Abstract
The mdx mouse lacks dystrophin and is a model of human Duchenne muscular dystrophy. Single mdx muscle fibres were isolated and subjected to a series of stretched (eccentric) contractions while measuring intracellular calcium concentration ([Ca2+]i) with fluo-3 and confocal microscopy. Following the stretched contractions there was a slow rise in resting [Ca2+]i and after 30 min both the [Ca2+]i during a tetanus (tetanic [Ca2+]i) and the tetanic force were reduced. Two blockers of stretch-activated channels, streptomycin and the spider venom toxin GsMTx4, prevented the rise of resting [Ca2+]i and partially prevented the decline of tetanic [Ca2+]i and force. Reducing extracellular calcium to zero also prevented the rise in resting [Ca2+]i and prevented some of the decline in tetanic [Ca2+]i and force. Patch-clamping experiments identified a stretch-activated channel in both wild-type and mdx myotubes which was blocked by GsMTx4. These data suggest that blockers of stretch-activated channels can ameliorate the force reduction following stretched contractions by reducing the influx of Ca2+ into the muscle. We therefore tested whether in intact mdx mice streptomycin, added to the drinking water, was capable of reducing muscle damage. mdx mice show a period of muscle damage from 20 to 40 days of life and fibres which regenerate from this damage display central nuclei. We measured the frequency of central nuclei in control mdx mice compared to streptomycin-treated mdx mice and showed that the incidence of central nuclei was significantly reduced by streptomycin treatment. This result suggests that blockers of stretch-activated channels may protect against muscle damage in the intact mdx mouse.
The mechanism of muscle damage in Duchenne muscular dystrophy and the mdx mouse remains uncertain. In both of these conditions, genetic mutations result in the absence of the protein dystrophin in muscle. Dystrophin is a cytoskeletal protein which links the contractile protein actin to a group of proteins (the dystrophin-associated glycoproteins) in the surface membrane (Blake et al. 2002). The functional role of dystrophin in normal muscles is unclear; two possibilities are as follows. (i) Dystrophin may have a structural role in maintaining the integrity of the surface membrane during the stress imposed by normal contractions, particularly those involving stretch. In support of this hypothesis dystrophic muscle is more susceptible to stretch-induced muscle damage (Moens et al. 1993; Petrof et al. 1993) and, further, expression of a mini-dystrophin in mdx muscle can reduce the increased susceptibility to damage (Deconinck et al. 1996). (ii) Dystrophin may be involved in the clustering or regulation of membrane-associated proteins including ion channels (Carlson, 1998). For instance the dystrophin-associated glycoproteins (Blake et al. 2002), NO synthase (Brenman et al. 1995), aquaporin (Frigeri et al. 2001), L-type Ca2+ channels (Friedrich et al. 2004), and stretch-activated channels (Franco-Obregon & Lansman, 2002) have each been shown to have altered expression or function in mdx muscles. Whether abnormal function in membrane proteins regulated by dystrophin contributes to the pathological muscle damage remains unclear.
Muscles which are stretched during contraction (eccentric contractions) are particularly liable to damage (Morgan & Allen, 1999; Warren et al. 2001) and, as noted above, this damage is exacerbated in the absence of dystrophin. Key pathological features of the damage are disordered sarcomeres (Fridén et al. 1981) and an increase in muscle permeability (McNeil & Khakee, 1992; Hamer et al. 2002). The increased membrane permeability causes (i) loss of soluble intracellular proteins such as creatine kinase (Newham et al. 1983), (ii) appearance of extracellular proteins or fluorescent dyes inside the muscle cells (Hamer et al. 2002) and (iii) increases in the concentration of intracellular ions which are normally maintained at low concentrations inside the cell (Fong et al. 1990; Yeung et al. 2003b). The mechanism of these pathological features is uncertain but a major candidate is increased intracellular calcium which is widely thought to be closely involved in the process of muscle damage, possibly by activating proteases (Turner et al. 1988; Goll et al. 2003).
There has been considerable interest in defining the mechanism(s) of the increase in Ca2+ influx which characterizes stretch-induced muscle damage. One hypothesis is that membrane tears, possibly caused by sarcomere inhomogeneities, cause the increase in Ca2+ influx. The fact that large proteins such as creatine kinase and albumin leave or enter the cell is a strong argument for large membrane defects which might result from membrane tears (McNeil & Khakee, 1992). However there are also studies which suggest that altered channel properties might be responsible for some aspects of the increased permeability. A Ca2+-permeable leak channel which is active in resting muscle and has a higher open probability in mdx muscle has been described by several groups (Franco & Lansman, 1990a; Fong et al. 1990). Franco-Obregon & Lansman (2002) have demonstrated that this channel can be activated or inactivated by stretch and propose that in dystrophic muscles these channels might act as a source of additional Ca2+ entry. There is also evidence for Ca2+ entry by channels which allow entry of Mn2+ and are activated by store depletion (Tutdibi et al. 1999; Vandebrouck et al. 2002); these channels were encoded by the transient receptor potential channel gene and seemed to have properties similar to the mechanosensitive channel discussed above (Vandebrouck et al. 2002).
In the first part of the present study we have concentrated on the cause and consequence of the increase in resting [Ca2+]i which occurs following stretched contractions (Balnave & Allen, 1995). Recently we showed that [Na+]i also rises following stretched contractions and this rise could be prevented by Gd3+ and streptomycin, agents which block stretch-activated channels (Yeung et al. 2003a, b). These findings led to the present study in which we test whether the increase in resting [Ca2+]i caused by stretch might also arise through stretch-activated channels.
In our earlier experiments on mdx fibres damaged by stretched contractions, we found that streptomycin and Gd3+ could partially reverse the fall of force (Yeung et al. 2003b). Because it is known that part of the fall in force after stretched contractions is caused by a fall in the [Ca2+]i during tetani (tetanic [Ca2+]i) (Balnave & Allen, 1995), in the present study we tested whether blockers of stretch-activated channels could prevent some of the fall of tetanic [Ca2+]i. Given the positive effects of streptomycin on isolated mdx muscles, in the second part of the study we have looked for evidence that streptomycin might reduce muscle damage in the intact mdx mouse. McBride et al. (2000) added streptomycin to the drinking water of rats and showed that sufficient streptomycin was absorbed to block the depolarizing effect of stretch-activated channels. In the present experiments we have shown that streptomycin, administered through the drinking water, can reduce the number of fibres with central nuclei in mdx muscles. Because central nuclei are a sign of fibres regenerating after damage (Coulton et al. 1988), this suggests that blockers of stretch-activated channels exert some protection against muscle damage in intact mdx mice.
Methods
Animals
mdx mice were supplied by ARC Perth, WA, Australia and from Jackson Laboratory, Bar Harbour, ME, USA. Wild-type mice were the C57BL/10ScSn strain in which the mdx mutation initially arose. These experiments were approved by the Animal Ethics Committee of the University of Sydney.
Studies on isolated mdx single muscle fibres
mdx mice (10–14 weeks old) were killed by cervical dislocation and single flexor brevis muscle fibres were dissected as previously described (Yeung et al. 2003b). Single fibres with clips attached to the tendons were mounted in the experimental chamber between an Akers AE 801 force transducer (SensorNor, Horten, Norway) and the lever of a motor (High speed length controller 322B Aurora Scientific Inc, Ontario, Canada). Platinum electrodes parallel to the muscle fibre allowed stimulation. With this apparatus, force could be measured during tetanic stimulation while known length changes were imposed on a single muscle fibre.
The dissection was performed in the following solution (mm): NaCl 136.5, KCl 5, CaCl2 1.8, MgCl2 0.5, NaH2PO4 0.4 and NaHCO3 11.9 (pH 8.0). During the experiment, the fibre was superfused with standard solution of the following composition (mm): NaCl 121, KCl 5, CaCl2 1.8, MgCl2 0.5, NaH2PO4 0.4, NaHCO3 24, glucose 5.5 and 0.2% fetal calf serum (Gibco). The solution was bubbled with 95% O2–5% CO2 (pH 7.4). Experiments on the isolated fibres were performed at room temperature (∼22°C).
The effect of stretch-activated channel blockers, gadolinium (Gd3+), streptomycin and GsMTx4 were investigated. Aqueous stock solutions of 1 m GdCl3, 100 mm streptomycin and 1 mm GsMTx4 were prepared and diluted immediately before use. Zero extracellular Ca2+ Tyrode solution was prepared by removing Ca2+ and adding 1 mm Mg2+ and 1 mm EGTA.
Experimental protocol
mdx muscle fibres were initially set to the length that produced maximal tetanic force (optimum length, Lo). Each muscle fibre was first subjected to 10 isometric tetani as control, followed by 10 stretched tetani (eccentric contractions). The muscle fibre was stimulated at 100 Hz, and tetani were 400 ms in duration with a rest period of 3.6 s between tetani (cycle, 4 s). The stretch was applied 200 ms after the start of stimulation and the fibre was stretched from Lo to Lo+ 40% over 100 ms (stretching velocity, 4 × Lo s−1). The fibre was returned to Lo between tetani. The 100 Hz force was measured at 10 and 30 min after the isometric controls and the stretched tetani (all at the original Lo).
Confocal imaging of [Ca2+]i using fluo-3
The fluorescent indicator fluo-3 (Molecular Probes, OR, USA) was used to measure [Ca2+]i. The membrane permeant acetoxymethyl ester (AM) version of fluo-3 (fluo-3-AM) was prepared as a 1 mm stock solution in dimethyl sulphoxide. Once dissection was complete, the muscle fibre was incubated in 0.5 ml of dissection solution containing 5 μm fluo-3-AM for 20 min at room temperature. After loading, the fibre was transferred to the experimental chamber and perfused with the standard solution for 30–45 min to allow hydrolysis to fluo-3. The experimental protocol was performed on the stage of an inverted confocal microscope (Leica TCS SL, Heidelberg, Germany) with the fibre positioned about 100–150 μm above the coverslip (thickness, 150 μm) which formed the base of the chamber. The fibre was examined with a water immersion objective (× 63; numerical aperture, 1.2), using a 488-nm illumination line at 25% maximum power and recording at wavelengths of 510–560 nm. This objective has a working distance of 220 μm above the coverslip.
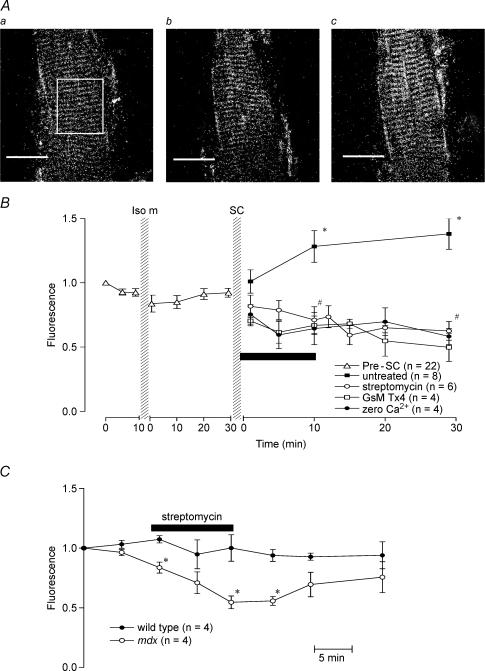
At high magnification in well-loaded fibres, the distribution of fluo-3 fluoresence shows a sarcomere-like pattern (see Fig. 1) as has been noted in previous studies (Tsugorka et al. 1995). This is thought to arise from binding of fluo-3 to sarcomeric structures but does not prevent the detection of superimposed [Ca2+]i-related signals (Tsugorka et al. 1995). In many preparations, dead fibres adjacent to the single live fibre showed varying degrees of punctate fluorescence. In addition regions of increased fluorescence probably attributable to nuclei were sometimes visible (either peripheral or central). Measurement of resting fluorescence was taken from a box over a large region of the fibre avoiding nuclei (see Fig. 1Aa) and any fluorescence from dead fibres. As far as possible the same region was used in all images but fibres sometimes moved particularly after stretched contractions so this was not always possible.
Figure 1. [Ca2+]i signals from mdx muscle fibres.
A, confocal images of fluo-3 fluorescence from one mdx muscle fibre under control conditions (a), following isometric contractions (b) and 30 min after stretched contractions (c). Fluorescence intensity was measured by outlining a region of the muscle fibre with a rectangle as indicated in a. Scale bars represent 50 μm. B, the effect of stretch-activated channel blockers and zero extracellular Ca2+ on resting [Ca2+]i following stretched contractions (SC) in mdx muscle fibres. The fluorescence intensity was normalized to the starting point of each experiment. Following stretched contractions in the untreated fibres, [Ca2+]i-dependent fluorescence was significantly higher than before the stretched contractions (*). Application of stretch-activated channel blockers, streptomycin or GsMTx4, or zero Ca2+ solution for 10 min following stretched contractions (as indicated by the horizontal bar) led to a reduction in [Ca2+]i. #Significant difference between the treated and untreated experiments. Bars represent s.e.m. C, effect of streptomycin on resting [Ca2+]i in wild-type and mdx muscle fibres. Muscle fibres were exposed to 200 μm streptomcyin for 10 min as indicated by the bar. *Significant difference between wild-type and mdx muscle fibres. Bars represent s.e.m.
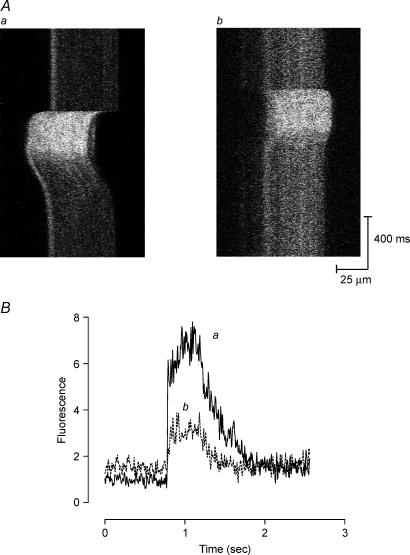
Tetani were imaged with the confocal microscope in xt mode; that is, repeated scanning of the same line with time displayed vertically (Fig. 2Aa and b). Fibres move both laterally and vertically during nominally isometric tetani and, where possible, the fibre position was adjusted so that the fibre could be imaged both at rest and during tetani. Some fibres moved too far and tetanic [Ca2+]i images could not be obtained. Because fluorescence was generally uniformly distributed we believe errors arising from this source should be small. For this reason n-values for tetanic [Ca2+]i are sometimes smaller than for resting [Ca2+]i.
Figure 2. Tetanic [Ca2+]i in one mdx muscle fibre following stretched contractions.
A, line scan images over a region of the muscle fibre during tetani were collected in xt mode under control conditions (a) and 30 min after stretched series (b). Movement artifacts cause the lateral movements of the signal during the contractions. B, spatially averaged fluorescence profiles corresponding to Aa and Ab, showing an elevated resting [Ca2+]i and a reduction in tetanic [Ca2+]i following stretched contractions.
Image processing and analysis were performed using NIH Image (Scion Corporation, MD, USA). Bleaching and/or dye loss did not appear to present problems under these conditions. It is difficult to calibrate fluo-3 signals and we present all data as the ratio of fluorescence to the resting level of fluorescence (F/Fo).
Patch clamping the stretch-activated channel
Flexor digitorm brevis muscles were isolated in 4 ml of 0.1% collagenase B in RPMI 1640 medium (Sigma-Aldrich, St Louis, MO, USA) for 2.5 h at room temperature. Blood vessels and tendons were removed, the muscle was rinsed with RPMI medium and incubated in 3 ml of RPMI medium plus 0.05% trypsin-EDTA for 15 min. The tissue was rinsed twice with divalent ion-free PBS. The tissue was triturated to release cells, spun at 1000 r.p.m., and the cells resuspended in wash medium. Cells were seeded onto mouse laminin-coated coverslips and incubated in 89% RPMI media, 10% heat-inactivated horse serum and 1% chick embryo extract. Myotubes began to form after 3–5 days. Myotubes were used in patch-clamp experiments between 6 and 12 days after plating.
Outside-out patches were formed so that GsMTx4 peptide could be tested on the extracellular surface. Outside-out patches are formed when the electrode is slowly withdrawn from a cell in whole-cell clamp mode. When the membrane pinches off, it reseals with the extracellular surface exposed on the outside of the pipette. An Axopatch 200B (Axon Instruments, CA, USA) was used for patch clamping, while experimental protocols and data acquisition were controlled by Axon Instruments pClamp9 software via a Digidata 1322A acquisition system. Currents were sampled at 10 kHz and low-pass filtered at 2 kHz through the 4-pole Bessel filter on the Axopatch 200B. All potentials are defined with respect to the extracellular surface. Electrodes were pulled on a Model PC-84 pipette puller (Brown-Flaming Instruments, CA, USA), painted with Sylgard 184 (Dow Corning Corp., Midland, MI, USA) and fire polished. Electrodes were filled with KCl saline (containing mm): KCl 140, EGTA 5, MgSO4 2, Hepes 10; pH 7.3 and had resistances ranging from 8 to 16 MΩ. Bath saline contained (mm): NaCl 140, KCl 5, CaCl2 2, MgCl2 0.5, glucose 6 and Hepes 10; pH 7.3. Pressure and suction were applied to the pipette by a HSPC-1 pressure clamp (ALA Scientific Instruments, NY, USA) controlled by the pClamp software. An ALA MP285 was used to perfuse outside-out patches. Normal perfusion saline contained (mm): NaCl 140, KCl 5, MgCl2 0.5 and Hepes 10; pH 7.3. Off-line data analysis was performed with Clampfit and Origin 6.1 software.
Studies on intact mdx mice
The aim of these experiments was to determine whether orally administered streptomycin could alter the development of muscle damage in the intact mdx mouse. mdx mice have a period of intense muscle damage around the age of 20–40 days, resulting in damage to muscle fibres followed by regeneration (McGeachie et al. 1993). Weaned mdx pups at the age of 18–19 days were divided into two groups; half were placed in cages with 4 g l−1 (3 mm) streptomycin in the drinking water (McBride et al. 2000) while the other half had normal drinking water. Wild-type mice were also given streptomycin-containing or normal drinking water.
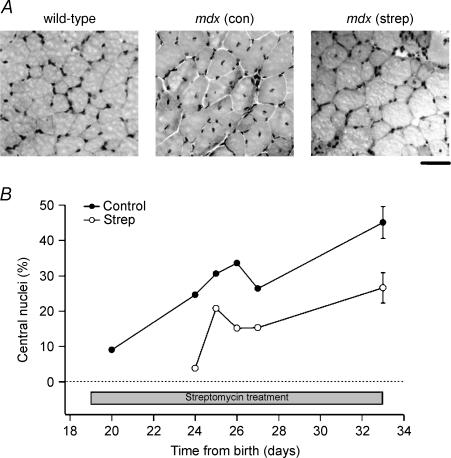
At various ages between 20 and 33 days, animals were killed by cervical dislocation and the extensor digitorum longus muscle (EDL) dissected free. Each muscle was attached to a cork pad on a steel frame, immersed in embedding medium (Tissue-Tek) before being snap-frozen in isopentane cooled in liquid N2. Frozen muscle sections (thickness, 10 μm) were fixed in 4% paraformaldehyde for 30 min and stained with haematoxylin and eosin. Sections were viewed under a microscope and digital images were taken and stored for later analysis. The number of cells containing central nuclei (see Fig. 6A) were expressed as a percentage of the total number of fibres (i.e. fibres with central or peripheral nuclei).
Figure 6. Central nuclei in streptomycin-treated and untreated mdx mice.
A, haematoxylin and eosin-stained cross-sections showing the location of nuclei (peripheral or central) from wild-type and mdx mice given either normal (con) or streptomycin-treated (strep) drinking water for 2 weeks. Scale bar, 50 μm. B, the percentage of muscle fibres with central nuclei from muscle cross-sections of treated and untreated mdx mice, plotted against time after birth of each animal. The period over which streptomycin was administered is indicated by the bar. At day 33 (14 days of streptomycin treatment), the values represent the mean ± s.e.m. from three untreated and four treated mice.
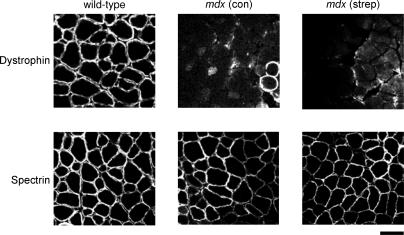
Streptomycin is an aminoglycoside antibiotic. It has been shown that other aminoglycosides, such as gentamicin, can result in some dystrophin expression in muscles of mdx mice, by suppressing the stop-codon mutation in the dystrophin gene (Barton-Davis et al. 1999). Therefore, in order to show that streptomycin was preventing muscle damage by blocking stretch-activated channels, it was important to demonstrate that dystrophin expression was not increased. Muscle cross-sections from mdx and wild-type muscles given either normal or streptomycin-treated drinking water for 7 or 14 days were used for immunohistochemical analysis of dystrophin and another membrane protein, spectrin, which is present in mdx muscle.
Cross-sections were first incubated in AffiniPure Fab Fragment IgG (Jackson ImmunoResearch Laboratories) at a concentration of 60 μg ml−1 in 2% bovine serum albumin (BSA) for 30 min, washed in PBS and blocked in 2% BSA for 15 min. Sections were then incubated for 2 h at room temperature with either dystrophin C-terminus (DYS2; Novocastra Laboratories) or spectrin (SPEC2; Novocastra Laboratories) primary antibodies, both diluted 1 : 100 in 2% BSA. After being washed in PBS, sections were incubated for 1 h with a Cy3-conjugated secondary antibody (Jackson Laboratories), which was diluted 1: 200 in 2% BSA. Images of cross-sections (see Fig. 7) were taken with a digital camera attached to a fluorescent microscope.
Figure 7. Dystrophin and spectrin staining of muscle sections.
Muscle cross-sections from wild-type and treated and untreated mdx mice (14 days of streptomycin treatment) showing dystrophin and spectrin staining. Note the two dystrophin-positive (revertant) fibres from the untreated mdx mouse muscle. Scale bar, 50 μm.
Results
Effect of stretched contractions on resting and tetanic [Ca2+]i in isolated single mdx muscle fibres
Figure 1A shows fluorescent images of fluo-3 distribution in a resting fibre (Fig. 1Aa), following isometric tetani (Fig. 1Ab) and following stretched contractions (Fig. 1Ac). There was no obvious change in intensity following isometric tetani while following stretched contractions the image becomes more intense indicating increased [Ca2+]i. Resting [Ca2+]i was estimated by measuring the intensity of fluorescence in a region which was less than the full width of the fibre and about one fibre diameter in length (see box in Fig. 1Aa). All subsequent measurements were normalized to the initial measurement. Figure 1B shows averaged data of this sort from 22 preparations before and after the isometric series. There was no significant change in resting [Ca2+]i after the 10 isometric tetani consistent with previous studies of short trains of isometric tetani. In contrast after the 10 stretched contractions (□), the resting [Ca2+]i showed a slow rise reaching 1.38 ± 0.12 (n = 8, P < 0.05) after 30 min. A rise in resting [Ca2+]i has also been observed in wild-type fibres after stretched contractions (Balnave & Allen, 1995; Ingalls et al. 1998).
At 30 min after the stretched contractions, isometric force was re-determined (with the muscle still at the previously determined Lo) and was reduced to 35 ± 4%(n = 4, P < 0.0001) of the original value. xt plots of fluorescence are shown in Fig. 2A, and Fig. 2Aa shows a typical tetanus; the pronounced movement to the left is a movement artifact (see Methods). Note the more intense line at the left of the resting image probably representing a nucleus. Figure 2Ab shows a tetanus from the same fibre 30 min after the stretched contractions and it is clear that the resting [Ca2+]i is higher, as established above, but the tetanic [Ca2+]i is lower. The spatially averaged time course from a region of the fibres is shown in Fig. 2B. In five such experiments, tetanic [Ca2+]i 30 min after the stretched contractions was reduced to 56 ± 2%(P < 0.0001) of the control. This confirms the reduction of tetanic [Ca2+]i following stretched contractions observed in wild-type muscles (Balnave & Allen, 1995). It is known that T-tubules are altered following stretched contractions (Takekura et al. 2001; Yeung et al. 2002) and this might impair inward transmission of the action potential and affect Ca2+ release in the centre of the fibre (Westerblad et al. 1990). To test, for this we examined whether the fluorescence in the centre of the fibre was lower than near the edges, but no such tendency was apparent (compare Fig. 2Aa and Ab). This suggests that impaired conduction in the T-tubules is not the cause of the reduced tetanic [Ca2+]i following stretched contractions.
Effect of blockers of stretch-activated channels on resting [Ca2+]i
Previously we have shown that Gd3+ and streptomycin, which are blockers of stretch-activated channels (Hamill & McBride, 1996), prevent the Na+ entry into wild-type and mdx fibres (Yeung et al. 2003a, b). We therefore tested whether the increase in resting [Ca2+]i following stretched contractions might also arise through Ca2+ entry from the extracellular space via stretch-activated channels.
In preliminary control experiments we examined the effect of streptomycin (200 μm) on resting [Ca2+]i in unperturbed fibres. In four wild-type fibres, streptomycin, applied for 10 min, had no significant effect on the resting [Ca2+]i (see Fig. 1C). In contrast when streptomycin was applied to resting mdx fibres, the ratio was reduced to 0.55 ± 0.05 after 10 min (significantly less than in wild-type fibres, P < 0.01) and partially recovered to 0.76 ± 0.13 after 20 min washoff. This result is similar to the finding with resting [Na+]i in wild-type and mdx fibres (Yeung et al. 2003b).
We then studied resting [Ca2+]i after the stretched contraction protocol. In one series of experiments, 100 μm streptomycin was added to the perfusate for the 10-min period immediately following the stretched contractions. Figure 1B shows that the resting [Ca2+]i showed a tendency to fall and by the end of 30 min was 0.63 ± 0.07 (n = 6), significantly less than that observed in the absence of streptomycin (P < 0.001).
In a second series of experiments, we tested the newly described stretch-activated channel blocker isolated from the venom of the spider Grammostola spatulata (Suchyna et al. 2000). We used synthetic GsMTx4 (Ostrow et al. 2003) and, because of limited availability, the GsMTx4 was added to the muscle bath (volume 200 μl) with the flow stopped to achieve a final concentration of 10 μm. After 10 min, flow was restarted washing away the GsMTx4. Figure 1B shows that the results are similar to streptomycin with the rise of [Ca2+]i prevented and resting [Ca2+]i reduced to 0.50 ± 0.11 (n = 4) by 30 min (significantly less than control, P < 0.001).
In a third series of experiments, zero extracellular Ca2+ solution was perfused for a 10-min period starting immediately after the end of the stretched tetani. As shown in Fig. 1B, this procedure eliminated the rise of [Ca2+]i and 30 min after the end of the stretched tetani, the fluo-3 fluorescence was reduced to 0.57 ± 0.07 (n = 4), significantly lower than the 1.38 ± 0.12 (n = 8) in the presence of extracellular Ca2+(P < 0.05). This results suggests that Ca2+ entry from the extracellular space is the source of the rise in [Ca2+]i.
We did not test Gd3+ using this protocol but in four preliminary experiments the stretched tetani were imposed, resting [Ca2+]i was allowed to rise for 30 min, and 20 μm Gd3+ was then applied for 10 min. Before application the resting fluo-3 ratio was 1.34 ± 0.15 while at the end of the 10-min exposure to Gd3+ the ratio was significantly reduced to 0.87 ± 0.07 (P < 0.05). Thus Gd3+ is also capable of reducing the increased resting [Ca2+]i after stretched contractions.
Effect of GsMTx4 on stretch-activated channels
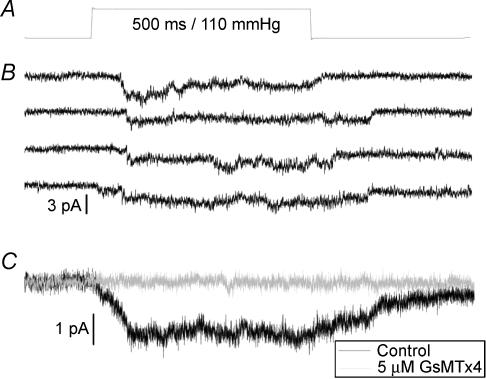
Given that GsMTx4 inhibits the rise in resting [Ca2+]i after stretched contractions we wanted to establish that it could block the stretch-activated channels in wild-type and mdx muscle. Synthetic GsMTx4 has been shown to block stretch-activated channels on rat astrocytes (Ostrow et al. 2003) and chick ventricular myocytes (Suchyna et al. 2004). Here we show that cation-selective stretch-activated channels with a unitary conductance of ∼1.8 pA at −60 mV, could be activated by pressure steps applied to an outside-out patch from a primary cultured mdx mouse myotube (Fig. 3B). These channels appear to be the same as those previously described in normal and mdx myotubes and myofibres (Franco-Obregon & Lansman, 1994). The average patch current produced by these stretch-activated channels is inhibited by 5 μm GsMTx4 (Fig. 3C). In two mdx myotubes the inhibition was 99 and 75% while in two wild-type fibres the inhibition was 98 and 96%. It is clear that GsMTx4 produces a substantial inhibition of this channel, and further experiments are underway to establish any difference in efficacy on patch and whole-cell currents from both wild-type and mdx myotubes.
Figure 3. GsMTx4 blocks stretch-activated channels in outside-out patches from mdx mouse myotubes.
Pressure step (A) applied for 500 ms every 2 s, to an outside-out patch from an mdx mouse myotube held at –60 mV, activates stretch-activated channels with a unitary current of 1.8 pA (B). Four representative records are shown prior to GsMTx4 application. Multiple steps are averaged (C) to produce the average patch current records in the absence (□, n = 11 pressure steps) and presence of GsMTx4 (▄, n = 11 pressure steps), showing that the peptide blocks these currents.
Effect of blockers of stretch-activated channels on tetanic [Ca2+]i and force
In previous studies we have shown that the reduced force after stretched contractions is partly caused by reduced sarcoplasmic reticulum (SR) Ca2+ release (Balnave & Allen, 1995) and that blocking stretch-activated channels with either Gd3+ or streptomycin can improve the recovery of force following stretch-induced damage (Yeung et al. 2003a, b). In the present study we sought to test whether interventions that limit the entry of Ca2+ into the fibre are capable of increasing the tetanic [Ca2+]i after stretch-induced damage.
In the first protocol we applied 100 μm streptomycin for 10 min immediately after the stretched contractions. In the two experiments from this series of six in which tetanic [Ca2+]i could be measured, this concentration of streptomycin did not appear to affect the tetanic [Ca2+]i measured 30 min after the stretched contractions. We therefore increased the concentration of streptomycin to 200 μm and applied it at the start of the stretched contractions and for 20 min after. Under these conditions, the tetanic [Ca2+]i measured 30 min after stretched contraction was 56 ± 2% under control conditions but in the treated case it was 73 ± 8%(n = 4, P < 0.05). Similarly the tetanic force was greater in the treated experiments (56 ± 5%) compared to the untreated controls (35 ± 4%; n = 4, P < 0.05).
In the second series of experiments we used 10 μm GsMTx4 applied for 20 min starting immediately after the stretched contractions. In the treated experiments tetanic force after the stretched contractions was 70 ± 8%(n = 4) of control while tetanic [Ca2+]i was 82 ± 2%(n = 4) of control. Both values are significantly greater than in the absence of GsMTx4 (Fig. 4; P < 0.01). GsMTx4 (10 μm) was significantly more effective (P < 0.05, unpaired t test) than either 100 or 200 μm streptomycin at increasing tetanic [Ca2+]i.
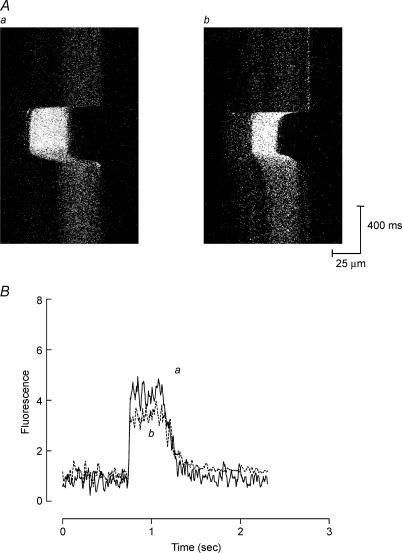
Figure 4. Effect of GsMTx4 on tetanic [Ca2+]i following stretched contractions in one mdx muscle fibre.
A, xt images of one mdx muscle fibre during tetani under control conditions (a) and 30 min after stretched contraction (b) in the presence of GsMTx4. B, spatially averaged fluorescence plots derived from images in Aa and b. Application of GsMTx4 immediately after stretched contractions prevented the rise in resting [Ca2+]i and prevented some reduction in tetanic [Ca2+]i (see Results).
In a third series of experiments, we explored the effect of zero extracellular calcium applied for 20 min immediately after the stretched contractions. In the treated experiments tetanic force after stretched contractions was 72 ± 8%(n = 4) while tetanic [Ca2+]i was 88 ± 4%(n = 4). Both values were significantly greater than those under untreated conditions (P < 0.05).
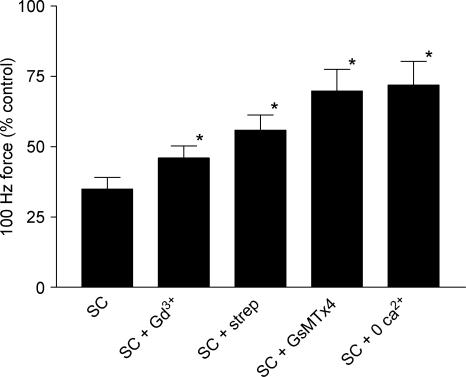
Figure 5 compares the recovery of tetanic force for the various procedures described here; the Gd3+ data are taken from our previous study in which an identical protocol was used (Yeung et al. 2003b). All the interventions reduced the decline of force after stretched contractions; zero extracellular calcium and GsMTx4 were the most effective.
Figure 5. Recovery of force following stretched contractions (SCs) in mdx muscle fibres.
Bars indicate 100 Hz force normalized to control tetani. Force recovery was significantly higher (*P < 0.05) in muscle fibres exposed to 20 μm gadolinium (SC + Gd3+), 200 μm streptomycin (SC + strep), 10 μm GsMTx4 (SC + GsMTx4) or zero extracellular [Ca2+]i (SC + 0 Ca2+) for 10 min following stretched contractions. Bars represent s.e.m.
Effect of oral streptomycin on central nuclei in intact mdx mice
Figure 6A illustrates haematoxylin and eosin sections of wild-type, mdx and streptomycin-treated mdx fibres. Note that in the wild-type muscle all fibres have peripheral nuclei. In the 33-day-old untreated mdx muscle there are more small fibres and many of these have central nuclei, indicating that the original fibre was damaged and had regenerated. In the 33-day-old treated (14 days streptomycin) there are some small fibres with central nuclei but fewer than in the age-matched untreated mdx muscle. Data collected from 18 mdx mice are shown in Fig. 6B. Note that at 20 days the untreated mdx mice have few central nuclei (∼9%) but these increase to 45 ± 5%(n = 3) by day 33. This result is similar to the results of other studies (Coulton et al. 1988; McGeachie et al. 1993). The streptomycin-treated animals have consistently lower levels of central nuclei compared to the untreated mdx mice of the same age. This is clearest at 33 days where the mean of four treated animals was 27 ± 4% (P < 0.05 compared to the untreated mdx muscles at 33 days). When these data was subjected to two-way ANOVA (time as one variable, treatment as the second), treatment with streptomycin significantly reduced the percentage of central nuclei (P < 0.01).
Dystrophin expression in streptomycin-treated animals
Aminoglycosides interfere with ribosomal translation of mRNA and can sometimes cause readthrough of premature stop codons. Thus in mdx mice, gentamicin has been shown to produce expression of dystrophin in some fibres (Barton-Davis et al. 1999) and this prospect has generated several clinical trials (e.g. Wagner et al. 2001). To establish whether the positive effects of streptomycin on central nuclei could have arisen by this mechanism we measured dystrophin expression by immunohistochemistry.
Figure 7 (wild-type) shows the standard immunohistological appearance of wild-type muscle stained for dystrophin. As expected mdx fibres show virtual absence of surface membrane-staining though we have intentionally shown a region with two revertant fibres (Wilton et al. 1997). Spectrin staining was normal in wild-type, mdx and mdx-treated fibres. The streptomycin treatment for 7 (not shown) and 14 days did not cause visible dystrophin expression. Thus under the present conditions, the changes in the numbers of regenerating fibres are not a consequence of dystrophin expression induced by streptomycin.
Discussion
Intracellular Ca2+ in the mdx muscle
Muscles from humans with muscular dystrophy and the mdx mouse have an elevated total Ca2+ (for review see Gillis, 1999), and increased activity of Ca2+-activated proteases is thought to represent one pathway for muscle damage (Turner et al. 1988; Blake et al. 2002). Nevertheless the pathway for Ca2+ entry in dystrophic muscle remains unclear. As indicated in the Introduction, there is considerable evidence linking membrane tears associated with stretched-contractions as a route for Ca2+ entry (McNeil & Khakee, 1992). How quickly such tears would remain permeable to small or large molecules is not entirely clear, though a recent study of intense light-induced membrane damage showed sealing over within 1 min and found that mdx fibres sealed over after damage at the same rate as wild-type fibres (Bansal et al. 2003).
Our studies point to Ca2+ entry via channels rather than membrane tears. First we have not been able to observe localized increases of either Ca2+ or Na+ following stretched contractions despite the measurements being made in the period where [Ca2+]i and [Na+]i are rising (Balnave et al. 1997; Yeung et al. 2003a). Of course there are many possible explanations for these negative findings such as rapid uptake of Ca2+ by SR which would minimize the rise in myoplasmic [Ca2+]i. Alternatively the increases might be too small or too transient or too infrequent to be detected by the methods used.
Second, we have previously shown that the rise in [Na+]i, and here show that the rise of [Ca2+]i, are prevented by a group of disparate substances which are all blockers of stretch-activated channels. Gd3+, a member of the lanthanide series, blocks stretch-activated channels at around 10–20 μm but also inhibits several other channels, albeit at higher concentrations, and binds to many physiological anions which can make its use problematic (Caldwell et al. 1998). Streptomycin has also been widely used as a blocker but it is relatively insensitive requiring a concentration of 50–200 μm in most studies (for review see Hamill & McBride, 1996). It also has a range of biological effects involving modification of ribosomal protein synthesis which are the basis of its antimicrobial activity. The mechanism of action on stretch-activated channels appears to be occlusion of the channel pore (Winegar et al. 1996). The recently described GsMTx4 is the most potent blocker yet described (Suchyna et al. 2000). In the present study GsMTx4 prevented Ca2+ entry at a concentration (10 μm) which is sufficient to inhibit the cation-selective swelling-activated channel in ventricular myocytes (Suchyna et al. 2000). The specificity of GsMTx4 has not been extensively studied but it appeared to have no effect on the voltage-sensitive currents in astrocytes (Suchyna et al. 2000), nor did it affect the action potential form or spontaneous firing rate of cultured rabbit atrial myocytes (Bode et al. 2000), nor a bacterial voltage-sensitive K+ channel (KVAP) (Ruta & MacKinnon, 2004). The gene for GsMTx4 has been cloned and the 34-amino acid peptide can be synthesized and has identical properties to the natural product (Ostrow et al. 2003). Mutational analysis may allow the active site to be defined and it may be possible to produce more active analogues. Alternatively, the enantiomeric d-peptide form of GsMTx4, which has at least the same efficacy as the wild-type l-peptide, may be a useful agent in its current state (Suchyna et al. 2004). d-peptides may be less susceptible to proteolysis and have lower immunogenicity.
Third, the observation that streptomycin lowers resting [Ca2+]i in the mdx muscle but not the wild-type suggests important differences in Ca2+ regulation and is similar to our earlier finding with Na+ measurements (Yeung et al. 2003b). In that study we showed that [Na+]i was higher in mdx fibres and the effect of stretch-activated channel blockers in the mdx was to lower [Na+]i back to close to the normal wild-type level. Thus both observations suggest that a class of channels, permeable to Na+ and Ca2+ and blocked by streptomycin and Gd3+, are substantially more active in resting muscles of the md x compared to wild-type mice.
The stretch-activated channel described in skeletal muscle by Franco & Lansman has several properties suggesting that it may contribute to Ca2+ entry following stretch-induced damage. Specifically it is permeable to both Na+ and Ca2+ (Franco & Lansman, 1990b) and is blocked by both Gd3+ and streptomycin (Franco et al. 1991; Winegar et al. 1996). Furthermore its activity is increased in muscles from mdx mice and Duchenne muscular dystrophy patients (Franco-Obregon & Lansman, 1994; Franco-Obregon & Lansman, 2002; Vandebrouck et al. 2001). In the present study the pressure sensitivity of the stretch-activated channel was lower than in previous studies but we believe this is because of two features of our approach. (i) Our data come from outside-out patches. When we activated channels in cell-attached mode the activation pressures were lower, between 30 and 70 mmHg. (ii) Our method of using short (0.5 s) pressure steps differs from most other studies that applied prolonged steady pressure. In order to activate channels in each step it is necessary to use pressures which are 30–40 mmHg above threshold (the pressure where channel activity is observed in 10–20% of sequential steps). The channels we show here have a conductance of ∼30 pS at −60 mV and are cation selective, similar to the mechanosensitive channels described in astrocytes (Suchyna et al. 2000) and normal and mdx muscle. This channel is active in wild-type and mdx muscle and is blocked by GsMTx4 in the micromolar range, which also prevents the rise of [Ca2+]i after stretched contractions. Thus our working hypothesis is that the absence of dystrophin in the mdx muscle increases the activity of a cation-selective stretch-activated channel which increases Ca2+ entry both in resting fibres and, particularly, following stretched contractions. The resulting increase in [Ca2+]i is proposed to activate various pathways which contribute to the muscle damage.
A recent study on mdx mice in which utrophin, which is closely related to dystrophin, was over-expressed led to different conclusions on the importance of stretch-activated channels in the pathology of dystrophic muscle (Squire et al. 2002). Increased expression of utrophin returned stretch-activated channel activity to normal but caused only a moderate improvement in necrotic foci, central nuclei and susceptibility to stretch-induced damage. The authors concluded that ‘these observations question the role of increased Ca2+ channel activity in initiating the dystrophic process’. We think an alternative interpretation of their experiments is that increased activity of Ca2+ channels appears capable of explaining a substantial component of dystrophic pathology which would be consistent with the interpretation of our experiments above.
What is the relationship between raised resting [Ca2+]i and reduced SR Ca2+ release?
It has been often observed in skeletal muscle that interventions which elevate resting [Ca2+]i can subsequently cause reduced Ca2+ release (Chin & Allen, 1996; Bruton et al. 1996). A particularly clear analysis was possible in skinned muscle with intact T-tubules and SR in which known elevations of Ca2+ could be applied and the resulting change in SR release quantified (Lamb et al. 1995). However the mechanism of this finding remains uncertain with Ca2+-activated proteases or mechanical stresses to the T-tubular–SR junction as two possibilities. The present study provides another example in which elevated resting [Ca2+]i is linked to a subsequent reduction of tetanic [Ca2+]i. The present experiments clearly demonstrate the functional link between elevated resting [Ca2+]i because when the elevation was prevented with stretch-activated channel blockers or reduced extracellular Ca2+, the decline in tetanic [Ca2+]i was ameliorated. However the mechanism of this functional link remains to be established.
Mechanism of action of streptomycin in the intact mdx mouse
Given that the stretch-activated blockers appear capable of reducing stretch-induced Na+ and Ca2+ influx and also improving the force production after stretch-induced damage, we were interested to explore whether these agents might exert some protection against in vivo muscle damage in the mdx mouse. We chose streptomycin for the present purpose because it has been widely used to treat bacterial infections and because McBride et al. (2000) showed that oral streptomycin could be absorbed by intact rats to a level which reduced the stretch-induced depolarization, presumably by blocking stretch-activated channels. In the present experiments, streptomycin administered by this route significantly lowered the incidence of central nuclei in young mdx mice. Our data do not establish whether the development of central nuclei were delayed or permanently reduced in magnitude, distinctions which would be of great importance clinically if these drugs were used to treat muscular dystrophy. Nevertheless, central nuclei are an established marker of previous damage and regeneration (Coulton et al. 1988; McGeachie et al. 1993). So these results are compatible with streptomycin reducing muscle damage in the intact mdx mouse as clearly shown in our single fibre experiments.
While promising, these results are clearly preliminary. Oral administration of streptomycin is simple and convenient but suffers from the problem that absorption from the gut is limited and possibly variable (Rake & Donovick, 1949). Other indicators of muscle damage such as creatine kinase release and fluorescent dye uptake need to be measured to confirm that muscle damage has been minimized and we need to establish the plasma concentration of streptomycin achieved. However, in support of our results, recent clinical trials of gentamicin in Duchenne muscular dystrophy patients showed that plasma creatine kinase levels were decreased in patients where no detectable dystrophin expression occurred (Wagner et al. 2001). Our interpretation would be that gentamicin was blocking stretch-activated channels and thereby lowering [Ca2+]i and reducing muscle damage.
Our results do establish convincingly that streptomycin is not acting by increasing expression of dystrophin although aminoglycoside antibiotics can produce such effects under some circumstances (Barton-Davis et al. 1999). Clearly in the acute, in vitro experiments the time course is far too fast for dystrophin expression to occur and the similar results with other stretch-activated channel blockers argue that channel blocking is the mechanism of action. In the intact animals, the 14-day time course could allow dystrophin expression to occur; however, there was no evidence of it based on immunohistochemistry.
Despite these limitations, the present results are sufficiently positive to encourage exploration of other routes of aminoglycoside administration and to perform more comprehensive tests of muscle damage. It would also be desirable to establish that muscle damage can be reduced by other drugs which block the stretch-activated channels. Even if aminoglycosides proved effective at reducing muscle damage it is doubtful if these drugs would be suitable for long-term treatment because of their toxic effects on vestibular, auditory and renal function. The newly described enantiomer of GsMTx4, synthesized from D amino acids and thought to be resistant to proteases, might be one such possibility (Suchyna et al. 2004).
Acknowledgments
This work was supported by the Australian Research Council. E.W.Y. acknowledges support from an Internal Competitive Research grant (A-PE65) from Hong Kong Polytechnic University. F.S. is supported by a National Institutes of Health grant.
References
- Balnave CD, Allen DG. Intracellular calcium and force in single mouse muscle fibres following repeated contractions with stretch. J Physiol. 1995;488:25–36. doi: 10.1113/jphysiol.1995.sp020943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balnave CD, Davey DF, Allen DG. Distribution of sarcomere length and [Ca2+]i in single fibres from mouse skeletal muscle following stretch-induced injury. J Physiol. 1997;502:649–659. doi: 10.1111/j.1469-7793.1997.649bj.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bansal D, Miyake K, Vogel SS, Groh S, Chen CC, Williamson R, McNeil PL, Campbell KP. Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature. 2003;423:168–172. doi: 10.1038/nature01573. [DOI] [PubMed] [Google Scholar]
- Barton-Davis ER, Cordier L, Shoturma DI, Leland SE, Sweeney HL. Aminoglycoside antibiotics restore dystrophin function to skeletal muscles of mdx mice. J Clin Invest. 1999;104:375–381. doi: 10.1172/JCI7866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blake DJ, Weir A, Newey SE, Davies KE. Function and genetics of dystrophin and dystrophin-related proteins in muscle. Physiol Rev. 2002;82:291–329. doi: 10.1152/physrev.00028.2001. [DOI] [PubMed] [Google Scholar]
- Bode F, Sachs F, Franz MR. Tarantula peptide inhibits atrial fibrillation. Nature. 2000;409:35. doi: 10.1038/35051165. [DOI] [PubMed] [Google Scholar]
- Brenman JE, Chao DS, Xia H, Aldape K, Bredt DS. Nitric oxide synthase complexed with dystrophin and absent from skeletal muscle sarcolemma in Duchenne muscular dystrophy. Cell. 1995;82:743–752. doi: 10.1016/0092-8674(95)90471-9. [DOI] [PubMed] [Google Scholar]
- Bruton JD, Lannergren J, Westerblad H. Effects of repetitive tetanic stimulation at long intervals on excitation–contraction coupling in frog skeletal muscle. J Physiol. 1996;495:15–22. doi: 10.1113/jphysiol.1996.sp021570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell RA, Clemo HF, Baumgarten CM. Using gadolinium to identify stretch-activated channels: technical considerations. Am J Physiol. 1998;275:C619–C621. doi: 10.1152/ajpcell.1998.275.2.C619. [DOI] [PubMed] [Google Scholar]
- Carlson CG. The dystrophinopathies: an alternative to the structural hypothesis. Neurobiol Dis. 1998;5:3–15. doi: 10.1006/nbdi.1998.0188. [DOI] [PubMed] [Google Scholar]
- Chin ER, Allen DG. The role of elevations in intracellular Ca2+ concentration in the development of low frequency fatigue in mouse single muscle fibres. J Physiol. 1996;491:813–824. doi: 10.1113/jphysiol.1996.sp021259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coulton GR, Morgan JE, Partridge TA, Sloper JC. The mdx mouse skeletal muscle myopathy: I. A histological, morphometric and biochemical investigation. Neuropathol Appl Neurobiol. 1988;14:53–70. doi: 10.1111/j.1365-2990.1988.tb00866.x. [DOI] [PubMed] [Google Scholar]
- Deconinck N, Ragot T, Marechal G, Perricaudet M, Gillis JM. Functional protection of dystrophic mouse (mdx) muscles after adenovirus-mediated transfer of a dystrophin minigene. Proc Natl Acad Sci U S A. 1996;93:3570–3574. doi: 10.1073/pnas.93.8.3570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fong PY, Turner PR, Denetclaw WF, Steinhardt RA. Increased activity of calcium leak channels in myotubes of Duchenne human and mdx mouse origin. Science. 1990;250:673–676. doi: 10.1126/science.2173137. [DOI] [PubMed] [Google Scholar]
- Franco A, Jr, Lansman JB. Calcium entry through stretch-inactivated ion channels in mdx myotubes. Nature. 1990a;344:670–673. doi: 10.1038/344670a0. [DOI] [PubMed] [Google Scholar]
- Franco A, Jr, Lansman JB. Stretch-sensitive channels in developing muscle cells from a mouse cell line. J Physiol. 1990b;427:361–380. doi: 10.1113/jphysiol.1990.sp018176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco A, Jr, Winegar BD, Lansman JB. Open channel block by gadolinium ion of the stretch-inactivated ion channel in mdx myotubes. Biophys J. 1991;59:1164–1170. doi: 10.1016/S0006-3495(91)82332-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco-Obregon A, Jr, Lansman JB. Mechanosensitive ion channels in skeletal muscle from normal and dystrophic mice. J Physiol. 1994;481:299–309. doi: 10.1113/jphysiol.1994.sp020440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franco-Obregon A, Lansman JB. Changes in mechanosensitive channel gating following mechanical stimulation in skeletal muscle myotubes from the mdx mouse. J Physiol. 2002;539:391–407. doi: 10.1113/jphysiol.2001.013043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridén J, Sjöström M, Ekblom B. A morphological study of delayed muscle soreness. Experientia. 1981;37:506–507. doi: 10.1007/BF01986165. [DOI] [PubMed] [Google Scholar]
- Friedrich O, Both M, Gillis JM, Chamberlain JS, Fink RH. Mini-dystrophin restores L-type calcium currents in skeletal muscle of transgenic mdx mice. J Physiol. 2004;555:251–265. doi: 10.1113/jphysiol.2003.054213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frigeri A, Nicchia GP, Nico B, Quondamatteo F, Herken R, Roncali L, Svelto M. Aquaporin-4 deficiency in skeletal muscle and brain of dystrophic mdx mice. FASEB J. 2001;15:90–98. doi: 10.1096/fj.00-0260com. [DOI] [PubMed] [Google Scholar]
- Gillis JM. Understanding dystrophinopathies: an inventory of the structural and functional consequences of the absence of dystrophin in muscles of the mdx mouse. J Muscle Res Cell Motil. 1999;20:605–625. doi: 10.1023/a:1005545325254. [DOI] [PubMed] [Google Scholar]
- Goll DE, Thompson VF, Li H, Wei W, Cong J. The calpain system. Physiol Rev. 2003;83:731–801. doi: 10.1152/physrev.00029.2002. [DOI] [PubMed] [Google Scholar]
- Hamer PW, McGeachie JM, Davies MJ, Grounds MD. Evans Blue Dye as an in vivo marker of myofibre damage: optimising parameters for detecting initial myofibre membrane permeability. J Anat. 2002;200:69–79. doi: 10.1046/j.0021-8782.2001.00008.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamill OP, McBride DW., Jr The pharmacology of mechanogated membrane ion channels. Pharmacol Rev. 1996;48:231–252. [PubMed] [Google Scholar]
- Ingalls CP, Warren GL, Williams JH, Ward CW, Armstrong RB. E-C coupling failure in mouse EDL muscle after in vivo eccentric contractions. J App Physiol. 1998;85:58–67. doi: 10.1152/jappl.1998.85.1.58. [DOI] [PubMed] [Google Scholar]
- Lamb GD, Junankar PR, Stephenson DG. Raised intracellular [Ca2+] abolishes excitation–contraction coupling in skeletal muscle fibres of rat and toad. J Physiol. 1995;489:349–362. doi: 10.1113/jphysiol.1995.sp021056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBride TA, Stockert BW, Gorin FA, Carlsen RC. Stretch-activated ion channels contribute to membrane depolarization after eccentric contractions. J App Physiol. 2000;88:91–101. doi: 10.1152/jappl.2000.88.1.91. [DOI] [PubMed] [Google Scholar]
- McGeachie JK, Grounds MD, Partridge TA, Morgan JE. Age-related changes in replication of myogenic cells in mdx mice: quantitative autoradiographic studies. J Neurol Sci. 1993;119:169–179. doi: 10.1016/0022-510x(93)90130-q. [DOI] [PubMed] [Google Scholar]
- McNeil PL, Khakee R. Disruptions of muscle fiber plasma membranes. Role in exercise-induced damage. Am J Pathol. 1992;140:1097–1109. [PMC free article] [PubMed] [Google Scholar]
- Moens P, Baatsen PH, Marechal G. Increased susceptibility of EDL muscles from mdx mice to damage induced by contractions with stretch. J Muscle Res Cell Motil. 1993;14:446–451. doi: 10.1007/BF00121296. [DOI] [PubMed] [Google Scholar]
- Morgan DL, Allen DG. Early events in stretch-induced muscle damage. J App Physiol. 1999;87:2007–2015. doi: 10.1152/jappl.1999.87.6.2007. [DOI] [PubMed] [Google Scholar]
- Newham DJ, Jones DA, Edwards RH. Large delayed plasma creatine kinase changes after stepping exercise. Muscle Nerve. 1983;6:380–385. doi: 10.1002/mus.880060507. [DOI] [PubMed] [Google Scholar]
- Ostrow KL, Mammoser A, Suchyna T, Sachs F, Oswald R, Kubo S, Chino N, Gottlieb PA. cDNA sequence and in vitro folding of GsMTx4, a specific peptide inhibitor of mechanosensitive channels. Toxicon. 2003;42:263–274. doi: 10.1016/s0041-0101(03)00141-7. [DOI] [PubMed] [Google Scholar]
- Petrof BJ, Shrager JB, Stedman HH, Kelly AM, Sweeney HL. Dystrophin protects the sarcolemma from stresses developed during muscle contraction. Proc Natl Acad Sci U S A. 1993;90:3710–3714. doi: 10.1073/pnas.90.8.3710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rake G, Donovick R. Absorption, distribution and excretion of streptomycin. In: Waksman SA, editor. Streptomycin; Nature and Practical Applications. Baltimore: Williams & Wilkins Co; 1949. pp. 236–253. [Google Scholar]
- Ruta V, MacKinnon R. Localization of the voltage-sensor toxin receptor on KvAP. Biochem. 2004;43:10071–10079. doi: 10.1021/bi049463y. [DOI] [PubMed] [Google Scholar]
- Squire S, Raymackers JM, Vandebrouck C, Potter A, Tinsley J, Fisher R, Gillis JM, Davies KE. Prevention of pathology in mdx mice by expression of utrophin: analysis using an inducible transgenic expression system. Hum Mol Genet. 2002;11:3333–3344. doi: 10.1093/hmg/11.26.3333. [DOI] [PubMed] [Google Scholar]
- Suchyna TM, Johnson JH, Hamer K, Leykam JF, Gage DA, Clemo HF, Baumgarten CM, Sachs F. Identification of a peptide toxin from Grammostola spatulata spider venom that blocks cation-selective stretch-activated channels. J Gen Physiol. 2000;115:583–598. doi: 10.1085/jgp.115.5.583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suchyna TM, Tape SE, Koeppe RE, Andersen OS, Sachs F, Gottlieb PA. Bilayer-dependent inhibition of mechanosensitive channels by neuroactive peptide enantiomers. Nature. 2004;430:235–240. doi: 10.1038/nature02743. [DOI] [PubMed] [Google Scholar]
- Takekura H, Fujinami N, Nishizawa T, Ogasawara H, Kasuga N. Eccentric exercise-induced morphological changes in the membrane systems involved in excitation–contraction coupling in rat skeletal muscle. J Physiol. 2001;533:571–583. doi: 10.1111/j.1469-7793.2001.0571a.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsugorka A, Rios E, Blatter LA. Imaging elementary events of calcium release in skeletal muscle cells. Science. 1995;269:1723–1726. doi: 10.1126/science.7569901. [DOI] [PubMed] [Google Scholar]
- Turner PR, Westwood T, Regen CM, Steinhardt RA. Increased protein degradation results from elevated free calcium levels found in muscle from mdx mice. Nature. 1988;335:735–738. doi: 10.1038/335735a0. [DOI] [PubMed] [Google Scholar]
- Tutdibi O, Brinkmeier H, Rudel R, Fohr KJ. Increased calcium entry into dystrophin-deficient muscle fibres of MDX and ADR-MDX mice is reduced by ion channel blockers. J Physiol. 1999;515:859–868. doi: 10.1111/j.1469-7793.1999.859ab.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandebrouck C, Duport G, Cognard C, Raymond G. Cationic channels in normal and dystrophic human myotubes. Neuromuscul Disord. 2001;11:72–79. doi: 10.1016/s0960-8966(00)00153-x. [DOI] [PubMed] [Google Scholar]
- Vandebrouck C, Martin D, Colson-Van Schoor M, Debaix H, Gailly P. Involvement of TRPC in the abnormal calcium influx observed in dystrophic (mdx) mouse skeletal muscle fibers. J Cell Biol. 2002;158:1089–1096. doi: 10.1083/jcb.200203091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner KR, Hamed S, Hadley DW, Gropman AL, Burstein AH, Escolar DM, Hoffman EP, Fischbeck KH. Gentamicin treatment of Duchenne and Becker muscular dystrophy due to nonsense mutations. Ann Neurol. 2001;49:706–711. [PubMed] [Google Scholar]
- Warren GL, Ingalls CP, Lowe DA, Armstrong RB. Excitation-contraction uncoupling: major role in contraction-induced muscle injury. Exerc Sport Sci Rev. 2001;29:82–87. doi: 10.1097/00003677-200104000-00008. [DOI] [PubMed] [Google Scholar]
- Westerblad H, Lee JA, Lamb AG, Bolsover SR, Allen DG. Spatial gradients of intracellular calcium in skeletal muscle during fatigue. Pflugers Arch. 1990;415:734–740. doi: 10.1007/BF02584013. [DOI] [PubMed] [Google Scholar]
- Wilton SD, Dye DE, Blechynden LM, Laing NG. Revertant fibres: a possible genetic therapy for Duchenne muscular dystrophy? Neuromuscul Disord. 1997;7:329–335. doi: 10.1016/s0960-8966(97)00058-8. [DOI] [PubMed] [Google Scholar]
- Winegar BD, Haws CM, Lansman JB. Subconductance block of single mechanosensitive ion channels in skeletal muscle fibers by aminoglycoside antibiotics. J Gen Physiol. 1996;107:433–443. doi: 10.1085/jgp.107.3.433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeung EW, Ballard HJ, Bourreau JP, Allen DG. Intracellular sodium in mammalian muscle fibers after eccentric contractions. J App Physiol. 2003a;94:2475–2482. doi: 10.1152/japplphysiol.01128.2002. [DOI] [PubMed] [Google Scholar]
- Yeung EW, Balnave CD, Ballard HJ, Bourreau JP, Allen DG. Development of T-tubular vacuoles in eccentrically damaged mouse muscle fibres. J Physiol. 2002;540:581–592. doi: 10.1113/jphysiol.2001.013839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeung EW, Head SI, Allen DG. Gadolinium reduces short-term stretch-induced muscle damage in isolated mdx mouse muscle fibres. J Physiol. 2003b;552:449–458. doi: 10.1113/jphysiol.2003.047373. [DOI] [PMC free article] [PubMed] [Google Scholar]