Abstract
Maize (Zea mays) emits a mixture of volatile compounds upon attack by the Egyptian cotton leafworm (Spodoptera littoralis). These substances, primarily mono- and sesquiterpenes, are used by parasitic wasps to locate the lepidopteran larvae, which are their natural hosts. This interaction among plant, lepidopteran larvae, and hymenopteran parasitoids benefits the plant and has been termed indirect defense. The committed step in the biosynthesis of the different skeletal types of mono- and sesquiterpenes is catalyzed by terpene synthases, a class of enzymes that forms a large variety of mono- and sesquiterpene products from prenyl diphosphate precursors. We isolated a terpene synthase gene, terpene synthase 1 (tps1), from maize that exhibits only a low degree of sequence identity to previously identified terpene synthases. Upon expression in a bacterial system, the encoded enzyme produced the acyclic sesquiterpenes, (E)-β-farnesene, (E,E)-farnesol, and (3R)-(E)-nerolidol, the last an intermediate in the formation of (3E)-4,8-dimethyl-1,3,7-nonatriene. Both (E)-β-farnesene and (3E)-4,8-dimethyl-1,3,7-nonatriene are prominent compounds of the maize volatile blend that is emitted after herbivore damage. The biochemical characteristics of the encoded enzyme are similar to those of terpene synthases from both gymnosperms and dicotyledonous angiosperms, suggesting that catalysis involves a similar electrophilic reaction mechanism. The transcript level of tps1 in the maize cv B73 was elevated after herbivory, mechanical damage, and treatment with elicitors. In contrast, the increase in the transcript level of the tps1 gene or gene homolog in the maize cv Delprim after herbivory was less pronounced, suggesting that the regulation of terpene synthase expression may vary among maize varieties.
Many plant species emit volatile compounds upon herbivore attack. These emissions often attract natural enemies of the herbivores, either predators or parasitoids, which use the volatiles as cues to locate their prey or hosts. The attraction of herbivore enemies by the plant, termed “indirect defense” (Dicke, 1999), has been shown to be beneficial to the plant (Kessler and Baldwin, 2001). For example, when maize (Zea mays) is attacked by lepidopteran larvae, such as one of the noctuid species of the genus Spodoptera, the blend of volatiles emitted attracts parasitic wasps, like Cotesia marginiventris, that oviposit on the surface of the larvae (Turlings et al., 1990). After hatching, the parasitoids develop inside the larvae and subsequently kill them. Because parasitized lepidopteran larvae do not procreate and feed less than unparasitized larvae (Hoballah and Turlings, 1999), it is assumed that the attraction of parasitoids is advantageous to maize. In keeping with the defensive function of the emitted volatiles, their release appears to be a specific response to herbivory that cannot be simulated by mechanical wounding of the leaves (Turlings et al., 1991). Factors that are present in the oral secretions of lepidopterans, like the fatty acid-amino acid conjugate volicitin, are necessary to trigger emission (Alborn et al., 1997).
The volatile blend of maize plants is dominated by terpenes (Turlings et al., 1991) and differs considerably among maize varieties (Gouinguene et al., 2001). The most common terpenes emitted by maize are the monoterpene linalool, and the sesquiterpenes, (E)-bergamotene, (E)-β-farnesene, and β-caryophyllene. Two terpenoids with an irregular acyclic carbon skeleton are also emitted, (3E)-4,8-dimethyl-1,3,7-nonatriene (DMNT) and (3E,7E)-4,8,12-trimethyl-1,3,7,11- tridecatetraene.
The biosynthesis of terpenes proceeds via the fusion of the activated 5-carbon isoprenoid units, isopentenyl diphosphate and dimethylallyl diphosphate, derived from the acetate/mevalonate or methylerythritol phosphate pathways (for review, see Chappell, 1995; Gershenzon and Kreis, 1999). The C5 units are condensed by prenyltransferases to form elongated linear prenyl diphosphates, among which are geranyl diphosphate (GPP, a C10 compound) and farnesyl diphosphate (FPP, a C15 compound), the precursors of mono- and sesquiterpenes, respectively. The prenyl diphosphates in turn undergo a wide range of cyclizations and other transformations to produce the various structural types of mono- and sesquiterpenes. These steps are catalyzed by terpene synthases, a very large group of enzymes with multiple representatives in all plant species studied so far (Facchini and Chappell, 1992; Steele et al., 1998; Wise et al., 1998). Terpene synthases also represent important regulatory steps in terpene formation after herbivore damage (Yin et al., 1997; Degenhardt and Gershenzon, 2000) or fungal attack (Facchini et al., 1992), making them a primary focus of research projects to study terpene biosynthesis and its function. Many terpene synthase genes that encode enzymes producing volatile products have been identified from gymnosperms and dicotyledonous angiosperms. This class of genes shares common structural features that are reflected in highly conserved amino acid residues throughout the encoded protein (Bohlmann et al., 1998). A very prominent feature of terpene synthases is an Asp-rich region, the DDxxD motif, which is involved in the binding of a divalent metal cofactor (Marrero et al., 1992).
By contrast to dicotyledons, only a few terpene synthase genes have been identified to date in monocotyledons. A gene with structural features common to many terpene synthases was found in the oil palm Elaeis oleifera (Shah and Cha, 2000) but remains otherwise uncharacterized. In maize, a diterpene synthase gene (an1) involved in the synthesis of gibberellins was identified by the analysis of gibberellin-sensitive transposon insertion mutants (Bensen et al., 1995). The corresponding enzyme converts geranylgeranyl diphosphate (GGPP, a C20 compound) to copalyl diphosphate and does not exhibit a pyrophosphorylase activity. Shen and coworkers (2001) recently described a second terpene synthase gene (stc1) in maize. This gene displayed elevated transcript levels in plants that were damaged mechanically or subjected to herbivory by lepidopteran larvae. Plants with null mutations in stc1 were missing a compound in their headspace reported as a naphthalene-type sesquiterpene, suggesting that stc1 is involved in the biosynthesis of this compound.
As part of a research program to study the function of volatile terpenes in maize anti-herbivore defense, we have begun to investigate the terpene synthases of this important crop plant. The isolated terpene synthase gene sequences should be valuable tools in obtaining maize lines that are altered in volatile production. Here, we report the isolation and characterization of a maize terpene synthase gene we have named terpene synthase 1 (tps1), which encodes an enzyme catalyzing the formation of three acyclic sesquiterpenes, (E)-β-farnesene, (3R)-(E)-nerolidol, and (E,E)-farnesol. Both (E)-β-farnesene and a metabolite of (E)-nerolidol, DMNT, are among the major volatile terpenes emitted by herbivore-damaged plants. Herbivory acts as a cue to induce transcript levels of tps1 in one of the two maize lines analyzed.
RESULTS
Cloning of the Maize Terpene Synthase tps1
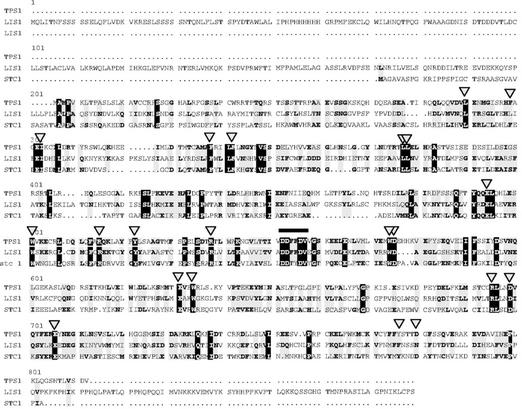
Initial attempts to clone maize terpene synthase genes by PCR with degenerate primers designed to highly conserved sequence elements of terpene synthase genes of gymnosperms or dicotyledonous angiosperms were unsuccessful. Therefore, we searched a proprietary maize expressed sequence tag (EST) database assembled by Pioneer Hi-Bred International (Des Moines, IA) and public databases (Gai et al., 2000) for ESTs with sequence similarity to known terpene synthases. Several such ESTs were selected and aligned with each other to form contigs. One 671-bp contig represented by two ESTs from the inbred maize cv B73, a line frequently used for genetic studies, was chosen for further study. The 5′ end of the contig was extended 1,510 bp by 5′ RACE-PCR using a cDNA library made from herbivore-induced maize cv B73 plants to obtain the complete cDNA. The cDNA contains an open reading frame (ORF) of 1,775 bp that encodes a protein with a predicted molecular mass of 67.4 kD (Fig. 1). No signal peptide was apparent in the N-terminal region of the protein. The tps1 cDNA sequence has only a low similarity to other terpene synthases based on comparison of the amino acid sequences of the encoded proteins. Most similar are the previously described maize terpene synthase gene stc1 (Shen et al., 2001) with 25.1% amino acid identity and the linalool synthase gene linalool synthase 1 (lis1) from the dicotyledon C. breweri (Dudareva et al., 1996) with 21.4% amino acid identity (Fig. 1). The identity to all other reported terpene synthases is less than 20%.
Figure 1.
Comparison of the deduced amino acid sequence of tps1 with other terpene synthase sequences of the highest similarity, the maize terpene synthase stc1 and a linalool synthase from Clarkia breweri (lis1). Amino acids identical in the three genes are marked by black boxes. Amino acids identical in two genes or representing conservative changes are highlighted with gray boxes. The highly conserved DDxxD region is marked with a bar, and single amino acids highly conserved among many terpene synthases are marked with a triangle.
Heterologous Expression and Product Identification
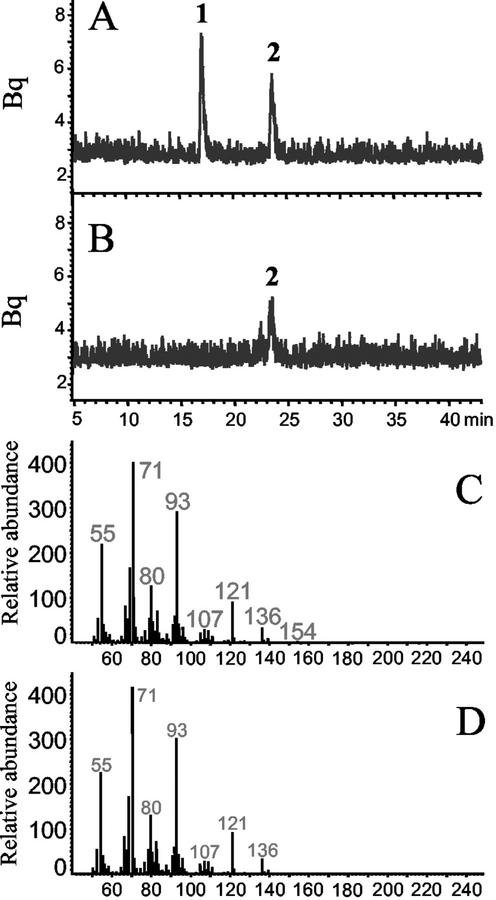
To confirm that tps1 encodes an enzyme with a terpene synthase activity, the complete ORF of the cDNA was overexpressed in Escherichia coli. The recombinant enzyme was incubated with the [1-3H]-labeled substrates GPP, FPP, and GGPP. The enzyme did not accept GGPP as a substrate (data not shown), converting only GPP and FPP to terpene products. In the presence of FPP, the enzyme catalyzed the formation of the acyclic sesquiterpenes (E)-β-farnesene (26% of total product), (E)-nerolidol (29%), and (E,E)-farnesol (45%; Fig. 2A). These products were identified by comparison of their retention times and mass spectra (Fig. 2, C, E, and G) with those of authentic standards (Fig. 2, D, F, and H) and with previously published mass spectra (Joulain and König, 1998). Analysis of the (E)-nerolidol product in comparison with (3R) and (3S)-(E)-nerolidol standards by gas chromatography (GC) on a chiral column revealed that >95% of the enzymatic product was the (3R)-enantiomer (Fig. 3A).
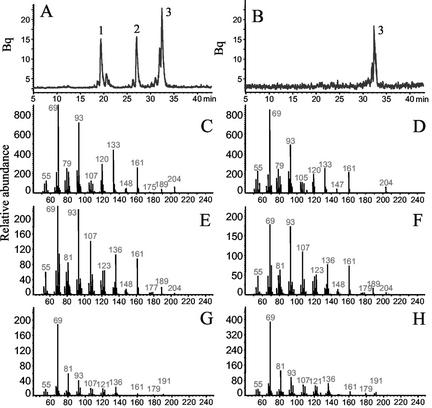
Figure 2.
Sesquiterpene products of TPS1. The enzyme was expressed in E. coli, extracted, and incubated with the substrate [1-3H]FPP. The resulting terpene products were separated by radio GC. The traces of the radio detector are shown for the products of the active enzyme (A) and the products formed by an extract of E. coli transformed with the expression vector lacking the tps1 gene (B). The products were identified as (E)-β-farnesene (1), (E)-nerolidol (2) and farnesol (3) by comparison of their retention times and mass spectra to those of authentic standards. Depicted are mass spectra of: (E)-β-farnesene, TPS1 product (C), and standard (D); (E)-nerolidol, TPS1 product (E), and standard (F); (E,E)-farnesol, TPS1 product (G), and standard (H).
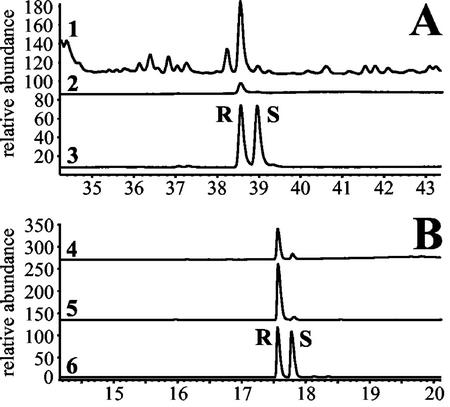
Figure 3.
Stereochemical analysis of TPS1 products. A, The flame ionization detector (FID) traces depict the nerolidol produced by the enzyme (1), a pure (3R)-(E)-nerolidol standard (2), and a racemic standard of (E)-nerolidol (3) after separation on a chiral column as described in the methods section. B, The traces show the enzymatically produced linalool (4), a (3R)-linalool standard (5), and a racemic standard of linalool (6).
The sesquiterpene alcohol (E,E)-farnesol was also formed in extracts of E. coli harboring only the expression vector and, thus, was considered to be at least partially a product of unspecific phosphatases in the bacterial extract (Fig. 2B). To test whether (E,E)-farnesol is also a true product of the enzyme encoded by tps1, we expressed the enzyme with a C-terminal 6× His tag and purified it on a nickel-nitrilotriacetate agarose column to >95% purity based on SDS-PAGE stained with Coomassie Blue. The sesquiterpene products formed by the purified, His-tagged protein were similar to those formed by the crude enzyme in the bacterial extract and included (E,E)-farnesol (Fig. 4). The shoulder on the trailing edge of the (E,E)-farnesol peak is (Z,E)-farnesol, an enzymatic product of (Z,E)-FPP, present in the (E,E)-FPP substrate as a 10% to 20% impurity. As further evidence that (E,E)-farnesol is a natural product of the TPS1 enzyme, this alcohol was found to be formed even in the presence of the phosphatase inhibitors sodium tungstate (5 mm) and sodium fluoride (1 mm) but was not formed after inactivation of the terpene synthase activity by heat or addition of 10 mm EDTA to the assay (data not shown). To exclude the possibility that a phosphatase activity had been copurified with the terpene synthase on the nickel-nitrilotriacetate agarose column, we repeated the purification procedure with an E. coli extract that did not contain His-tagged enzyme, but we failed to detect any phosphatase activity.
Figure 4.
Comparison of the TPS1 terpene products formed by a crude extract of E. coli transformed with tps1 (A) and the purified His-tagged TPS1 protein (B) after incubation with [1-3H]FPP. The radio detector traces from radio GC analysis show that similar amounts of (E)-β-farnesene (1), (E)-nerolidol (2), and (E,E)-farnesol (3) are produced in both assays.
In the presence of GPP, TPS1 catalyzed the formation of the acyclic monoterpenes linalool and geraniol (Fig. 5A). Linalool was identified by comparison of its retention time and mass spectrum (Fig. 5C) with those of a linalool standard (Fig. 5D) and previously published mass spectra (Joulain and König, 1998). A chiral analysis of the linalool product showed that the (3R)-enantiomer was formed almost exclusively (Fig. 3B). Geraniol, like (E,E)-farnesol, was shown to be a natural product of TPS1, following assay of the His-tagged enzyme purified as described above (data not shown).
Figure 5.
Monoterpene products of TPS1. The enzyme was expressed in E. coli, extracted, and incubated with the substrate [1-3H]GPP. The resulting terpene products were separated by radio GC. The traces of the radio detector are shown for the products of the active enzyme (A) and the products formed by extract of E. coli transformed with the expression vector lacking the tps1 gene (B). The products were determined as linalool (1) and geraniol (2). Linalool was identified by comparison of its retention time and mass spectrum (C) with those of an authentic linalool standard (D).
Biochemical Characterization
Because very little information is available about the biochemical properties of the terpene synthases of monocotyledons, we characterized the basic enzymatic features of TPS1. The enzyme exhibited a catalytic optimum at pH 7.0, with half-maximal activities at pH 6.0 and 7.7. The optimal reaction temperature for the enzyme was between 30°C and 37°C. The Km values with GPP and FPP were 1.1 and 1.0 μm, respectively, similar to those found for terpene synthases from nearly all other plants. However, despite the similar Km for these two substrates, the in vitro rate of sesquiterpene formation (19 nmol g−1 total protein) under saturating substrate concentration was about four times higher than the rate of monoterpene formation (4.4 nmol g−1 total protein).
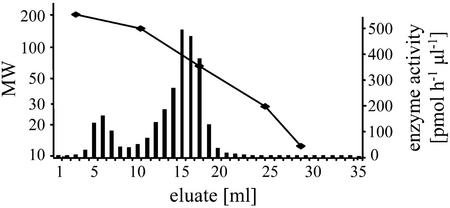
Gel permeation chromatography was used to determine the mass of the active enzyme as 71 ± 5 kD (Fig. 6). This coincides with a mass of 67.4 kD predicted by the amino acid sequence, suggesting that the full-length protein was expressed in E. coli and was not significantly processed or modified after translation. There was also a small peak of activity at approximately 140 kD, indicating that catalytically active homodimers were present in the E. coli extract.
Figure 6.
Size determination of TPS1. The recombinant protein was separated on a size exclusion column. The bars represent the terpene synthase activity of the eluted fractions, and the line indicates the Mr of the fraction determined with standard proteins.
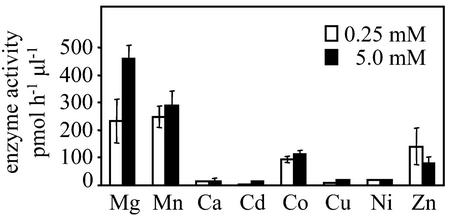
A divalent metal ion cofactor is required for enzyme activity (Fig. 7). Of the divalent cations tested, Mg2+ and Mn2+ ions gave substantial activity, with Mg2+ at 5 mm giving the highest activity measured. The Km values were 470 and 26 μm for Mg2+ and Mn2+, respectively.
Figure 7.
Metal cofactors affect the enzymatic activity of TPS1. The catalytic activity of the partially purified enzyme extract was measured in the presence of various divalent metal ions at 0.25 or 5.0 mm. Means and se of triplicate assays were shown.
TPS1 Products and Volatile Terpene Emission
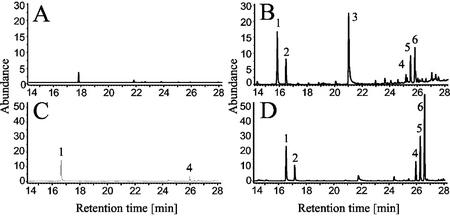
To find out whether the terpene products of TPS1 are constituents of the terpene blend emitted by maize, we analyzed the volatiles emitted from 2-week-old plants after herbivore damage. When plants of the maize cv B73 were attacked by larvae of the lepidopteran Egyptian cotton leafworm (Spodoptera littoralis), (E)-β-farnesene and (3R)-linalool were among the major volatiles released within several hours after damage (Fig. 8, A and B). Both terpenes were also emitted by the hybrid maize cv Delprim (Fig. 8, C and D), which releases higher amounts of terpene volatiles than most other varieties (Gouinguene et al., 2001). However, only traces of (E)-nerolidol, and no (E,E)-farnesol or geraniol, were detected in the headspace of both maize varieties.
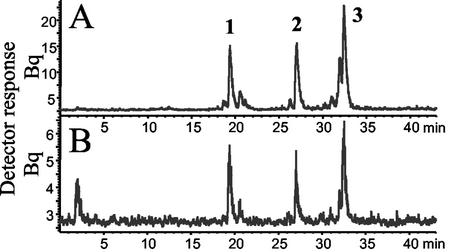
Figure 8.
Volatiles released by maize cvs B73 and Delprim after herbivore attack. Volatiles from 2-week-old plants of maize cv B73 (A and B) and maize cv Delprim (C and D) were collected after no herbivore treatment (A and C) or after 16 h of feeding by Egyptian cotton leafworm (B and D). After separation by GC, the FID traces show: 1, linalool; 2:, DMNT; 3, indole; 4, (E)-α-bergamotene; 5, β-caryophyllene; 6, (E)-β-farnesene.
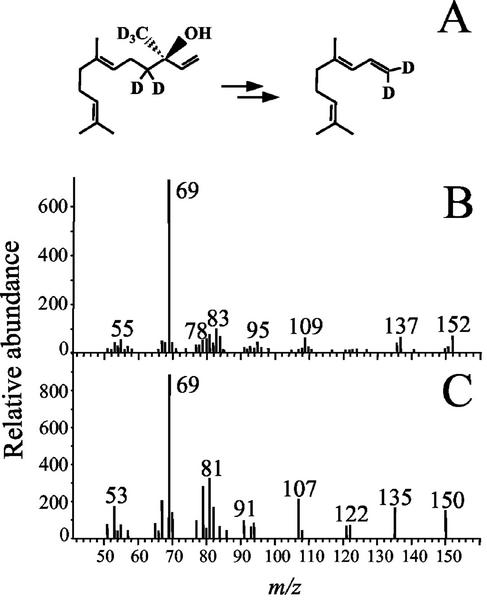
(E)-Nerolidol has been demonstrated to be a pathway intermediate in the formation of DMNT, an irregular terpene with an 11-carbon skeleton, which is a prominent component of the maize cvs B73 and Delprim volatile blends released after herbivore damage (Degenhardt and Gershenzon, 2000). Therefore, the (E)-nerolidol produced by TPS1 might not volatilize directly because of its rapid conversion to DMNT (Donath and Boland, 1994). To test whether the 3R stereoisomer of (E)-nerolidol produced by TPS1 can be converted to DMNT, we fed deuterated [4-2H2, 15-2H3]-(3R)-nerolidol (Gäbler et al., 1991) to cut stems of maize. The labeled compound was taken up by the plants and converted to [1-2H2]DMNT, which was subsequently released into the headspace (Fig. 9). The absence of (E,E)-farnesol or geraniol emission may be attributable to their further metabolism or accumulation. However, accumulated mono- or sesquiterpenes have not been previously found in maize tissues in greater than trace amounts (Buttery et al., 1978).
Figure 9.
Conversion of (3R)-(E)-nerolidol to DMNT in maize. A, [4-2H2, 15-2H3]-(3R)-(E)-nerolidol was administered in aqueous solution to maize cuttings and head space volatiles were analyzed by GC-MS. B, The mass spectrum of the resulting DMNT shows a molecular ion with a m/z of 152, indicating the presence of two deuterium atoms. C, The mass spectrum of DMNT with natural isotopic abundance.
Transcript Levels of tps1 Are Induced by Herbivory in Some Genotypes
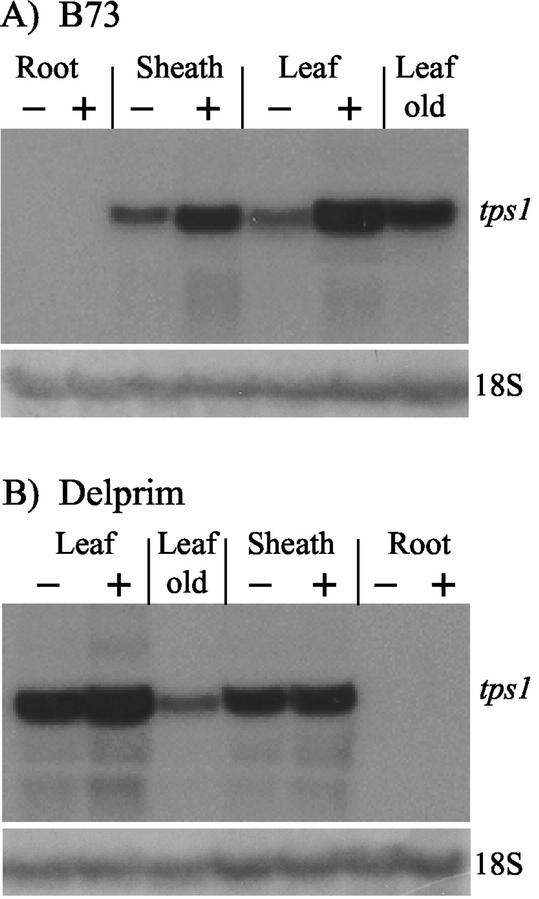
Two different probes were used to detect the level of tps1 transcript in RNA-blot experiments: one representing a part of the coding region, and the other the 3′-untranslated region of tps1. Both probes detected only a single band of about 1.7 kb in both maize cvs B73 and Delprim with comparable densities (data not shown), indicating that the signals seen are specific and represent tps1 and its homolog in maize cv Delprim. Transcripts were detected in the leaf and sheath tissue of 2-week-old uninjured maize plants, but none was found in the roots. After 16 h of herbivory by Egyptian cotton leafworm larvae, transcript levels in the leaves of maize cv B73 were induced about 8-fold. The tps1 transcript levels in the maize cv Delprim were surprisingly high even in uninjured plants and were only marginally elevated by herbivory (Fig. 10). In older plants (3 months), moderate levels of transcript were present in leaves of both herbivore-treated and untreated plants of both cultivars.
Figure 10.
Transcript levels of tps1 in different organs and developmental stages of maize cvs B73 (A) and Delprim (B) after herbivore-damage (+) or in undamaged controls (−). RNA was isolated from plants that were 2 weeks old except for the “leaf old” sample, which was isolated from 3-month-old herbivore-treated plants. The bottom panels show hybridization with an 18S rRNA probe as a control for equal RNA loading.
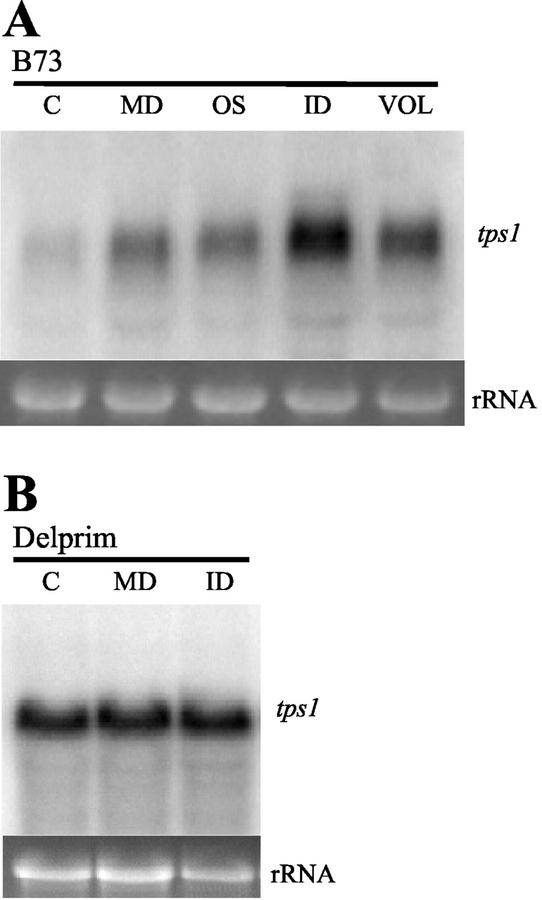
To test which stimuli increase the steady-state level of tps1 mRNA in young plants of maize cv B73, we compared herbivore damage with mechanical damage and treatment with two previously identified elicitors of volatile terpene biosynthesis, regurgitant of Egyptian cotton leafworm and the fatty acid-amino acid conjugate, volicitin (Alborn et al., 1997). Sixteen hours after the initiation of herbivore damage, there was a significant elevation in transcript level (Fig. 11). Mechanically damaged leaves both with and without the elicitor treatments accumulated transcripts to a lesser degree. In maize cv Delprim, the tps1 transcript levels were only marginally affected by the damage treatments.
Figure 11.
The effect of mechanical damage and insect damage on tps1 transcript levels. The transcript level of tps1 was measured in 2-week-old plants of maize cv B73 (A) or maize cv Delprim (B) at 16 h after mechanical damage (MD), feeding by Egyptian cotton leafworm larvae (ID), mechanical damage and application of Egyptian cotton leafworm oral secretions (OS), or mechanical damage and application of volicitin (VOL), with C being the untreated control. As a check of equal RNA loading, the bottom panel shows the 18S rRNA band of each extract on an ethidium bromide-stained agarose gel.
DISCUSSION
The emission of terpene volatiles after herbivore damage is thought to serve as an important part of the indirect defense of maize by attracting predators and parasitoids of the herbivore. To get a better understanding of the defensive function of volatile terpenes in maize and how the formation of volatiles is regulated, we have begun to study the terpene synthases of this species, an enzyme class that catalyzes the committed step in the formation of the different structural classes of terpenes. We isolated a terpene synthase gene from a cDNA library obtained from herbivore-damaged maize leaves that produces three sesquiterpenes, the olefin (E)-β-farnesene and the alcohols (3R)-(E)-nerolidol and (E,E)-farnesol. (E)-β-Farnesene is a prominent component of the herbivore-induced volatile blend of maize, whereas (E)-nerolidol is an intermediate in the formation of DMNT, another major component of this blend.
We named this gene tps1 according to the nomenclature used for terpene synthase genes of several other plant species. The designation also conforms to the standards for maize genetics nomenclature. According to the nomenclature introduced by Trapp and Croteau (2001), the gene would be referred to as ZmfEβfar, ZmfEneroh, or ZmfEEfaroh.
TPS1 Has Properties Similar to Those of Other Terpene Synthases
After expression of the recombinant protein in a bacterial system, TPS1 was found to convert FPP to a mixture of three acyclic sesquiterpene products, (E)-β-farnesene, (E)-nerolidol, and (E,E)-farnesol. Although the formation of multiple products is an unusual feature of enzymes in general, it has often been described for terpene synthases (e.g. Colby et al., 1993; Crock et al., 1997; Wise et al., 1998). The expressed TPS1 was also capable of converting GPP to (3R)-linalool and geraniol in vitro. However, it is likely to function as a sesquiterpene synthase in vivo because it lacks an N-terminal signal peptide for chloroplast targeting suggesting residence in the cytoplasm where FPP is likely to be present in much higher concentration than GPP (Gershenzon and Kreis, 1999; Newman and Chappell, 1999). In addition, the rate of sesquiterpene formation in vitro is four times higher than the rate of monoterpene generation. The ability of sesquiterpene synthases to use GPP as a substrate in vitro is not unprecedented (Crock et al., 1997; Steele et al., 1998).
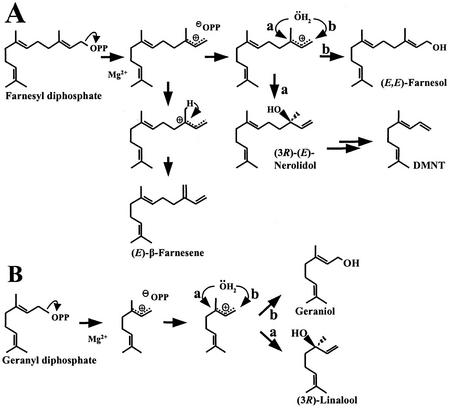
The catalytic mechanism of mono- and sesquiterpene synthases proceeds via a succession of carbocationic intermediates whose formation is initiated by dephosphorylation of GPP or FPP, respectively (Croteau, 1987; Cane, 1990). In the case of TPS1, initial generation of the charge-delocalized carbocation is followed by capture of water forming the sesquiterpene alcohols (3R)-(E)-nerolidol or (E,E)-farnesol. Deprotonation of this cation yields (E)-β-farnesene (Fig. 12).
Figure 12.
Proposed reaction mechanisms for the formation of sesquiterpene (A) and monoterpene (B) products by TPS1.
In this paper, we have described the biochemical characteristics of a terpene synthase from a monocotyledon for the first time, to our knowledge. The basic features of maize TPS1, including pH optimum, Km for the diphosphate substrate, and the requirement for a divalent metal as cofactor are similar to those of other terpene synthases (Rajaonarivony et al., 1992; Williams et al., 1998), despite the low degree of sequence homology to other members of this enzyme class. TPS1 can use Mg2+ or Mn2+ as a cofactor in vitro. Although the Km for Mn2+ is about 18-fold lower than that for Mg2+, the enzyme is likely to employ Mg2+ in vivo because the concentration of Mg2+ in plant cells is on average 50- to 100-fold higher than that of Mn2+ (Marschner, 1998). The ratio of the enzymatic products is the same regardless of whether Mg2+ or Mn2+ serves as a cofactor (data not shown).
The tps1 Sequence Is Only Distantly Related to Those of Other Terpene Synthases
The isolated terpene synthase gene displays a low overall similarity to previously isolated terpene synthases, but the deduced protein contains numerous amino acids throughout its sequence that are highly conserved among members of this gene class (Fig. 1). The most characteristic element is an Asp-rich DDxxD motif in the C-terminal part of the gene (amino acids 333–337) that is involved in the binding of the divalent metal cofactor (Starks et al., 1997). The common ancestry of terpene synthases has often been suggested (McGarvey and Croteau, 1995; Bohlmann et al., 1998; Trapp and Croteau, 2001), because many features of these enzymes and their reaction mechanisms are conserved. Nevertheless, the sequence variation within this gene class is very large, which is reflected in the low amino acid identities between tps1 and other terpene synthase genes. The most closely related gene is the recently identified terpene synthase stc1 from maize, which is reported to be involved in the formation of a naphthalene-type sesquiterpene olefin (Shen et al., 2001). Nevertheless, the amino acid sequence identity shared with tps1 is low (25.1%) and exists mostly in areas that are highly conserved among all plant terpene synthases. Because tps1 shares a similarly low amino acid identity (21.4%) with a terpene synthase from C. breweri, a dicotyledon, and because no maize-specific conserved regions are apparent between tps1, stc1, and an1, the maize terpene synthases gene family likely has a polyphyletic origin. The isolation of additional members of this family is necessary to get a better understanding of the evolution of terpene synthases.
Terpene synthases with catalytic functions similar to TPS1 have been isolated from other plant species, including (E)-β-farnesene synthase from peppermint (Mentha piperita; Crock et al., 1997) and Citrus junos (Maruyama et al., 2001). However, these genes exhibit very little (< 20%) similarity to tps1. A similar divergence between sequence similarity and catalytic function was described for some gymnosperm terpene synthases, where genes with very low similarity encode proteins catalyzing the same reaction (Bohlmann et al., 1998), whereas genes with high similarity (80%) encode proteins catalyzing different reactions (Steele et al., 1998). It is assumed that unrelated terpene synthases forming similar products are the result of convergent evolution (Bohlmann et al., 1998; Cseke et al., 1998). If so, terpene synthases like TPS1 that catalyze reactions with simple mechanisms can be expected to arise more frequently from evolutionary convergence than terpene synthases with more complex mechanisms.
It is noteworthy that the lis1 gene of C. breweri, which also encodes an enzyme with a simple reaction mechanism (Fig. 12), shares sequence identity with tps1. Both enzymes produce linalool, but the lis1 gene product does not form the corresponding sesquiterpene alcohol, nerolidol (Dudareva et al., 1996; J. Degenhardt and J. Gershenzon, unpublished data). A genomic analysis of C. breweri lis1 suggests that this gene is a composite sequence of recent origin (Dudareva et al., 1996; Cseke et al., 1998). The N-terminal section of lis1 is thought to be related to the N terminus of copalyl diphosphate synthase, whereas the C-terminal end is homologous to those of many terpene synthases. The tps1 gene exhibits 25% amino acid identity to the C-terminal one-half of lis1 but has almost no similarity to the N terminus.
Transcript Levels of tps1 Are Regulated by Herbivory in Some Cultivars
Because terpene synthases constitute a key regulatory step of terpene biosynthesis (Vögeli and Chappell, 1988; Degenhardt and Gershenzon, 2000) and the expression of these enzymes is often regulated on the level of transcription (Facchini and Chappell, 1992; Yin et al., 1997; Shen et al., 2001), we anticipated that the steady-state mRNA levels of tps1 in maize would be elevated in response to herbivory. Herbivory by Egyptian cotton leafworm increased tps1 transcript concentrations about 8-fold over the basal level in leaves of the maize cv B73.
Mechanical damage led to a much less pronounced increase in tps1 transcript level, but this increase could be enhanced somewhat by the application of herbivore oral secretions or volicitin to the site of the mechanical wounding. A much stronger activation by volicitin was observed with the maize terpene synthase stc1 (Shen et al., 2001), suggesting that its expression is controlled differently. Volicitin also induces the expression of Igl, a gene encoding indole-3-glycerol phosphate lyase that catalyzes the formation of indole (Frey et al., 2001), a prominent non-terpene volatile released by herbivore-damaged maize (Fig. 8).
The maize hybrid cv Delprim also emits high concentrations of volatiles after herbivore attack but, in contrast to maize cv B73, the transcript level of the tps1 homolog is constitutively high, increasing only marginally after herbivore attack. The high level of transcript in undamaged plants is especially surprising because these plants emit only very low constitutive levels of (E)-β-farnesene or DMNT. The results point toward either a posttranscriptional regulation of tps1 or the corresponding enzyme, or to a control of flux at an earlier step of the pathway. The differential regulation of transcript levels among maize varieties could be attributable to sequence differences in the respective promoter region or to more general differences in the regulation of transcription among different maize cultivars. Interestingly, maize cv Delprim was reported to have an herbivore-inducible enzyme activity for (3S)-(E)-nerolidol formation in in vitro studies (Degenhardt and Gershenzon, 2000). Because TPS1 produces predominantly the (3R)-enantiomer, both enantiomers may be involved in DMNT formation in this variety in vivo because they are both readily converted to DMNT in maize (Fig. 9; Degenhardt and Gershenzon, 2000). Further investigation of DMNT formation in the hybrid maize cv Delprim is under way.
tps1 May Have a Role in Plant Defense
The identification of genes involved in the biosynthesis of two important maize volatiles provides a valuable molecular tool to study the function of volatile terpene biosynthesis in this species. Although the ecological function of herbivore-induced volatiles has often been investigated for the volatile blend as a whole (for review, see Turlings and Benrey, 1998; Dicke, 1999), the specific role of (E)-β-farnesene, DMNT, and other substances within the blend is still unclear. For example, aphids emit (E)-β-farnesene as an alarm pheromone (Pickett and Griffith, 1980), which causes nearby conspecifics to disperse from the area and thus avoid predation. It has been hypothesized that plant-derived (E)-β-farnesene interferes with aphid signaling, although the presence of low levels of (E)-β-farnesene does not necessarily deter settling and feeding by all aphid species (Pickett et al., 1992). Herbivore-induced emissions of maize volatiles repel the corn leaf aphid Rhopalosiphum maidis, but this behavior has not been directly linked to plant-derived (E)-β-farnesene (Bernasconi et al., 1998). The role of plant-derived (E)-β-farnesene as an alarm pheromone in maize might be reduced by the presence of (−)-β-caryophyllene in the volatile blend, which inhibits the alarm response (Dawson et al., 1984; Al Abassi et al., 2000). Furthermore, plant-emitted (E)-β-farnesene did not cause a habituation of pea aphids (Acyrthosiphon pisum), which would disrupt their alarm communication (Petrescu et al., 2001). Thus, the role of (E)-β-farnesene in direct defense against aphids and in indirect defense against herbivores requires further study.
Compared with (E)-β-farnesene, even less is known about the ecological function of DMNT, the irregular C11 terpene olefin for which (3R)-(E)-nerolidol produced by TPS1 is a precursor. DMNT is emitted by many plant species after herbivore attack (Gäbler et al., 1991) and could therefore be a reliable volatile signal for arthropod predators or parasitoids to locate their respective prey or hosts. The role of the third TPS1 product, (E,E)-farnesol, in plant defense is as yet unclear. This is curiously the first characterized sesquiterpene synthase that has been shown to produce (E,E)-farnesol. This acyclic alcohol has been implicated as an endogenous regulator of terpene metabolism in animals, based on its acceleration of the degradation of 3-hydroxy-3-methyl glutaryl-CoA reductase, a key regulatory enzyme of the acetate-mevalonate route of terpene biosynthesis (Goldstein and Brown, 1990; Correll et al., 1994; Meigs and Simoni, 1997). Although such a regulatory role of farnesol has not been demonstrated in plants, enzyme activities that could be involved in controlling the concentration of farnesol by altering its interconversion with FPP have been reported (Thai et al., 1999; Nah et al., 2001). Thus, it is conceivable that herbivore-induced production of (E,E)-farnesol by TPS1 modulates the rate of formation of other terpenoids in a way that alters flux into the biosynthesis of damage-induced volatiles. The genetic manipulation of maize terpene synthesis using tps1 and other members of the terpene synthase gene family should allow testing of this idea as well as the generation of plants with modified terpene emission profiles for ecological studies.
MATERIALS AND METHODS
Plant and Insect Material
Seeds of the maize (Zea mays) inbred line B73 were provided by KWS Seeds (Einbeck, Germany), whereas seeds of the maize hybrid cv Delprim were obtained from Delley Samen und Pflanzen (Delley, Switzerland). Plants were grown in commercially available potting soil in a climate-controlled chamber with a 16-h photoperiod, 1 mmol (m2)−1 s−1 of photosynthetically active radiation, a temperature cycle of 22°C/18°C (day/night), and 65% relative humidity. Twelve- to 15-d-old-plants (20–30 cm high, four–five expanded leaves) were used in all experiments. Eggs of Egyptian cotton leafworm (Spodoptera littoralis Boisd. [Lepidoptera: Noctuidae]) were obtained from Aventis (Frankfurt) and were reared on an artificial wheat germ diet (Heliothis mix, Stonefly Industries, Bryan, TX) for about 10 to 15 d at 22°C under an illumination of 750 μmol m−2 s−1. For the herbivory treatments, three third instar larvae were enclosed on the middle portion of each plant in a cage made of two halves of a petri dish (9-cm diameter) with a circle cut out of each side and covered with gauze to allow for ventilation (Röse et al., 1996). For mechanical damage, the undersides of two young leaves were lightly scraped with a razor blade to each side of the midvein in an area of about 2 × 10 mm. For elicitor treatment, 5 μL of Egyptian cotton leafworm regurgitant or 5 μL of volicitin, 0.6 mg mL−1 in water, was applied to each wounded area (Turlings et al., 1990).
cDNA Library Construction
Ten-day-old maize plants of the cv B73 were subjected to herbivory by Egyptian cotton leafworm for 4 h. One gram of leaf material was ground in a mortar to a fine powder in liquid nitrogen and was added to 10 mL of Trizol Reagent (Invitrogen, Carlsbad, CA). The mixture was treated with a Polytron (Kinematika AG, Luzern, Switzerland) for 1 min and incubated for 3 min on ice. Total RNA was isolated according to manufacturer's instructions. From about 80 μg of total RNA, the mRNA was isolated using poly(T)-coated ferromagnetic beads (Dynal Biotech, Oslo). The mRNA was transcribed into cDNA while constructing a Marathon RACE library according to manufacturer's instructions (BD Biosciences Clontech, Palo Alto, CA).
Isolation of a Maize Terpene Synthase cDNA
Sequences with high similarity to plant terpene synthases were identified in BLAST searches of the Maize Genome Database (Gai et al., 2000) and an EST database maintained by Pioneer Hi-Bred International. Several maize ESTs from the maize cv B73 that contained fragments of a putative terpene synthase gene were aligned to each other to form a 671-bp contig. Sequence alignments were performed with the DNASTAR suite of programs (DNASTAR, Inc., Madison, WI). The identified contig was cloned, sequenced, and extended for 1,510 bp toward the 5′ end by the Marathon RACE procedure (BD Biosciences Clontech) with a cDNA library from herbivore-induced leaves of the maize cv B73. The complete sequence contains an ORF of 1,775 bp and was deposited in GenBank (http://www.ncbi.nlm.nih.gov) with the accession number AF529266.
Heterologous Expression of Terpene Synthases
The ORF of tps1 was cloned as an NdeI-BamHI fragment and inserted into the bacterial expression vector pSBET using the “sticky-end PCR” method of Zeng (1998). For expression with a C-terminal 6× His tag, the ORF was cloned as a NcoI-BamHI fragment into the expression vector pQE60. The constructs were introduced into the Escherichia coli strain BL21 (DE3) and fully sequenced to avoid errors introduced by DNA amplification. When pQE60 was used as expression vector, the plasmid pSBET including the araU gene for tRNAarg4 was cotransformed. Liquid cultures of the bacteria harboring the expression constructs were grown at 37°C to an OD600 of 0.6. Then, isopropyl-β-thiogalactopyranoside was added to a final concentration of 1 mm, and the cultures were incubated for 20 h at 18°C. The cells were collected by centrifugation and disrupted by a 4 × 30 s treatment with a sonicator (Bandelin UW2070, Berlin) in chilled extraction buffer (50 mm MOPSO, pH 7.0, with 5 mm MgCl2, 5 mm sodium ascorbate, 0.5 mm phenylmethylsulfonyl fluoride, 5 mm dithiothreitol, and 10% [v/v] glycerol). The cell fragments were removed by centrifugation at 14,000g, and the supernatant was desalted into assay buffer (10 mm MOPSO, pH 7.0, 1 mm dithiothreitol, and 10% [v/v] glycerol) by passage through a Econopac 10DG column (Bio-Rad, Hercules, CA). The His-tagged enzymes were further purified on a nickel-nitrilotriacetate agarose column (Qiagen, Heidelberg) according to manufacturer's instructions.
Assay for Terpene Synthase Activity
If not mentioned otherwise, each 1-mL assay contained 200 μL of the bacterial extract in assay buffer with 10 μm [1-3H]GPP or [1-3H]-(E,E)-FPP (37 GBq mol−1, American Radiolabeled Chemicals, St. Louis; [1-3H]-(E,E)-FPP contained 10% to 20% of the (Z,E)-isomer), 20 mm MgCl2, 0.2 mm MnCl2, 0.2 mm NaWO4, and 0.1 mm NaF in a Teflon-sealed, 7-mL screw-capped glass test tube. The assay was overlaid with 0.5 mL of pentane to trap volatile products and incubated for 3 h at 30°C. The reaction was stopped by mixing with 2 mL of diethyl ether, and the resulting pentane-ether layer was removed and passed through a short column of MgSO4 or NaSO4 in a Pasteur pipette. The volume of the extracted organic phase was adjusted to 2 mL. For product identification, a set of standard monoterpene and sesquiterpene alcohols (20 μg each of linalool, geraniol, (Z)-nerolidol, (E)-nerolidol, and (E,E)-farnesol, obtained from Sigma-Aldrich, Deisenhofen, Germany) was added to the organic phase. A 100-μL portion of the organic phase was taken for measurement of radioactivity by liquid scintillation counting in 5 mL of Lipoluma cocktail (Packard Bioscience, Groningen, The Netherlands) using a Packard Tricarb 2300TR liquid scintillation counter (3H efficiency = 61%). The remaining organic phase was concentrated to approximately 40 μL for radio GC analysis and product identification. For the determination of metal ion cofactors, Km values, and effects of temperature and pH, a scaled down assay of 100-μL volume was used. The pH optimum was determined in buffers from pH 6.0 to 9.0. Assay results are reported as the mean of three independent replicate assays, and each experiment was repeated two to three times with similar results. The Km values and the pH optimum were determined using seven to nine substrate concentrations with three repetitions each. The enzyme activity was stable for at least 1 week when stored at −80°C. The protein concentration of the extract was determined by the method of Bradford (1976) using the Bio-Rad reagent with bovine serum albumin as standard.
Product Identification
Radio GC was performed on a gas chromatograph (H2 carrier gas, 2 mL min−1; splitless injector temperature, 220°C; injection volume, 2 μL; model 6890, Hewlett-Packard, Palo Alto, CA) with a Chrompack CP-SIL-5 CB-MS column ([5%-phenyl]-methylpolysiloxane, 25-m × 0.25-mm i.d. × 0.25-μm film thickness; Varian Medical Systems, Palo Alto, CA) or a DB-WAX column (polyethylene glycol, 30-m × 0.25-mm i.d. × 0.25-μm film thickness, J&W Scientific, Folsom, CA). Temperature was programmed from 40°C (3-min hold) at 5°C min−1 to 240°C (10-min hold). The outlet of the column was connected to a thermal conductivity detector (TCD) followed by a radioactivity detector (Raga 92, Raytest, Straubenhardt, Germany). Within the radioactivity detector, the eluent (9 mL min−1) was passed through a conversion reactor filled with platinum chips at 740°C. After supplementation with a quench gas, methane (12 mL min−1), the eluent entered two proportional counting tubes (10- and 2-mL volume), connected in series. TCD and radioactivity detector outputs were processed with the Hewlett-Packard Chemstation data system. Radioactive compounds were identified by comparison of their retention times with those of the unlabeled standards recorded with the TCD. The amount of radioactivity in each peak was determined as a percentage of total sample radioactivity (measured by liquid scintillation counting) by comparing peak areas within each run. To correct for losses during sample concentration and injection, peak area was standardized by comparison with the area of the co-injected, unlabeled standards detected with the TCD.
GC-MS was performed on a Hewlett-Packard 6890 gas chromatograph (carrier gas He at 1 mL min−1; column, injection parameters, and temperature program as described above) coupled to a Hewlett-Packard 5973 quadrupole-type mass selective detector with transfer line temperature of 230°C, source temperature of 230°C, quadrupole temperature of 150°C, ionization potential of 70 eV, and a scan range of 50 to 400 atomic mass units. Terpene products from the heterologous expression assays were identified by comparison of retention times with those of authentic standards and by comparison of spectra with those of the standards and the Wiley and National Institute of Standards and Technology libraries. All experiments were repeated at least once to verify the results.
The enantiomers of (E)-nerolidol were separated and identified by radio GC and GC-FID using a heptakis (2,6-di-O-methyl-3 O-pentyl)-β-cyclodextrin column (30-m × 0.25-mm × 0.2-μm film, Machery and Nagel, Düren, Germany) operated with H2 (2 mL min−1) as carrier, splitless injection (220°C, 2 μL volume), and a temperature program from 40°C (1-min hold) at 2.3°C min−1 to 160°C (10-min hold). Standards of (3S)- and (3R)-(E)-nerolidol were kindly provided by Wilhelm Boland (Max-Planck-Institute for Chemical Ecology, Jena, Germany).
Protein Size Determination
Crude bacterial extract (0.5 mL) was separated by FLPC on a Superdex-200 16/6 gel filtration column in MOPSO buffer at pH 7.0. The calibration was performed with a mixture of dextran blue, sweet potato (Ipomoea batatas) β-amylase, yeast alcohol dehydrogenase, bovine serum albumin, bovine carbonic anhydrase, and horse cytochrome c.
Plant Volatile Collection and Analysis
An automated collection system (Analytical Research Systems, Gainesville, FL) based on the design of Heath and Manukian (1994) was employed to analyze plant headspace volatiles. In brief, the aerial portion of a living, potted maize plant was placed in a large glass cylinder (50-cm high × 20-cm diameter) whose base was fitted with two adjustable blades that closed loosely around the stem. Air that had been passed through a charcoal-infused medium for purification and moistened to a relative humidity of 65% entered the chamber from above at a rate of 5 L min−1. After sweeping over the plant, the air exited the chamber through one of a series of collection traps, 150- × 5-mm diameter glass tubes containing 75 mg of Super Q (80/100 mesh, Alltech, Deerfield, IL) arrayed around the base of the chamber. Air was drawn through the traps at a rate of 1 L min−1 by an automated flow controller that could switch from one trap to the next at a designated time. The remaining air escaped through the opening around the stem providing a positive pressure barrier against the entrance of ambient air. The entire volatile collection system was contained in a controlled environment chamber (VB1014/S, Voetsch, Balingen, Germany) set at 25°C, 75% relative humidity, 16-h photoperiod, and 750 μmol m−2 s−1 photosynthetically active radiation.
All collections were performed between 11 am and 1 pm to avoid differences attributable to diurnal rhythms. After the 2-h collection period, the trap was rinsed with 0.2 mL of dichloromethane and the sample was analyzed by GC. A Hewlett-Packard model 6890 instrument was employed with a DB-WAX column (specifications and temperature program as described above) using H2 (2 mL min−1) as carrier, splitless injection (injector temperature, 220°C; injection volume, 2 μL), an FID (250°C), and a temperature program from 40°C (3-min hold) at 5°C min−1 to 240°C (3-min hold). Compounds were identified by comparison of retention times and mass spectra (obtained as described above) with those of authentic standards.
Northern Blotting
Plant RNA was prepared with the RNeasy plant mini kit (Qiagen) according the manufacturer's instructions. Two fragments were used as a probe, the first 682 bp of the coding sequence generated by PCR with the primers 5′-GCCATGCCAGTGAAGCTGACTCCTGC-3′ and 5′-GTAGACGGTCCAATGTGGTGTAGAAG-3′ and a 150-bp fragment of the 3′-untranslated region of the gene amplified with the primers 5′-TTTTTAACGCAAGCCCAAGAGAGC-3′ and 5′-AAAGGAACTAAGCGTCGATGTGATGT-3′. The probes were labeled with [32P]ATP using the Strip-EZ PCR procedure (Ambion, Austin, TX). Blotting on a Nytran-Plus nylon membrane (Schleicher & Schuell, Dassel, Germany), hybridization, and washing were carried out following standard procedures. The blots were exposed to BioMax MS1 film (Eastman-Kodak, Rochester, NY) with an intensifying screen. To verify an even loading of RNA, the blots were hybridized with a 32P-labeled 520-bp fragment of the maize 18S rRNA, which was amplified with the primers 18Sfwd (5′-GTATGGTCGCAAGGCTGAAAC-3′) and 18Srev (5′-TCACCGGACCATTCAATCG-3′). The blots were scanned with a Storm 840 PhosphoImager (Molecular Dynamics, Sunnyvale, CA) for quantification.
Distribution of Materials
Upon request, all novel materials described in this publication will be made available in a timely manner for noncommercial research purposes, subject to the requisite permission from third-party owners of parts of the material. Obtaining permission from the third party owner is the responsibility of the requester.
ACKNOWLEDGMENTS
We are grateful to Robert B. Meeley at Pioneer Hi-Bred International for access to the maize EST libraries, EST supply, and other valuable support. We are also indebted to KWS Seeds for maize cv B73 seeds, Aventis for Egyptian cotton leafworm, and our colleague Wilhelm Boland (Max-Planck-Institute for Chemical Ecology, Jena, Germany) for the donation of volicitin and the deuterated nerolidol substrate and the nerolidol standards.
Footnotes
This work was supported by the Claussen-Simon foundation (fellowship to J.D.).
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.008326.
LITERATURE CITED
- Al Abassi S, Birkett MA, Petterson J, Pickett JA, Wadhams LJ, Woodcock CM. Response of the seven-spot ladybird to an aphid alarm pheromone and an alarm pheromone inhibitor is mediated by paired olfactory cells. J Chem Ecol. 2000;26:1765–1771. [Google Scholar]
- Alborn HT, Turlings TCJ, Jones TH, Stenhagen G, Loughrin JH, Tumlinson JH. An elicitor of plant volatiles from beet armyworm oral secretion. Science. 1997;276:945–949. [Google Scholar]
- Bensen RJ, Johal GS, Crane VC, Tossberg JT, Schnable PS, Meeley RB, Briggs SP. Cloning and characterization of the maize An1 gene. Plant Cell. 1995;7:75–84. doi: 10.1105/tpc.7.1.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernasconi ML, Turlings TCJ, Ambrosetti L, Bassetti P, Dorn S. Herbivore-induced emissions of maize volatiles repel the corn leaf aphid, Rhopalosiphum maidis. Entomol Exp App. 1998;87:133–142. [Google Scholar]
- Bohlmann J, Meyer-Gauen G, Croteau R. Plant terpenoid synthases: molecular biology and phylogenetic analysis. Proc Natl Acad Sci USA. 1998;95:4126–4133. doi: 10.1073/pnas.95.8.4126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Buttery RG, Ling LC, Chan BG. Volatiles of corn kernels and husks: possible corn ear worm attractants. J Agric Food Chem. 1978;26:866–869. [Google Scholar]
- Cane DE. Enzymatic formation of sesquiterpenes. Chem Rev. 1990;90:1089–1103. [Google Scholar]
- Chappell J. The biochemistry and molecular biology of isoprenoid metabolism. Plant Physiol. 1995;107:1–6. doi: 10.1104/pp.107.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colby SM, Alonso WR, Katahira EJ, McGarvey DJ, Croteau R. 4S-limonene synthase from the oil glands of spearmint (Mentha spicata): cDNA isolation, characterization, and bacterial expression of the catalytically active monoterpene cyclase. J Biol Chem. 1993;268:23016–23024. [PubMed] [Google Scholar]
- Correll CC, Ng L, Edwards PA. Identification of farnesol as the non-sterol derivative of mevalonic acid required for the accelerated degradation of 3-hydroxy-3-methylglutaryl-coenzyme A reductase. J Biol Chem. 1994;269:17390–17393. [PubMed] [Google Scholar]
- Crock J, Wildung M, Croteau R. Isolation and bacterial expression of a sesquiterpene synthase cDNA clone from peppermint (Mentha × piperita, L.) that produces the aphid alarm pheromone (E)-β-farnesene. Proc Natl Acad Sci USA. 1997;94:12833–12838. doi: 10.1073/pnas.94.24.12833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croteau R. Biosynthesis and catabolism of monterpenoids. Chem Rev. 1987;87:929–954. [Google Scholar]
- Cseke L, Dudareva N, Pichersky E. Structure and evolution of linalool synthase. Mol Biol Evol. 1998;15:1491–1498. doi: 10.1093/oxfordjournals.molbev.a025876. [DOI] [PubMed] [Google Scholar]
- Dawson GW, Griffith DC, Pickett JA, Smith MC, Woodcock CM. Natural inhibition of aphid alarm pheromone. Entomol Exp Appl. 1984;36:197–199. [Google Scholar]
- Degenhardt J, Gershenzon J. Demonstration and characterization of (E)-nerolidol synthase from maize: a herbivory-inducible terpene synthase participating in (3E)-4,8-dimethyl-1,3,7-nonatriene biosynthesis. Planta. 2000;210:815–822. doi: 10.1007/s004250050684. [DOI] [PubMed] [Google Scholar]
- Dicke M. Evolution of induced indirect defense of plants. In: Harwell CD, Trollian R, editors. The Ecology and Evolution of Inducible Defenses. Princeton, NJ: Princeton University Press; 1999. pp. 62–88. [Google Scholar]
- Donath J, Boland W. Biosynthesis of acyclic homoterpenes in higher plants parallels steroid hormone metabolism. J Plant Physiol. 1994;143:473–478. [Google Scholar]
- Dudareva N, Cseke L, Blanc VL, Pichersky E. Evolution of floral scent in Clarkia: novel patterns of S-linalool synthase gene expression in the C. breweri flower. Plant Cell. 1996;8:1137–1148. doi: 10.1105/tpc.8.7.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Facchini PJ, Chappell J. Gene family for an elicitor-induced sesquiterpene cyclase in tobacco. Proc Natl Acad Sci USA. 1992;89:11088–11092. doi: 10.1073/pnas.89.22.11088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frey M, Stettner C, Pare PW, Schmelz EA, Tumlinson JH, Gierl A. An herbivore elicitor activates the gene for indole emission in maize. Proc Natl Acad Sci USA. 2001;97:14801–14806. doi: 10.1073/pnas.260499897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gäbler A, Boland W, Preiss U, Simon H. Stereochemical studies on homoterpene biosynthesis in higher plants: mechanistic, phylogenetic and ecological aspects. Helv Chem Acta. 1991;74:173–1789. [Google Scholar]
- Gai XW, Lal S, Xing LQ, Brendel V, Walbot V. Gene discovery using the maize genome database ZmDB. Nucleic Acids Res. 2000;28:94–96. doi: 10.1093/nar/28.1.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gershenzon J, Kreis W. Biosynthesis of monoterpenes, sesquiterpenes, diterpenes, sterols, cardiac glycosides and steroid saponins. In: Wink M, editor. Biochemistry of Plant Secondary Metabolism, Annual Plant Reviews. Vol. 2. Sheffield, UK: Sheffield Academic Press; 1999. pp. 222–229. [Google Scholar]
- Goldstein JL, Brown MS. Regulation of the mevalonate pathway. Nature. 1990;343:425–430. doi: 10.1038/343425a0. [DOI] [PubMed] [Google Scholar]
- Gouinguene S, Degen T, Turlings TCJ. Variability in herbivore-induced odour emissions among maize cultivars and their wild ancestors (teosinte) Chemoecology. 2001;11:9–16. [Google Scholar]
- Heath B, Manukian A. An automated system for use in collecting volatile chemicals released from plants. J Chem Ecol. 1994;18:1209–1226. doi: 10.1007/BF02059600. [DOI] [PubMed] [Google Scholar]
- Hoballah MEF, Turlings TCJ. Experimental evidence that plants under caterpillar attack may benefit from attracting parasitoids. Evol Ecol Res. 1999;3:553–565. [Google Scholar]
- Joulain D, König WA. The atlas of spectral data of sesquiterpene hydrocarbons. E.B.-Verlag, Germany: Hamburg; 1998. [Google Scholar]
- Kessler A, Baldwin IT. Defensive function of herbivore-induced plant volatile emissions in nature. Science. 2001;291:2141–2144. doi: 10.1126/science.291.5511.2141. [DOI] [PubMed] [Google Scholar]
- Marschner H. Mineral Nutrition of Higher Plants. Ed 2. San Diego: Academic Press; 1998. [Google Scholar]
- Marrero PF, Poulter CD, Edwards PA. Effects of site-directed mutagenesis of the highly conserved aspartate residues in domain II of fanesyl diphosphate synthase activity. J Biol Chem. 1992;267:21873–21878. [PubMed] [Google Scholar]
- Maruyama T, Ito M, Honda G. Molecular cloning, functional expression and characterization of (E)-β-farnesene synthase from Citrus junos. Biol Pharm Bull. 2001;24:1171–1175. doi: 10.1248/bpb.24.1171. [DOI] [PubMed] [Google Scholar]
- McGarvey DJ, Croteau R. Terpenoid metabolism. Plant Cell. 1995;7:1015–1026. doi: 10.1105/tpc.7.7.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meigs TE, Simoni RD. Farnesol as a regulator of HMG-CoA reductase degradation: characterization and role of farnesyl pyrophosphatase. Arch Biochem Biophys. 1997;34:1–9. doi: 10.1006/abbi.1997.0200. [DOI] [PubMed] [Google Scholar]
- Nah J, Song S-J, Back K. Partial charaterization of farnesyl and geranylgeranyl diphosphatases induced in rice seedlings by UV-C radiation. Plant Cell Physiol. 2001;42:864–867. doi: 10.1093/pcp/pce102. [DOI] [PubMed] [Google Scholar]
- Newman JD, Chappell J. Isoprenoid biosynthesis in plants: carbon partitioning within the cytoplasmic pathway. Crit Rev Biochem Mol Biol. 1999;34:95–106. doi: 10.1080/10409239991209228. [DOI] [PubMed] [Google Scholar]
- Petrescu AS, Mondor EB, Roitberg BD. Subversion of alarm communication: do plants habituate aphids to their own alarm signals? Can J Zool. 2001;79:737–740. [Google Scholar]
- Pickett JA, Griffith DC. Composition of aphid alarm pheromones. J Chem Ecol. 1980;6:349–360. [Google Scholar]
- Pickett JA, Wadhams LJ, Woodcock CM, Hardie J. The chemical ecology of aphids. Annu Rev Entomol. 1992;37:67–90. [Google Scholar]
- Rajaonarivony JIM, Gershenzon J, Miyazaki J, Croteau R. Characterization and mechanism of 4S limonene synthase, a monoterpene cyclase from the glandular trichomes of peppermint. Arch Biochem Biophys. 1992;299:77–82. doi: 10.1016/0003-9861(92)90543-6. [DOI] [PubMed] [Google Scholar]
- Röse USR, Manukian A, Heath RR, Tumlinson JH. Volatile semiochemicals released from undamaged cotton leaves. Plant Physiol. 1996;111:487–495. doi: 10.1104/pp.111.2.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah FH, Cha TS. A mesocarp- and species-specific cDNA clone from oil palm encodes for sesquiterpene synthase. Plant Sci. 2000;154:153–160. doi: 10.1016/s0168-9452(99)00270-8. [DOI] [PubMed] [Google Scholar]
- Shen B, Zheng Z, Dooner HK. A maize sesquiterpene cyclase gene induced by insect herbivory and volicitin: characterization of wild-type and mutant alleles. Proc Natl Acad Sci USA. 2001;97:14801–14806. doi: 10.1073/pnas.240284097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starks CM, Back K, Chappell J, Noel JP. Structural basis for cyclic terpene biosynthesis by tobacco 5-epi-aristolochene synthase. Science. 1997;277:1815–1820. doi: 10.1126/science.277.5333.1815. [DOI] [PubMed] [Google Scholar]
- Steele CL, Crock J, Bohlmann J, Croteau R. Sesquiterpene synthases from grand fir (Abies grandis): comparison of constitutive and wound-inducible activities, and cDNA isolation, characterization, and bacterial expression of δ-selinene synthase and γ-humulene synthase. J Biol Chem. 1998;273:2078–2089. doi: 10.1074/jbc.273.4.2078. [DOI] [PubMed] [Google Scholar]
- Thai L, Rush JS, Maul JE, Devarenne T, Rodgers DL, Chappell J, Waechter CJ. Farnesol is utilized for isoprenoid biosynthesis in plant cells via farnesyl pyrophosphate formed by successive monophosphorylation reactions. Proc Natl Acad Sci USA. 1999;96:13080–13085. doi: 10.1073/pnas.96.23.13080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trapp SC, Croteau RB. Genomic organization of plant terpene synthases and molecular evolutionary implications. Genetics. 2001;158:811–832. doi: 10.1093/genetics/158.2.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turlings TCJ, Benrey B. Effects of plant metabolites on behavior and development of parasitic wasps. Ecoscience. 1998;5:321–333. [Google Scholar]
- Turlings TCJ, Tumlinson JH, Heath RB, Proveaux AT, Doolittle RE. Isolation and identification of allelochemicals that attract the larval parasitoid, Cotesia marginiventris (Cresson), to the microhabitat of one of its hosts. J Chem Ecol. 1991;17:2235–2251. doi: 10.1007/BF00988004. [DOI] [PubMed] [Google Scholar]
- Turlings TCJ, Tumlinson JH, Lewis WJ. Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science. 1990;250:1251–1253. doi: 10.1126/science.250.4985.1251. [DOI] [PubMed] [Google Scholar]
- Vögeli U, Chappell J. Induction of sesquiterpene cyclase and suppression of squalene synthetase activities in plant cell cultures treated with fungal elicitor. Plant Physiol. 1988;88:1291–1296. doi: 10.1104/pp.88.4.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams DC, McGarvey DJ, Katahira EJ, Croteau R. Truncation of limonene synthase preprotein provides a fully active “pseudomature” form of this monoterpene cyclase and reveals the function of the amino-terminal arginine pair. Biochemistry. 1998;37:12213–12220. doi: 10.1021/bi980854k. [DOI] [PubMed] [Google Scholar]
- Wise ML, Savage TJ, Katahira E, Croteau R. Monoterpene synthases from common sage (Salvia officinalis) J Biol Chem. 1998;273:14891–14899. doi: 10.1074/jbc.273.24.14891. [DOI] [PubMed] [Google Scholar]
- Yin S, Mei L, Newman J, Back K, Chappell J. Regulation of sesquiterpene cyclase gene expression. Plant Physiol. 1997;115:437–451. doi: 10.1104/pp.115.2.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng G. Sticky-end PCR: a new method for subcloning. BioTechniques. 1998;25:206–208. doi: 10.2144/98252bm05. [DOI] [PubMed] [Google Scholar]