Abstract
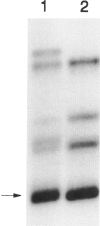
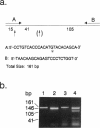
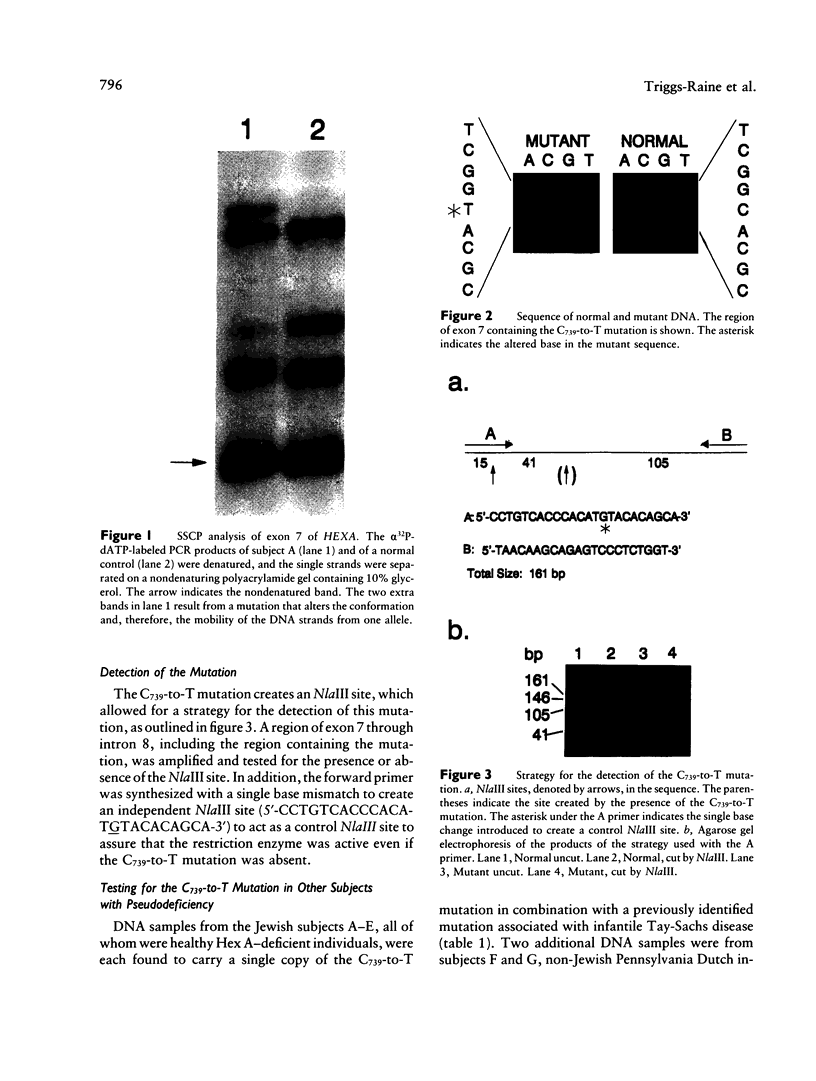
Deficiency of β-hexosaminidase A (Hex A) activity typically results in Tay-Sachs disease. However, healthy subjects found to be deficient in Hex A activity (i.e., pseudodeficient) by means of in vitro biochemical tests have been described. We analyzed the HEXA gene of one pseudodeficient subject and identified both a C739-to-T substitution that changes Arg247→Trp on one allele and a previously identified Tay-Sachs disease mutation on the second allele. Six additional pseudodeficient subjects were found to have the C739-to-T mutation. This allele accounted for 32% (20/62) of non-Jewish enzyme-defined Tay-Sachs disease carriers but for none of 36 Jewish enzyme-defined carriers who did not have one of three known mutations common to this group. The C739-to-T allele, together with a “true” Tay-Sachs disease allele, causes Hex A pseudodeficiency. Given both the large proportion of non-Jewish carriers with this allele and that standard biochemical screening cannot differentiate between heterozygotes for the C739-to-T mutations and Tay-Sachs disease carriers, DNA testing for this mutation in at-risk couples is essential. This could prevent unnecessary or incorrect prenatal diagnoses.
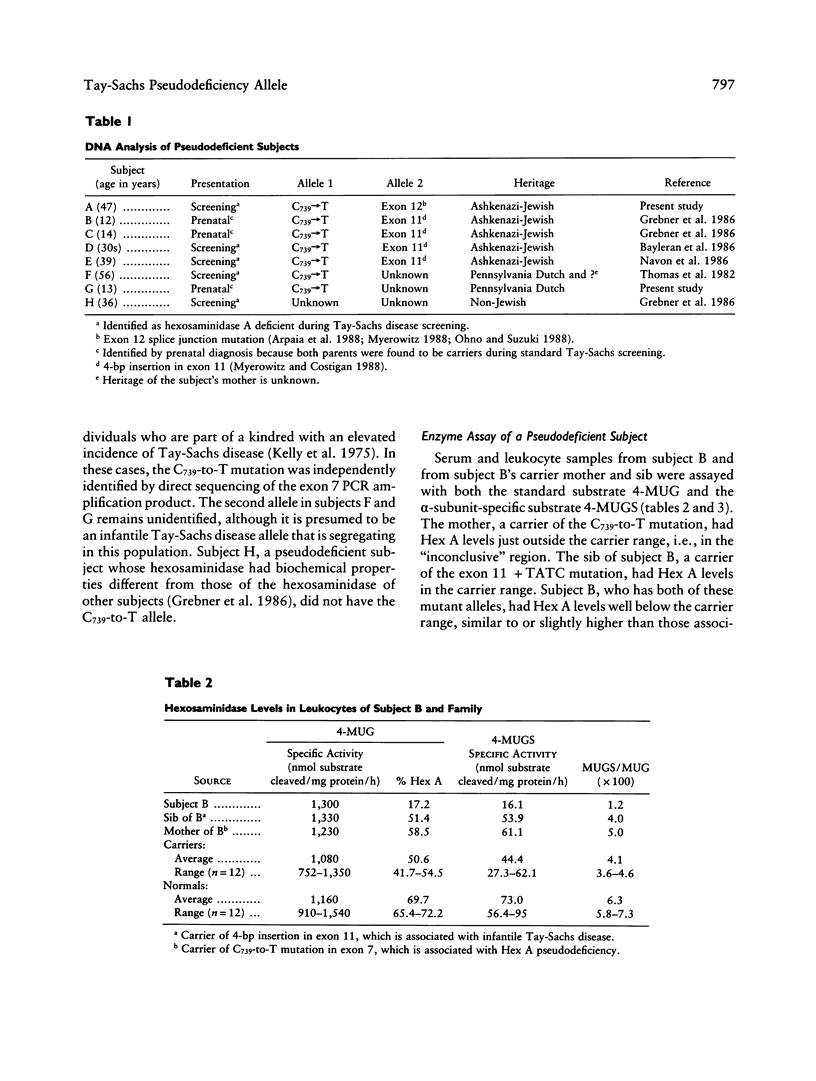
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akli S., Chelly J., Lacorte J. M., Poenaru L., Kahn A. Seven novel Tay-Sachs mutations detected by chemical mismatch cleavage of PCR-amplified cDNA fragments. Genomics. 1991 Sep;11(1):124–134. doi: 10.1016/0888-7543(91)90109-r. [DOI] [PubMed] [Google Scholar]
- Arpaia E., Dumbrille-Ross A., Maler T., Neote K., Tropak M., Troxel C., Stirling J. L., Pitts J. S., Bapat B., Lamhonwah A. M. Identification of an altered splice site in Ashkenazi Tay-Sachs disease. Nature. 1988 May 5;333(6168):85–86. doi: 10.1038/333085a0. [DOI] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Geiger B., Arnon R. Chemical characterization and subunit structure of human N-acetylhexosaminidases A and B. Biochemistry. 1976 Aug 10;15(16):3484–3493. doi: 10.1021/bi00661a014. [DOI] [PubMed] [Google Scholar]
- Gravel R. A., Triggs-Raine B. L., Mahuran D. J. Biochemistry and genetics of Tay-Sachs disease. Can J Neurol Sci. 1991 Aug;18(3 Suppl):419–423. doi: 10.1017/s0317167100032583. [DOI] [PubMed] [Google Scholar]
- Grebner E. E., Mansfield D. A., Raghavan S. S., Kolodny E. H., d'Azzo A., Neufeld E. F., Jackson L. G. Two abnormalities of hexosaminidase A in clinically normal individuals. Am J Hum Genet. 1986 Apr;38(4):505–514. [PMC free article] [PubMed] [Google Scholar]
- Grebner E. E., Tomczak J. Distribution of three alpha-chain beta-hexosaminidase A mutations among Tay-Sachs carriers. Am J Hum Genet. 1991 Mar;48(3):604–607. [PMC free article] [PubMed] [Google Scholar]
- Greenberg D. A., Kaback M. M. Estimation of the frequency of hexosaminidase a variant alleles in the American Jewish population. Am J Hum Genet. 1982 May;34(3):444–451. [PMC free article] [PubMed] [Google Scholar]
- Hoar D. I., Haslam D. B., Starozik D. M. Improved direct molecular diagnosis and rapid fetal sexing. Prenat Diagn. 1984 Jul-Aug;4(4):241–247. doi: 10.1002/pd.1970040402. [DOI] [PubMed] [Google Scholar]
- Kelly T. E., Chase G. A., Kaback M. M., Kumor K., McKusick V. A. Tay-Sachs disease: high gene frequency in a non-Jewish population. Am J Hum Genet. 1975 May;27(3):287–291. [PMC free article] [PubMed] [Google Scholar]
- Kelly T. E., Reynolds L. W., O'Brien J. S. Segregation within a family of two mutant alleles for hexosaminidase A. Clin Genet. 1976 May;9(5):540–543. doi: 10.1111/j.1399-0004.1976.tb01609.x. [DOI] [PubMed] [Google Scholar]
- Kytzia H. J., Sandhoff K. Evidence for two different active sites on human beta-hexosaminidase A. Interaction of GM2 activator protein with beta-hexosaminidase A. J Biol Chem. 1985 Jun 25;260(12):7568–7572. [PubMed] [Google Scholar]
- Landels E. C., Ellis I. H., Fensom A. H., Green P. M., Bobrow M. Frequency of the Tay-Sachs disease splice and insertion mutations in the UK Ashkenazi Jewish population. J Med Genet. 1991 Mar;28(3):177–180. doi: 10.1136/jmg.28.3.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahuran D., Lowden J. A. The subunit and polypeptide structure of hexosaminidases from human placenta. Can J Biochem. 1980 Apr;58(4):287–294. doi: 10.1139/o80-038. [DOI] [PubMed] [Google Scholar]
- Myerowitz R. Splice junction mutation in some Ashkenazi Jews with Tay-Sachs disease: evidence against a single defect within this ethnic group. Proc Natl Acad Sci U S A. 1988 Jun;85(11):3955–3959. doi: 10.1073/pnas.85.11.3955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myrianthopoulos N. C., Aronson S. M. Population dynamics of Tay-Sachs disease. I. Reproductive fitness and selection. Am J Hum Genet. 1966 Jul;18(4):313–327. [PMC free article] [PubMed] [Google Scholar]
- Navon R., Argov Z., Frisch A. Hexosaminidase A deficiency in adults. Am J Med Genet. 1986 May;24(1):179–196. doi: 10.1002/ajmg.1320240123. [DOI] [PubMed] [Google Scholar]
- Neote K., Bapat B., Dumbrille-Ross A., Troxel C., Schuster S. M., Mahuran D. J., Gravel R. A. Characterization of the human HEXB gene encoding lysosomal beta-hexosaminidase. Genomics. 1988 Nov;3(4):279–286. doi: 10.1016/0888-7543(88)90116-4. [DOI] [PubMed] [Google Scholar]
- O'Brien J. S., Tennant L., Veath M. L., Scott C. R., Bucknall W. E. Characterization of unusual hexosaminidase A (HEX A) deficient human mutants. Am J Hum Genet. 1978 Nov;30(6):602–608. [PMC free article] [PubMed] [Google Scholar]
- Ohno K., Suzuki K. A splicing defect due to an exon-intron junctional mutation results in abnormal beta-hexosaminidase alpha chain mRNAs in Ashkenazi Jewish patients with Tay-Sachs disease. Biochem Biophys Res Commun. 1988 May 31;153(1):463–469. doi: 10.1016/s0006-291x(88)81247-6. [DOI] [PubMed] [Google Scholar]
- Orita M., Suzuki Y., Sekiya T., Hayashi K. Rapid and sensitive detection of point mutations and DNA polymorphisms using the polymerase chain reaction. Genomics. 1989 Nov;5(4):874–879. doi: 10.1016/0888-7543(89)90129-8. [DOI] [PubMed] [Google Scholar]
- Paw B. H., Tieu P. T., Kaback M. M., Lim J., Neufeld E. F. Frequency of three Hex A mutant alleles among Jewish and non-Jewish carriers identified in a Tay-Sachs screening program. Am J Hum Genet. 1990 Oct;47(4):698–705. [PMC free article] [PubMed] [Google Scholar]
- Paw B. H., Wood L. C., Neufeld E. F. A third mutation at the CpG dinucleotide of codon 504 and a silent mutation at codon 506 of the HEX A gene. Am J Hum Genet. 1991 Jun;48(6):1139–1146. [PMC free article] [PubMed] [Google Scholar]
- Petersen G. M., Rotter J. I., Cantor R. M., Field L. L., Greenwald S., Lim J. S., Roy C., Schoenfeld V., Lowden J. A., Kaback M. M. The Tay-Sachs disease gene in North American Jewish populations: geographic variations and origin. Am J Hum Genet. 1983 Nov;35(6):1258–1269. [PMC free article] [PubMed] [Google Scholar]
- Proia R. L. Gene encoding the human beta-hexosaminidase beta chain: extensive homology of intron placement in the alpha- and beta-chain genes. Proc Natl Acad Sci U S A. 1988 Mar;85(6):1883–1887. doi: 10.1073/pnas.85.6.1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proia R. L., Soravia E. Organization of the gene encoding the human beta-hexosaminidase alpha-chain. J Biol Chem. 1987 Apr 25;262(12):5677–5681. [PubMed] [Google Scholar]
- Robinson D., Stirling J. L. N-Acetyl-beta-glucosaminidases in human spleen. Biochem J. 1968 Apr;107(3):321–327. doi: 10.1042/bj1070321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava S. K., Wiktorowicz J. E., Awasthi Y. C. Interrelationship of hexosaminidases A and B: conformation of the common and the unique subunit theory. Proc Natl Acad Sci U S A. 1976 Aug;73(8):2833–2837. doi: 10.1073/pnas.73.8.2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka A., Ohno K., Sandhoff K., Maire I., Kolodny E. H., Brown A., Suzuki K. GM2-gangliosidosis B1 variant: analysis of beta-hexosaminidase alpha gene abnormalities in seven patients. Am J Hum Genet. 1990 Feb;46(2):329–339. [PMC free article] [PubMed] [Google Scholar]
- Thomas G. H., Raghavan S., Kolodny E. H., Frisch A., Neufeld E. F., O'Brien J. S., Reynolds L. W., Miller C. S., Shapiro J., Kazazian H. H., Jr Nonuniform deficiency of hexosaminidase A in tissues and fluids of two unrelated individuals. Pediatr Res. 1982 Mar;16(3):232–237. doi: 10.1203/00006450-198203000-00014. [DOI] [PubMed] [Google Scholar]
- Triggs-Raine B. L., Akerman B. R., Clarke J. T., Gravel R. A. Sequence of DNA flanking the exons of the HEXA gene, and identification of mutations in Tay-Sachs disease. Am J Hum Genet. 1991 Nov;49(5):1041–1054. [PMC free article] [PubMed] [Google Scholar]
- Triggs-Raine B. L., Feigenbaum A. S., Natowicz M., Skomorowski M. A., Schuster S. M., Clarke J. T., Mahuran D. J., Kolodny E. H., Gravel R. A. Screening for carriers of Tay-Sachs disease among Ashkenazi Jews. A comparison of DNA-based and enzyme-based tests. N Engl J Med. 1990 Jul 5;323(1):6–12. doi: 10.1056/NEJM199007053230102. [DOI] [PubMed] [Google Scholar]
- Vidgoff J., Buist N. R., O'Brien J. S. Absence of -N-acetyl-D-hexosaminidase A activity in a healthy woman. Am J Hum Genet. 1973 Jul;25(4):372–381. [PMC free article] [PubMed] [Google Scholar]