Abstract
Artemisia annua plants produce a broad range of volatile compounds, including monoterpenes, which contribute to the characteristic fragrance of this medicinal species. A cDNA clone, QH6, contained an open reading frame encoding a 582-amino acid protein that showed high sequence identity to plant monoterpene synthases. The prokaryotically expressed QH6 fusion protein converted geranyl diphosphate to (−)-β-pinene and (−)-α-pinene in a 94:6 ratio. QH6 was predominantly expressed in juvenile leaves 2 weeks postsprouting. QH6 transcript levels were transiently reduced following mechanical wounding or fungal elicitor treatment, suggesting that this gene is not directly involved in defense reaction induced by either of these treatments. Under a photoperiod of 12 h/12 h (light/dark), the abundance of QH6 transcripts fluctuated in a diurnal pattern that ebbed around 3 h before daybreak (9th h in the dark phase) and peaked after 9 h in light (9th h in the light phase). The contents of (−)-β-pinene in juvenile leaves and in emitted volatiles also varied in a diurnal rhythm, correlating strongly with mRNA accumulation. When A. annua was entrained by constant light or constant dark conditions, QH6 transcript accumulation continued to fluctuate with circadian rhythms. Under constant light, advanced cycles of fluctuation of QH6 transcript levels were observed, and under constant dark, the cycle was delayed. However, the original diurnal pattern could be regained when the plants were returned to the normal light/dark (12 h/12 h) photoperiod. This is the first report that monoterpene biosynthesis is transcriptionally regulated in a circadian pattern.
In plants, many aspects of circadian and photoperiodic behaviors are regulated by an endogenous clock that helps plants to anticipate and adapt to the daily and seasonal fluctuations in light and temperature (McWatters et al., 2001; Yanovsky and Kay, 2001). Functional genomic studies of Arabidopsis show many genes to be controlled by the clock, and their functions oscillate rhythmically (Harmer et al., 2000; Kreps et al., 2000; Schaffer et al., 2001).
It is well documented that genes involved in primary metabolic processes such as photosynthesis are circadianly regulated (Sugiyama et al., 2001). For example, in crassulacean acid metabolism plants, the gene for phosphoenolpyruvate carboxylase kinase is regulated by a circadian oscillator to enhance enzyme activity in the dark (Bakrim et al., 2001). In other branches of metabolism, attention has been paid to the circadian pattern of plant hormones such as abscisic acid (Thompson et al., 2000), or photoprotective pigments, including phenylpropanoids (Borevitz et al., 2000). However, reports on the biosynthetic rhythm of terpenes, the biggest group of plant secondary metabolites, has been limited to in planta chemical analysis and emission profiles of flowers, grasses, shrubs, and trees (Bertin et al., 1997; Hansen et al., 1997; Helsper et al., 1998; He et al., 2000a, 2000b).
In nature, plants are the main source of volatile organic compounds (VOCs) emitted to the atmosphere, among which some terpenoids and benzenoic compounds are released in diurnal or nocturnal patterns (Loreto et al., 1996; Staudt et al., 1997; Kolosova et al., 2001). β-Pinene is one of the most widely detected VOCs (Geron et al., 2000), and in some plants, α-pinene, β-pinene, and sabinene may account for more than 80% of the monoterpenes emitted (Bertin et al., 1997). In Quercus ilex grown under natural conditions, terpene emission was found to peak at noon, with the quantity shifting with light intensity and temperature (Bertin et al., 1997; Staudt and Bertin, 1998). The emission of α- and β-pinene in laboratory tests peaked in as short as 30 min after the light was switched on (Loreto et al., 1996). Because this plant has no special storage tissue (such as the oil glands of peppermint, Mentha piperita) for terpenoids produced, the emission pattern implies fluctuations in the activity of specific enzymes, which could be controlled at any step from transcription to posttranslational modification, or in the availability of substrate.
The first committed reactions in the formation of the various classes of terpenes are catalyzed by terpene synthases, which convert acyclic diphosphate substrates (geranyl diphosphate [GPP], farnesyl diphosphate [FPP], geranylgeranyl diphosphate [GGPP], etc.) into acyclic or cyclic terpenes (compare with Greenhagen and Chappell, 2001). These terpene products may be further modified by other enzymes, such as P450 monooxygenases, leading to various terpenoid derivatives (Schuler, 1996). Monoterpenes, which are formed from GPP by monoterpene synthases, are widely present in plants (Croteau, 1987), and some of them account for the flower scent of plants (e.g. Borg-Karlson et al., 1994). In different developmental stages of peppermint, coincident temporal change in enzyme activities, enzyme protein levels, and steady-state transcript abundances of monoterpene synthases was demonstrated, indicating that most of the monoterpene synthases in this plant are regulated at the level of gene expression (Gershenzon et al., 2000; McConkey et al., 2000).
Artemisia annua is an annual herb widely distributed in Asia, Europe, and North America. Different from most of the other plants in the Asteraceae family, the pollination of A. annua is mediated by wind or insects (McVaugh, 1984). The plant is rich in terpenoids (van Geldre et al., 1997; Tan et al., 1998). Among these are the sesquiterpene lactone artemisinin, which is one of the most efficient drugs against Plasmodium species involved in malaria (van Agtmael et al., 1999), and monoterpenes camphor, 1, 8-cineole, α-pinene, and β-pinene, etc. (Ahmad and Misra, 1994), which give the plant a sweet scent. A key enzyme for artemisinin synthesis, amorpha-4, 11-diene synthase, has recently been purified, and its cDNA was cloned (Bouwmeester et al., 1999; Mercke et al., 2000). At least two monoterpene synthases (QH1 and QH5) were proved to be inducible at the transcriptional level by mechanical wounding. In vitro assay showed that both of them converted GPP into (3R)-linalool, which, however, could not be detected from the plant extract (Jia et al., 1999). Here, we report a new A. annua monoterpene synthase cDNA, QH6, which encodes a (−)-β-pinene synthase. This is the first reported terpene synthase of which transcripts and enzymatic products were shown to fluctuate in a circadian pattern.
RESULTS
Cloning and Functional Analysis of QH6
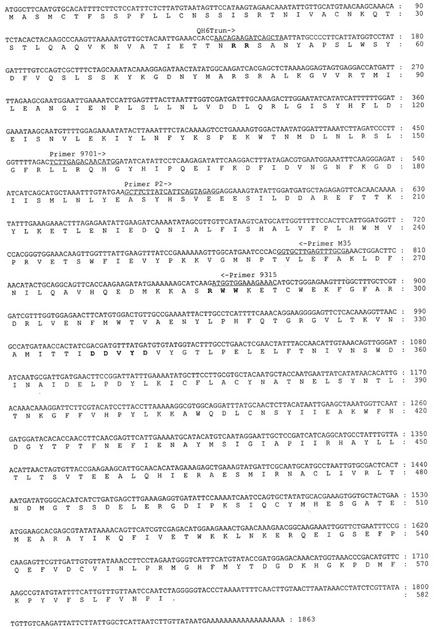
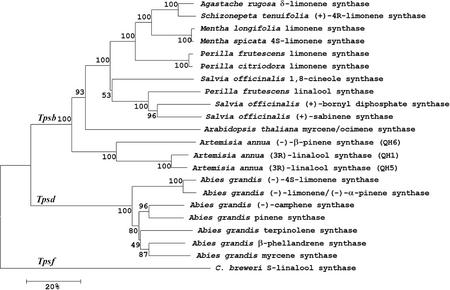
The isolated clone, QH6, contained a cDNA insert of 1,863 bp with an open reading frame encoding a 582-amino acid protein, of which the calculated Mr was 67 kD and the calculated pI was 5.7. R286W287W288 and D337D338X339X340D341 motifs, which are conserved in terpene synthases (Stofer Vogel et al., 1996), were also present in QH6 (Fig. 1). As in other monoterpene synthases, a transit peptide sequence upstream to the absolutely conserved R48R49 was found (Fig. 1) that may function in targeting the protein into plastids (Williams et al., 1998). Sequence analysis showed that the deduced protein was closest to monoterpene synthases from other angiosperms (Tpsb subfamily; Bohlmann et al., 1998; Fig. 2). QH6 showed 57.1% and 57.2% sequence identities, respectively, with another two monoterpene synthases, QH1 and QH5, from A. annua (Jia et al., 1999), but had only 32.5% with a β-pinene synthase (U87909), and 31.6% with a (−)-limonene/(−)-α-pinene synthase (AF139207) from the gymnosperm grand fir (Abies grandis).
Figure 1.
Nucleotide and deduced amino acid sequences of QH6. The conserved motifs are in boldface. Positions and directions of primers used are underlined and labeled. The HindIII site, before which the fragment is used as template for probe labeling in northern blotting, is in italic (AAGCTT, position 566–571).
Figure 2.
Phylogenetic analysis of monoterpene synthases of which the enzymatic functions reported. The neighbor-joining method was used for the protein sequences. Bootstrap values were labeled besides the branches, and the scale bar indicates 20% sequence divergence.
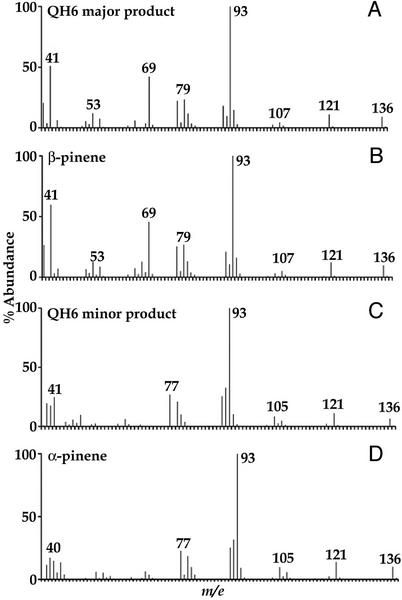
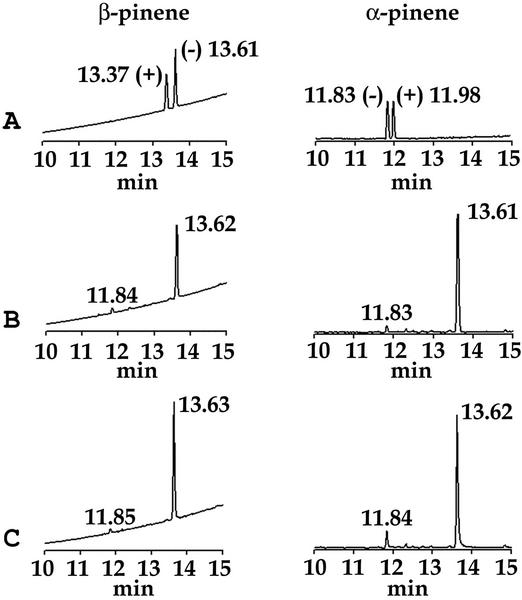
The recombinant plasmid pET32b/QH6, in which the transit peptide sequence had been truncated, resulted in successful expression of QH6 fusion protein in Escherichia coli after isopropyl β-d-thiogalactoside induction. With GPP as substrate, the fusion protein catalyzed the formation of two products in the ratio of 94:6, as revealed by two peaks in gas chromatography (GC). Mass spectra (MS) showed these to be β-pinene (the major product) and α-pinene (the minor product; Fig. 3). By coinjection with authentic standards for GC, both products were found to be (−)-enantiomers (Fig. 4). These results were supported by 1H nuclear magnetic resonance (NMR) spectra of the products. The signals from the major product were 4.625 (br s, 1H), 4.556 (br s, 1H), 2.539 (m, 1H), 2.455 (t, 5.4 Hz, 1H), 2.316 (dtd, 9.9 Hz, 5.8 Hz, 1.2 Hz, 1H), 2.248 (m, 1H), 1.975 (m, 1H), 1.84 (m, 1H), 1.82 (m, 1H), 1.420 (d, 9.8 Hz, 1H), 1.236 (s, 3H), and 0.720 (s, 3H), and those from the minor product were 5.185 (t of sextet, 3 Hz, 1.5 Hz, 1H), 2.162 (d of sextet, 17.5 Hz, 2.5 Hz, 1H), 1.931 (td, 5.6 Hz, 1.5 Hz, 1H), 1.658 (br q, approximately 2 Hz, 3H), 1.151 (d, 8.5 Hz, 1H), and 0.835 (s, 3H). These signals are within experimental error of literature values (Badjah-Hadj-Ahmed et al., 1992). These in vitro results indicate that QH6 encodes a β-pinene synthase, which produces α-pinene as a minor product. The 94:6 ratio of β-:α-pinene in in vitro assay was similar to that measured from A. annua leaf extract (92:8), but higher than that from stems (89:11) or inflorescences (79:21).
Figure 3.
Identification of the major enzymatic product as β-pinene, and the minor as α-pinene. MS are of the major product (A), authentic β-pinene (B), minor product (C), and authentic α-pinene (D).
Figure 4.
Enantiomer-selective GC identification of α- and β-pinene produced by the recombinant enzyme are (−)-enantiomers. Left, Identification of the major product; right, identification of the minor product. A, A mixture of (+)- and (−)-authentic standards; B, enzymatic products; and C, coinjection of enzymatic products with authentic (−)-enantiomer.
QH6 Transcripts in Different Tissues
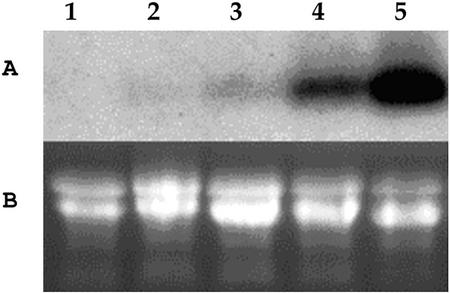
Reverse transcriptase-PCR detected a high level of QH6 transcripts in juvenile leaves (2 weeks postsprouting and not fully expanded), and a lower level in stem, mature leaves (fully expanded), and inflorescences (data not shown). Northern-blot analysis also showed that QH6 transcripts were mainly present in juvenile leaves; much lower, but detectable levels were found in mature leaves, inflorescences, and stems (Fig. 5).
Figure 5.
Transcript abundance of QH6 in different organs of A. annua A, Northern blot; B, ethidium bromide-stained gel for monitoring equal amount of total RNA loaded per lane. Each of 20 μg of total RNA was isolated from roots (lane 1), stems (lane 2), inflorescences (lane 3), mature leaves (lane 4), and juvenile leaves (lane 5). Twenty micrograms of total RNA was loaded per lane.
Circadian Pattern of QH6 Gene Expression
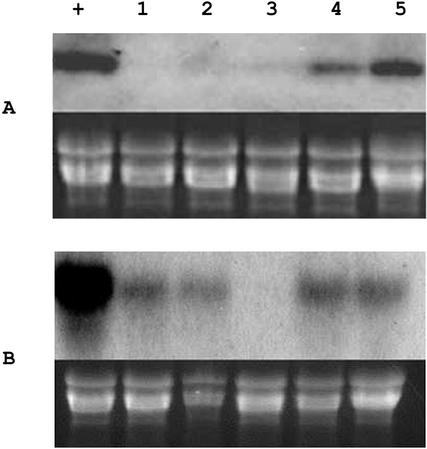
Many plant monoterpene synthases, including QH1 and QH5 of A. annua (Jia et al., 1999), show an increase in steady-state mRNA level due to mechanical wounding (Steele et al., 1998). However, for the (−)-β-pinene synthase QH6, the wounding-treatment of juvenile leaves caused a suppression, rather than an induction, of the steady-state level of transcripts. During the first 2 d postwounding, the QH6 transcripts in juvenile leaves were almost undetectable; at the 3rd d, the mRNA started to accumulate and reached a level comparable with the original at the 5th d postwounding (Fig. 6A). Similar to mechanical wounding, treatment with the fungal elicitor also suppressed the accumulation of QH6 transcripts in juvenile leaves, although the effect was more significant and longer lasting (Fig. 6B). Without these treatments, QH6 transcripts in the leaves collected in the same light phase did not show significant fluctuation, as can be seen in Figure 7A. For mature leaves in which the QH6 transcript abundance was much lower, no induction was detected by either of the treatments (data not shown). The treatment of juvenile leaves brought no systemic induction to the neighboring leaves, regardless of their age (data not shown).
Figure 6.
Effects of mechanical wounding (approximately 40% damage of upper leaf surface; A) and Verticillium dahliae elicitor treatment (1 μg of Suc equivalent mL−1; B) on the steady-state level of QH6 transcripts. In each panel, an ethidium bromide-stained gel was used for monitoring equal amount of total RNA loaded. RNA samples were isolated from juvenile leaves, which were before (+) and at 1 to 5 d (lanes 1–5) posttreatment.
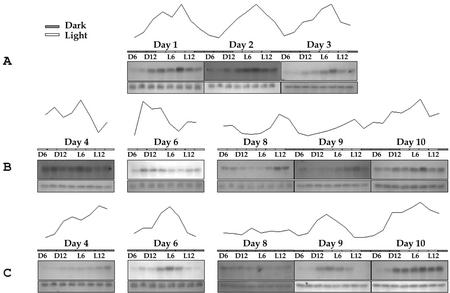
Figure 7.
Circadian pattern of transcriptional regulation of QH6. A, Fluctuation of steady-state level of QH6 transcripts with day-night shifting (d 1–3); B, when plants were entrained by LL photoperiod, the oscillation pattern was in advanced cycles (d 4–8); and C, entrained by DD photoperiod, the oscillation was in delayed cycles (d 4–8). When plants were released from the entrainments, the original diurnal pattern of QH6 transcript fluctuation was resumed (d 9 and 10 in B and C). For each of the northern blots, the signal strength of QH6 was normalized against that of ubiquitin extension protein (UEP; internal control), and the relative percentage was plotted.
We then investigated if other environmental factors could affect QH6 expression. Examination of the diurnal pattern of QH6 expression by northern-blot analysis showed that the steady-state mRNA level fluctuated with the day-night rhythm. With a 12 h/12 h (light/dark, LD) photoperiod, the QH6 mRNA level was generally higher in the day (L) than in the night (D). However, the mRNA level started to increase around 9th h in the dark phase (D9), approximately 3 h prior to the light phase. This accumulation reached its peak level in 12 h, i.e. at the 9th h in light (L9; Fig. 7A).
A key issue is whether plants of A. annua could still maintain a circadian pattern of QH6 transcript accumulation when entrained in constant light (LL) or constant dark (DD; Fig. 7). After the plants were moved to a LL regimen for 3 d (d 6 of Fig. 7), the fluctuation was still clearly detectable, with the peak level appearing at D9, about 12 h ahead that of the original cycle at L9. The rhythm was further accelerated, as in d 8, two peaks appeared at around D9 and L12, respectively (Fig. 7B). When plants were entrained in DD, the transcripts accumulated much slower, and the level did not reach the peak until phase D3 in d 4. At the 3rd d under DD (d 6), the peak was delayed by about 15 h, and it was delayed by 12 h on d 8. In addition, the oscillation magnitude was reduced after 5 d of DD treatment (Fig. 7C). Therefore, the fluctuation cycles were advanced under LL and were delayed under DD conditions. When plants were returned from LL to LD, the peak of QH6 transcript abundance stayed at phase L12 in the 1st d, and then went back to its original phase L9 on the day after (Fig. 7B). For plants from DD, the fluctuation also returned to its original phase on the 2nd d under LD (Fig. 7C).
Diurnal Pattern of (−)-β-Pinene in Planta Content and Emission
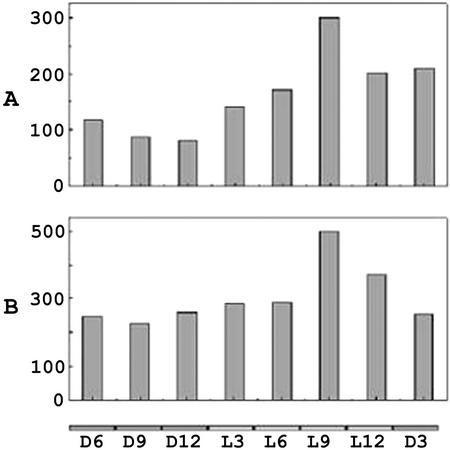
Under LD conditions, contents of β-pinene in the VOCs collected and the leaf extract fluctuated, and peaked at L9 (Fig. 8), the time phase at which QH6 transcripts reached maximum. This indicates a close correlation between (−)-β-pinene formation and QH6 gene expression, suggesting that (−)-β-pinene synthase activity is mainly regulated at the transcriptional level. In the volatiles collected, a much lower amount of (−)-β-pinene was emitted within 6 h before daybreak (D9 and D12), when the mRNA level was just past its lowest level. At the same time, the in planta content of (−)-β-pinene was still clearly detectable in leaves (Fig. 8).
Figure 8.
Diurnal fluctuation of β-pinene content. A, β-Pinene in total VOCs collected in each 3-h time phase (nanograms per plant); B, β-pinene in volatiles extracted from juvenile leaves (nanograms per gram of fresh weight). Time phases in which samples were collected are labeled at the bottom.
DISCUSSION
The Enzymatic Products of (−)-β-Pinene Synthase, QH6
Although β-pinene is one of the most prevalent components of VOCs (Geron et al., 2000), no enzyme has been reported so far that produces only β-pinene. All pinene synthases produce multiple products in vitro with GPP as substrate. For examples, the pinene synthase from grand fir produces (−)-α- and (−)-β-pinene, at a ratio of 42:58 (Bohlmann et al., 1997), and the (−)-pinene synthase from sage (Salvia officinalis) produces an assortment of five monoterpenes: (−)-camphene, (−)-α-pinene, (−)-β-pinene, myrcene, and (−)-limonene (Gambliel and Croteau, 1984). However, the enzyme cloned from A. annua revealed in vitro a great preference for (−)-β-pinene, which represented 94% of the product, much higher than that produced by its counterpart in grand fir or sage. Furthermore, the β-/α-ratios detected from the in vitro assay and from different plant tissues were different, and this may suggest the presence of another pinene synthase that produces more α-pinene in this plant.
Spatial and Temporal Characteristics of (−)-β-Pinene Biosynthesis
QH6 transcripts are organ specific, being mainly present in green tissues of leaves and stems. These organs bear glandular trichomes, which could theoretically serve for storing volatiles. QH6 transcripts and the (−)-β-pinene itself (data not shown) cannot be detected from roots in this work. In a developmental manner, juvenile leaves, which are no older than 2 weeks postsprouting, have the highest steady-state level of QH6 mRNA. This is similar to the findings from peppermint in which monoterpenes were found to be synthesized only in 12- to 20-d-old leaves that were not yet fully expanded (Mihaliak et al., 1991; Gershenzon et al., 2000; McConkey et al., 2000). This rapid decline during development is supported by Tellez et al. (1999), who found that in leaves of 4-month-old A. annua plants, no β-pinene could be detected, whereas α-pinene represented as much as 26.7% of the total volatiles extracted.
It is interesting to note that the QH6 mRNA level and in planta (−)-β-pinene content displayed a circadian pattern of variation, and their peak levels even appeared in the same time phase (L9). This suggests that the biosynthesis of (−)-β-pinene in A. annua is largely controlled at the transcriptional level. Such a regulatory pattern is similar to that of monoterpene biosynthesis in peppermint glandular trichomes (McConkey et al., 2000), which showed coincidental temporal changes in steady-state transcript abundance and enzyme activities. Furthermore, the similar fluctuation of (−)-β-pinene emission suggests that this monoterpene is probably not stored in glandular trichomes for long periods of time, but is released directly after synthesis from glandular or nonglandular tissues. Although α- and β-pinene were reported to take part in direct plant defense against herbivores and pathogens, allelopathy, and plant pollination (Langenheim, 1994), little is known about the biological or ecological function of (−)-β-pinene in A. annua. In contrast to QH1 and QH5, which could be induced by wounding, QH6 transcription was suppressed by mechanical wounding or fungal elicitation. It seems unlikely that (−)-β-pinene synthase is involved in the chemical defense of A. annua. One possible explanation for this suppression is that those up-regulated “defensive enzymes” such as QH1 and QH5 would have more substrate (GPP) available, if other, possibly nondefensive, monoterpene synthases were down-regulated. A similar result was obtained with ponderosa pine (Pinus ponderosa) in which it was shown that an increase in limonene content was accompanied by the decreased levels of α-pinene, β-pinene, 3-carene, and myrcene (Sturgeon, 1979).
Circadian Rhythm of Transcriptional Regulation of (−)-β-Pinene Synthase
Under LD photoperiod, the fluctuation of QH6 transcripts showed an obviously diurnal pattern, which peaked shortly after noon (L9), although in our experiment, the light intensity did not change in the entire light phase. Because the transcripts started to accumulate well before entering the light phase (at D9), QH6 transcription seems not directly triggered by light, but controlled by internal oscillation signals. This is consistent with our findings that the circadian pattern persisted when the plants were transferred by LL or DD photoperiod, although the circadian oscillation of QH6 transcripts became accelerated (LL) or decelerated (DD). It has been proposed that a circadian rhythm is determined by three factors: a free-running period, which is determined by the nature of the gene in certain plants, temperature compensation of the free-running period, and the entrainment of the oscillator through photic and nonphotic time cues (zeitgeber; McWatters et al., 2001). The (possible) only explanation for the varied oscillation frequency of QH6 transcript abundance reported herein is the zeitgeber from LL or DD entrainment signals of A. annua.
It should be mentioned that (−)-β-pinene is not the only example of a plant volatile that can be synthesized and emitted in a diurnal mode. Floral volatiles from rose (Rosa hybrida L. cv Honesty; Helsper et al., 1998) and snapdragon (Antirrhinum majus; Kolosova et al., 2001), as well as damage-induced volatiles of cotton (Gossypium hirsutum; Loughrin et al., 1994), are emitted in a clear circadian rhythm. Delfine et al. (2000) and Sharkey (1996) pointed that monoterpene might improve thermotolerance of photosynthesis in leaves and all isoprenoids are thought to protect membrane from denaturation. It is possible that a peak amount of pinene, as found in this study for A. annua, is synthesized and released for protective purpose when light intensity and temperature are reaching their maximal values under nature conditions.
Isopentenyl diphosphate (IPP) is the biosynthetic precursor of all the terpenoids. In Arabidopsis, transcripts of DXS, the first enzyme in the pathway to IPP biosynthesis in plastids (Lichtenthaler, 1999), vary in a diurnal pattern that peaks at 4 h in the light phase (Estévez et al., 2000; Harmer et al., 2000). DXR generates IPP precursors and was thought to catalyze the critical step in the entire pathway (Mahmoud and Croteau, 2001). With A. annua, we found that both QH6, one of the monoterpene synthases, and DXR, a key enzyme of the plastid isoprenoid pathway, are up-regulated in the later light phase (S. Lu and X.-Y. Chen, unpublished data) when the biological clock in plant is set to allocating assimilated carbohydrates to different pathways (Harmer et al., 2000). At this point, it is still unclear whether the QH6 transcription is directly regulated by an intrinsic circadian clock or by some intermediates in the pathway downstream of DXR, e.g. IPP or GPP. It remains to be learned what signal regulates DXR and QH6 transcription.
MATERIALS AND METHODS
Plant Material
Seeds of Artemisia annua were collected from Guilin, Guangxi Province of China and were planted in the green house at 25°C under a light intensity of 150 μmol m−2 s−1 with 12 h/12 h L/D regime. Leaves at 2 weeks postsprouting that were not yet fully expanded were designated as “juvenile” and those at 6 to 8 weeks postsprouting were designated as “mature.” For stems, only the green tissue was sampled. The inflorescences used in this work did not include their axes and stipules. For transcriptional analyses, juvenile leaves were used, unless otherwise specifically noted.
Plant Treatments
For mechanical wounding, the upper leaf surfaces of plants growing under LD were rubbed with fine sandpaper to approximately 40% surface damage. Elicitation treatment was performed by spreading Verticillium dahliae elicitor preparation at 1 μg of Suc equivalent mL−1 (Liu et al., 1999) on the upper surfaces of leaves. Leaves were collected before, and at 1 to 5 d after each treatment, at time phase L9.
For circadian analysis, a group of cultivated plants were entrained under LD. One group was kept under LD as control, and the rest were divided for two treatments. One was removed to LL, and the other to DD. After 5 d, all of these treated plants were returned to the original LD cycles. Juvenile leaves were collected from 3 d prior to different photoperiod treatments to 1 d after returning to LD, at 3-h intervals.
In each of the triplicate experiments, the plants were destroyed once a leaf was sampled.
Isolation of cDNA
PCR was conducted on the cDNA library of A. annua with the degenerate primers 9701 and 9315 (Jia et al., 1999). A number of cDNA fragments of terpene synthase candidates were obtained. From the sequence of one of the PCR amplicons, specific forward primer P2 (5′-GCTTCTTATCATTCAGTAGAGG-3′) and reverse primer M35 (5′-TCGCAAACTCAAGCACC-3′) were designed for screening the library by a PCR-mediated method, as previously described (Jia et al., 1999). The final isolated clone was named QH6 and the insert was sequenced.
Prokaryotic Expression
A forward primer QH6Trun (5′-ATGAATTCCATGAACAGAAGATCAGCTA-3′), containing an EcoRI cloning site (underlined), was designed for truncating the transit peptide sequence just before the nucleotide sequence of R48R49 motif (boldface). With this and the universal reverse primer T7 (5′-GTAATACGACTCACTATAGGGC-3′) and QH6 as template, the open reading frame was modified by PCR with Pfu DNA polymerase. The product was digested by EcoRI and XhoI and was ligated into pET-32b. The resulting plasmid, named pET32b/QH6, was confirmed by sequencing and was transferred into Escherichia coli BL21(DE3).
The transformed E. coli cells were cultured overnight at 37°C in Luria-Bertani (LB) medium supplemented with 100 μg mL−1 ampicillin. A 500-μL aliquot of the saturated culture was used to inoculate 50 mL of fresh LB medium containing 100 μg mL−1 ampicillin. After adding 1 mm isopropyl β-d-thiogalactoside, the induced culture was allowed to grow at 22°C overnight. The cells were pelted and resuspended in 10 mL of cold assay buffer (Jia et al., 1999) and were disrupted by sonication. After centrifugation, the supernatant was then transferred to a glass tube and the insoluble fraction was resuspended in 10 mL of the same assay buffer.
Enzyme Assay
To assay the terpenoid synthase activity of the bacterial extract, the supernatant was adjusted to a final concentration of 10 mm MgCl2, 20 μm MnCl2, and 30 μm GPP or FPP. Each reaction mixture was then covered with 2 mL of pentane to trap volatile products, sealed, and incubated at 30°C for 3 h. The reaction mixture was extracted with pentane (3 × 2 mL). The remaining aqueous phase was subsequently extracted with diethyl ether (3 × 2 mL). The pentane extracts were combined and passed through a silica gel column (3 cm × 5 mm i.d.) covered with MgSO4 (1 cm) to yield the terpene hydrocarbon fraction. The ether extract was passed through the same silica column to provide an oxygenated terpene fraction. The pentane extract was concentrated to 500 μL under a nitrogen stream. The solvent in the ether extract was evaporated similarly to near dryness and was redissolved in 500 μL of ethyl acetate. Both samples were analyzed by GC and GC-MS. Product from E. coli BL21 (DE3) expressing empty pET32b vector was included as a negative control.
To obtain sufficient product for NMR analysis, a large-scale preparation was conducted by inoculating 750 μL of an overnight culture into 750 mL of LB medium containing 100 μg mL−1 ampicillin. The culture was prepared similarly to the small-scale assay, except that the supernatant of the sonicate was adjusted to a final concentration of 10 mm MgCl2, 20 μm MnCl2, and 200 μm GPP. Cyclopentane (30 mL) was used instead of pentane to minimize the solvent interference in subsequent NMR analysis. After incubation at 30°C overnight, the reaction mixture was extracted with cyclopentane (3 × 100 mL). The combined organic extracts were passed through a silica gel column (8 × 2 cm i.d.) overlaid with MgSO4 (2 cm) to yield the terpene hydrocarbon fraction. A 2-μL aliquot was analyzed by GC and the total amount of product was estimated to be approximately 1 mg based on the peak area-to-weight ratio calculated from the (−)-β-pinene standard. The solvent was evaporated under a stream of nitrogen to near dryness for NMR analysis.
GC, GC-MS, and NMR Analyses
To isolate A. annua essential oil, stems, inflorescences, and mature leaves were ground in liquid nitrogen to fine powder and were then extracted with hexane twice. The organic extracts were combined and were passed through a column of silica gel 60 (EM Science, Gibbstown, NJ) overlaid with MgSO4, and were concentrated under a nitrogen stream. For emitted volatiles, a collecting device according to Jansen et al. (1999) with about 10 L of headspace and an air flow of 0.2 L min−1 was used, and the plants were grown under normal cultivation conditions as mentioned above. The released chemicals were trapped by 20 mL of hexane, which was finally condensed under nitrogen stream to 200 μL for GC analysis. The authentic (−)-β-pinene was diluted with hexane in series and was then used for quantifying (−)-β-pinene contents in samples.
GC analyses were performed on a 6890 system (Hewlett-Packard, Palo Alto, CA) equipped with a Rtx-5 capillary column (30-m × 0.25-mm i.d., 0.10 μm df; Restek, Bellefonte, PA). Separation conditions were the following: injection port 200°C, flame ionization detector 250°C, a split ratio of 39:1 for plant extracts and enzymatic products, and splitless for volatile collections, and helium flow at 20 cm s−1 (0.6 mL min−1). The temperature program was 50°C for 5 min, an increase to 250°C (10°C min−1), and 250°C for 5 min. GC-MS was performed on a 5890A instrument (Hewlett-Packard) equipped with a DB-5 ms column (60-m × 0.25-mm i.d., 0.10 μm df; J&W Scientific, Folsom, CA). Separation was achieved with splitless injection at 200°C, helium flow at 30 cm s−1 (1 mL min−1), and the identical temperature program as above. Mass spectra (m/z 35–500) were obtained on a ZAB-HF reverse-geometry double-focusing instrument at 70 eV with an electron-impact ion source (200°C). The accelerating voltage was 8 kV and the resolution was 1,000 (10% valley). Enantioselective GC was performed on a GC-9A instrument (Shimadzu, Tokyo) with a CycloSil-B capillary column containing a modified β-cyclodextrin stationary phase (30-m × 0.25-mm i.d., 0.25 μm df; J&W Scientific). The separation of chiral components was achieved with injection port and flame ionization detector at 200°C, helium flow at 29 cm s−1 (0.8 mL min−1), a 36:1 split ratio, and the following temperature program: 50°C for 1 min and a 5°C min−1 ramp to 200°C, where the temperature was held for 10 min. Authentic (+)-α-pinene, (−)-α-pinene, (+)-β-pinene, and (−)-β-pinene from Sigma-Aldrich (St. Louis) were used as standards. The QH6 product was also coinjected with authentic samples to determine the pinene stereochemistries.
1H NMR spectra were obtained on a AMX500 spectrometer (500.1 MHz for 1H; Bruker, Billerica, MA) at 25°C in CDCl3 solution (approximately 10 mm) and were referenced to internal tetramethylsilane.
Transcriptional Analysis
RNA was extracted according to Fütterer et al. (1995). A total of 2 μg of RNA was used as template for reverse transcription by using a Reverse Transcription System (Promega, Madison, WI) with oligo (dT)15 as the primer. As an internal control, transcripts of UEP were detected by a forward primer UbiF2 (5′-CTTGGGGGAAGACGGGC-3′) and a reverse primer UbiR2 (5′-GCCAAGATTCAGGACAAGGAAGG-3′). Primers P2 and M35 were used to analyze QH6 transcripts.
For northern-blot analysis, a total of 20 μg of RNA was loaded per lane, and an ethidium bromide-stained gel or a duplicated blot probed for UEP transcripts was used to monitor the amount of RNA loaded. A fragment containing the first 566 bp of QH6 (released by EcoRI and HindIII digestion of pBK-CMV/QH6), which showed a relatively lower sequence identity with QH1 or QH5 (Jia et al., 1999), was 32P labeled by the Prime-a-Gene System (Promega) to detect QH6 transcripts on the membrane. The blots were hybridized, washed as described (Ausubel et al., 1995), and exposed to x-ray film at −70°C. The images were collected with GeneGenius gel documentation system and were then digitalized with GeneTools software (Syngene, Cambridge, UK). Corresponding data from UEP transcripts were used for normalization.
ACKNOWLEDGMENTS
We thank Prof. Rodney Croteau for help with the entire project. We are also grateful to Dr. William Wilson for his expert assistance in NMR, to Drs. Yong-Ping Huang and Hong-Chun Zhou for their assistance in chemical analysis, and to Elizabeth A. Hart, Hala G. Schepmann, and Dr. Xiang-Jun Zhou for helpful discussions.
Footnotes
This work was supported by the National Natural Science Foundation of China (grant nos. 39925005 and 30030020 to X.-Y.C.), by the National Institutes of Health (grant no. AI41598), and by the Robert A. Welch Foundation (grant no. C–1323 to S.P.T.M.).
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.006544.
LITERATURE CITED
- Ahmad A, Misra LN. Terpenoids from Artemisia annua and constituents of its essential oil. Phytochemistry. 1994;37:183–186. [Google Scholar]
- Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K. Current Protocols in Molecular Biology. New York: John Wiley & Sons; 1995. [Google Scholar]
- Badjah-Hadj-Ahmed AY, Meklati BY, Waton H, Pham QT. Structural studies in the bicyclo [3.1.1]heptane series by H-1 and C-13 NMR. Magn Reson Chem. 1992;30:807–816. [Google Scholar]
- Bakrim N, Brulfert J, Vidal J, Chollet R. Phosphoenolpyruvate carboxylase kinase is controlled by a similar signaling cascade in CAM and C4 plants. Biochem Biophys Res Commun. 2001;286:1158–1162. doi: 10.1006/bbrc.2001.5527. [DOI] [PubMed] [Google Scholar]
- Bertin N, Staudt M, Hansen U, Seufert G, Ciccioli P, Foster P, Fugit JL, Torres L. Diurnal and seasonal course of monoterpene emission from Quercus ilex (L.) under natural conditions—application of light and temperature algorithms. Atmos Environ. 1997;31:135–144. [Google Scholar]
- Bohlmann J, Meyer-Gauen G, Croteau R. Plant terpenoid synthases: molecular biology and phylogenetic analysis. Proc Natl Acad Sci USA. 1998;95:4126–4133. doi: 10.1073/pnas.95.8.4126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohlmann J, Steele CL, Croteau R. Monoterpene synthases from grand fir (Abies grandis): cDNA isolation, characterization, and functional expression of myrcene synthase, (−)-(4S)-limonene synthase, and (−)-(1S, 5S)-pinene synthase. J Biol Chem. 1997;272:21784–21792. doi: 10.1074/jbc.272.35.21784. [DOI] [PubMed] [Google Scholar]
- Borevitz JO, Xia Y, Blount J, Dixon RA, Lamb C. Activation tagging identifies a conserved MYB regulator of phenylpropanoid biosynthesis. Plant Cell. 2000;12:2383–2394. doi: 10.1105/tpc.12.12.2383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borg-Karlson A-K, Valterova I, Nilsson A. Volatile compounds from flowers of six species in the family Apiaceae: bouquets for different pollinators. Phytochemistry. 1994;35:111–119. [Google Scholar]
- Bouwmeester HJ, Wallaart TE, Janssen MHA, van Loo B, Jansen BJM, Posthumus MA, Schmidt CO, de Kraker J-W, König WA, Franssen MCR. Amopha-4,11-diene synthase catalyses the first probable step in artemisinin biosynthesis. Phytochemistry. 1999;52:843–854. doi: 10.1016/s0031-9422(99)00206-x. [DOI] [PubMed] [Google Scholar]
- Croteau R. Biosynthesis and metabolism of monoterpenes. Chem Rev. 1987;87:929–954. [Google Scholar]
- Delfine S, Csiky O, Seufert G, Loreto F. Fumigation with exogenous monoterpenes of a non-isoprenoid-emitting oak (Quercus suber): monoterpene acquisition, translocation, and effect on the photosynthetic properties at high temperatures. New Phytol. 2000;146:27–36. [Google Scholar]
- Estévez JM, Cantero A, Romero C, Kawaide H, Jiménez LF, Kuzuyama T, Seto H, Kamiya Y, León P. Analysis of the expression of CLA1, a gene that encodes the 1-deoxyxylulose 5-phosphate synthase of the 2-C-methyl-d-erythritol-4-phosphate pathway in Arabidopsis. Plant Physiol. 2000;124:95–103. doi: 10.1104/pp.124.1.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fütterer J, Gisel A, Iglesias V, Kloti A, Kost B, Mittelsten Scheid O, Neuhaus G, Neuhaus-Url G, Schrott M, Shillito R . et al. Standard molecular techniques for the analysis of transgenic plants. In: Potrykus I, Spangenberg G, editors. Gene Transfer to Plants. Berlin: Springer-Verlag; 1995. [Google Scholar]
- Gambliel H, Croteau R. Pinene cyclases I and II: two enzymes from sage (Salvia officinalis) which catalyze stereospecific cyclization of geranyl pyrophosphate to monoterpene olefins of opposite configuration. J Biol Chem. 1984;259:740–748. [PubMed] [Google Scholar]
- Geron C, Rasmussen R, Arnts RR, Guenther A. A review and synthesis of monoterpene speciation from forests in the United States. Atmos Environ. 2000;34:1761–1781. [Google Scholar]
- Gershenzon J, McConkey ME, Croteau RB. Regulation of monoterpene accumulation in leaves of peppermint. Plant Physiol. 2000;122:205–213. doi: 10.1104/pp.122.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenhagen B, Chappell J. Molecular scaffolds for chemical wizardry: learning nature's rule for terpene cyclases. Proc Natl Acad Sci USA. 2001;98:13479–13481. doi: 10.1073/pnas.261562898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen U, Van Eijk J, Bertin N, Staudt M, Kotzias D, Seufert G, Fugit J-L, Torres L, Cecinato A, Brancaleoni E et al. Biogenic emissions and CO2 gas exchange investigated on four Mediterranean shrubs. Atmos Environ. 1997;31:157–166. [Google Scholar]
- Harmer SL, Hogenesch JB, Straume M, Chang H-S, Han B, Zhu T, Wang X, Kreps JA, Kay SA. Orchestrated transcription of key pathways in Arabidopsis by the circadian clock. Science. 2000;290:2110–2113. doi: 10.1126/science.290.5499.2110. [DOI] [PubMed] [Google Scholar]
- He C, Murray F, Lyons T. Monoterpene and isoprene emission from 15 Eucalyptus species in Australia. Atmos Environ. 2000a;34:645–665. [Google Scholar]
- He C, Murray F, Lyons T. Seasonal variations in monoterpene emissions from Eucalyptus species. Chemosph Global Change Sci. 2000b;2:65–76. [Google Scholar]
- Helsper JPFG, Davies JA, Bouwmeester HJ, Krol AF, van Kampen MH. Circadian rhythmicity in emission of volatile compounds by flowers of Rosa hybrida L. cv. Honesty Planta. 1998;207:88–95. [Google Scholar]
- Jansen R, De Serves C, Romero R. Emission of isoprene and carbonyl compounds from a boreal forest and wetland in Sweden. Agric Forest Met. 1999;98–99:671–681. [Google Scholar]
- Jia J-W, Crock J, Lu S, Croteau R, Chen X-Y. (3R)-Linalool synthase from Artemisia annua L.: cDNA isolation, characterization, and wound induction. Arch Biochem Biophys. 1999;372:143–149. doi: 10.1006/abbi.1999.1466. [DOI] [PubMed] [Google Scholar]
- Kolosova N, Gorenstein N, Kish CM, Dudareva N. Regulation of circadian methyl benzonate emission in diurnally and nocturnally emitting plants. Plant Cell. 2001;13:2333–2347. doi: 10.1105/tpc.010162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreps JA, Muramatsu T, Furuya M, Kay SA. Fluorescent differential display identifies circadian clock regulated genes in Arabidopsis thaliana. J Biol Rhythms. 2000;5:208–217. doi: 10.1177/074873040001500302. [DOI] [PubMed] [Google Scholar]
- Langenheim JH. Higher plant terpenoids: a phytocentric overview of their ecological roles. J Chem Ecol. 1994;20:1223–1280. doi: 10.1007/BF02059809. [DOI] [PubMed] [Google Scholar]
- Lichtenthaler HK. The 1-deoxy-d-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plant. Annu Rev Plant Physiol Plant Mol Biol. 1999;55:47–65. doi: 10.1146/annurev.arplant.50.1.47. [DOI] [PubMed] [Google Scholar]
- Liu CJ, Heinstein P, Chen X-Y. Expression pattern of genes encoding farnesyl diphosphate synthase and sesquiterpene cyclase in cotton suspension-cultured cells treated with fugal elicitors. Mol Plant-Microbe Interact. 1999;12:1095–1104. doi: 10.1094/MPMI.1999.12.12.1095. [DOI] [PubMed] [Google Scholar]
- Loreto F, Ciccioli P, Brancaleoni E, Cecinato A, Frattoni M, Sharkey T. Different sources of reduced carbon contribute to form three classes of terpenoid emitted by Quercus ilex L. leaves. Proc Natl Acad Sci USA. 1996;93:9966–9969. doi: 10.1073/pnas.93.18.9966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loughrin JH, Manukian A, Heath RR, Turlings TCJ, Tumlinson J. Diurnal cycle of emission of induced volatile terpenoids by herbivore-injured cotton plants. Proc Natl Acad Sci USA. 1994;91:11836–11840. doi: 10.1073/pnas.91.25.11836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahmoud SS, Croteau RB. Metabolic engineering of essential oil yield and composition in mint by altering expression of deoxyxylulose phosphate reductoisomerase and menthofuran synthase. Proc Natl Acad Sci USA. 2001;98:8915–8920. doi: 10.1073/pnas.141237298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McConkey ME, Geshenzon J, Croteau RB. Developmental regulation of monoterpene biosynthesis in glandular trichomes of peppermint. Plant Physiol. 2000;122:215–223. doi: 10.1104/pp.122.1.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McVaugh R. Compositae. In: Anderson WR, editor. Flora Novo-Galiciana: A Descriptive Account of the Vascular Plants of Western Mexico. Vol. 12. Ann Arbor: University of Michigan Press; 1984. [Google Scholar]
- McWatters HG, Roden LC, Staiger D. Picking out parallels: plant circadian clocks in context. Phil Trans R Soc Lond B. 2001;356:1735–1743. doi: 10.1098/rstb.2001.0936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercke P, Bengtsson M, Bouwmeester HJ, Posthumus MA, Brodelius PE. Molecular cloning, expression, and characterization of amorpha-4,11-diene synthase, a key enzyme of artemisinin biosynthesis in Artemisia annua L. Arch Biochem Biophys. 2000;381:173–180. doi: 10.1006/abbi.2000.1962. [DOI] [PubMed] [Google Scholar]
- Mihaliak CA, Gershenzon J, Croteau R. Lack of rapid monoterpene turnover in rooted plants: implications for theories of plant-chemical defense. Oecologia. 1991;87:373–376. doi: 10.1007/BF00634594. [DOI] [PubMed] [Google Scholar]
- Schaffer R, Landgraf J, Accerbi M, Simon V, Larson M, Wisman E. Microarray analysis of diurnal and circadian-regulated genes in Arabidopsis. Plant Cell. 2001;13:113–123. doi: 10.1105/tpc.13.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuler MA. Plant cytochrome P450 monooxygenases. Crit Rev Plant Sci. 1996;15:235–284. [Google Scholar]
- Sharkey TD. Isoprene synthesis in plants and animals. Endeavour. 1996;20:24–28. doi: 10.1016/0160-9327(96)10014-4. [DOI] [PubMed] [Google Scholar]
- Staudt M, Bertin N. Light and temperature dependence of the emission of cyclic and acyclic monoterpenes from holm oak (Quercus ilex L.) leaves. Plant Cell Environ. 1998;21:385–395. [Google Scholar]
- Staudt M, Bertin N, Hansen U, Seufert G, Ciccioli P, Foster P, Frenzel B, Fugit J-L. Seasonal and diurnal patterns of monoterpene emissions from Pinus pinea (L.) under field. Atmos Environ. 1997;31:145–156. [Google Scholar]
- Steele CL, Katoh S, Bohlmann J, Croteau R. Regulation of oleoresinosis in grand fir (Abies grandis): differential transcriptional control of monoterpene, sesquiterpene, and diterpene synthase genes in response to wounding. Plant Physiol. 1998;116:1497–1504. doi: 10.1104/pp.116.4.1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stofer Vogel B, Wildung MR, Vogel G, Croteau R. Abietadiene synthase from grand fir (Abies grandis): cDNA isolation, characterization, and bacterial expression of a bifunctional diterpene cyclase involved in resin acid biosynthesis. J Biol Chem. 1996;271:23262–23268. doi: 10.1074/jbc.271.38.23262. [DOI] [PubMed] [Google Scholar]
- Sturgeon KB. Monoterpene variation in ponderosa pine xylem resin related to western pine-beetle predation. Evolution. 1979;33:803–814. doi: 10.1111/j.1558-5646.1979.tb04736.x. [DOI] [PubMed] [Google Scholar]
- Sugiyama N, Izawa T, Oikawa T, Shimamoto K. Light regulation of circadian clock-controlled gene expression in rice. Plant J. 2001;26:607–615. doi: 10.1046/j.1365-313x.2001.01063.x. [DOI] [PubMed] [Google Scholar]
- Tan RX, Zheng WF, Tang HQ. Biologically active substances from the genus Artemisia. Plant Med. 1998;64:295–302. doi: 10.1055/s-2006-957438. [DOI] [PubMed] [Google Scholar]
- Tellez MR, Canel C, Rimando AM, Duck SO. Differential accumulation of isoprenoids in glanded and glandless Artemisia annua L. Phytochemistry. 1999;52:1035–1040. [Google Scholar]
- Thompson AJ, Jackson AC, Parker RA, Morpeth DR, Burbidge A, Taylor IB. Abscisic acid biosynthesis in tomato: regulation of zeaxanthin epoxidase and 9-cis-epoxycarotenoid dioxygenase mRNAs by light/dark cycles, water stress and abscisic acid. Plant Mol Biol. 2000;42:833–845. doi: 10.1023/a:1006448428401. [DOI] [PubMed] [Google Scholar]
- van Agtmael MA, Eggelte TA, van Boxtel CJ. Artemisinin drugs in the treatment of malaria: from medicinal herb to registered medication. Trends Pharmacol Sci. 1999;20:199–205. doi: 10.1016/s0165-6147(99)01302-4. [DOI] [PubMed] [Google Scholar]
- van Geldre E, Vergauwe A, van den Eeckhout E. State of the art of the production of the antimalarial compound artemisinin in plants. Plant Mol Biol. 1997;33:199–209. doi: 10.1023/a:1005716600612. [DOI] [PubMed] [Google Scholar]
- Williams DC, McGarvey DJ, Katahira EJ, Croteau R. Truncation of limonene synthase preprotein provides a fully active “Pseudomature” form of this monoterpene cyclase and reveals the function of the amino-terminal arginine pair. Biochemistry. 1998;37:12213–12220. doi: 10.1021/bi980854k. [DOI] [PubMed] [Google Scholar]
- Yanovsky MJ, Kay SA. Signaling networks in the plant circadian system. Curr Opin Plant Biol. 2001;4:429–435. doi: 10.1016/s1369-5266(00)00196-5. [DOI] [PubMed] [Google Scholar]