Abstract
Vasoconstrictor responses to sympathetic nerve stimulation and their sensitivity to metabolic modulation reportedly differ in fast-twitch and slow-twitch muscles, but the underlying mechanisms are not known. Both α1- and α2-adrenoceptors mediate these vascular responses in fast-twitch muscle, while their roles in slow-twitch muscle are less well defined. In this study, the phosphorylation of smooth muscle myosin regulatory light chain (smRLC) was measured as an index of vasoconstriction in slow-twitch soleus muscles and fast-twitch extensor digitorum longus (EDL) muscles isolated from C57BL/6J mice. In soleus muscles, incubation with phenylephrine (PE) or UK 14,304 to selectively activate α1- or α2-adrenoceptors resulted in concentration-dependent increases in smRLC phosphorylation. To evaluate metabolic modulation of these responses, vasodilator pathways previously implicated in such modulation in fast-twitch muscle were activated in soleus muscles by treatment with the nitric oxide (NO) donor nitroprusside or the ATP-sensitive potassium (KATP) channel opener cromakalim. Both drugs inhibited responses to UK 14,304, but not to PE. The effect of nitroprusside to antagonize UK 14,304 responses was prevented by inhibition of guanylyl cyclase or by blockade of KATP channels, but not by blockade of other potassium channels. Results were similar in EDL muscles. These data provide the first evidence for α2-adrenoceptor-mediated constriction in slow-twitch muscle, and show that it is sensitive to modulation by NO via a cGMP-dependent mechanism that requires KATP channel activation. Based on the similar findings in soleus and EDL muscles, fibre type does not appear to determine the innate vascular response to α1- or α2-adrenoceptor activation.
Autonomic neural control of skeletal muscle blood flow is dominated by the sympathetic nerves, which densely innervate feed arteries and arterioles throughout the muscle microcirculation (Marshall, 1982; Fleming et al. 1989). Much of the current knowledge about sympathetic control is derived from studies of animal muscles composed predominantly of fast-twitch fibres, and of human muscles composed of a more uniform mixture of fast- and slow-twitch fibres. In these muscles, sympathetic vasoconstriction is mediated by both the α1- and α2-adrenoceptor subtypes (Marshall, 1982; Ohyanagi et al. 1991; Dinenno et al. 2002). Animal studies suggest that these receptors play distinct functional roles in the muscle microcirculation, with the α1-adrenoceptors mainly controlling the large resistance arterioles, and the α2-adrenoceptors controlling the small precapillary arterioles (Faber, 1988; Ohyanagi et al. 1991). The α2-adrenoceptors therefore may be optimally positioned to regulate capillary perfusion and distribute intramuscular blood flow according to the metabolic needs of the skeletal muscle cells.
In this regard, vasoconstriction mediated by α2-adrenoceptors appears to be particularly sensitive to inhibition by metabolic disturbances provoked, for example, by muscle hypoxia, ischaemia, or contraction (Anderson & Faber, 1991; McGillivray-Anderson & Faber, 1991; Thomas et al. 1994; Tateishi & Faber, 1995b; Buckwalter et al. 2001; Wray et al. 2004). We and others have reported that the blunted α-adrenergic vasoconstriction observed in these conditions is mediated in part by the activation of vasodilatory pathways involving either nitric oxide (NO) or the hyperpolarizing, metabolically regulated ATP-sensitive potassium (KATP) channels (Ohyanagi et al. 1992; Tateishi & Faber, 1995a; Thomas et al. 1997; Thomas & Victor, 1998; Buckwalter et al. 2004; Keller et al. 2004). A mechanistic link between these signalling pathways has been shown in studies of large conduit arteries in which NO and its downstream effector cGMP can activate KATP channels (Murphy & Brayden, 1995; Wu et al. 1999), although it is not clear if a similar interaction also occurs in the muscle microcirculation.
In contrast to the extensive characterization of α-adrenoceptor-mediated vasoconstriction and its modulation in fast-twitch muscle, relatively little is known about these responses in slow-twitch muscle. The limited data that have been reported suggest that α-adrenergic control of blood flow may differ substantially in muscles with different fibre type profiles. For example, the vasoconstrictor responses to sympathetic nerve activation are reduced in slow-twitch muscles compared to fast-twitch muscles at rest (Folkow & Halicka, 1968; Hilton et al. 1970; Gray, 1971), and may be less susceptible to attenuation in slow-twitch muscles than in fast-twitch muscles during contraction (Thomas et al. 1994). The reasons for these differential responses in slow-twitch and fast-twitch muscles are not known, but could potentially be mediated by differences in the vasoconstrictor responses evoked by either α1- or α2-adrenoceptors, and/or by differences in the susceptibility of these vasoconstrictor responses to metabolic modulation. In particular, a large gap in our understanding of vascular regulation in slow twitch muscle stems from the current lack of information about the role of the α2-adrenoceptors.
Therefore, our primary goal in this study was to evaluate α1- and α2-adrenoceptor-mediated vasoconstrictor responses and their modulation in the microcirculation of muscles composed predominantly of slow-twitch or fast-twitch fibres. To avoid the potentially confounding effects of differences in basal blood flow or sympathetic neural tone in muscles with different fibre type profiles, we performed ex vivo studies using slow-twitch soleus muscles and fast-twitch extensor digitorum longus (EDL) muscles isolated from the mouse hindlimb. To evaluate in situ microvascular responses to direct activation of α1- or α2-adrenoceptors, we measured the phosphorylation of smooth muscle myosin regulatory light chain (smRLC), which is a key biochemical event mediating the activation of myosin ATPase and smooth muscle contraction (Kamm & Stull, 1985; Stull et al. 1991; Somlyo & Somlyo, 1994). Finally, to evaluate the susceptibility of α-adrenoceptor responses to metabolic modulation, we exposed muscles to an NO donor or KATP channel agonist to activate specific vasodilator pathways that have previously been implicated in such modulation.
Methods
All of the procedures used in this study were approved by the Institutional Animal Care and Use Committee at the University of Texas Southwestern Medical Center at Dallas.
Experimental protocol
Male and female C57BL/6J mice (The Jackson Laboratory) aged 12–16 weeks were killed with sodium pentobarbital (250 mg kg−1, i.p.). The EDL (mass, 7–14 mg) and soleus (mass, 8–16 mg) muscles were isolated and removed from each hindlimb. Each muscle was secured at one end to an isometric force transducer and at the other end to a fixed support in a tissue bath containing oxygenated (95% O2–5% CO2) physiological saline solution (PSS; pH 7.6, 30°C) composed of (mm) 120.5 NaCl, 4.8 KCl, 1.2 MgSO4, 20.4 NaHCO3, 1.6 CaCl2, 1.2 NaH2PO4, 10 glucose and 1 pyruvate. Muscle tension was adjusted to maintain 1 g of resting tension. Muscles were equilibrated for 30–45 min prior to any drug treatment.
Muscles were treated for 60 s with the α1-adrenoceptor agonist phenylephrine (PE, 1 or 10 μm) or the α2-adrenoceptor agonist UK 14,304 (2 or 20 μm). Preliminary experiments indicated that a 60 s exposure produced maximal smRLC phosphorylation (data not shown). In some experiments, the NO donor sodium nitroprusside (10 nm) or the KATP channel opener cromakalim (7 μm) were added during the last 30 s of the α-adrenoceptor agonist exposure. In other experiments, muscles were pretreated for 20 min with antagonists to KATP channels (glibenclamide, 10 μm), voltage-dependent K+ channels (Kv; 4-aminopyridine, 5 mm), large conductance Ca2+-activated K+ channels (KCa; iberiotoxin, 100 nm), small conductance KCa channels (apamin, 100 nm), or soluble guanylyl cyclase (1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one, ODQ; 50 μm). Drug concentrations were based on previously published data (Ohyanagi et al. 1992; Tateishi & Faber, 1995a) and preliminary experiments from our laboratory. Drugs were added directly to the tissue baths to achieve the given final concentrations. At the end of each experiment, muscles were snap-frozen by tongs prechilled in liquid nitrogen and stored at −80°C for subsequent analysis.
Measurement of smRLC phosphate content
Measurements were performed as previously described (Persechini et al. 1986; Grange et al. 2001). Briefly, frozen muscles were weighed and then homogenized in 500 μl ice-cold 10% trichloroacetic acid (TCA)–acetone solution containing 1 mm dithiothreitol (DTT). After centrifugation at 735 g for 2 min, pellets were washed three times with 500 μl ethyl ether, air-dried for 20 min, and resuspended in 300 μl of an 8 m urea sample buffer containing (mm): 18.5 Tris, 20.4 glycine, 9.2 DTT, and 4.6 EDTA, pH 8.6. Samples were saturated with urea crystals and shaken for 1 h at room temperature to ensure complete protein solubilization.
Extraction of smRLC was performed by adding ice-cold 95% ethanol drop-wise to 25% of the final volume. Samples were incubated on ice for 20 min and centrifuged at 4000 g for 7 min. An equal volume of 20% TCA–2 mm DTT–water was added to the supernatant fraction and samples were incubated on ice for 20 min. After centrifugation at 4000 g for 10 min, pellets were resuspended in 100 μl of 8 m urea sample buffer (minus Tris, plus saturated sucrose and 0.004% bromophenol blue). Samples were solubilized by saturation with urea crystals and pH was maintained by adding 2.5 m Tris base (pH 11.0). Samples were shaken vigorously for 1 h at room temperature and stored at −80°C.
Samples (20 μl) were loaded onto a 10% polyacrylamide gel and electrophoresed at 400 V for 60 min using a running buffer of 214 mm Tris and 266 mm glycine and an identical cathode buffer supplemented with 2 mm thioglycolate and 2 mm DTT. Proteins were transferred to polyvinylidene fluoride (PVDF) membranes (Millipore) at 25 V for 60 min. Membranes were washed briefly in 100% methanol, air-dried for ∼20 min, and fixed in 0.4% gluteraldehyde for 30 min. Membranes were then blocked in 5% Amersham Liquid Block for 60 min at room temperature, and incubated overnight at 4°C with a primary monoclonal antibody (ascites diluted 1: 10 000; provided by Kathy Trybus, University of Vermont, Burlington, VT) in 0.5% Amersham Block–PBS that recognizes smooth muscle, but not skeletal muscle, myosin RLC (Lau et al. 1998). Membranes were washed and incubated for 60 min at room temperature with goat antimouse IgG(H + l) conjugated with alkaline phosphatase (1: 10 000; Southern Biotechnology Associates). Phosphorylated and non-phosphorylated smRLC bands were visualized by chemiluminescence (CSPD substrate and Sapphire enhancer, Tropix) according to the manufacturer's instructions. The ratio of phosphorylated smRLC to total smRLC was determined by densitometry (Molecular Analyst, Bio-Rad) and reported as moles Pi per mole of smRLC.
Chemicals
UK 14,304 and glibenclamide were purchased from Research Biochemicals International. All other chemicals were purchased from Sigma. Stock solutions of phenylephrine, nitroprusside, prazosin, yohimbine, 4-aminopyridine, iberiotoxin and apamin were dissolved in dH2O. Stock solutions of UK 14,304, cromakalim, glibenclamide and ODQ were dissolved in 40% DMSO. Final tissue bath concentrations of DMSO were less than 0.1%.
Statistical analysis
Differences in smRLC phosphate content among treatments were analysed using a one-way ANOVA and Scheffé's post hoc tests. P values < 0.05 were considered statistically significant. Data are expressed as means ± s.e.m.
Results
Direct activation of α2- or α1-adrenoceptors increases smRLC phosphorylation in soleus and EDL
To evaluate vasoconstrictor responses to α-adrenoceptor activation, the ratio of phosphorylated smRLC to total smRLC was measured in extracts of mouse soleus and EDL muscles as illustrated in Fig. 1. As shown in Fig. 2, treating muscles for 60 s with the α2-adrenoceptor agonist UK 14,304 elicited concentration-dependent increases in smRLC phosphorylation. The ratio of phosphorylated smRLC to total smRLC increased from a basal level of 0.16 ± 0.03 in soleus muscles to a maximum of 0.47 ± 0.03, and from 0.15 ± 0.03 in EDL muscles to a maximum of 0.41 ± 0.05. Likewise, treatment with the α1-adrenoceptor agonist phenylephrine increased smRLC phosphorylation from 0.11 ± 0.03 in soleus muscles to a maximum of 0.45 ± 0.06, and from 0.13 ± 0.03 in EDL muscles to a maximum of 0.43 ± 0.12. In preliminary experiments, responses to UK 14,304 or phenylephrine were prevented by pretreatment with the α2-adrenoceptor antagonist yohimbine or the α1-adrenoceptor antagonist prazosin, respectively (data not shown).
Figure 1. Regulation of vascular smooth muscle contraction by myosin phosphorylation.
Activation of α-adrenoceptors (α-AR) increases intracellular Ca2+, which binds to calmodulin and activates myosin light chain (MLC) kinase, resulting in phosphorylation of the 20 kDa regulatory light chains of myosin and smooth muscle contraction. G, heterotrimeric G protein.
Figure 2. Effects of α2- and α1-adrenoceptor agonists on smooth muscle myosin regulatory light chain (smRLC) phosphorylation in slow-twitch soleus and fast-twitch EDL muscles.
A, representative Western blots of skeletal muscle extracts showing clear separation between phosphorylated smRLC (lower bands) and non-phosphorylated smRLC (upper bands) in response to the α2-adrenoceptor agonist UK 14,304 and the α1-adrenoceptor agonist phenylephrine. B, results of quantitative densitometry of Western blots showing that both UK 14,304 (black bars) and phenylephrine (grey bars) increased smRLC phosphorylation in a concentration-dependent manner in soleus and EDL muscles. *P < 0.05 versus 0 μm drug; n≥ 4 muscles per concentration.
Preferential inhibition of α2-adrenoceptor-mediated smRLC phosphorylation by nitroprusside or cromakalim in soleus and EDL
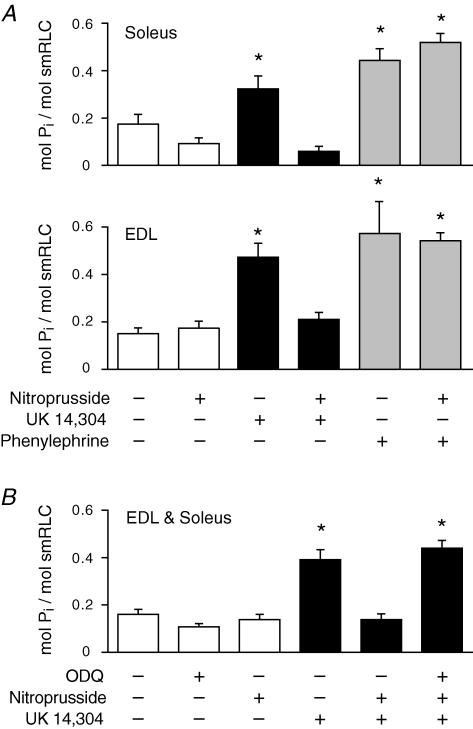
A concentration of nitroprusside (10 nm) that would be expected to yield NO in the low physiological range had no significant effect on basal phosphorylation of smRLC in soleus or EDL muscles (Fig. 3). Nitroprusside inhibited the increases in smRLC phosphorylation in response to 20 μm UK 14,304 in both soleus and EDL muscles, but it had no effect on the increases in smRLC phosphorylation induced by 10 μm phenylephrine in either type of muscle. The inhibitory effect of nitroprusside on UK 14,304-mediated smRLC phosphorylation was prevented by treating muscles with ODQ, an inhibitor of soluble guanylyl cyclase (Fig. 3).
Figure 3. Differential effect of the NO donor nitroprusside to inhibit the smRLC phosphorylation response to activation of α2- and α1-adrenoceptors.
A, nitroprusside inhibited the increases in smRLC phosphorylation induced by UK 14,304 (20 μm), but not by phenylephrine (10 μm). Results were similar for soleus and EDL muscles. *P < 0.05 versus no drug; n≥ 4 muscles per treatment. B, effect of the soluble guanylyl cyclase inhibitor ODQ to prevent the inhibitory action of nitroprusside on UK 14,304-induced smRLC phosphorylation. Results were not significantly different in soleus and EDL muscles. Pooled data are shown. *P < 0.05 versus no drug; n≥ 10 muscles per treatment.
The KATP channel opener cromakalim had no significant effect on basal phosphorylation of smRLC in resting soleus or EDL muscles. The increases in smRLC phosphorylation in response to UK 14,304, but not phenylephrine, were inhibited by cromakalim in both soleus and EDL muscles (Fig. 4).
Figure 4. Differential effect of the KATP channel opener cromakalim to inhibit the smRLC phosphorylation response to activation of α2- and α1-adrenoceptors.
Cromakalim inhibited the increases in smRLC phosphorylation induced by UK 14,304 (20 μm), but not by phenylephrine (10 μm). Results were similar for soleus and EDL muscles. *P < 0.05 versus no drug; n≥ 4 muscles per treatment.
The effect of nitroprusside to inhibit α2-adrenoceptor-mediated smRLC phosphorylation requires KATP channels, but not Kv or KCa channels
Increases in smRLC phosphorylation in response to UK 14,304 were not affected by pretreatment of soleus or EDL muscles with glibenclamide (Fig. 5). As expected, glibenclamide prevented the inhibitory effect of cromakalim on UK 14,304-induced increases in smRLC phosphorylation. Glibenclamide also prevented the inhibitory effect of nitroprusside on UK 14,304-induced increases in smRLC phosphorylation (Fig. 5).
Figure 5. Effect of pretreatment with the KATP channel blocker glibenclamide to block the inhibitory action of either cromakalim or nitroprusside on α2-adrenoceptor-induced smRLC phosphorylation.
Glibenclamide prevented the inhibitory action of either cromakalim or nitroprusside on UK 14,304-induced smRLC phosphorylation. Results were similar for soleus and EDL muscles. *P < 0.05 versus glibenclamide alone; n≥ 4 muscles per treatment.
In contrast to the effect of glibenclamide, blockade of Kv channels with 4-aminopyridine, large conductance KCa channels with iberiotoxin, or small conductance KCa channels with apamin, did not alter the ability of nitroprusside to inhibit UK 14,304-induced increases in smRLC phosphorylation (Fig. 6). Pooled data from soleus and EDL muscles are shown in Fig. 6 as the responses in the two types of muscle were not statistically different for any of these treatments.
Figure 6. Effects of pretreatment with K+ channel antagonists on the inhibitory interaction between nitroprusside and α2-adrenoceptor-induced smRLC phosphorylation.
The effect of nitroprusside to inhibit UK 14,304-induced increases in smRLC phosphorylation was prevented by the KATP channel blocker glibenclamide, but not by the Kv channel blocker iberiotoxin, the large conductance KCa channel blocker 4-aminopyridine, or the small conductance KCa channel blocker apamin. Results were not significantly different in soleus and EDL muscles. Pooled data are shown. *P < 0.05 versus K+ channel blocker alone; n≥ 4 muscles per treatment.
Discussion
Although α-adrenergic control of the microcirculation has been fairly well characterized in fast-twitch muscle (Faber, 1988; Anderson & Faber, 1991; McGillivray-Anderson & Faber, 1991; Ohyanagi et al. 1991, 1992; Tateishi & Faber, 1995a, b), much less is known about such regulation in slow-twitch muscle. In this study, we were particularly interested in probing the role of α2-adrenoceptors, which had not previously been examined in slow-twitch muscle. Using changes in smooth muscle myosin phosphorylation to evaluate in situ vascular responses in mouse hindlimb muscles, we now provide novel functional evidence for α2-adrenoceptors in the microcirculation of slow-twitch muscle. We also show that the vascular responses mediated by these receptors are sensitive to inhibition by a low physiological concentration of NO acting selectively via KATP channels, demonstrating the potential for metabolic modulation of α2-adrenergic vasoconstriction in slow-twitch muscle.
Our finding that the increases in smRLC phosphorylation evoked by either the α2-adrenoceptor agonist UK 14,304 or the α1-adrenoceptor agonist phenylephrine were similar in soleus and EDL muscles suggests that fibre type composition is not a principal determinant of the vascular response to α-adrenoceptor activation. In a recent in vitro study of second-order resistance arterioles isolated from rat skeletal muscles with varying proportions of fast-twitch and slow-twitch fibres, Aaker & Laughlin reached the same conclusion regarding α1-adrenergic vasoconstriction (Aaker & Laughlin, 2002). However, in that study α2-adrenergic vasoconstriction could not be evaluated as the vessels did not respond to UK 14,304, either because α2-adrenoceptors are of less functional importance in the control of the large second-order arterioles (Faber, 1988) or because their sensitivity is selectively reduced after in vitro isolation (Ikeoka & Faber, 1993). An advantage of our experimental preparation is that the muscle microvascular network remained intact in its native environment and we were therefore able to observe α2-adrenoceptor responses. It remains to be seen if the spatial distribution of the α-adrenoceptors in the microcirculation of slow-twitch muscle is the same as that of fast-twitch muscle, where both α1- and α2-adrenoceptors control the large proximal arterioles and large venules, while the α2-adrenoceptors control the small distal arterioles (Faber, 1988).
The outcome of these ex vivo studies indicating a lack of association between α-adrenergic vasoconstrictor responses and muscle fibre type composition would appear to conflict with previous in vivo studies in which sympathetic vasoconstrictor responses were blunted in quiescent slow-twitch versus fast-twitch muscles (Folkow & Halicka, 1968; Hilton et al. 1970; Gray, 1971). Based on our data, we suggest that the results of the in vivo studies cannot be explained by a reduced intrinsic efficacy of either α1- or α2-adrenoceptors in slow-twitch muscle. Other factors that might contribute to this differential sympathetic responsiveness in slow-twitch and fast-twitch muscles but have yet to be explored include variations in sympathetic innervation or in α-adrenoceptor densities or reserves.
Another important aspect of our study was to evaluate the sensitivity of α-adrenoceptor-mediated constrictor responses to metabolic modulation in muscles with different fibre type compositions. To do so, we used drugs to directly activate specific vasodilator pathways that have been implicated in the metabolic attenuation of α-adrenergic vasoconstriction during hypoxia or muscle contraction. This ensured that soleus and EDL muscles were exposed to equivalent vasodilator species and concentrations, which would not have been the case if hypoxia or muscle contraction had been used to stimulate the production of endogenous vasodilator substances given the dissimilar metabolic profiles of slow-twitch and fast-twitch muscles.
Using this strategy, we found that the increase in smRLC phosphorylation in response to α2-, but not α1-, adrenoceptor activation was highly susceptible to modulation by the NO donor nitroprusside and the KATP channel opener cromakalim in soleus muscle. These results provided the first evidence that α2-adrenergic vasoconstriction in slow-twitch muscle is potentially subject to metabolic modulation. Similar results were obtained for the EDL muscle, suggesting that fibre type composition does not influence the intrinsic sensitivity of α-adrenergic vasoconstriction to such modulation. These results also confirm at the biochemical level previous studies showing metabolic inhibition of α2-adrenergic vasoconstriction in fast-twitch or mixed skeletal muscles using conventional measurements of blood flow or blood vessel diameter (Anderson & Faber, 1991; McGillivray-Anderson & Faber, 1991; Ohyanagi et al. 1992; Thomas et al. 1994; Tateishi & Faber, 1995a, b; Buckwalter et al. 2001; Rosenmeier et al. 2003; Wray et al. 2004).
These new data would appear to differ from the results of our previous in vivo study in which we reported that sympathetic vasoconstriction was attenuated in the rat hindlimb during contractions of the fast-twitch gastrocnemius, but not slow-twitch soleus, muscles (Thomas et al. 1994). In the present study, the effects of well-defined, uniform vasoconstrictor (α-adrenoceptor agonists) and vasodilator (NO, KATP channels) stimuli were evaluated in individual muscles. In contrast, in the previous study, the effects of relatively less specific constrictor (sympathetic nerve stimulation) and dilator (muscle contraction) stimuli were evaluated in the intact hindlimb. Having identified a specific set of conditions in which α2-adrenergic vasoconstriction can be modulated in the isolated soleus muscle, a future challenge is to determine if the findings from this reductionist ex vivo preparation are applicable to vascular regulation in slow-twitch muscles in vivo.
Use of this reductionist preparation also allowed us to probe the mechanism by which NO attenuates α2-adrenergic vasoconstriction in the skeletal muscle microcirculation. In previous in vivo studies in rats, we reported that the effect of hindlimb contraction to attenuate sympathetic vasoconstriction was equally impaired by solo or combined inhibition of NO synthase activity or KATP channel activity (Thomas et al. 1997; Thomas & Victor, 1998). Although those data suggested a potential interaction between NO and KATP channels, we could not definitively exclude a role for other K channels because of concerns about the specificity of the high systemic dose of the KATP channel blocker used in that study. In the present study, we overcame that limitation by using selective concentrations of inhibitors of the major classes of K channels that have been implicated in vascular control. These new data show that the effect of the NO donor nitroprusside to antagonize α2-adrenergic vasoconstriction was mediated specifically by activation of KATP channels, but not by Kv or KCa channels. This interaction might involve cGMP-mediated activation of the KATP channel, as has been reported in earlier studies using conduit blood vessels (Murphy & Brayden, 1995; Wu et al. 1999).
In contrast to the results of our study, nitroprusside has previously been shown to attenuate α1-adrenoceptor-mediated increases in smRLC phosphorylation in the fast-twitch EDL muscle (Grange et al. 2001). These disparate findings are probably due to the 1000-fold difference in the concentration of nitroprusside used in these studies (10 nmversus 10 μm), and most likely reflect concentration-dependent effects of NO to activate multiple cellular signalling pathways leading to vasorelaxation. In addition to activating KATP channels (Murphy & Brayden, 1995; Wu et al. 1999), NO is reported to activate KCa channels (Robertson et al. 1993; Bolotina et al. 1994), reduce Ca2+ influx (Blatter & Wier, 1994), reduce the Ca2+ sensitivity of the contractile proteins (Sauzeau et al. 2000), and enhance cytosolic removal of Ca2+ by Ca2+-ATPase (Cornwell et al. 1991).
In general, our data indicate that smRLC phosphorylation is a sensitive biological endpoint that can be used to study α-adrenergic regulation of the muscle microcirculation. Because smRLC phosphorylation reflects the activity of both myosin light chain kinase (MLCK) and phosphatase (MLCP) (Kamm & Stull, 1985; Kimura et al. 1996; Pfitzer, 2001; Somlyo & Somlyo, 2003), additional studies are needed to determine how the vasoconstrictor and vasodilator stimuli used in our study interact with these regulatory pathways. For example, ligand binding to α-adrenoceptors increases cytosolic Ca2+ (Guimaraes & Moura, 2001) which activates the Ca2+–calmodulin-dependent MLCK (Kamm & Stull, 1985), while NO and KATP channels have the opposite effect of reducing cytosolic Ca2+ (Brayden, 2002; Schlossmann et al. 2003) which inhibits MLCK. Furthermore, α-adrenoceptor activation stimulates the GTPase Rho and its effector Rho kinase (Carter et al. 2002; Crowley et al. 2002; Damron et al. 2002) which inhibits MLCP (Kimura et al. 1996), while NO has the opposite effect of inhibiting Rho/Rho kinase and activating MLCP (Sauzeau et al. 2000; Etter et al. 2001). Inhibition of Rho kinase has recently been shown to attenuate α2-adrenergic vasoconstriction (Carter et al. 2002), raising the possibility that reliance on this pathway could contribute to the enhanced susceptibility of α2-adrenoceptor responses to inhibition by NO.
In summary, the new findings of this study are that both α1- and α2-adrenoceptors can evoke constrictor responses in the microcirculation of slow-twitch muscle, and that the α2-adrenoceptor responses are particularly sensitive to inhibition by NO via a cGMP-dependent mechanism that requires KATP channel activation. Similar findings in fast-twitch muscle confirmed previous studies (Faber, 1988; Ohyanagi et al. 1992; Tateishi & Faber, 1995a; Thomas et al. 1997; Thomas & Victor, 1998), and suggested that skeletal muscle fibre type composition is not a determinant of the innate vascular responsiveness to α-adrenoceptor activation. Based on these findings, we speculate that previously reported differences in vascular regulation in fast-twitch and slow-twitch muscles (Folkow & Halicka, 1968; Hilton et al. 1970; Gray, 1971; Thomas et al. 1994) are probably mediated by mechanisms upstream of the α-adrenoceptors. These could include differences in the vascular innervation or activity of sympathetic nerves, in the densities or functional reserves of vascular α-adrenoceptors, or in the local production and accumulation of muscle metabolites.
Acknowledgments
We thank Drs James Stull and Robert Grange for their assistance with the smRLC phosphorylation measurements. This work was supported by NIH grants HL06296 and AR051034.
References
- Aaker A, Laughlin MH. Diaphragm arterioles are less responsive to α1-adrenergic constriction than gastrocnemius arterioles. J Appl Physiol. 2002;92:1808–1816. doi: 10.1152/japplphysiol.01152.2001. [DOI] [PubMed] [Google Scholar]
- Anderson KM, Faber JE. Differential sensitivity of arteriolar α1- and α2-adrenoceptor constriction to metabolic inhibition during rat skeletal muscle contraction. Circ Res. 1991;69:174–184. doi: 10.1161/01.res.69.1.174. [DOI] [PubMed] [Google Scholar]
- Blatter LA, Wier WG. Nitric oxide decreases [Ca2+]i in vascular smooth muscle by inhibition of the calcium current. Cell Calcium. 1994;15:122–131. doi: 10.1016/0143-4160(94)90051-5. [DOI] [PubMed] [Google Scholar]
- Bolotina VM, Najibi S, Palacino JJ, Pagano PJ, Cohen RA. Nitric oxide directly activates calcium-dependent potassium channels in vascular smooth muscle. Nature. 1994;368:850–853. doi: 10.1038/368850a0. [DOI] [PubMed] [Google Scholar]
- Brayden JE. Functional roles of KATP channels in vascular smooth muscle. Clin Exp Pharmacol Physiol. 2002;29:312–316. doi: 10.1046/j.1440-1681.2002.03650.x. [DOI] [PubMed] [Google Scholar]
- Buckwalter JB, Naik JS, Valic Z, Clifford PS. Exercise attenuates α-adrenergic-receptor responsiveness in skeletal muscle vasculature. J Appl Physiol. 2001;90:172–178. doi: 10.1152/jappl.2001.90.1.172. [DOI] [PubMed] [Google Scholar]
- Buckwalter JB, Taylor JC, Hamann JJ, Clifford PS. Role of nitric oxide in exercise sympatholysis. J Appl Physiol. 2004;97:417–423. doi: 10.1152/japplphysiol.01181.2003. Discussion 416. [DOI] [PubMed] [Google Scholar]
- Carter RW, Begaye M, Kanagy NL. Acute and chronic NOS inhibition enhances α2-adrenoreceptor-stimulated RhoA and Rho kinase in rat aorta. Am J Physiol Heart Circ Physiol. 2002;283:H1361–H1369. doi: 10.1152/ajpheart.01101.2001. [DOI] [PubMed] [Google Scholar]
- Cornwell TL, Pryzwansky KB, Wyatt TA, Lincoln TM. Regulation of sarcoplasmic reticulum protein phosphorylation by localized cyclic GMP-dependent protein kinase in vascular smooth muscle cells. Mol Pharmacol. 1991;40:923–931. [PubMed] [Google Scholar]
- Crowley CM, Lee CH, Gin SA, Keep AM, Cook RC, Van Breemen C. The mechanism of excitation-contraction coupling in phenylephrine-stimulated human saphenous vein. Am J Physiol Heart Circ Physiol. 2002;283:H1271–H1281. doi: 10.1152/ajpheart.01129.2001. [DOI] [PubMed] [Google Scholar]
- Damron DS, Kanaya N, Homma Y, Kim SO, Murray PA. Role of PKC, tyrosine kinases, and Rho kinase in alpha-adrenoreceptor-mediated PASM contraction. Am J Physiol Lung Cell Mol Physiol. 2002;283:L1051–L1064. doi: 10.1152/ajplung.00345.2001. [DOI] [PubMed] [Google Scholar]
- Dinenno FA, Eisenach JH, Dietz NM, Joyner MJ. Post-junctional α-adrenoceptors and basal limb vascular tone in healthy men. J Physiol. 2002;540:1103–1110. doi: 10.1113/jphysiol.2001.015297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etter EF, Eto M, Wardle RL, Brautigan DL, Murphy RA. Activation of myosin light chain phosphatase in intact arterial smooth muscle during nitric oxide-induced relaxation. J Biol Chem. 2001;276:34681–34685. doi: 10.1074/jbc.M104737200. 10.1074/jbc.M104737200. [DOI] [PubMed] [Google Scholar]
- Faber JE. In situ analysis of α-adrenoceptors on arteriolar and venular smooth muscle in rat skeletal muscle microcirculation. Circ Res. 1988;62:37–50. doi: 10.1161/01.res.62.1.37. [DOI] [PubMed] [Google Scholar]
- Fleming BP, Gibbins IL, Morris JL, Gannon BJ. Noradrenergic and peptidergic innervation of the extrinsic vessels and microcirculation of the rat cremaster muscle. Microvasc Res. 1989;38:255–268. doi: 10.1016/0026-2862(89)90004-6. 10.1016/0026-2862(89)90004-6. [DOI] [PubMed] [Google Scholar]
- Folkow B, Halicka HD. A comparison between ‘red’ and ‘white’ muscle with respect to blood supply, capillary surface area and oxygen uptake during rest and exercise. Microvasc Res. 1968;1:1–14. 10.1016/0026-2862(68)90002-2. [Google Scholar]
- Grange RW, Isotani E, Lau KS, Kamm KE, Huang PL, Stull JT. Nitric oxide contributes to vascular smooth muscle relaxation in contracting fast-twitch muscles. Physiol Genomics. 2001;5:35–44. doi: 10.1152/physiolgenomics.2001.5.1.35. [DOI] [PubMed] [Google Scholar]
- Gray SD. Responsiveness of the terminal vascular bed in fast and slow skeletal muscles to α-adrenergic stimulation. Angiologica. 1971;8:285–296. doi: 10.1159/000157902. [DOI] [PubMed] [Google Scholar]
- Guimaraes S, Moura D. Vascular adrenoceptors: an update. Pharmacol Rev. 2001;53:319–356. [PubMed] [Google Scholar]
- Hilton SM, Jeffries MG, Vrbova G. Functional specializations of the vascular bed of soleus. J Physiol. 1970;206:543–562. doi: 10.1113/jphysiol.1970.sp009030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeoka K, Faber JE. ANG II reverses selective inhibition of α2-adrenoceptor sensitivity after in vitro isolation of arterioles. Am J Physiol. 1993;265:H1988–H1995. doi: 10.1152/ajpheart.1993.265.6.H1988. [DOI] [PubMed] [Google Scholar]
- Kamm KE, Stull JT. The function of myosin and myosin light chain kinase phosphorylation in smooth muscle. Annu Rev Pharmacol Toxicol. 1985;25:593–620. doi: 10.1146/annurev.pa.25.040185.003113. 10.1146/annurev.pa.25.040185.003113. [DOI] [PubMed] [Google Scholar]
- Keller DM, Ogoh S, Greene S, Olivencia-Yurvati A, Raven PB. Inhibition of KATP channel activity augments baroreflex-mediated vasoconstriction in exercising human skeletal muscle. J Physiol. 2004;561:273–282. doi: 10.1113/jphysiol.2004.071993. 10.1113/jphysiol.2004.071993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura K, Ito M, Amano M, Chihara K, Fukata Y, Nakafuku M, Yamamori B, Feng J, Nakano T, Okawa K, Iwamatsu A, Kaibuchi K. Regulation of myosin phosphatase by Rho and Rho-associated kinase (Rho-kinase) Science. 1996;273:245–248. doi: 10.1126/science.273.5272.245. [DOI] [PubMed] [Google Scholar]
- Lau KS, Grange RW, Chang WJ, Kamm KE, Sarelius I, Stull JT. Skeletal muscle contractions stimulate cGMP formation and attenuate vascular smooth muscle myosin phosphorylation via nitric oxide. FEBS Lett. 1998;431:71–74. doi: 10.1016/s0014-5793(98)00728-5. 10.1016/S0014-5793(98)00728-5. [DOI] [PubMed] [Google Scholar]
- McGillivray-Anderson KM, Faber JE. Effect of reduced blood flow on α1- and α2-adrenoceptor constriction of rat skeletal muscle microvessels. Circ Res. 1991;69:165–173. doi: 10.1161/01.res.69.1.165. [DOI] [PubMed] [Google Scholar]
- Marshall JM. The influence of the sympathetic nervous system on individual vessels of the microcirculation of skeletal muscle of the rat. J Physiol. 1982;332:169–186. doi: 10.1113/jphysiol.1982.sp014408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy ME, Brayden JE. Nitric oxide hyperpolarizes rabbit mesenteric arteries via ATP-sensitive potassium channels. J Physiol. 1995;486:47–58. doi: 10.1113/jphysiol.1995.sp020789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohyanagi M, Faber JE, Nishigaki K. Differential activation of α1- and α2-adrenoceptors on microvascular smooth muscle during sympathetic nerve stimulation. Circ Res. 1991;68:232–244. doi: 10.1161/01.res.68.1.232. [DOI] [PubMed] [Google Scholar]
- Ohyanagi M, Nishigaki K, Faber JE. Interaction between microvascular α1- and α2-adrenoceptors and endothelium-derived relaxing factor. Circ Res. 1992;71:188–200. doi: 10.1161/01.res.71.1.188. [DOI] [PubMed] [Google Scholar]
- Persechini A, Kamm KE, Stull JT. Different phosphorylated forms of myosin in contracting tracheal smooth muscle. J Biol Chem. 1986;261:6293–6299. [PubMed] [Google Scholar]
- Pfitzer G. Invited review: regulation of myosin phosphorylation in smooth muscle. J Appl Physiol. 2001;91:497–503. doi: 10.1152/jappl.2001.91.1.497. [DOI] [PubMed] [Google Scholar]
- Robertson BE, Schubert R, Hescheler J, Nelson MT. cGMP-dependent protein kinase activates Ca-activated K channels in cerebral artery smooth muscle cells. Am J Physiol. 1993;265:C299–C303. doi: 10.1152/ajpcell.1993.265.1.C299. [DOI] [PubMed] [Google Scholar]
- Rosenmeier JB, Dinenno FA, Fritzlar SJ, Joyner MJ. α1- and α2-adrenergic vasoconstriction is blunted in contracting human muscle. J Physiol. 2003;547:971–976. doi: 10.1113/jphysiol.2002.037937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauzeau V, Le Jeune H, Cario-Toumaniantz C, Smolenski A, Lohmann SM, Bertoglio J, Chardin P, Pacaud P, Loirand G. Cyclic GMP-dependent protein kinase signaling pathway inhibits RhoA-induced Ca2+ sensitization of contraction in vascular smooth muscle. J Biol Chem. 2000;275:21722–21729. doi: 10.1074/jbc.M000753200. 10.1074/jbc.M000753200. [DOI] [PubMed] [Google Scholar]
- Schlossmann J, Feil R, Hofmann F. Signaling through NO and cGMP-dependent protein kinases. Ann Med. 2003;35:21–27. doi: 10.1080/07853890310004093. 10.1080/07853890310004093. [DOI] [PubMed] [Google Scholar]
- Somlyo AP, Somlyo AV. Signal transduction and regulation in smooth muscle. Nature. 1994;372:231–236. doi: 10.1038/372231a0. 10.1038/372231a0. [DOI] [PubMed] [Google Scholar]
- Somlyo AP, Somlyo AV. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev. 2003;83:1325–1358. doi: 10.1152/physrev.00023.2003. [DOI] [PubMed] [Google Scholar]
- Stull JT, Gallagher PJ, Herring BP, Kamm KE. Vascular smooth muscle contractile elements. Cellular regulation. Hypertension. 1991;17:723–732. doi: 10.1161/01.hyp.17.6.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tateishi J, Faber JE. ATP-sensitive K+ channels mediate α2D-adrenergic receptor contraction of arteriolar smooth muscle and reversal of contraction by hypoxia. Circ Res. 1995a;76:53–63. doi: 10.1161/01.res.76.1.53. [DOI] [PubMed] [Google Scholar]
- Tateishi J, Faber JE. Inhibition of arteriole α2- but not α1-adrenoceptor constriction by acidosis and hypoxia in vitro. Am J Physiol. 1995b;268:H2068–H2076. doi: 10.1152/ajpheart.1995.268.5.H2068. [DOI] [PubMed] [Google Scholar]
- Thomas GD, Hansen J, Victor RG. Inhibition of α2-adrenergic vasoconstriction during contraction of glycolytic, not oxidative, rat hindlimb muscle. Am J Physiol. 1994;266:H920–H929. doi: 10.1152/ajpheart.1994.266.3.H920. [DOI] [PubMed] [Google Scholar]
- Thomas GD, Hansen J, Victor RG. ATP-sensitive potassium channels mediate contraction-induced attenuation of sympathetic vasoconstriction in rat skeletal muscle. J Clin Invest. 1997;99:2602–2609. doi: 10.1172/JCI119448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas GD, Victor RG. Nitric oxide mediates contraction-induced attenuation of sympathetic vasoconstriction in rat skeletal muscle. J Physiol. 1998;506:817–826. doi: 10.1111/j.1469-7793.1998.817bv.x. 10.1111/j.1469-7793.1998.817bv.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray DW, Fadel PJ, Smith ML, Raven P, Sander M. Inhibition of α-adrenergic vasoconstriction in exercising human thigh muscles. J Physiol. 2004;555:545–563. doi: 10.1113/jphysiol.2003.054650. 10.1113/jphysiol.2003.054650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CC, Chen SJ, Yen MH. Cyclic GMP regulates cromakalim-induced relaxation in the rat aortic smooth muscle: role of cyclic GMP in KATP-channels. Life Sci. 1999;64:2471–2478. doi: 10.1016/s0024-3205(99)00204-0. 10.1016/S0024-3205(99)00204-0. [DOI] [PubMed] [Google Scholar]