of certain polyunsaturated fatty acids (such as arachidonic acid), neuroprotectants (such as riluzole) and volatile and gaseous general anaesthetics (such as halothane and nitrous oxide). It has recently been reported that TREK-1 channels are also affected by oxygen concentrations, and that at the levels of hypoxia that occur in the normal human brain, the channels greatly change their properties and, for example, lose their ability to be modulated by arachidonic acid and internal acidosis. These reports seriously challenge the idea that TREK-1 is a target for general anaesthetics and neuroprotectants. However, in this report we show that TREK-1 is not oxygen sensitive, and its ability to be activated by anaesthetics, arachidonic acid and internal acidosis remains unaltered under conditions of hypoxia. We further show that the protocol used by previous workers to prepare hypoxic solutions of arachidonic acid results in the removal of the compound from solution.
TREK-1 (also known as KCNK2) is a member of the most recently discovered family of two-pore-domain K+ channels, so called because they contain two pore-forming domains in their primary sequence. The class of mammalian two-pore-domain K+ channel subunits now includes 15 members, which are thought to dimerize to form functional channels. Human TREK-1 channels are highly expressed in many regions of the central nervous system and are generally believed to play a critical role in controlling neuronal excitability (Goldstein et al. 2001; Patel & Honoré, 2001; Lesage, 2003).
A remarkable feature of the TREK-1 channel is its sensitivity to a wide variety of endogenous and exogenous modulators. These include cell volume, with hyperosmolarity closing the channel, temperature, with heat opening the channel (with a Q10 of about 7), and internal acidosis, which also opens the channel. In addition, a number of compounds potently activate TREK-1, including lysophospholipids, polyunsaturated fatty acids and certain general anaesthetics and neuroprotectants. Among these are particular drugs, such as the neuroprotectant riluzole (Duprat et al. 2000), and volatile (Patel et al. 1999) and gaseous (Gruss et al. 2004) general anaesthetics, that have been proposed to act, at least in part, by activating TREK-1 channels.
The possibility that TREK-1 activation might explain the actions of these drugs has been challenged recently by reports (Miller et al. 2003, 2004) that, under the conditions of relative hypoxia that exist in the brain, TREK-1 channels are insensitive to the actions of arachidonic acid and, by implication, other neuroprotectants and general anaesthetics. If true, these observations would seem to rule out a role for TREK-1 channels in neuroprotection and general anaesthesia.
During the course of experiments (Gruss et al. 2004) on the activation of TREK-1 by anaesthetic gases (such as nitrous oxide and xenon) we observed that the activation by 100% xenon, and therefore in the absence of oxygen, was at least as large at that observed at 80% xenon. This implied that either xenon was exceptional in being able to activate TREK-1 under hypoxic conditions, or that the findings (Miller et al. 2003, 2004) showing that the properties of TREK-1 were greatly affected by hypoxia were incorrect.
Because of the potential importance of this question we have re-investigated the sensitivity of TREK-1 to oxygen and the extent to which the channel can be modulated by anaesthetics, arachidonic acid and internal acidosis. We present data that contradict some recent publications (Miller et al. 2003, 2004) that have reported that TREK-1 is oxygen sensitive. Moreover, we provide an explanation for the apparent inability of arachidonic acid to activate TREK-1 under hypoxic conditions. We conclude that TREK-1 channels might still play an important role in the actions of certain neuroprotectants and general anaesthetics.
Methods
Tissue culture
Cells from the human embryonic kidney cell line HEK 293, stably transfected with human TREK-1, were maintained in 5% CO2–95% air in a humidified incubator at 37°C in growth media (89.3% Dulbecco's modified Eagle's medium, 8.9% heat-inactivated fetal bovine serum and 1.8% of a stock solution of geneticin (20 mg ml−1)). When the cells were 80% confluent they were split and plated onto glass coverslips coated with poly d-lysine (1 mg ml−1) and stored at 37°C in a humidified incubator in 5% CO2–95% air. The cells were used during the next 5 days for electrophysiological experiments. Alternatively, HEK 293 cells were transiently transfected using the calcium phosphate method. One microgram of cDNA encoding the human TREK-1 subunit was added to each 35 mm well in a multiwell plate, and 1 μg of a plasmid encoding the cDNA of green fluorescent protein was included to identify cells expressing TREK-1 K+ channels. Following a 24 h incubation period at 3% CO2–97% air the cells were rinsed with saline and fresh growth medium was added to the wells. The cells were incubated at 37°C with 5% CO2–95% air for 12–72 h before electrophysiological measurements were made. HEK 293 cells stably expressing human TREK-1 channels (Meadows et al. 2000) and the human TREK-1 clone in the pcDNA 3.1 vector were kindly provided by Dr Helen Meadows at GlaxoSmithKline, Stevenage, Hertfordshire, UK.
Solution preparation
Solutions for the experiments were prepared by bubbling with pure oxygen, nitrogen or xenon through fine sintered-glass bubblers in either 250 ml or 500 ml Dreschel bottles filled with external saline (see ‘Electrophysiology’). During bubbling, the solutions were continuously stirred and held at room temperature. The solutions were bubbled for at least 30 min. Gas chromatography was used to show that this was sufficient time to achieve equilibrium. Mixing nitrogen- and oxygen-saturated solutions in a 4: 1 ratio formed the normoxic control saline solution with partial pressures of the dissolved gases nominally equivalent to 80% atm nitrogen and 20% atm oxygen. Hypoxic solutions were formed with nitrogen-saturated saline. The normoxic and hypoxic solutions were drawn up into a glass syringe via polytetrafluoro ethylene tubing from the bottom of the bubbled Dreschel bottles.
The equivalent partial pressure of oxygen (PO2) was measured using a Clark oxygen-sensitive electrode (OX100 sensor connected to a pA2000 meter, Unisense, Aarhus, Denmark). The equivalent PO2 in the electrophysiology bath was measured every 10 s and the data recorded to a hard disk using pCLAMP 6 software (Axon Instruments, Union City, CA, USA). The oxygen meter was calibrated before each use with 100% and 0% oxygen and a linear relationship assumed. Our hypoxic solutions typically had a PO2 of about 3% atm oxygen (this corresponds to 22.8 mmHg or 3 kPa). The normoxic solutions contained a fixed percentage of oxygen corresponding to, nominally, 20% atm.
Internal acidosis was achieved by exposing the cells to 10 mm propionate in our control extracellular solution (see below), but with the NaCl concentration reduced by 10 mm to 125 mm. Weak acids such as propionate have been shown (Bonnet et al. 2000) to produce a transient reduction in the intracellular pH, which in turn has been shown to activate TREK-1 channels (Maingret et al. 1999). Alternatively, internal acidosis was achieved by filling the electrodes with the intracellular solution (see below) titrated to pH 6.6 using KOH.
Solutions of 10 μm arachidonic acid in saline were made up using 100 mm stock solutions in ethanol. An appropriate volume of this stock solution was introduced into the bottom of a syringe reservoir before a polypropylene float was added. The syringe was then immediately back-filled with either hypoxic or normoxic extracellular saline solutions. All extracellular solutions, including control solutions, used in the arachidonic acid experiments contained a final concentration of 1.7 mm ethanol.
Solutions containing the volatile anaesthetic halothane (0.53 mm) were prepared as volume fractions of a saturated aqueous solution. The saturated concentration of halothane was taken to be 17.5 mm (Raventós, 1956). Back-filling the syringe reservoirs with an appropriate volume of a saturated halothane solution followed by an appropriate volume of a hypoxic or a normoxic solution formed the halothane solution. Eighty per cent xenon-saturated solutions were prepared by combining a saturated solution of xenon with 20% of either a nitrogen- or oxygen-saturated solution. Back-filling prevented the entry of oxygen into the solutions. The polypropylene floats prevented the loss or gain of any significant amount of the gases to the solutions throughout the course of the experiments. Polytetrafluoroethylene tubing and nylon valves were used throughout to connect the reservoirs to the recording bath.
Xenon (99.9%; research grade) was obtained from Air Products (Basingstoke, Hampshire, UK); nitrogen and oxygen were obtained from BOC (Guilford, Surrey, UK). All other chemicals were obtained from Sigma (Poole, Dorset, UK).
Electrophysiology
The composition of the control extracellular solution was (mm): 135 NaCl, 5 KCl, 1.2 MgCl, 5 Hepes, 2.5 CaCl2, 10 d-glucose, 30 sucrose titrated to pH 7.4 with NaOH, except for the experiments which included 10 mm propionate for which the NaCl concentration was reduced by 10 mm to 125 mm. Glass microelectrodes were pulled from thick-walled borosilicate glass capillaries (GC150F-7.5; Harvard Apparatus, Edenbridge, Kent, UK) using a two-stage vertical puller (PP-830; Narishige, Tokyo, Japan). Fire-polished pipettes were back-filled with 0.2 μm-filtered intracellular solution containing (mm): 10 NaCl, 117 KCl, 2 MgCl2, 11 Hepes, 11 EGTA, 1 CaCl2, 2 Na2ATP titrated to pH 7.2 or pH 6.6 (for some of the internal acidosis experiments) with KOH. Pipette resistances ranged from 3 to 5 MΩ. Series resistance was compensated by 80%. Voltage-clamp recordings were made using the whole-cell recording configuration. Cells were held at −80 mV and 2.69 s voltage ramps from −120 to −50 or 0 mV were recorded every 10 s using pCLAMP 6. The percentage activation elicited by the different agents was calculated relative to the average of the controls taken before and after the application of the agents.
Recordings were digitized at 10 kHz and the output of the patch-clamp amplifier (Axopatch 200A; Axon Instruments) was recorded on a computer hard disk via an analog to digital converter (Digidata 1200, Axon Instruments). The data were then analysed using Clampfit (Axon Instruments).
All electrophysiological experiments were carried out at room temperature (21–23°C). Values are given throughout as means ± s.e.m. Statistical significance was assessed using Student's paired t test.
Measurement of arachidonic acid concentrations
Samples (200 μl) of arachidonic acid in external saline were transferred to a vial containing 800 μl ethanol, then 20 μl of this solution was injected onto a Vydac C18 HPLC column (2.1 mm inner diameter, 250 mm length, Grace Vydac, USA) in a high performance liquid chromatography/mass spectrometer (Agilent 1100 Series, Agilent Technologies Deutschland GmbH, Germany) using a mobile phase (0.02% formic acid, 20% acetonitrile and water) at a flow rate of 0.2 ml min−1 at 25°C. The arachidonic acid was then eluted from the column using a linear gradient (over 30 min) of 0.02% formic acid, 90% acetonitrile and water and detected using a quadrupole mass spectrometer in electrospray ionization mode tuned for the negatively charged arachidonic acid ion (m/q= 303.2). Data were analysed using the Agilent Chemstation software.
Results
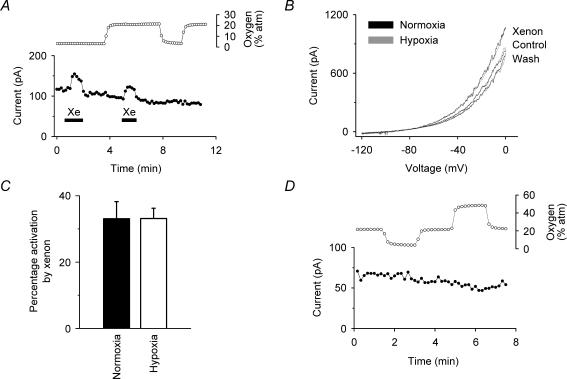
We first investigated whether or not the activation of TREK-1 by xenon that we had observed under normoxic conditions (Gruss et al. 2004), still occurred under conditions of relative hypoxia. Whole-cell recordings of human TREK-1 channels stably expressed in HEK 293 cells showed essentially the same degree of activation by 80% xenon under relative hypoxia as under normoxia. The data in Fig. 1A show the whole-cell hTREK-1 current at −50 mV when xenon (80%) was transiently applied. The presence of xenon caused a rapid and reversible increase in current under both normoxia and hypoxia. The measured oxygen concentration in the electrophysiology bath is plotted above the whole-cell current and shows that, during relative hypoxia, the PO2 was ∼3% atm. The current–voltage curves in Fig. 1B show that the activation by xenon under normoxia and hypoxia overlay almost exactly. Figure 1C gives a summary of the data which shows that the activation (33 ± 5%) under normoxia is not significantly different (at 99% confidence) from the activation (33 ± 3%) under hypoxia (means ± s.e.m.; n = 6). Figure 1D shows that the baseline TREK-1 K+ current changed insignificantly (2.6 ± 2.2%; n = 4) during relative hypoxia (3% atm) and that an increase in oxygen partial pressure to 50% atm was without significant effect.
Figure 1. Effects of oxygen on the modulation of human TREK-1.
A, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that the activation of hTREK-1 by xenon (80%) was comparable under normoxia (20% atm oxygen) and hypoxia (3% atm oxygen). The oxygen concentration in the electrophysiology bath, given as an equivalent partial pressure, is shown above the current recording (^). Xenon was transiently applied as indicated by the filled bars. B, current–voltage (I–V) curves show that the activation of TREK-1 by xenon was essentially identical under normoxia (21.3 ± 0.6% oxygen) and hypoxia (3.8 ± 0.2% oxygen). The normoxia I–V curves are normalized to the hypoxia control I–V curve to compensate for rundown. C, summary of the effects of xenon on TREK-1 currents under normoxia and hypoxia from whole-cell recordings at –50 mV. Xenon (80% atm) activated TREK-1 under normoxia and hypoxia by 33 ± 5% and 33 ± 3% (means ± s.e.m.; n = 6), respectively. D, current recordings under voltage clamp (•) (at –50 mV, derived from standard voltage ramps) showing that the equivalent partial pressure of oxygen in the solution had no significant effect on the baseline potassium current. For these stably transfected cells, the current at –50 mV under voltage clamp is almost exclusively due to the TREK-1 channel (see, for example Gruss et al. 2004).
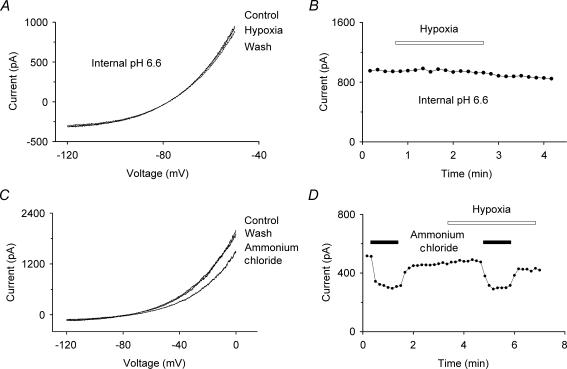
We next investigated if the TREK-1 K+ current, when activated by internal acidosis, was sensitive to hypoxia, as reported (Miller et al. 2004). We found that, as previously shown (Maingret et al. 1999), the TREK-1 K+ current increased significantly (86 ± 17% activation; n = 6) following internal acidosis caused by external perfusion with 10 mm propionate (Fig. 2). However, we found that the activation (75 ± 13%;n = 6) under conditions of relative hypoxia (2.9 ± 0.1% atm oxygen) was not significantly different (at 99% confidence). Although the extent of acidosis was unknown, based on the effects of pH on TREK-1 on inside-out patches (Maingret et al. 1999), this degree of activation is consistent with a decrease of the intracellular pH to about 6.8 units. We performed complementary experiments in which cells were patched using electrodes filled with internal solutions titrated to pH 6.6. Such internal acidification causes a rapid run-up in current and we tested the effects of relative hypoxia (2% atm oxygen) on the whole-cell current after it had stabilized. We found that hypoxia did not significantly affect this activated current (0.3 ± 1%;n = 4) (see Fig. 3A and B).
Figure 2. Effects of hypoxia on the modulation of hTREK-1 by internal acidosis caused by 10 mM propionate.
A, I–V curves show the activation of TREK-1 by internal acidosis under hypoxia. B, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that the activation of TREK-1 by propionate (Pro, 10 μm) was comparable under normoxia (22.3 ± 0.4% atm oxygen) and hypoxia (2.9 ± 0.1% atm oxygen). Propionate was transiently applied as indicated by the filled bars. C, summary of the effects of propionate (10 μm) on TREK-1 currents under normoxia and hypoxia from whole-cell recordings at –50 mV. Propionate activated TREK-1 under normoxia and hypoxia by 86 ± 17% and 75 ± 13% (means ± s.e.m.; n = 6), respectively.
Figure 3. Effects of hypoxia on the pH modulation of TREK-1.
A, I–V curves show no modulation by hypoxia (2% atm oxygen) of the TREK-1 current activated by an internal pH of 6.6. B, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that hypoxia had no effect on the TREK-1 current after activation by an internal pH of 6.6. C, I–V curves show the inhibition of TREK-1 during alkalinization by ammonium chloride (10 mm) under hypoxia. D, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that the inhibition of TREK-1 by ammonium chloride (10 mm) was comparable under normoxia (22 ± 0.4% atm oxygen) and hypoxia (2 ± 0.1% atm oxygen). Ammonium chloride was transiently applied as indicated by the filled bars.
We also attempted to use the ammonium chloride washout method (Roos & Boron, 1981), in which cells are washed with 10 mm ammonium chloride and then returned to normal saline, at which point there is a transient internal acidification. While we observed the expected inhibition of the current due to alkalinization when the ammonium chloride was applied, we did not observe the transient activation that should have occurred due to the internal acidosis following washout. We attribute this to an inhibition of the potassium current by either intracellular ammonia or ammonium ions, as has been reported for other potassium channels (Schlatter et al. 1994). However, we were able to show that internal alkalinization caused the same degree of inhibition, 45 ± 6% and 43 ± 5%, under normoxia (22 ± 0.4% atm) and hypoxia (2 ± 0.1% atm), respectively (n = 4) (see Fig. 3C and D).
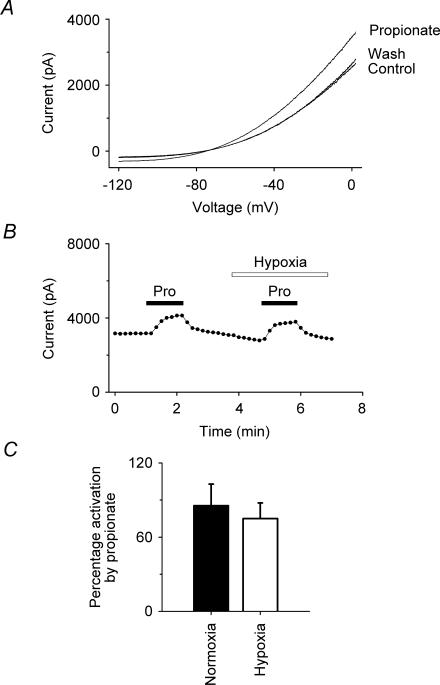
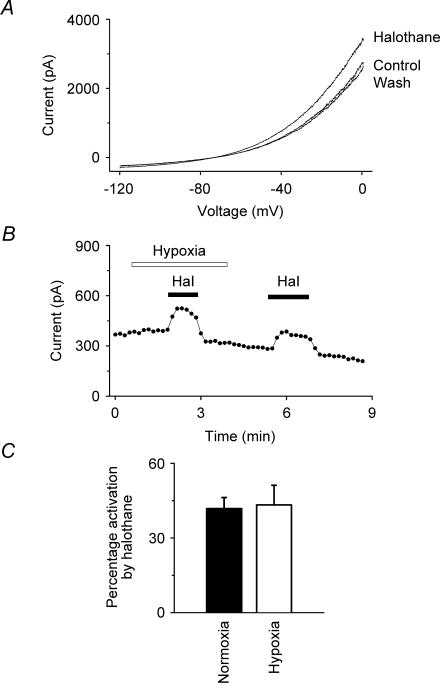
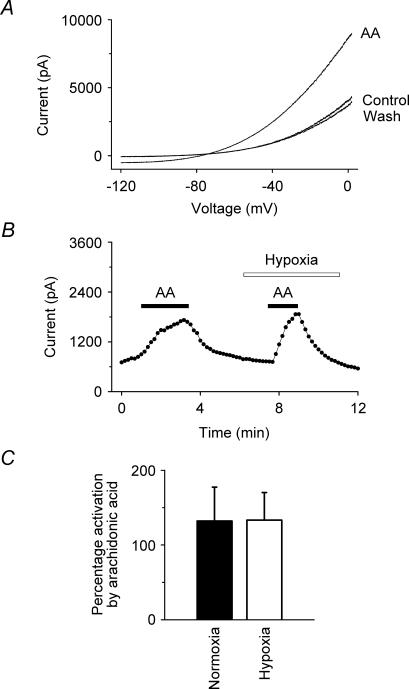
We further investigated the effects of hypoxia on the extent to which TREK-1 could be modulated by other agents to which it is sensitive, namely the inhalational general anaesthetic halothane and the polyunsaturated fatty acid, arachidonic acid. The data in Fig. 4 show that halothane (0.53 mm) activates the hTREK-1 K+ current reversibly under hypoxia as well as under normoxia. Figure 4C gives a summary of the data for halothane which shows that the activations observed under normoxia (42 ± 4%) and hypoxia (43 ± 8%) are not significantly different (at 99% confidence; n = 6). Figure 5 shows equivalent data for arachidonic acid (10 μm). As with halothane, the activation under normoxia (132 ± 45%) by arachidonic acid was not significantly different from that under hypoxia (133 ± 37%) (at 99% confidence; n = 6).
Figure 4. Effects of hypoxia on the activation of hTREK-1 by halothane.
A, I–V curves show the activation of TREK-1 by halothane (0.53 mm) under hypoxia. B, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that the activation of TREK-1 by halothane (Hal, 0.53 mm) was comparable under normoxia (20% atm oxygen) and hypoxia (3% atm oxygen). Halothane was transiently applied as indicated by the filled bars. C, summary of the effects of halothane (0.53 mm) on TREK-1 currents under normoxia and hypoxia from whole-cell recordings at –50 mV. Halothane activated TREK-1 under normoxia and hypoxia by 42 ± 4% and 43 ± 8% (means ± s.e.m.; n = 6), respectively.
Figure 5. Effects of hypoxia on the activation of hTREK-1 by arachidonic acid.
A, current–voltage (I–V) curves show the activation of TREK-1 by arachidonic acid (AA, 10 μm) under hypoxia. B, voltage-clamp currents (•) (at –50 mV, derived from standard voltage ramps) showing that the activation of TREK-1 by arachidonic acid (10 μm) was comparable under normoxia (21.7 ± 0.7% oxygen) and hypoxia (4.1 ± 0.4% oxygen). Arachidonic acid was transiently applied as indicated by the filled bars. C, summary of the effects of arachidonic acid (10 μm) on TREK-1 currents under normoxia and hypoxia from whole-cell recordings at –50 mV. Arachidonic acid (10 μm) activated TREK-1 under normoxia and hypoxia by 132 ± 45% and 133 ± 37% (means ± s.e.m.; n = 6), respectively.
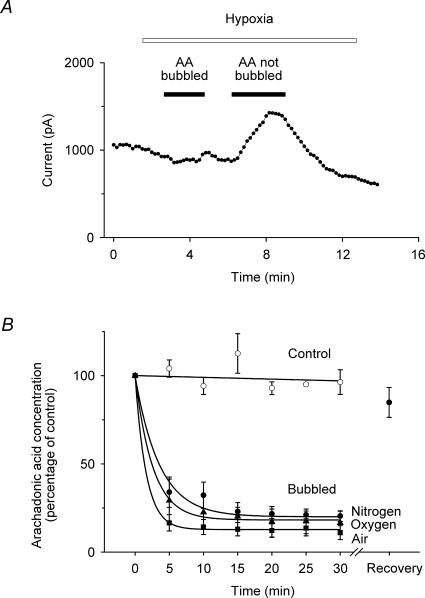
Because these results were so different to those that had been published previously (Miller et al. 2003, 2004), we explored different protocols for preparing hypoxic solutions containing arachidonic acid. We soon discovered that the bubbling process per se was critical and that, if the solutions were first bubbled with nitrogen and the arachidonic acid was added last, such solutions gave robust activations of TREK-1 currents (see Figs 6A and 5). However, if the arachidonic acid was added first and then the solutions were bubbled with nitrogen (the protocol used by previous workers), such arachidonic acid solutions elicited little or no activation of TREK-1 currents (Fig. 6A). We suspected that the arachidonic acid was simply being removed from solution by the bubbling process and we investigated this using HPLC/mass spectrometry. We first showed (Fig. 6B) that 10 μm arachidonic acid maintained its concentration in aqueous solution under exactly the conditions in which our gas solutions were prepared, excepting only that the solutions were not bubbled. If, however, the arachidonic acid was added to the aqueous solution prior to bubbling, then the concentration in solution dropped very rapidly during the bubbling process such that the majority of the arachidonic acid was lost after just 5 min (Fig. 6B). This was the case when the solutions were bubbled by nitrogen, oxygen or air. After this process, a faint smear was visible on the surface of the Dreschel bottle which we took to be arachidonic acid that had been taken out of solution. If fresh, nitrogen-bubbled, external saline was then added and the Dreschel bottle vigorously shaken, then this solution was able to give substantial activations of TREK-1 currents (data not shown). In separate experiments, we used ethanol to recover the arachidonic acid that had been ‘lost’ during the bubbling process and were able to show using HPLC/mass spectrometry that 85 ± 9% could be recovered from the glassware (Fig. 6B).
Figure 6. Effects of bubbling on arachidonic acid concentration in solution.
A, whole-cell current recordings (at –50 mV from standard voltage ramps) show that arachidonic acid (10 μm) caused a marked activation of TREK-1 under hypoxic conditions (3% oxygen) when it was added to the solution after bubbling with nitrogen (AA not bubbled), but caused little or no activation of TREK-1 currents when the arachidonic acid was added first and then the solution bubbled (AA bubbled). B, arachidonic acid (10 μm) remained in solution when stirred (^) but was rapidly removed from solution if the solution was bubbled with nitrogen (•), oxygen (▴) or air (▪) (see Methods). The arachidonic acid can be shown to have been deposited on the glassware because when the bottle was rinsed with ethanol 85 ± 9% of the arachidonic acid could be recovered after bubbling with nitrogen (see Recovery data point in B).
Discussion
Since their discovery, two-pore-domain potassium channels have emerged as one of the most richly modulated groups of ion channels (Goldstein et al. 2001; Patel & Honoré, 2001; Lesage, 2003). Sensitive to many different intracellular and extracellular chemical modulators, as well as to temperature and cell volume, the various members of the family provide countless ways in which neuronal excitability can be regulated. In addition to their normal physiological functions, many of which have, no doubt, yet to be determined, some members of the family have been identified as likely drug targets. In particular, the TREK channels have been proposed to be a target for volatile (Patel et al. 1999) and gaseous (Gruss et al. 2004) general anaesthetics, as well as neuroprotective drugs such as riluzole (Duprat et al. 2000) and certain polyunsaturated fatty acids (Lesage et al. 2000).
The plausibility of the TREK channels acting as targets for these drugs in the brain is seriously challenged by reports (Miller et al. 2003, 2004) that, under conditions of relative hypoxia, TREK-1 loses much of its ability to be modulated by agents such as arachidonic acid. Because the human brain operates under relatively hypoxic conditions, with PO2 levels varying between 3 and 6% (Hoffman et al. 1996; Dings et al. 1998), these reports are incompatible with TREK-1 being a significant target for either anaesthetics or other neuroactive drugs.
We report here, however, that we have been unable to reproduce the published findings (Miller et al. 2003, 2004) that appear to demonstrate that TREK-1 is oxygen sensitive. Not only do we find that increasing or decreasing oxygen concentrations have no effect on TREK-1 baseline currents, but more importantly, we show that the channel can still be activated by volatile and gaseous general anaesthetics, by arachidonic acid and by internal acidosis, to exactly the same extent under normoxia and hypoxia. We also demonstrate that the techniques used in previous studies (Miller et al. 2003, 2004) to prepare hypoxic solutions containing arachidonic acid would have removed the arachidonic acid from solution. The mechanism by which this happens is easy to imagine. Long-chain fatty acids are highly surface-active compounds and will tend to concentrate at air–water interfaces when these are present. During the bubbling, a very large surface area of gas–water interface is created and this is likely to effectively sequester the arachidonic acid from solution, bringing it to the surface. This is consistent with the deposit we observed at the top of the Dreschel bottle after bubbling arachidonic acid solutions. We cannot offer an explanation for why TREK-1 appeared to be inhibited by hypoxia in the previous studies (Miller et al. 2003), or why hypoxia appeared to change the response to internal acidosis (Miller et al. 2004); it seems possible that the explanation lies in the protocols used for preparing or applying the solutions.
In summary, we think it unlikely that the activity of the TREK-1 channel, or its ability to be modulated by endogenous or exogenous chemicals, is significantly affected by the oxygen concentration, although we can only state this with confidence for the agents and conditions that we have studied, namely activation by the volatile and gaseous anaesthetics, arachidonic acid and internal acidosis. We thus conclude that TREK-1 channels remain a plausible target for such general anaesthetics and neuroprotectants. In corroboration, we note that the recent demonstration (Heurteaux et al. 2004) that TREK-1 knock-out mice have a significantly reduced sensitivity for volatile general anaesthetics gives additional support for this view.
Acknowledgments
We thank Raquel Yustos for excellent technical support, Alistair Mathie and Stephen Brickley for helpful comments on the manuscript and the Medical Research Council, Carburos Metálicos and Air Products and Chemicals Inc. for support. A.J.C. holds a BBSRC case studentship. Marco Gruss was a Deutsche Forschungsgemeinschaft Fellow.
References
- Bonnet U, Bingmann D, Wiemann M. Intracellular pH modulates spontaneous and epileptiform bioelectric activity of hippocampal CA3-neurones. Eur Neuropsychopharmacol. 2000;10:97–103. doi: 10.1016/s0924-977x(99)00063-2. [DOI] [PubMed] [Google Scholar]
- Dings J, Meixensberger J, Jager A, Roosen K. Clinical experience with 118 brain tissue oxygen partial pressure catheter probes. Neurosurgery. 1998;43:1082–1095. doi: 10.1097/00006123-199811000-00045. 10.1097/00006123-199811000-00045. [DOI] [PubMed] [Google Scholar]
- Duprat F, Lesage F, Patel AJ, Fink M, Romey G, Lazdunski M. The neuroprotective agent riluzole activates the two P domain K+ channels TREK-1 and TRAAK. Mol Pharmacol. 2000;57:906–912. 10.1097/00006123-199811000-00045. [PubMed] [Google Scholar]
- Goldstein SA, Bockenhauer D, O'Kelly I, Zilberberg N. Potassium leak channels and the KCNK family of two-P-domain subunits. Nat Rev Neurosci. 2001;2:175–184. doi: 10.1038/35058574. 10.1038/35058574. [DOI] [PubMed] [Google Scholar]
- Gruss M, Bushell TJ, Bright DP, Lieb WR, Mathie A, Franks NP. Two-pore-domain K+ channels are a novel target for the anesthetic gases xenon, nitrous oxide, and cyclopropane. Mol Pharmacol. 2004;65:443–452. doi: 10.1124/mol.65.2.443. 10.1124/mol.65.2.443. [DOI] [PubMed] [Google Scholar]
- Heurteaux C, Guy N, Laigle C, Blondeau N, Duprat F, Mazzuca M, Lang-Lazdunski L, Widmann C, Zanzouri M, Romey G, Lazdunski M. TREK-1, a K+ channel involved in neuroprotection and general anesthesia. EMBO J. 2004;23:2684–2695. doi: 10.1038/sj.emboj.7600234. 10.1038/sj.emboj.7600234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman WE, Charbel FT, Edelman G. Brain tissue oxygen, carbon dioxide, and pH in neurosurgical patients at risk for ischemia. Anesth Analg. 1996;82:582–586. doi: 10.1097/00000539-199603000-00027. 10.1097/00000539-199603000-00027. [DOI] [PubMed] [Google Scholar]
- Lesage F. Pharmacology of neuronal background potassium channels. Neuropharmacology. 2003;44:1–7. doi: 10.1016/s0028-3908(02)00339-8. 10.1016/S0028-3908(02)00339-8. [DOI] [PubMed] [Google Scholar]
- Lesage F, Terrenoire C, Romey G, Lazdunski M. Human TREK2, a 2P domain mechano-sensitive K+ channel with multiple regulations by polyunsaturated fatty acids, lysophospholipids, and Gs, Gi, and Gq protein-coupled receptors. J Biol Chem. 2000;275:28398–28405. doi: 10.1074/jbc.M002822200. 10.1074/jbc.M002822200. [DOI] [PubMed] [Google Scholar]
- Maingret F, Patel AJ, Lesage F, Lazdunski M, Honoré E. Mechano- or acid stimulation, two interactive modes of activation of the TREK-1 potassium channel. J Biol Chem. 1999;274:26691–26696. doi: 10.1074/jbc.274.38.26691. 10.1074/jbc.274.38.26691. [DOI] [PubMed] [Google Scholar]
- Meadows HJ, Benham CD, Cairns W, Gloger I, Jennings C, Medhurst AD, Murdock P, Chapman CG. Cloning, localisation and functional expression of the human orthologue of the TREK-1 potassium channel. Pflugers Arch. 2000;439:714–722. doi: 10.1007/s004249900235. 10.1007/s004240050997. [DOI] [PubMed] [Google Scholar]
- Miller P, Kemp PJ, Lewis A, Chapman CG, Meadows HJ, Peers C. Acute hypoxia occludes hTREK-1 modulation: re-evaluation of the potential role of tandem P domain K+ channels in central neuroprotection. J Physiol. 2003;548:31–37. doi: 10.1113/jphysiol.2003.040048. 10.1097/00006123-199811000-00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller P, Peers C, Kemp PJ. Polymodal regulation of hTREK1 by pH, arachidonic acid, and hypoxia: physiological impact in acidosis and alkalosis. Am J Physiol Cell Physiol. 2004;286:C272–C282. doi: 10.1152/ajpcell.00334.2003. 10.1152/ajpcell.00334.2003. [DOI] [PubMed] [Google Scholar]
- Patel AJ, Honoré E. Properties and modulation of mammalian 2P domain K+ channels. Trends Neurosci. 2001;24:339–346. doi: 10.1016/s0166-2236(00)01810-5. 10.1016/S0166-2236(00)01810-5. [DOI] [PubMed] [Google Scholar]
- Patel AJ, Honoré E, Lesage F, Fink M, Romey G, Lazdunski M. Inhalational anesthetics activate two-pore-domain background K+ channels. Nat Neurosci. 1999;2:422–426. doi: 10.1038/8084. 10.1038/8084. [DOI] [PubMed] [Google Scholar]
- Raventós J. The action of fluothane – a new volatile anaesthetic. Br J Pharmacol. 1956;11:394–410. doi: 10.1111/j.1476-5381.1956.tb00007.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roos A, Boron WF. Intracellular pH. Physiol Rev. 1981;61:296–434. doi: 10.1152/physrev.1981.61.2.296. [DOI] [PubMed] [Google Scholar]
- Schlatter E, Haxelmans S, Hirsch J, Leipziger J. pH dependence of K+ conductances of rat cortical collecting duct principal cells. Pflugers Arch. 1994;428:631–640. doi: 10.1007/BF00374587. 10.1007/BF00374587. [DOI] [PubMed] [Google Scholar]