Abstract
In Saccharomyces cerevisiae meiosis, recombination occurs frequently between sequences at the same location on homologs (allelic recombination) and can take place between dispersed homologous sequences (ectopic recombination). Ectopic recombination occurs less often than does allelic, especially when homologous sequences are on heterologous chromosomes. To account for this, it has been suggested that homolog pairing (homolog colocalization and alignment) either promotes allelic recombination or restricts ectopic recombination. The latter suggestion was tested by examining ectopic recombination in two cases where normal interhomolog relationships are disrupted. In the first case, one member of a homolog pair was replaced by a homoeologous (related but not identical) chromosome that has diverged sufficiently to prevent allelic recombination. In the second case, ndj1 mutants were used to delay homolog pairing and synapsis. Both circumstances resulted in a substantial increase in the frequency of ectopic recombination between arg4-containing plasmid inserts located on heterologous chromosomes. These findings suggest that, during normal yeast meiosis, progressive homolog colocalization, alignment, synapsis, and allelic recombination restrict the ability of ectopically located sequences to find each other and recombine. In the absence of such restrictions, the meiotic homology search may encompass the entire genome.
Keywords: homolog pairing, sequence divergence, NDJ1
During prophase of meiosis I, chromosomes move from a state of relative dispersal to a highly organized paired conformation. Sequences on one parental homolog are brought close to corresponding sequences on the other, resulting in end-to-end homolog alignment. We will refer to this progressive process as homolog pairing. Pairing culminates at pachytene, when coaligned homologs are tightly synapsed by the synaptonemal complex (reviewed in ref. 1). Homolog juxtaposition can also occur in nonmeiotic cells and ranges in extent from the close homolog alignment seen in Drosophila (2) to the intermittent colocalization of short chromosome segments seen in mitotic and premeiotic Saccharomyces cerevisiae cells (3, 4). In S. cerevisiae, segmental colocalization disappears during meiotic S phase. Restoration of colocalization is nonuniform and gradual, with homolog segments colocalized or even synapsed when the bulk of chromosomes appear to be dispersed (4, 5). In other organisms, end-to-end homolog alignment can occur at times when most chromosomal sequences are still separated by a significant distance (6–9). Alignment at a distance may also occur in Saccharomyces, but the nonuniform homolog pairing seen in this organism precludes its detection.
Meiosis is also characterized by elevated frequencies of genetic recombination, including both nonreciprocal (gene conversion) and reciprocal (crossing-over) events. Meiotic recombination in S. cerevisiae (and most likely in other organisms) is initiated via the formation and subsequent repair of double-strand DNA breaks (DSBs) induced early in meiosis I prophase (1, 10–13). Recombination usually occurs at levels sufficient to ensure at least one crossover per homolog pair and is needed to ensure correct homolog disjunction during the first meiotic division (14). The contribution of recombination to meiotic homolog colocalization and alignment remains an issue of some controversy. Recombination is clearly dispensable for pairing in Drosophila and in Caenorhabditis elegans, inasmuch as recombination-null mutants display apparently normal synapsis at pachytene (12, 13). In contrast, S. cerevisiae Rec− mutants display defects in homolog pairing and synapsis (1, 4, 15, 16). It remains to be determined whether these defects reflect an obligate role for recombination in homolog pairing, a checkpoint-mediated block to meiotic progression in the absence of recombination, or a structural role for recombination proteins or intermediates in synaptonemal complex assembly (17–19).
Aspects of the relationship between meiotic recombination and homolog pairing are revealed by studies of ectopic recombination (20–24). Ectopic recombination occurs during S. cerevisiae meiosis but is limited by chromosomal context. For example, we have shown that recombination between arg4 inserts located on heterologous chromosomes is 10- to 20-fold reduced relative to interhomolog recombination between inserts at allelic loci (22). Inserts located on homologs at sites about 20 kb apart recombine as efficiently as do allelic inserts, and the likelihood of ectopic recombination declines with increasing distance between insert loci. These results were interpreted as suggesting that most meiotic recombination occurs at a time when homologs are colocalized and are in end-to-end alignment.
Two possible explanations could account for the reduced efficiency of ectopic relative to allelic recombination seen in S. cerevisiae meiosis. The first explanation suggests that multiple interhomolog interactions are needed for efficient recombination between homologous sequences; the lack of such interactions between heterologous chromosomes would reduce the likelihood of interheterolog ectopic recombination. The second explanation suggests that homologous sequences, unless otherwise constrained, would find each other and efficiently recombine irrespective of their location. In this view, homolog pairing and synapsis would reduce the ability of dispersed sequences to find each other during meiosis, while preserving the ability of allelic sequences to recombine.
We examined meiotic recombination between dispersed copies of the ARG4 gene in two situations where normal interhomolog interactions might be disrupted. In the first, ectopic recombination was measured in hybrid S. cerevisiae strains containing one or two homoeologous chromosomes from Saccharomyces carlsbergensis. These homoeologous chromosomes are 15% diverged in coding sequences, with even greater differences seen in intergenic regions (ref. 25; T. Nilsson-Tillgren, personal communication). This sequence divergence severely reduces (by at least 100-fold) rates of mitotic and meiotic recombination between diverged S. cerevisiae and S. carlsbergensis sequences (26–28). Furthermore, studies using LacI-green fluorescent protein fusions bound to the arms of chromosomes V from S. cerevisiae and S. carlsbergensis indicate that these two diverged chromosomes do not undergo end-to-end alignment in meiosis I prophase (R. Boumil, B. Kemp, and D. Dawson, personal communication). Consistent with this lack of alignment and recombination, homoeologous S. cerevisiae–S. carlsbergensis chromosome pairs display a level of nondisjunction typical of that produced by the distributive disjunction system that segregates single pairs of nonhomologous chromosomes (refs. 29 and 30 and B. Kemp and D. Dawson, personal communication; unpublished data).
Ectopic recombination was also examined in strains homozygous for a loss-of-function ndj1 mutation. Ndj1p is produced early in meiosis and is located primarily at chromosome ends. ndj1 mutants display defects in telomere clustering (bouquet stage), which normally occurs early in meiosis I prophase (ref. 31; H. Scherthan, personal communication). Perhaps as a result, homolog colocalization is reduced early in meiosis I prophase (H. Scherthan, personal communication), and complete synapsis is delayed relative to wild type (32, 33). Genetic studies of haploid meiosis in ndj1 strains are also consistent with a defect in homolog pairing (34). Nevertheless, allelic gene conversion and crossing over occur at near-normal frequencies in ndj1 mutants (32, 33).
We report here that both of these disruptions in normal homolog interactions result in a significant increase in ectopic recombination between sequences on affected chromosomes. This finding is consistent with the view that meiotic homolog pairing and synapsis, accompanied by allelic recombination, serve to limit an otherwise promiscuous homology search, thus actively discouraging ectopic interactions between homologous sequences on heterologous chromosomes.
Materials and Methods
Strains.
All yeast strains (the complete list will be supplied upon request) are congenic to SK1 (35). The S. carlsbergensis chromosome V (abbreviated Vcarl), marked with ilv1, was introduced into SK1 by S. Priebe and M. Resnick (National Institute on Environmental Health Sciences, Triangle Park, NC), who performed five backcrosses; we performed five more backcrosses. All hybrid diploids containing chromosome Vcarl were homozygous for an unlinked single-locus modifier, not present in SK1, that is necessary for germination of ilv1 spores. The S. carlsbergensis chromosome IIIcarl was provided in an SK1 background by C. Newlon (University of Medicine and Dentistry, New Jersey Medical School, Newark, NJ) and was introduced by using standard crosses. URA3-arg4 inserts on S. cerevisiae chromosomes (at LEU2, MAT, CHA1, URA3, PHO11, PHO12, and PUT2) have been described (22, 36) and were introduced during backcrossing. A MAT∷URA3-arg4-bgl insert on chromosome IIIcarl was created by transformation as described (37), except that an EcoNI/BglII digest was used to direct integration. ppx1∷URA3-arg4 inserts were made by using plasmids pMJ443 and pMJ444, which contain chromosome VIII sequences from bp 501400 to bp 503284 at the EcoRI site of PMJ113 and PMJ115, respectively (36), and were inserted at PPX1 as described (22). Diploids are homozygous for lys2 ho∷LYS2 and for arg4-nsp,bgl at the normal ARG4 locus. All except those with IIIcarl are leu2-K/leu2-R. Strains containing the rad50S (rad50-K81I) allele (38) were constructed as described (36). Haploid ndj1∷KanMX6 strains were obtained by transformation with a digest of plasmid MCB253, a gift of M. Conrad and M. Dresser (Oklahoma Medical Research Foundation, Oklahoma City, OK).
Genetic Methods.
ARG4 and LEU2 recombination frequencies and efficiencies were determined as described (22). At least two independent determinations were made. Crossover-associated ARG4 recombinants were detected in allelic crosses with inserts at MAT by scoring for nonmating haploids (39) and in LEU2 × URA3 or PPX1 × URA3 ectopic crosses by pulsed-field gel analysis (22). In diploids used to determine crossover association, the two inserts had the same orientation relative to their centromeres, thus allowing the recovery of viable translocations containing crossover-associated ARG4 recombinants.
Species-Specific PCR.
Primers used to detect unique sequences at the left end of chromosome V of S. cerevisiae (Vcere) were TGTTGCTCAGTTAACTGCCGAGGC (bp 16373–16396) and GTTGGTTCTCCAGAGGGAAAGGC (bp 17262–17243); primers for the right end were CGATCAAATCGTGGCAGCGG (bp 566235–566254) and CTCCAGTGTTCTGAGCACCG (bp 566641–566622). Coordinates are from the chromosome V sequence [J. M. Cherry, C. Ball, K. Dolinski, S. Dwight, M. Harris, J. C. Matese, G. Sherlock, G. Binkley, H. Jin, S. Weng, and D. Botstein (2000) Saccharomyces Genome Database, http://genome-www.stanford.edu/Saccharomyces/].
DSB Quantitation.
DSBs were detected in DNA from meiotic rad50S/rad50S diploids by Southern blotting, as described (36). A Fuji BAS2000 phosphorimager and macbas 2.5 software were used to quantify radioactivity on blots.
Results
The experiments reported here examine the relationship between meiotic homolog pairing and meiotic recombination, specifically the impact of interhomolog interactions on ectopic recombination. We determined the effect of either homoeology or an ndj1 mutation on the frequency of ARG4 spores produced by ectopic recombination between arg4 mutant alleles (arg4-nsp and arg4-bgl) present on dispersed pairs of an 8-kb URA3-arg4 insert (Fig. 1a). DSBs occur at three places in the insert at all loci, and comparisons of DSB and ARG4 recombinant frequencies indicate that most if not all meiotic ARG4 recombinants are induced by breaks formed in the insert (22, 36, 40). Under normal circumstances, a DSB break formed during meiosis can be repaired by interhomolog allelic recombination, by sister chromatid recombination, or by ectopic recombination with homologous sequences if they are present elsewhere in the genome. The current study used inserts that were hemizygous (present on only one member of a homolog or homoeolog pair) and were located on heterologous chromosomes (Fig. 1b). These inserts could not directly participate in allelic pairing or recombination. Thus the experiments described below specifically address the issue of how interhomolog interactions, occurring outside the 8-kb insert, affect the ability of sequences within that insert to encounter and recombine with an insert located elsewhere in the genome.
Figure 1.
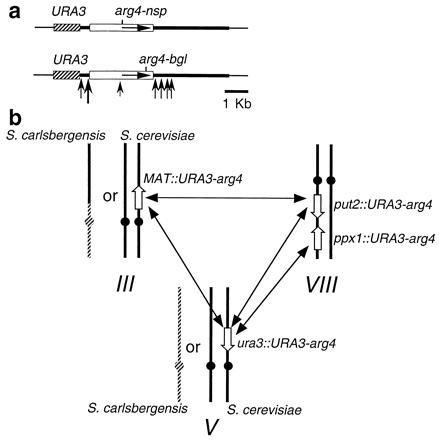
Experimental design. (a) Structure of the URA3-arg4 recombination substrates. Thick line, pBR322 sequences; hatched box, 1.2-kb HindIII URA3 fragment; gray box, 3.3-kb PstI arg4 fragment containing either arg4-nsp or arg4-bgl; thin lines, flanking genomic sequences; horizontal arrows, direction of ARG4 transcription; vertical arrows, meiosis-induced DSBs seen in all inserts (36). (b) Ectopic recombination was measured between pairs of arg4 inserts (open arrows, indicating insert orientation) on chromosomes III, V, and VIII. Ectopic recombination was measured in three different diploid types: (i) nonhybrid diploids with only S. cerevisiae chromosomes (solid lines), (ii) diploids with a single homoeologous S. carlsbergensis chromosome V, and (iii) diploids with homoeologous copies of S. carlsbergensis chromosomes III and V. Hatched lines on S. carlsbergensis chromosomes indicate regions of divergence from S. cerevisiae.
Homoeologous Chromosomes Increase the Frequency of Ectopic Recombination.
To examine the effect of homoeology on ectopic meiotic recombination, we measured the frequency of ARG4 spores produced by ectopic recombination between URA3-arg4 inserts located on chromosome III at MAT, on chromosome V at URA3, or on chromosome VIII at PUT2. Ectopic recombination was measured in three types of diploids: nonhybrid control strains with two copies each of chromosome Vcere and chromosome IIIcere; hybrid strains containing a single homoeologous chromosome Vcere/Vcarl pair; and hybrid strains containing both homoeologous chromosome pairs Vcere/Vcarl and IIIcere/IIIcarl (Fig. 1b). Chromosomes Vcarl and Vcere are diverged along their entire length and do not exhibit end-to-end alignment or recombine with each other during meiosis I prophase (refs. 27 and 28; R. Boumil, B. Kemp, and D. Dawson, personal communication). The absence of meiotic recombination between Vcarl and Vcere was confirmed in our genetic background, using as markers an ilv1 mutation on Vcarl and the unique sequences closest to each end of Vcere. Only parental marker configurations were observed among 72 ILV1 and 59 ilv1 spore colonies, indicating that crossing over between Vcere and Vcarl occurs in less than 3% of meioses. Chromosomes IIIcarl and IIIcere diverge in the region from the left telomere to within 1 kb of the MAT locus but are homologous at MAT and in the remainder of the chromosome (ref. 28; M.L., unpublished data). Diploids hybrid for chromosomes IIIcere and IIIcarl do not undergo meiotic recombination in the homoeologous region to the left of MAT, whereas recombination is normal in the homologous region to the right of MAT. For example, crossovers between MAT and THR4 occur at nearly identical frequencies in hybrid IIIcere/IIIcarl and in nonhybrid IIIcere/IIIcere strains (28).
In the presence of a homoeologous chromosome Vcarl, the frequency of recombination between inserts on chromosomes V and III increased by about 3-fold relative to nonhybrid controls (Table 1). Ectopic recombination between inserts on chromosomes V and VIII also increased in Vcere/Vcarl hybrids, by about 5-fold. Ectopic recombination between inserts on chromosomes III and VIII was not affected by the presence of a homoeologous chromosome Vcarl. Thus the presence of diverged chromosome V homoeolog caused a specific increase in the ability of inserts on chromosome Vcere to participate in ectopic recombination, without affecting the global level of ectopic recombination between inserts on unrelated chromosomes.
Table 1.
Effect of homoeologous chromosomes on ectopic recombination
| Location (chromosome–locus)
|
f(ARG4) × 103* | Relative to nonhybrid | |
|---|---|---|---|
| arg4-nsp | arg4-bgl | ||
| Nonhybrid strains | |||
| III–MAT | V–ura3 | 0.25 ± 0.04 | |
| V–ura3 | III–MAT | 0.40 ± 0.10 | |
| VIII–put2 | V–ura3 | 0.15 ± 0.02 | |
| V–ura3 | VIII–put2 | 0.15 ± 0.02 | |
| VIII–ppx1 | V–ura3 | 0.36 ± 0.04 | |
| V–ura3 | VIII–ppx1 | 0.32 ± 0.01 | |
| III–MAT | VIII–put2 | 0.31 ± 0.08 | |
| VIII–put2 | III–MAT | 0.65 ± 0.1 | |
| Homoeologous chromosome V | |||
| III–MAT | V–ura3 | 0.82 ± 0.06 | 3.3 |
| V–ura3 | III–MAT | 1.1 ± 0.17 | 2.8 |
| VIII–put2 | V–ura3 | 0.98 ± 0.1 | 6.5 |
| V–ura3 | VIII–put2 | 0.72 ± 0.17 | 4.8 |
| VIII–ppx1 | V–ura3 | 1.9 ± 0.36 | 5.3 |
| V–ura3 | VIII–ppx1 | 1.7 ± 0.19 | 5.3 |
| III–MAT | VIII–put2 | 0.34 ± 0.06 | 1.1 |
| VIII–put2 | III–MAT | 0.67 ± 0.06 | 1.0 |
| Homoeologous chromosomes V and III | |||
| III–MAT | V–ura3 | 1.70 ± 0.05 | 6.8 |
| V–ura3 | VIII–put2 | 0.81 ± 0.11 | 5.4 |
| III–MAT | VIII–put2 | 0.69 ± 0.09 | 2.2 |
arg4 mutant alleles were present on hemizygous copies of a URA3-arg4 insert (see Fig. 1) at MAT (chromosome III), at URA3 (chromosome V), at PUT2 (chromosome VIII), or at PPX1 (chromosome VIII). Strains with homoeologous chromosomes V or III contain one chromosome each from S. cerevisiae and S. carlsbergensis. The arg4 insert was always present on the S. cerevisiae chromosome.
ARG4 spores/total viable spores, mean ± standard deviation.
A further test of the effect of homoeology on ectopic recombination used double-hybrid strains with both Vcere/Vcarl and IIIcere/IIIcarl homoeologous chromosome pairs (Fig. 1b). Ectopic recombination between inserts on chromosomes V and III was further increased, by about 7-fold relative to nonhybrid control strains and by about 2-fold relative to strains containing a single Vcere/Vcarl pair (Table 1). Ectopic recombination between inserts on chromosomes III and VIII also increased by about 2-fold relative to nonhybrid control strains or Vcere/Vcarl strains. In contrast, ectopic recombination between inserts on chromosomes V and VIII showed no further increase beyond the 5-fold increase conferred by the homoeologous chromosome pair Vcere/Vcarl. This confirms the conclusion that a homoeologous chromosome affects only events involving sequences located on its partner.
A further control for global effects measured recombination between the leu2-K and leu2-R mutant alleles, present in all strains with two copies of chromosome IIIcere. LEU2 recombinant frequencies in nonhybrid control strains (3.2 × 10−3 ± 0.6 × 10−3) did not differ significantly from those in Vcere/Vcarl hybrids (2.9 × 10−3 ± 0.6 × 10−3).
DSBs in the URA3-arg4 Insert Are Not Affected by a Homoeologous Chromosome.
An alternative explanation for the above results is that recombination initiates more frequently within arg4 inserts when they are opposite a homoeologous partner. To test this possibility, we measured DSBs within a hemizygous ura3∷URA3-arg4 insert in rad50S derivatives of hybrid Vcarl/Vcere and nonhybrid Vcere/Vcere strains. Neither the pattern nor the relative frequency of DSBs in the insert was altered by the presence of a homoeologous chromosome Vcarl (Fig. 2). Similar results were obtained when the same DNA samples were used to examine DSBs at a control locus, the YCR47c-ARE1 region on chromosome III (data not shown).
Figure 2.
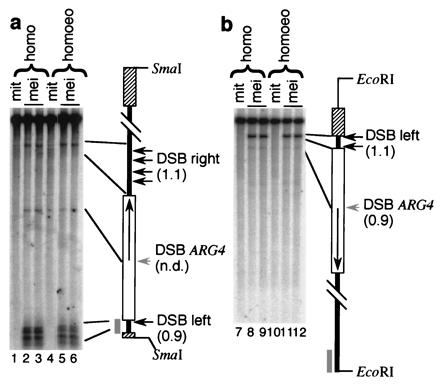
DSBs at ura3∷URA3-arg4 in normal and hybrid diploids. Southern blots contain DNA from diploids with a hemizygous ura3∷URA3-arg4 insert and homozygous for the rad50-K81I mutation, containing either two S. cerevisiae chromosomes V (homo) or an S. cerevisiae and an S. carlsbergensis chromosome V (homoeo). DNA samples from premeiotic cells (mit, lanes 1, 4, 7, and 10) and cells 5 and 6 h after induction of meiosis (mei, 5 h, lanes 2, 5, 8, and 11; mei, 6 h, lanes 3, 6, 9, and 12) were restriction enzyme-digested, displayed on 0.5% agarose gels, transferred to membranes, and hybridized with radioactive probe (36). (a) Restriction enzyme, SmaI; probe, pBR322 HindIII–BamHI fragment. (b) Restriction enzyme, EcoRI; probe, pBR322 PstI–EcoRI fragment. Horizontal arrows, meiosis-induced DSBs at ARG4 (DSB ARG4) and in pBR322 sequences on either side of ARG4 sequences (DSB left and DSB right). Numbers in parentheses are ratios of DSB frequencies for each site (6-h samples, DSB-homoeo/DSB-homo; n.d., not determined). In the insert diagrams, thick lines, pBR322; hatched boxes, URA3; open boxes, ARG4; thin lines, Ty sequences; thick gray lines, probe. A Ty element inserted in URA3 provides one of the EcoRI sites in B (T.-C. Wu and M.L., unpublished data).
Recombination Between URA-arg4 Inserts in Hybrid Strains Is Associated with Crossing Over.
Meiotic recombination between URA3-arg4 inserts is frequently accompanied by crossing over, in the case of ectopic recombination producing reciprocal translocations (22). We examined the effect of homoeology on the fraction of crossover-associated ARG4 recombinants in two circumstances: allelic recombination between MAT∷URA3-arg4 inserts in IIIcere/IIIcere nonhybrids and in IIIcere/IIIcarl hybrids, and ectopic recombination between inserts on chromosomes Vcere (at URA3)and VIIIcere (at PPX1) in a hybrid diploid containing a Vcere/Vcarl homoeologous pair. Crossover-associated MAT∷URA3-ARG4 allelic recombinants were recovered at similar frequencies from IIIcere/IIIcere and IIIcere and IIIcarl strains (63/105 and 137/198, respectively). V–VIII translocations were recovered in 23% (42/175) of ARG4 ectopic recombinants between inserts at URA3 and PPX1 in a Vcere/Vcarl hybrid. Correcting for spore inviability due to unbalanced translocation segregation (22), this result corresponds to 56% of ectopic recombinants being crossover associated, in good agreement with values obtained for ectopic recombination in nonhybrid diploids [42–66% (22)].
The ndj1 Mutation Increases the Efficiency of Ectopic Recombination Between Sequences on Heterologous Chromosomes.
ndj1 mutants display a delay in early meiotic homolog colocalization and in homolog synapsis, although allelic meiotic recombination occurs at approximately wild-type frequencies (refs. 31–33; H. Scherthan, personal communication). To examine the impact of these defects on ectopic recombination, we determined frequencies of allelic and ectopic recombination between arg4 inserts in wild-type and in ndj1 strains (Table 2). Ectopic recombination was expressed in terms of the efficiency of ectopic recombination, a measurement that corrects for marker- and locus-specific effects by combining ectopic and allelic recombination frequencies into a single term (22).
Table 2.
Effect of ndj1 on allelic and ectopic meiotic recombination
| Insert location | ndj1 | NDJ1† | ndj1/NDJ1 |
|---|---|---|---|
| Allelic recombination, f(ARG4) × 103* | |||
| LEU2 | 19 | 19 | 1.0 |
| URA3 | 8.1 | 5.7 | 1.4 |
| CHA1 | 5.0 | 4.7 | 1.1 |
| PHO11 | 3.0 | 4.8 | 0.6 |
| PHO12 | 1.3 | 1.6 | 0.8 |
| MAT | 7.4 | 9.4 | 0.8 |
| Mean | 0.9 ± 0.3 | ||
| Ectopic recombination, Eab‡ | |||
| On heterologous chromosomes | |||
| MAT × URA3 | 0.29 | 0.12 | 2.4 |
| LEU2 × URA3 | 0.29 | 0.13 | 2.2 |
| PHO11 × PHO12 | 0.51 | 0.24 | 2.1 |
| PHO11 × CHA1 | 0.59 | 0.27 | 2.2 |
| PHO12 × CHA1 | 0.57 | 0.28 | 2.0 |
| Mean | 2.2 ± 0.1 | ||
| On homologs | |||
| LEU2 × CHA1 | 0.6 | 0.56 | 1.1 |
| LEU2 × MAT | 0.5 | 0.48 | 1.1 |
| MAT × CHA1 | 0.62 | 0.4 | 1.6 |
| Mean | 1.3 ± 0.3 | ||
Arg+ spores/total spores from strains with URA3-arg4 inserts at allelic locations.
Data from ref. 22.
Eab, efficiency of ectopic recombination (22). Eab for a pair of insert loci is the sum of the ectopic recombination frequencies for both marker orientations divided by the sum of the two relevant allelic recombination frequencies, corrected for loss of recombinants on lethal crossover-associated rearrangements. For allelic inserts, Eab = 1; Eab < 1 indicates that two inserts recombine less frequently than would be expected if they were at allelic positions.
The efficiency of ectopic recombination between arg4 inserts on heterologous chromosomes was consistently 2.0- to 2.4-fold greater in ndj1 mutants compared with wild-type cells. Ectopic recombination between dispersed inserts on homologs was less affected, with no significant increase in recombination between inserts at LEU2 and CHA1 (separated by about 70 kb) or LEU2 and MAT (separated by about 105 kb), and a 1.6-fold increase in the efficiency of ectopic recombination between MAT∷URA3-arg4 and cha1∷URA3-arg4 (separated by about 175 kb). Allelic recombination frequencies were similar in ndj1 and wild-type strains, with modest locus-specific deviations from equality in both directions.
Calculations of ectopic recombination efficiency include a correction for ARG4 recombinants lost because of the segregation of crossover-associated reciprocal translocations (22). It was therefore important to show that the ndj1 mutation does not alter the fraction of ARG4 recombinants accompanied by crossing over. This was done for allelic recombination between MAT∷URA3-arg4 inserts and for ectopic recombination between inserts at LEU2 and at URA3. About 60% of MAT∷URA3-ARG4 allelic recombinants were associated with crossovers (63/105 in NDJ1, 66/106 in ndj1); 25–30% of viable ARG4 ectopic recombinants between leu2∷URA3-arg4 and ura3∷URA3-arg4 were associated with III–V reciprocal translocation (26/99 in NDJ1, 28/98 in ndj1). These data further support the suggestion that the increase in ectopic recombination seen in ndj1 mutants reflects increased interactions between dispersed homologous sequences, rather than a general effect on recombination initiation or on intermediate resolution.
Discussion
Previous studies have shown that, in S. cerevisiae, ectopic recombination between homologous sequences dispersed on heterologous chromosomes occurs less frequently than recombination between sequences at allelic locations on homologs (20–22). In the present study, we measured ectopic recombination between URA3-arg4 inserts under two circumstances where normal interhomolog interactions are disrupted but where most meiotic products are recovered as viable spores. In the first case, chromosome arm pairing and allelic recombination on chromosome V were abolished by replacing a S. cerevisiae chromosome with a diverged S. carlsbergensis chromosome. This resulted in an increase (from 3- to 6-fold) in ectopic recombination frequencies, but only when at least one of the recombining inserts was on the S. cerevisiae partner of the homoeologous chromosome pair. In the second case, a delay in homolog pairing and synapsis caused by loss of NDJ1 function was accompanied by a 2- to 2.4-fold increase in the efficiency of interheterolog ectopic recombination relative to allelic recombination. Similar increases in ectopic recombination have been reported for rad17, rad24, mec1, and hop2 mutants, all of which confer defects in homolog synapsis but which also result in substantial meiotic lethality (41–43). Taken together, these results are most consistent with the suggestion that, in normal S. cerevisiae meiosis, interhomolog interactions prevent recombination between dispersed homologous sequences and that disruption of these interactions relieves the normal restriction on ectopic recombination.
Why Does the Frequency of Ectopic Recombination Increase in S. cerevisiae/S. carlsbergensis Hybrids and in ndj1 Diploids?
We believe that our data can be most economically explained by suggesting that meiotic recombination and chromosome pairing proceed along pathways that normally may be temporally linked by the assembly of common structures or by checkpoint mechanisms (1, 15) but have the potential to proceed independently. In normal yeast meiosis, homolog colocalization, alignment, recombination, and synapsis have the effect of reducing chromosome mobility as meiosis progresses. The pairing–synapsis process, which may be facilitated by early steps in allelic recombination, reduces the ability of dispersed sequences to encounter each other, thus creating a barrier to ectopic recombination that is not imposed on allelic events. According to this view, most ectopic recombination should occur during the early stages of homolog pairing, when interhomolog associations are either loose or transient. Delays or defects in pairing and/or synapsis would create a longer time period for dispersed sequences to encounter one another, thus resulting in increased frequencies of ectopic recombination. Sequence divergence between S. carlsbergensis and S. cerevisiae homoeologs may cause such a pairing defect or delay, either by preventing allelic recombination itself or by interfering with other homology-dependent interactions. Similarly, in ndj1 mutants, a general delay in onset of homolog pairing would result in a corresponding increase in opportunities for interaction between sequences dispersed on heterologs.
While favoring the above interpretation, we recognize that the pairing defects conferred by homoeology or loss of NDJ1 function are not completely understood, and the relationship of these defects to meiotic recombination has not been fully established. Although the extent of homoeology between S. cerevisiae and S. carlsbergensis chromosomes is sufficient to block meiotic recombination, the effect of divergence on other interchromosomal transactions remains to be determined. In other organisms, most notably cereal plants, diverged chromosomes can pair early in meiosis I and may even synapse if fully homologous partners are absent (44). A similar situation may hold for Saccharomyces, inasmuch as fully synapsed late pachytene nuclei can be detected in cerevisiae/carlsbergensis and cerevisiae/paradoxus single chromosome-hybrid strains (A.S.H.G. and R. M. Speed, unpublished data; R. H. Borts, personal communication). It remains possible that abnormal chromosome structures formed in both cerevisiae/carlsbergensis hybrids and ndj1 mutants permit more flexibility in recombination partner choice, thus allowing increased ectopic recombination throughout or even late in meiosis I prophase. Temporal analysis of chromosome pairing and synapsis relative to the molecular events of meiotic recombination in hybrid strains and in ndj1 mutants should distinguish between this and the above-mentioned possibility.
A Genome-Wide Homology Search?
The most striking increase in ectopic recombination observed was between MAT∷URA3-arg4 and ura3∷URA3-arg4 inserts in double-hybrid IIIcere/IIIcarl and Vcere/Vcarl diploids. Once adjusted for crossover-associated recombinant lethality, these ectopic events occurred at a frequency (3.4 × 10−3) roughly two-thirds that seen for allelic events at URA3, and more than one-third that seen for allelic events at MAT, and well within the range of allelic frequencies seen at other arg4 inserts (1.2 × 10−3 to 1.9 × 10−2) (Fig. 3). Chromosome IIIcarl is a composite chromosome, with MAT defining the most centromere-proximal point of homology with IIIcere. Interactions in the homologous parts of IIIcere and IIIcarl may partially restrict the ability of the MAT∷URA3-arg4 insert to engage in ectopic recombination and thus may account for the modest increase in ectopic recombination seen for MAT∷URA3-arg4 inserts opposite IIIcarl, versus the greater increase seen for URA3∷URA3-arg4 inserts opposite the fully homoeologous Vcarl.
Figure 3.
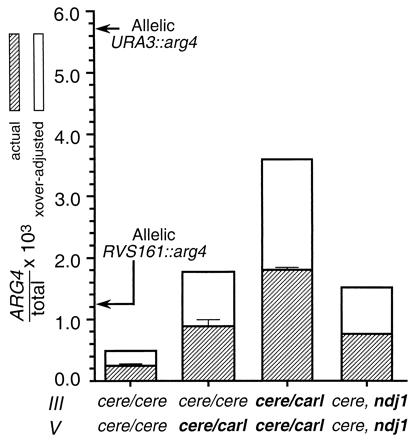
Summary of homolog/homoeolog effects on MAT∷URA3-arg4-nsp × ura3:URA3-arg4-bgl recombination. Hatched bars, uncorrected frequencies of ARG4 spores; open bars, frequencies corrected for loss of recombinants associated with a crossover, assuming that half of all ARG4 recombinants are crossover associated (22). The MAT∷URA3-arg4 and ura3∷URA3-arg4 inserts face in opposite directions relative to their respective centromeres; crossover-associated recombinants will reside on lethal rearrangements. For purposes of comparison, frequencies of allelic recombination at two insert loci are presented (40, 53).
The high level of MAT∷URA3-arg4 × URA3∷URA3-arg4 ectopic recombination seen in double-hybrid IIIcere/IIIcarl and Vcere/Vcarl diploids suggests that, if unimpeded, the meiotic recombination homology search can encompass the entire genome, as has been shown for DSB repair during vegetative yeast growth (45). However, it is also possible that centromere-mediated chromosome orientation (46, 47) or region-specific nuclear addressing (48) fortuitously place the insert loci (MAT, URA3, PUT2, and PPX1) in the same nuclear subcompartment. Were this true, a homology search encompassing only part of the nucleus could generate our results. We believe this to be unlikely, because inserts at other locations in nonhybrid diploids display interheterolog ectopic recombination efficiencies similar to those seen in the current study (ref. 22; A.S.H.G., unpublished observations). In fact, nuclear addressing and a genome-wide homology search are not mutually exclusive; extensive meiotic nuclear motions seen in many organisms (reviewed in ref. 1) might rearrange the nucleus sufficiently to allow a genome-wide homology search.
Meiotic Pairing and Recombination: Different Strategies in Different Species.
It has been suggested that multiple, dispersed, homology-dependent DNA–DNA interactions initiate and advance homolog colocalization and alignment in S. cerevisiae (49). This presents the possibility of frequent contact between heterologous chromosomes and may explain the relatively modest (10- to 20-fold) differences between ectopic and allelic recombination frequencies seen in this organism (22). In contrast, Drosophila, C. elegans, and Schizosaccharomyces pombe all appear to use recombination-independent mechanisms to align homologs (12, 13, 50), and ectopic recombination is several orders of magnitude less frequent than allelic recombination (24, 51, 52). To account for these differences, we suggest that the amount of ectopic recombination seen in an organism reflects the timing of recombination initiation relative to homolog colocalization and alignment. In Saccharomyces, ectopic interactions can occur unless they are locked out by the multiple allelic interactions involved in homolog pairing. In the other organisms cited above, recombination-independent initiation of homolog association uses specific sites on each chromosome, and recombination is most likely initiated in the context of already-paired homologs.
In summary, our data support the suggestion that ectopic recombination is restricted by the homolog colocalization and coalignment events that occur during Saccharomyces cerevisiae meiosis. A variety of homolog pairing strategies may be used in diverse species, possibly in response to differences in genome structure and complexity. However, we expect that homolog colocalization, alignment, and synapsis will perform a similar function in all organisms, namely, to prevent recombination between repeated sequences, thus preventing deleterious rearrangements, and to ensure the faithful transmission of a complete genome to gametes.
Acknowledgments
We thank M. Resnick, S. Priebe, C. Newlon, M. Conrad, and M. Dresser for strains; M. Resnick for helpful discussions; R. H. Borts, T. Nillsson-Tilgren, R. Boumil, B. Kemp, D. Dawson, and H. Scherthan for personal communications; T. Allers, V. Borde, D. Chattoraj, N. Kleckner, C. Klee, B. Thomas, and M. Yarmolinsky for comments that improved the manuscript; and Ayesha Johnson for superb technical assistance. This research was funded in part by a Wellcome Trust Grant to A.S.H.G.
Abbreviations
- DSB
double-strand DNA break
- IIIcere
chromosome III of Saccharomyces cerevisiae
- IIIcarl
chromosome III of Saccharomyces carlsbergensis
- Vcere
chromosome V of Saccharomyces cerevisiae
- Vcarl
chromosome V of Saccharomyces carlsbergensis.
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Zickler D, Kleckner N. Annu Rev Genet. 1998;32:619–697. doi: 10.1146/annurev.genet.32.1.619. [DOI] [PubMed] [Google Scholar]
- 2.Fung J C, Marshall W F, Dernburg A, Agard D A, Sedat J W. J Cell Biol. 1998;141:5–20. doi: 10.1083/jcb.141.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Burgess S M, Kleckner N, Weiner B M. Genes Dev. 1999;13:1627–1641. doi: 10.1101/gad.13.12.1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weiner B M, Kleckner N. Cell. 1994;77:977–991. doi: 10.1016/0092-8674(94)90438-3. [DOI] [PubMed] [Google Scholar]
- 5.Padmore R, Cao L, Kleckner N. Cell. 1991;66:1239–1256. doi: 10.1016/0092-8674(91)90046-2. [DOI] [PubMed] [Google Scholar]
- 6.Albini S M, Jones G H. Chromosoma. 1987;95:324–338. [Google Scholar]
- 7.Scherthan H, Loidl J, Schuster T, Schweizer D. Chromosoma. 1992;101:590–595. doi: 10.1007/BF00360535. [DOI] [PubMed] [Google Scholar]
- 8.Schwarzacher T. Symp Soc Exp Biol. 1996;50:71–75. [PubMed] [Google Scholar]
- 9.Barlow A L, Hulten M A. Zygote. 1998;6:27–38. doi: 10.1017/s0967199400005050. [DOI] [PubMed] [Google Scholar]
- 10.Lichten M, Goldman A S. Annu Rev Genet. 1995;29:423–444. doi: 10.1146/annurev.ge.29.120195.002231. [DOI] [PubMed] [Google Scholar]
- 11.Smith K N, Nicolas A. Curr Opin Genet Dev. 1998;8:200–211. doi: 10.1016/s0959-437x(98)80142-1. [DOI] [PubMed] [Google Scholar]
- 12.McKim K S, Green-Marroquin B L, Sekelsky J J, Chin G, Steinberg C, Khodosh R, Hawley R S. Science. 1998;279:876–878. doi: 10.1126/science.279.5352.876. [DOI] [PubMed] [Google Scholar]
- 13.Dernburg A F, McDonald K, Moulder G, Barstead R, Dresser M, Villeneuve A M. Cell. 1998;94:387–398. doi: 10.1016/s0092-8674(00)81481-6. [DOI] [PubMed] [Google Scholar]
- 14.Nicklas R B. Science. 1997;275:632–637. doi: 10.1126/science.275.5300.632. [DOI] [PubMed] [Google Scholar]
- 15.Roeder G S. Genes Dev. 1997;11:2600–2621. doi: 10.1101/gad.11.20.2600. [DOI] [PubMed] [Google Scholar]
- 16.Loidl J, Klein F, Scherthan H. J Cell Biol. 1994;125:1191–1200. doi: 10.1083/jcb.125.6.1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xu L, Kleckner N. EMBO J. 1995;14:5115–5128. doi: 10.1002/j.1460-2075.1995.tb00194.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lydall D, Nikolsky Y, Bishop D K, Weinert T. Nature (London) 1996;383:840–843. doi: 10.1038/383840a0. [DOI] [PubMed] [Google Scholar]
- 19.Xu L, Weiner B M, Kleckner N. Genes Dev. 1997;11:106–118. doi: 10.1101/gad.11.1.106. [DOI] [PubMed] [Google Scholar]
- 20.Jinks-Robertson S, Petes T D. Proc Natl Acad Sci USA. 1985;82:3350–3354. doi: 10.1073/pnas.82.10.3350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lichten M, Borts R H, Haber J E. Genetics. 1987;115:233–246. doi: 10.1093/genetics/115.2.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goldman A S, Lichten M. Genetics. 1996;144:43–55. doi: 10.1093/genetics/144.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murti J R, Bumbulis M, Schimenti J C. Genetics. 1994;137:837–843. doi: 10.1093/genetics/137.3.837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Virgin J B, Bailey J P. Genetics. 1998;149:1191–1204. doi: 10.1093/genetics/149.3.1191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Theis J F, Yang C, Schaefer C B, Newlon C S. Genetics. 1999;152:943–952. doi: 10.1093/genetics/152.3.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Resnick M A, Zgaga Z, Hieter P, Westmoreland J, Fogel S, Nilsson-Tillgren T. Mol Gen Genet. 1992;234:65–73. doi: 10.1007/BF00272346. [DOI] [PubMed] [Google Scholar]
- 27.Nilsson-Tillgren T, Gjermansen C, Holmberg S, Petersen J G L, Kielland-Brandt M C. Carlsberg Res Commun. 1986;51:309–326. [Google Scholar]
- 28.Nilsson-Tillgren T, Gjermansen C, Petersen J G L, Kielland-Brandt M C, Holmberg S. Carlsberg Res Commun. 1981;46:65–76. [Google Scholar]
- 29.Dawson D S, Murray A W, Szostak J W. Science. 1986;234:713–717. doi: 10.1126/science.3535068. [DOI] [PubMed] [Google Scholar]
- 30.Kaback D B. Curr Genet. 1989;15:385–392. doi: 10.1007/BF00376792. [DOI] [PubMed] [Google Scholar]
- 31.Trelles-Sticken E, Loidl J, Scherthan H. J Cell Sci. 1999;112:651–658. doi: 10.1242/jcs.112.5.651. [DOI] [PubMed] [Google Scholar]
- 32.Chua P R, Roeder G S. Genes Dev. 1997;11:1786–1800. doi: 10.1101/gad.11.14.1786. [DOI] [PubMed] [Google Scholar]
- 33.Conrad M N, Dominguez A M, Dresser M E. Science. 1997;276:1252–1255. doi: 10.1126/science.276.5316.1252. [DOI] [PubMed] [Google Scholar]
- 34.Rockmill B, Roeder G S. Genes Dev. 1998;12:2574–2586. doi: 10.1101/gad.12.16.2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kane S M, Roth R. J Bacteriol. 1974;118:8–14. doi: 10.1128/jb.118.1.8-14.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wu T-C, Lichten M. Genetics. 1995;140:55–66. doi: 10.1093/genetics/140.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Goyon C, Lichten M. Mol Cell Biol. 1993;13:373–382. doi: 10.1128/mcb.13.1.373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alani E, Padmore R, Kleckner N. Cell. 1990;61:419–436. doi: 10.1016/0092-8674(90)90524-i. [DOI] [PubMed] [Google Scholar]
- 39.Borts R H, Lichten M, Hearn M, Davidow L S, Haber J E. Cold Spring Harbor Symp Quant Biol. 1984;49:67–76. doi: 10.1101/sqb.1984.049.01.010. [DOI] [PubMed] [Google Scholar]
- 40.Borde V, Wu T-C, Lichten M. Mol Cell Biol. 1999;19:4832–4842. doi: 10.1128/mcb.19.7.4832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Leu J Y, Chua P R, Roeder G S. Cell. 1998;94:375–386. doi: 10.1016/s0092-8674(00)81480-4. [DOI] [PubMed] [Google Scholar]
- 42.Leu J-Y. Ph.D. thesis. New Haven, CT: Yale Univ.; 1999. [Google Scholar]
- 43.Grushcow J M, Holzen T M, Park K J, Weinert T, Lichten M, Bishop D K. Genetics. 1999;153:607–620. doi: 10.1093/genetics/153.2.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gillies C B. Theor Appl Genet. 1987;74:430–438. doi: 10.1007/BF00289817. [DOI] [PubMed] [Google Scholar]
- 45.Haber J E, Leung W Y. Proc Natl Acad Sci USA. 1996;93:13949–13954. doi: 10.1073/pnas.93.24.13949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jin Q, Trelles-Sticken E, Scherthan H, Loidl J. J Cell Biol. 1998;141:21–29. doi: 10.1083/jcb.141.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hayashi A, Ogawa H, Kohno K, Gasser S M, Hiraoka Y. Genes Cells. 1998;3:587–601. doi: 10.1046/j.1365-2443.1998.00215.x. [DOI] [PubMed] [Google Scholar]
- 48.Marshall W F, Straight A, Marko J F, Swedlow J, Dernburg A, Belmont A, Murray A W, Agard D A, Sedat J W. Curr Biol. 1997;7:930–939. doi: 10.1016/s0960-9822(06)00412-x. [DOI] [PubMed] [Google Scholar]
- 49.Kleckner N, Weiner B. Cold Spring Harbor Symp Quant Biol. 1993;58:553–565. doi: 10.1101/sqb.1993.058.01.062. [DOI] [PubMed] [Google Scholar]
- 50.Hiraoka Y. Genes Cells. 1998;3:405–413. doi: 10.1046/j.1365-2443.1998.00205.x. [DOI] [PubMed] [Google Scholar]
- 51.Hipeau-Jacquotte R, Brutlag D L, Bregegere F. Mol Gen Genet. 1989;220:140–146. doi: 10.1007/BF00260868. [DOI] [PubMed] [Google Scholar]
- 52.Zetka M, Rose A. Trends Genet. 1995;11:27–31. doi: 10.1016/s0168-9525(00)88983-0. [DOI] [PubMed] [Google Scholar]
- 53.Wu T-C, Lichten M. Genetics. 1995;140:55–66. doi: 10.1093/genetics/140.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]


