Abstract
We report on the first detection of an AmpC-type Ambler class C 1 (ACC-1) β-lactamase in Citrobacter freundi isolated from a patient also harboring ACC-1-producing Escherichia coli and Klebsiella pneumoniae. We propose a simple cefoxitin-based double-disk synergy test (DDST) for the specific detection of ACC-1 in members of the family Enterobacteriaceae, including natural AmpC producers, in association with a cloxacillin-based DDST as a first-line AmpC-type β-lactamase screening test.
β-Lactamase production is the main mechanism underlying resistance to β-lactam antibiotics in gram-negative bacteria. β-Lactamase genes are often located on plasmids or transposons, making them readily transmissible among different species. During the last decade plasmid-mediated AmpC-type β-lactamases have been increasingly isolated worldwide and have been linked to treatment failure (27, 33). Plasmid-mediated AmpC β-lactamases are derived from the chromosomally encoded AmpC β-lactamases produced by various bacteria, such as Aeromonas (5, 18, 19), Citrobacter freundii (4, 10, 35), Enterobacter (9, 28), and Morganella morganii (2, 14). In 1999 a novel plasmid-borne AmpC β-lactamase was isolated from Klebsiella pneumoniae in Germany (3). Unlike other plasmidic AmpCs, it had low levels of activity against cephamycins. The authors proposed the name Ambler class C 1 (ACC-1). It was subsequently shown that ACC-1 was derived from the chromosome-borne AmpC of Hafnia alvei (25). ACC-1 has so far been isolated only in Germany (3), France (6, 16, 25, 26), Spain (24), and Tunisia (21); the French isolates originated from Tunisia (6, 16, 25, 26). ACC-1 is carried by K. pneumoniae (3, 6, 25, 26), Escherichia coli (6, 16, 24), Proteus mirabilis (16), and Salmonella enterica serotypes Mbandaka and Livingstone (21, 31). Here we describe the simultaneous isolation of three bacterial species, each of which carries ACC-1, from the same stool sample of a 17-year-old patient. To our knowledge this is the first time that the ACC-1 β-lactamase has been found in Citrobacter freundii. We also describe a new cefoxitin-based double-disk synergy test (DDST) for specific ACC-1 detection.
The patient, a teenage boy, was admitted to the Robert-Debré Hospital (Paris, France) in November 2005 for surgical treatment of a leaking jejuno-jejunal anastomosis after intestinal grafting. He had received multiple courses of antibiotics, including broad-spectrum cephalosporins.
The same stool culture yielded three different ceftazidime-resistant members of the family Enterobacteriaceae: 108 CFU/g Escherichia coli (strain Ec1), 109 CFU/g Klebsiella pneumoniae (strain Kp1), and 106 CFU/g Citrobacter freundii (strain Cf1). Antimicrobial susceptibility was determined by the disk diffusion method on Mueller-Hinton agar (Bio-Rad, Marnes-La-Coquette, France), as recommended by the Clinical and Laboratory Standards Institute (formerly NCCLS). The three isolates were susceptible to cefoxitin and cefepime but had decreased susceptibilities to ceftazidime and cefotaxime. No synergy was observed between clavulanic acid and ceftazidime, cefotaxime, or cefepime in the double-disk synergy test. The MICs of cefoxitin, cefotaxime, ceftriaxone, ceftazidime, cefepime, imipenem, piperacillin, and piperacillin-tazobactam were determined by the Etest diffusion method (AB Biodisk, Solna, Sweden) (Table 1), as recommended by the manufacturer.
TABLE 1.
In vitro susceptibilities (MICs) of the three stool isolates, the E. coli recipient strain (J53), and the resulting transconjugants
| Antimicrobial(s) | MIC (μg/ml)
|
||||||
|---|---|---|---|---|---|---|---|
| E. coli (Ec1) | K. pneumoniae (Kp1) | C. freundii (Cf1) | J53 | TC Ec | TC Kp | TC Cf | |
| Cefoxitin | 6 | 6 | 8 | 2 | 2 | 2 | 2 |
| Cefotaxime | 12 | 24 | 16 | 0.032 | 3 | 4 | 4 |
| Ceftriaxone | 12 | 16 | 16 | 0.023 | 4 | 4 | 4 |
| Ceftazidime | 24 | 24 | 24 | 0.047 | 4 | 4 | 12 |
| Cefepime | 0.5 | 1 | 0.38 | 0.016 | 0.125 | 0.19 | 0.19 |
| Imipenem | 0.25 | 0.25 | 0.19 | 0.125 | 0.125 | 0.125 | 0.125 |
| Piperacillin | 192 | >256 | 64 | 1 | 32 | 32 | 32 |
| Piperacillin + tazobactam | 32 | 64 | 16 | 1 | 6 | 6 | 6 |
Strains Ec1, Kp1, and Cf1 were conjugated with rifampin-resistant, amoxicillin-susceptible Escherichia coli strain J53, as described previously (1). The transconjugants (TCs; TC Ec, TC Kp, and TC Cf, respectively) displayed the same patterns of β-lactam resistance as the donor strains. PCR and sequencing were used to assess the three isolates and their transconjugants for the presence of blaACC-1 and blaTEM-1, as described previously (6, 25). Pulsed-field gel electrophoresis showed that strains Ec1 and Kp1 were unrelated to previous French isolates harboring blaACC-1 (6, 23) (data not shown).
The cefoxitin susceptibilities of the ACC-1-producing strains were recently attributed to ACC-1 β-lactamase inhibition by cefoxitin itself (17). We therefore determined the ceftazidime and cefepime MICs for strains Ec1, Kp1, and Cf1 and also for 15 isolates of the Enterobacteriaceae expressing a broad range of AmpC-type β-lactamases by using the Etest diffusion method (AB Biodisk) on agar plates containing cefoxitin concentrations of 0.25, 1, and 4 μg/ml (2, 6, 13, 20, 21, 23, 30, 36). The results confirmed the powerful inhibitory action of cefoxitin and its specificity for ACC-1, as cefoxitin did not reduce the ceftazidime or cefepime MICs for the other strains tested. Cefoxitin concentrations below 0.25 μg/ml were not inhibitory. We used these results to develop a DDST with cefoxitin and ceftazidime or cefotaxime for the detection of ACC-1. We used a distance of only 2 cm between the centers of the two disks, as in previous studies, cefoxitin showed no particular synergy with broad-spectrum cephalosporins against ACC-1-expressing isolates at a distance of 3 cm, possibly owing to inadequate cefoxitin concentrations (Table 2).
TABLE 2.
Ceftazidime and cefepime MICs determined by Etest diffusion method on agar plates with or without cefoxitin
| Organism | Reference | AmpC type | Ceftazidime MIC (μg/ml) with cefoxitin at concn (μg/ml) of:
|
Cefepime MIC (μg/ml) with cefoxitin at concn (μg/ml) of:
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 1 | 0.25 | 0 | 4 | 1 | 0.25 | 0 | |||
| Klebsiella pneumoniae (Kp1) | This study | ACC-1 | 0.094 | 0.75 | 24 | 24 | 0.023 | 0.38 | 1 | 1 |
| Escherichia coli (Ec1) | This study | ACC-1 | No growth | 0.25 | 16 | 24 | No growth | 0.032 | 0.25 | 0.5 |
| Citrobacter freundii (Cf1) | This study | ACC-1 | No growth | 0.25 | 12 | 24 | No growth | 0.032 | 0.19 | 0.38 |
| Salmonella serotype Mbandaka | 21 | ACC-1 | No growth | 2 | 256 | >256 | No growth | 0.047 | 0.25 | 1 |
| Escherichia coli | 6 | ACC-1 | No growth | 8 | 64 | 64 | No growth | 0.38 | 0.75 | 0.75 |
| Klebsiella pneumoniae | 6 | ACC-1 | No growth | 1 | 48 | 64 | No growth | 0.125 | 0.5 | 0.75 |
| Klebsiella pneumoniae | 26 | ACC-1 | 1 | 24 | 64 | 256 | 0.125 | 0.38 | 0.5 | 0.75 |
| Klebsiella pneumoniae | 13 | CMY-2 | 3 | 4 | 3 | 4 | 0.047 | 0.064 | 0.047 | 0.064 |
| Salmonella serotype Senftenberg | 20 | CMY-2b | 24 | 24 | 24 | 24 | 0.19 | 0.25 | 0.25 | 0.25 |
| Klebsiella pneumoniae | 36 | DHA-1 | 32 | 16 | 8 | 4 | 0.094 | 0.064 | 0.064 | 0.064 |
| Klebsiella pneumoniae | 30 | MOX-2 | 48 | 48 | 48 | 48 | 0.5 | 0.38 | 0.5 | 0.5 |
| Klebsiella pneumoniae | 23 | FOX-3 | 32 | 48 | 32 | 32 | 0.25 | 0.25 | 0.25 | 0.25 |
| Hafnia alvei | This study | Derepressed ampC | No growth | 2 | 4 | 16 | No growth | 0.016 | 0.032 | 0.064 |
| Citrobacter freundii | This study | Derepressed ampC | >256 | >256 | >256 | >256 | 0.38 | 0.38 | 0.38 | 0.38 |
| Enterobacter cloacae | This study | Derepressed ampC | >256 | >256 | >256 | >256 | 0.032 | 0.032 | 0.032 | 0.032 |
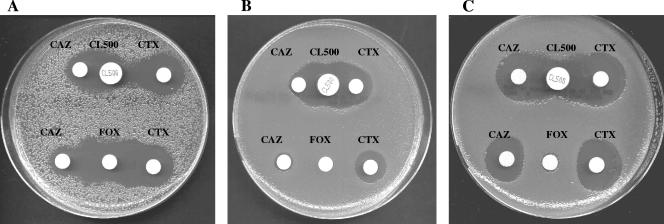
A DDST with 500-μg cloxacillin disks (Rosco Diagnostica, Denmark) was applied to the 15 strains mentioned above, as recommended by the manufacturer. Cloxacillin was synergistic with both ceftazidime and cefotaxime against all the strains (Fig. 1). In contrast, cefoxitin was synergistic with cefotaxime and ceftazidime only against ACC-1-expressing strains (Fig. 1A). As expected (15), cefoxitin induced resistance to ceftazidime for the DHA-1-expressing strain (Fig. 1C).
FIG. 1.
Double-disk synergy test on Mueller-Hinton agar plates with broad-spectrum cephalosporins and either cloxacillin (upper set of disks) or cefoxitin (lower set of disks) applied to three AmpC-carrying strains (CAZ, ceftazidime; CTX, cefotaxime; FOX, cefoxitin; CL500, cloxacillin at 500 μg). The distances between the disks were optimized according to the inhibition diameters, as described in the text. Cloxacillin is synergistic with both ceftazidime and cefotaxime for the three strains. (A) Citrobacter freundii harboring ACC-1 (strain Cf1) for which cefoxitin is synergistic with cefotaxime and ceftazidime; (B) Citrobacter freundii with chromosomal ampC derepression for which no synergy is observed between cefoxitin and either cefotaxime or ceftazidime; (C) Klebsiella pneumoniae harboring DHA-1 (36) for which cefoxitin induces resistance to cefotaxime and ceftazidime.
Detection of plasmidic AmpC is an emerging challenge for clinical laboratories, especially in bacteria that naturally expressing AmpC, like Citrobacter freundii. Various homemade detection tests have been proposed, but none has been widely adopted (7, 8, 11, 12, 22, 32, 34).
Previous reports of ACC-1-expressing isolates (3, 6, 16, 21, 24-26) mention no particular interactions between antibiotic disks. Indeed, since no ampR motif was identified on blaACC-1 (3), cefoxitin was not an inducer by the double-disk test (3, 25). The cefoxitin DDST that we have described here is specific for the ACC-1 β-lactamase, which, to our knowledge, is the only plasmidic AmpC to be inhibited by cefoxitin (29). This cefoxitin DDST method readily identified ACC-1 even in natural AmpC producers like Citrobacter freundii. As ACC-1-expressing strains have an atypical resistance profile (notably, cefoxitin susceptibility, ceftazidime resistance, and cefepime susceptibility), the cefoxitin DDST can be used as a second-line test to confirm the expression of ACC-1. We did not test this DDST with strains carrying both ACC-1 and other wide-spectrum β-lactamases (e.g., extended-spectrum β-lactamases, other plasmid-mediated AmpC β-lactamases, or derepressed ampC). In such strains, the synergy between cefoxitin and ceftazidime provided by DDST may be more difficult to visualize.
The DDST with a 500-μg cloxacillin disk successfully identified AmpC expression in all 15 positive control strains tested here. However, whereas the manufacturer recommends one disk-to-disk distance (1 cm edge to edge), we adapted this distance using the inhibition diameter of the cephalosporin (center-to-center distance = inhibition radius + 1 cm) tested to obtain the most obvious synergy picture possible (Fig. 1).
In conclusion, we report on the first isolation of a Citrobacter freundii strain expressing the ACC-1 β-lactamase and propose a cefoxitin DDST in association with the DDST with a 500-μg cloxacillin disk for identifying this enzyme in isolates of the family Enterobacteriaceae, including natural ampC carriers. The synergy between cefoxitin and ceftazidime or cefotaxime reveals ACC-1 β-lactamase expression, whereas induction is observed with inducible AmpC β-lactamases such as DHA-1. This DDST can easily be performed on the same agar plate by the DDST with a 500-μg cloxacillin disk, which is specific for AmpC-type β-lactamases. These two associated DDSTs may be simple and cost-effective first-line tests for AmpC-type β-lactamase determination, indicating the presence of an AmpC-type β-lactamase, and further providing a specific means of detection of the ACC-1 β-lactamase.
Acknowledgments
This work was supported in part by grants from Faculté de Médecine Pierre et Marie Curie, Université Paris VI, and from the European Community (sixth PCRD contract LSHM-CT 2003-503335).
Footnotes
Published ahead of print on 13 September 2006.
REFERENCES
- 1.Arlet, G., M. Rouveau, I. Casin, P. J. Bouvet, P. H. Lagrange, and A. Philippon. 1994. Molecular epidemiology of Klebsiella pneumoniae strains that produce SHV-4 beta-lactamase and which were isolated in 14 French hospitals. J. Clin. Microbiol. 32:2553-2558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barnaud, G., G. Arlet, C. Verdet, O. Gaillot, P. H. Lagrange, and A. Philippon. 1998. Salmonella enteritidis: AmpC plasmid-mediated inducible beta-lactamase (DHA-1) with an ampR gene from Morganella morganii. Antimicrob. Agents Chemother. 42:2352-2358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bauernfeind, A., I. Schneider, R. Jungwirth, H. Sahly, and U. Ullmann. 1999. A novel type of AmpC beta-lactamase, ACC-1, produced by a Klebsiella pneumoniae strain causing nosocomial pneumonia. Antimicrob. Agents Chemother. 43:1924-1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bauernfeind, A., I. Stemplinger, R. Jungwirth, and H. Giamarellou. 1996. Characterization of the plasmidic beta-lactamase CMY-2, which is responsible for cephamycin resistance. Antimicrob. Agents Chemother. 40:221-224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bauernfeind, A., I. Stemplinger, R. Jungwirth, R. Wilhelm, and Y. Chong. 1996. Comparative characterization of the cephamycinase blaCMY-1 gene and its relationship with other beta-lactamase genes. Antimicrob. Agents Chemother. 40:1926-1930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bidet, P., B. Burghoffer, V. Gautier, N. Brahimi, P. Mariani-Kurkdjian, A. El-Ghoneimi, E. Bingen, and G. Arlet. 2005. In vivo transfer of plasmid-encoded ACC-1 AmpC from Klebsiella pneumoniae to Escherichia coli in an infant and selection of impermeability to imipenem in K. pneumoniae. Antimicrob. Agents Chemother. 49:3562-3565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Black, J. A., E. S. Moland, and K. S. Thomson. 2005. AmpC disk test for detection of plasmid-mediated AmpC beta-lactamases in Enterobacteriaceae lacking chromosomal AmpC beta-lactamases. J. Clin. Microbiol. 43:3110-3113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Black, J. A., K. S. Thomson, J. D. Buynak, and J. D. Pitout. 2005. Evaluation of beta-lactamase inhibitors in disk tests for detection of plasmid-mediated AmpC beta-lactamases in well-characterized clinical strains of Klebsiella spp. J. Clin. Microbiol. 43:4168-4171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bradford, P. A., C. Urban, N. Mariano, S. J. Projan, J. J. Rahal, and K. Bush. 1997. Imipenem resistance in Klebsiella pneumoniae is associated with the combination of ACT-1, a plasmid-mediated AmpC beta-lactamase, and the foss of an outer membrane protein. Antimicrob. Agents Chemother. 41:563-569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bret, L., C. Chanal-Claris, D. Sirot, E. B. Chaibi, R. Labia, and J. Sirot. 1998. Chromosomally encoded ampC-type beta-lactamase in a clinical isolate of Proteus mirabilis. Antimicrob. Agents Chemother. 42:1110-1114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Coudron, P. E. 2005. Inhibitor-based methods for detection of plasmid-mediated AmpC beta-lactamases in Klebsiella spp., Escherichia coli, and Proteus mirabilis. J. Clin. Microbiol. 43:4163-4167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coudron, P. E., E. S. Moland, and K. S. Thomson. 2000. Occurrence and detection of AmpC beta-lactamases among Escherichia coli, Klebsiella pneumoniae, and Proteus mirabilis isolates at a veterans medical center. J. Clin. Microbiol. 38:1791-1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Decre, D., C. Verdet, L. Raskine, H. Blanchard, B. Burghoffer, A. Philippon, M. J. Sanson-Le-Pors, J. C. Petit, and G. Arlet. 2002. Characterization of CMY-type beta-lactamases in clinical strains of Proteus mirabilis and Klebsiella pneumoniae isolated in four hospitals in the Paris area. J. Antimicrob. Chemother. 50:681-688. [DOI] [PubMed] [Google Scholar]
- 14.Fortineau, N., L. Poirel, and P. Nordmann. 2001. Plasmid-mediated and inducible cephalosporinase DHA-2 from Klebsiella pneumoniae. J. Antimicrob. Chemother. 47:207-210. [DOI] [PubMed] [Google Scholar]
- 15.Gaillot, O., C. Clement, M. Simonet, and A. Philippon. 1997. Novel transferable beta-lactam resistance with cephalosporinase characteristics in Salmonella enteritidis. J. Antimicrob. Chemother. 39:85-87. [DOI] [PubMed] [Google Scholar]
- 16.Girlich, D., A. Karim, C. Spicq, and P. Nordmann. 2000. Plasmid-mediated cephalosporinase ACC-1 in clinical isolates of Proteus mirabilis and Escherichia coli. Eur. J. Clin. Microbiol. Infect. Dis. 19:893-895. [DOI] [PubMed] [Google Scholar]
- 17.Girlich, D., T. Naas, S. Bellais, L. Poirel, A. Karim, and P. Nordmann. 2000. Biochemical-genetic characterization and regulation of expression of an ACC-1-like chromosome-borne cephalosporinase from Hafnia alvei. Antimicrob. Agents Chemother. 44:1470-1478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gonzalez Leiza, M., J. C. Perez-Diaz, J. Ayala, J. M. Casellas, J. Martinez-Beltran, K. Bush, and F. Baquero. 1994. Gene sequence and biochemical characterization of FOX-1 from Klebsiella pneumoniae, a new AmpC-type plasmid-mediated beta-lactamase with two molecular variants. Antimicrob. Agents Chemother. 38:2150-2157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Horii, T., Y. Arakawa, M. Ohta, S. Ichiyama, R. Wacharotayankun, and N. Kato. 1993. Plasmid-mediated AmpC-type beta-lactamase isolated from Klebsiella pneumoniae confers resistance to broad-spectrum beta-lactams, including moxalactam. Antimicrob. Agents Chemother. 37:984-990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Koeck, J. L., G. Arlet, A. Philippon, S. Basmaciogullari, H. V. Thien, Y. Buisson, and J. D. Cavallo. 1997. A plasmid-mediated CMY-2 beta-lactamase from an Algerian clinical isolate of Salmonella senftenberg. FEMS Microbiol. Lett. 152:255-260. [DOI] [PubMed] [Google Scholar]
- 21.Makanera, A., G. Arlet, V. Gautier, and M. Manai. 2003. Molecular epidemiology and characterization of plasmid-encoded beta-lactamases produced by Tunisian clinical isolates of Salmonella enterica serotype Mbandaka resistant to broad-spectrum cephalosporins. J. Clin. Microbiol. 41:2940-2945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Manchanda, V., and N. P. Singh. 2003. Occurrence and detection of AmpC beta-lactamases among gram-negative clinical isolates using a modified three-dimensional test at Guru Tegh Bahadur Hospital, Delhi, India. J. Antimicrob. Chemother. 51:415-418. [DOI] [PubMed] [Google Scholar]
- 23.Marchese, A., G. Arlet, G. C. Schito, P. H. Lagrange, and A. Philippon. 1998. Characterization of FOX-3, an AmpC-type plasmid-mediated beta-lactamase from an Italian isolate of Klebsiella oxytoca. Antimicrob. Agents Chemother. 42:464-467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Miro, E., B. Mirelis, F. Navarro, L. Matas, M. Gimenez, and C. Rabaza. 2005. Escherichia coli producing an ACC-1 class C β-lactamase isolated in Barcelona, Spain. Antimicrob. Agents Chemother. 49:866-867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nadjar, D., M. Rouveau, C. Verdet, L. Donay, J. Herrmann, P. H. Lagrange, A. Philippon, and G. Arlet. 2000. Outbreak of Klebsiella pneumoniae producing transferable AmpC-type beta-lactamase (ACC-1) originating from Hafnia alvei. FEMS Microbiol. Lett. 187:35-40. [DOI] [PubMed] [Google Scholar]
- 26.Ohana, S., V. Leflon, E. Ronco, M. Rottman, D. Guillemot, S. Lortat-Jacob, P. Denys, G. Loubert, M. H. Nicolas-Chanoine, J. L. Gaillard, and C. Lawrence. 2005. Spread of a Klebsiella pneumoniae strain producing a plasmid-mediated ACC-1 AmpC beta-lactamase in a teaching hospital admitting disabled patients. Antimicrob. Agents Chemother. 49:2095-2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pai, H., C. I. Kang, J. H. Byeon, K. D. Lee, W. B. Park, H. B. Kim, E. C. Kim, M. D. Oh, and K. W. Choe. 2004. Epidemiology and clinical features of bloodstream infections caused by AmpC-type-beta-lactamase-producing Klebsiella pneumoniae. Antimicrob. Agents Chemother. 48:3720-3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Papanicolaou, G. A., A. A. Medeiros, and G. A. Jacoby. 1990. Novel plasmid-mediated beta-lactamase (MIR-1) conferring resistance to oxyimino- and alpha-methoxy beta-lactams in clinical isolates of Klebsiella pneumoniae. Antimicrob. Agents Chemother. 34:2200-2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Philippon, A., G. Arlet, and G. A. Jacoby. 2002. Plasmid-determined AmpC-type beta-lactamases. Antimicrob. Agents Chemother. 46:1-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Raskine, L., I. Borrel, G. Barnaud, S. Boyer, B. Hanau-Bercot, J. Gravisse, R. Labia, G. Arlet, and M. J. Sanson-Le-Pors. 2002. Novel plasmid-encoded class C beta-lactamase (MOX-2) in Klebsiella pneumoniae from Greece. Antimicrob. Agents Chemother. 46:2262-2265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rhimi-Mahjoubi, F., M. Bernier, G. Arlet, Z. B. Jemaa, P. Jouve, A. Hammami, and A. Philippon. 2002. Identification of plasmid-encoded cephalosporinase ACC-1 among various enterobacteria (Klebsiella pneumoniae, Proteus mirabilis, Salmonella) isolated from a Tunisian hospital (Sfax 997-2000). Pathol. Biol. (Paris) 50:7-11. [DOI] [PubMed] [Google Scholar]
- 32.Shahid, M., A. Malik, M. Agrawal, and S. Singhal. 2004. Phenotypic detection of extended-spectrum and AmpC beta-lactamases by a new spot-inoculation method and modified three-dimensional extract test: comparison with the conventional three-dimensional extract test. J. Antimicrob. Chemother. 54:684-687. [DOI] [PubMed] [Google Scholar]
- 33.Song, W., E. S. Moland, N. D. Hanson, J. S. Lewis, J. H. Jorgensen, and K. S. Thomson. 2005. Failure of cefepime therapy in treatment of Klebsiella pneumoniae bacteremia. J. Clin. Microbiol. 43:4891-4894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Thomson, K. S., Z. A. Mejglo, G. N. Pearce, and T. J. Regan. 1984. 3- Dimensional susceptibility testing of beta-lactam antibiotics. J. Antimicrob. Chemother. 13:45-54. [DOI] [PubMed] [Google Scholar]
- 35.Tzouvelekis, L. S., E. Tzelepi, and A. F. Mentis. 1994. Nucleotide sequence of a plasmid-mediated cephalosporinase gene (blaLAT-1) found in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 38:2207-2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Verdet, C., Y. Benzerara, V. Gautier, O. Adam, Z. Ould-Hocine, and G. Arlet. 2006. Emergence of DHA-1-producing Klebsiella spp. in the Parisian region: genetic organization of the ampC and ampR genes originating from Morganella morganii. Antimicrob. Agents Chemother. 50:607-617. [DOI] [PMC free article] [PubMed] [Google Scholar]