Abstract
This study shows that solitary, dormant human cancer cells, retrieved from metastasis-free organs of animals carrying spontaneously metastatic primary tumors, can reactivate their tumorigenic and metastatic potency. The tumors were produced by MDA-MB-435 CL16 breast cancer cells permanently labeled with green fluorescent protein and the neomycin resistance gene. This enabled unequivocal identification of tumor cells emerging from organ explants cultured in neomycin to eliminate nonneoplastic host cells. Rescued cells resumed proliferation and generated lines that were tumorigenic and metastatic in fresh animals. All resulting primary and secondary tumors were uniformly labeled. Cells recovered from bone marrows and spleens, where there were no metastases, were as tumorigenic and metastatic as cells recovered from lungs and lymph nodes, which are the preferred sites of colonization for this tumor line. This evidence that malignant growth of disseminated cancer cells is suspended indefinitely by microenvironmental conditions in metastasis-free organs, although it is still active in others of the same host, shows that neoplastic progression can be arrested and has far-reaching biological and clinical implications. Specifically, it predicts the existence of natural, nonimmune host mechanisms that stimulate or inactivate tumor growth in different anatomical sites, which may be exploitable for therapeutic benefit.
Clinical and experimental studies on humans and animals have established that metastasizing tumors preferentially establish secondary colonies in certain organs and fail to grow in others.1–6 These nonrandom patterns of colonization are clinically predictable and are related to the site of origin of the primary tumor. Ultimately, the growth of the secondary tumors leads to failure of the vital organs because of their replacement by tumor tissue, and this is the major cause of death in cancer patients.4 Many studies7–9 have demonstrated that numerous blood-borne tumor cells transit the pulmonary capillary bed and enter the systemic circulation,10 which distributes them to all organs. Therefore, disseminating cells from the same primary tumor fail to grow in most organs although they do make secondary tumors in some favored sites. Dissemination of cancer cells by vascular channels is a necessary precondition for tumor metastasis but is not sufficient to ensure the formation of secondary tumors in distant organs. Consequently, disseminated cancer cells (DCCs) are notthe same as metastases. To understand the underlying mechanisms involved in these events, it is essential to distinguish clearly between the presence of metastases and that of disseminated cells with unfulfilled potential.
Little is known about the fate, life expectancy, and malignant potential of the DCCs that come to rest in organs where metastases are notformed. This present investigation was conducted to determine whether they soon die and are eliminated or whether they commonly survive for long periods. This is important because, although breast cancer commonly metastasizes to the lungs, bones, lymph nodes, and liver,1,11 distant recurrences in other organs may occasionally become evident many years after resection of the primary tumor. In such examples of delayed recurrence, the cancer cell must have departed from the primary tumor before it was resected. It is presumed to have then entered a dor-mant state, until some signal reactivated tumorigenic growth.12–14 To design therapy that is effective but does not overtreat patients, we need more information on the frequency, lifespan, and tumorigenic potency of such sequestered tumor cells and the molecular mechanisms that keep them quiescent. Conversely, knowledge of the molecular environmental signals favoring secondary growth in other “permissive” organs could allow the design of new therapeutic antagonists.
The recent introduction of techniques to permanently label cells and their progeny and to follow their behavior in vivo12,15,16 now makes such investigation possible. Initial studies with such methods have enabled us to establish that many tumor cells that are spontaneously disseminated from undisturbed tumors can lie dormant but viable in the lungs of mice that had no metastases for up to a quarter of the lifetime of the host.16 However, in animals with multiple pulmonary and/or lymph node metastases, the fate of disseminated tumor cells in metastasis-free organs remained unknown. If it could be convincingly shown that they regularly survive in substantial numbers, the reasons for their biological quiescence could be of potential therapeutic value. In the current work, we use cell lines descended from a matched, isogenic pair of highly metastatic (M-4A4) and weakly metastatic (NM-2C5) cell lines labeled with green (GFP) or red (RFP) fluorescent protein, cloned from the human breast cancer cell line MDA-MB-435,17,18 to investigate this issue. Our experimental protocol examined the destinations, fate, and malignant potential of cells that have disseminated spontaneously from undisturbed primary tumors generated by these lines in the mammary glands of SCID mice. The focus was to determine whether labeled tumor cells could be seen and retrieved from organs that are routinely metastasis-free and, if so, to investigate their viability, tumorigenicity, and metastatic potential. The results unequivocally demonstrated that many quiescent tumor cells do survive in all organs tested, even after the excision of the primary tumor, and that the cell populations that they generate on reactivating their proliferative potential are equally as tumorigenic and metastatic as cells obtained from the original tumors or from their metastases. From previous work16 we know that such cells can survive in the extravascular compartment of tissues for up to 6 months after tumor excision. Additionally, we have confirmed that nonmetastatic tumors also disseminated many cells that lay dormant in several organs other than the lungs. These cells were viable and tumorigenic after retrieval and reinoculation into the mammary gland but were still nonmetastatic although capable of dissemination, demonstrating that disseminated cells require conducive stroma from specific organs to make tumors.
These data provide direct evidence that the widespread persistence of disseminated tumor cells for extended periods of time after tumor excision is not a rare phenomenon and continues to represent a potential hazard to the host. Conversely, the tight control exerted over the growth of undoubtedly malignant tumor cells, in many organs, by natural, nonimmune environmental conditions, demonstrates that tumorigenic growth and behavior is not irreversible and is potentially vulnerable to novel therapeutic approaches that exploit mechanisms holding tumor cells dormant.
Materials and Methods
Cell Culture and Fluorescent Labeling
Breast cancer cell lines NM-2C5 and M-4A4 CL16, derived by single cell cloning17 from the MDA-MB-435 cell line, were used in this study. Their metastatic potential (high metastatic potential for M-4A4 CL16 cell line; low metastatic potential for the NM-2C5 cell line) have been described previously.17–19 These cell lines were engineered to constitutively express GFP and neomycin resistance genes by transduction with retroviral vectors.16 A few experiments were conducted with RFP-labeled cells to ensure that the fluorescent signals observed were not attributable to autofluorescence and to confirm that the labels were not inducing unusual behavior.
Spontaneous Metastasis Assay
Anesthetized batches of female SCID/B17 mice, 8 weeks old, were inoculated with 1 × 106 labeled tumor cells suspended in 1:1 mixture of extracellular matrix from the Engelbreth-Holm-Swarm sarcoma (Sigma Chemical Co., St. Louis, MO) and culture medium. Injections (0.1 ml/gland) were made under direct vision of the surgically exposed mammary gland through a dissecting stereoscope (Carl Zeiss, Thornwood, NY). A few experiments were conducted in nude/BalbC mice to confirm the results in another host strain. Results of spontaneous metastasis assays were evaluated at necropsy (see below).
Fluorescence Microscopy and Histology
The surfaces of fresh organs excised at necropsy were examined for presence of fluorescent cells with a Leica fluorescence stereomicroscope (model MZFL III; Leica Microsystems, Bannockburn, IL). At this time point, 12 weeks after orthotopic tumor cell inoculation, primary tumors averaged 2 cm in diameter and glowed bright apple green or rose red under appropriate illumination. GFP fluorescence at 488 nm was best distinguished from tissue autofluorescence using a single excitation dual emission 35002 mzfl filter from Chroma (Rockingham, VT), whereas specific RFP fluorescence was best assessed using an eGFP/DsRed dual excitation and emission filter (51019 set, Chroma). Fragments of organs were then frozen in OCT, cryosectioned (10 μm thickness), and scanned for individual fluorescent cancer cells deeper within the organs. Adjacent 5-μm H&E-stained sections were examined with a standard brightfield microscope for parallel histopathological assessment of tumor cell location in the tissues.
Cell Retrieval Experiments
Several internal organs, confirmed to be metastasis-free by fluorescence microscopy, were excised from tumor-bearing donor mice (M-4A4 CL16 and NM-2C5) at necropsy, 3 months after tumor cell inoculation. These included liver, spleen, lungs, lymph nodes, kidneys, and adrenals. In addition the marrow was flushed with Dulbecco’s modified Eagle’s medium through a fine gauge needle, from the long bones (femur and humerus) after excision of the proximal and distal ends. In animals known to have pulmonary metastases, the abdominal viscera were sampled first, to avoid contamination by spillage of loose tumor cells from the lungs and pleural cavity. The thoracic cavity was then opened with fresh surgical instruments and the lungs and pleura were examined in both white and blue (488 nm) light for metastases. Metastases and primary tumors were excised to obtain control cells. All samples were minced individually and explants cultured in RPMI 1640 containing 10% newborn calf serum. Peripheral blood cultures were also plated in the same type of medium. After 72 hours, tissue debris was removed, and plastic-adherent cells were subcultured in selective medium that contained 200 μg/ml of G418 for 1 week, after which the concentration was increased to 400 μg/ml for 1 week and then to 600 μg/ml. G418 was removed 2 weeks later and replaced with standard culture medium. Viable recovered cells at this time were 100% GFP-positive. After collection of tissues for culture, a fragment was frozen in OCT compound (Sakura, Torrance, CA) for histopathological analysis.
Testing of Tumorigenicity and Metastatic Capability of Retrieved Cancer Cells
Cancer cells retrieved from the spleen, bone marrow, and other organs (see Results) were grown, after selection in G418 in vitro, until labeled stable cell lines were obtained from each site. Each DCC-derived tumor line was then reinoculated in the mammary fat pads of fresh batches of SCID mice. These recipients of inoculated DCC lines were euthanized at 12 weeks after inoculation, when the primary tumor had reached a diameter of ∼2 cm. At necropsy, all of the major organs were examined by fluorescence microscopy for metastatic deposits and disseminated tumor cells.
Primary Cultures of Mouse Organs and Derivation of Conditioned Medium
Normal cells from spleens, bone marrow, and lungs were obtained by culture of minced organ fragments in RPMI 1640 medium containing 20% fetal calf serum for 1 week, after which the cells were used immediately in co-culture experiments or cryopreserved. By this time hematological cells and epithelial cells had disappeared and the cultures consisted only of adherent mesenchymal cells, which could easily be distinguished from tumor cells in co-cultures because the latter were labeled with GFP. Organ-conditioned media were obtained from explant cultures of healthy organs in serum-free RPMI 1640 for 48 hours as described in Horak and colleagues.20 We determined that this method yields protein concentrations in the range of 11 to 14 mg/ml for all organs tested. Supernatants were centrifuged at 900 × g to remove debris and either used immediately or aliquoted before freezing at −20°C. Results were similar with organ-conditioned media cryopreserved for differing periods up to 3 months.
In the absence of knowledge of the chemical composition of a possible mediator, the organ-conditioned media were diluted with serum-free medium, and doses were standardized by dose response curves as described in Horak and colleagues.20 The optimum concentration of total protein in the medium was found to be 0.75 mg/ml. Assays were conducted in triplicate in 96-well plates with 1000 tumor cells in 100 μl of organ-conditioned media per well.
Culture of Tumor Cells with Conditioned Media or Normal Cells
Co-culture experiments involved plating 1 × 103 tumor cells/well in flat-bottomed 96-well plates with serum-free organ-conditioned media (100 μl) or with fivefold numbers of normal mesenchymal cells from the lungs, spleens, or marrow of tumor-free SCID mice. Types of tumor cells tested included DCC cultures retrieved from the spleen, lymph nodes, bone marrow, peripheral blood, liver, and kidney and control cells from lung metastases and primary tumors. After 5 days culture of tumor cells with conditioned media or normal cells, commercial MTT21 and BrdU assays were performed to evaluate changes in cell numbers and proliferation (Supplemental Methods section, see http://ajp.amjpathol.org). For co-culture experiments, separate visual counts of GFP-labeled tumor cells and unlabeled normal cells were performed with a hemocytometer, to measure the effects on the tumor cell population and the normal cells separately. In these experiments the normal cells were in fivefold excess to simulate conditions experienced by dormant tumor cells in vivo and to compare results with data from the MTT and BrdU assays above.
Results
Metastatic Proficiency of Tumor Cell Lines
The metastatic proficiencies of the labeled cell lines used in this work were greatly different from each other (Table 1). Line M-4A4 CL16 was chosen because of its reliable pattern (ie, frequency and burden) of heavy metastatic spread to the lungs and lymph nodes and the rarity of metastases in other sites. It was derived from its metastatic parent clone M-4A4 by five successive cycles of selection and reinoculation of cells from spontaneous metastases in immunocompromised mice,19 to increase its metastatic potency. Conversely the NM-2C5 line is only very weakly metastatic but is locally tumorigenic in the mammary gland in 100% of animals. The metastatic proficiencies of cell lines derived from retrieved DCCs are described below.
Table 1.
Tumorigenicity and Metastatic Proficiencies of Parental Cell Lines
| Cell line | M-4A4 CL16 | NM-2C5 | |
|---|---|---|---|
| Number of mice injected | 32 | 8 | 26 |
| Formation of primary tumor | 32 | 8 | 26 |
| Primary tumor resection (yes/no) | No | Yes | No |
| Formation of metastases | |||
| Lung | 32* | 8* | 6† |
| Lymph node | 28‡ | 6‡ | 16§ |
| Liver | 0 | 1 | 0 |
| Pancreas | 2 | 0 | 0 |
| Ovary | 1 | 0 | 0 |
| Adrenal gland | 1 | 0 | 0 |
More than 100/animal.
Less than five/animal.
Fifty to one hundred confluent thoracic nodal metastases and several regional nodes positive.
Single regional node and no thoracic metastases.
Detection and Retrieval of DCCs from Metastasis-Free Internal Organs
Fluorescence Microscopy
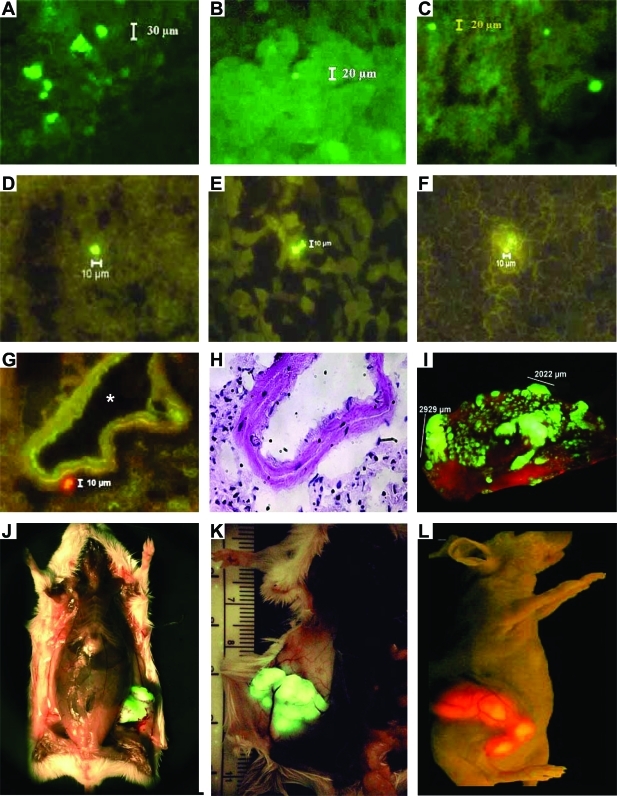
Fluorescence microscopy of the surfaces of these organs revealed scattered single, labeled human tumor cells, or small clumps of two or three cells, in many host organs. Occasional clumps of 3 to 10 cells were seen in the pulmonary capillaries and less frequently in some other organs (eg, spleen, where the blood flow is slow). These were not vascularized and were not micrometastatic colonies. The presence of clumps was not a regular feature, and their slightly higher incidence in the lungs than in other organs may reflect the greater dose of cells reaching this organ owing to circulatory anatomy. In animals inoculated orthotopically with the metastatic M-4A4 CL16 cell line, we routinely saw many individual cells in the lungs, lymph nodes, spleen, liver, and kidneys (Figure 1, A–C), although metastases from the tumors in the breast of these animals were only present in the lungs and lymph nodes. In the lungs, solitary labeled cells were present between the metastatic deposits. In animals carrying mammary tumors generated by NM-2C5 cells, we observed disseminated individual cancer cells only in the lungs, lymph nodes, spleen, and liver. Similar results were observed with tumors generated by the same cell lines labeled with red fluorescent protein, confirming the reproducibility of the finding of long-term persistence of cancer cells in many organs where metastases have never been seen in our detailed pathological studies in vivo with these lines.
Figure 1-6935.
DCCs and tumors and metastases formed by retrieved cells. A–C: Scattered fluorescing cancer cells detected by microscopy of the surface of the spleen (A), kidney (B), and liver (C). D–F: Frozen sections show individual cancer cells deeper within the spleen (D), kidney (E), and liver (F). Fluorescence microscopy confirmed that labeled cancer cells (G) lay outside blood vessels (asterisk) in the tissues, although they were not identifiable in adjacent H&E-stained sections (H). In G a RFP-labeled tumor cell is bright red, background autofluorescence is pale green. Subsequent panels show pulmonary metastases (I) and mammary carcinomas generated by reinoculated fluorescent DCCs, retrieved from spleen (J) and bone marrow (K and L).
Histopathology and Necropsy Correlations
Fluorescence microscopy of frozen sections demonstrated that many single, labeled tumor cells are also scattered throughout the tissues deep in the interior of the organs including the lungs, lymph nodes, spleen, liver, and kidneys (Figure 1, D–F). These cells could not be identified in adjacent frozen sections stained with hematoxylin and eosin (H&E) nor in high-resolution light microscopy of H&E-stained paraffin sections of these organs, demonstrating the value of the fluorescent labeling method for tracking disseminating tumor cells. The resolution attainable by light microscopy of frozen sections was not always sufficient to establish unequivocally whether individual labeled cells were intra- or extravascular in location, but in some instances the histological evidence that the tumor cell had departed from the blood and entered the tissues of the host organs was convincing (Figure 1, G and H). Further supporting evidence was obtained from the tumor excision experiments described below.
Histopathologically, all primary tumors were poorly differentiated mammary adenocarcinomas. Pulmonary metastases were located either inside large blood vessels, around the blood vessels, or invading the lung parenchyma. Under the fluorescent microscope, a section of a metastasis presents a heterogeneous population of tumor cells, showing bright fluorescent areas interspersed with less bright areas, invading weakly autofluorescent host tissue.
Retrieval and Culture of Disseminated Cells
Although the fluorescent proteins are very sensitive and reliable markers for the detection of solitary disseminated tumor cells resting in many organs where metastases did not form, the technique does not confirm their continuing viability or tumorigenic potential. To evaluate their viability and proliferative capability, we attempted to retrieve the tumor cells from various metastasis-free organs. After culture of minced organ fragments in medium containing the neomycin analog G418, we reproducibly obtained pure populations of fluorescent cancer cells retrieved not only from the lungs but also from the peripheral blood, the liver, spleen, kidneys, lymph nodes, and bone marrow of animals carrying M-4A4 CL16 tumors. From 17 mice carrying labeled metastatic tumors (M-4A4 CL16), we retrieved DCCs from blood, lymph nodes, spleen, bone marrow, liver, and kidneys of differing numbers of animals as shown (Table 2). These cells grew progressively and eventually (after 6 to 8 weeks) became confluent in the culture flasks, and we derived permanent DCC lines from them. From 19 animals bearing orthotopic, labeled NM-2C5 tumors, we were able to culture tumor cells from the lungs, blood, and bone marrow (Table 2). These cells also took ∼6 to 8 weeks to become confluent and grow for multiple passages in vitro.
Table 2.
Incidence of Retrieval and Culture of Cancer Cells from Blood and Various Organs of Mice Inoculated in the Mammary Gland with Tumor Cells of M4A4 and NM2C5 Lineages
| M4A4 cell lineage |
NM2C5 lineage |
|||
|---|---|---|---|---|
| Primary tumor not resected | Primary tumor resected | Primary tumor not resected | Primary tumor resected | |
| Total no. of mice | 17 | 10 | 19 | 8 |
| Blood | 12* | 0 | 1 | 0 |
| Lungs | † | † | 10 | 8 |
| Lymph nodes | 8 | 8 | 0 | 8 |
| Spleen | 11 | 5 | 0 | 4 |
| Bone marrow | 6 | 5 | 1 | 5 |
| Liver | 1 | 0 | 0 | 0 |
| Kidneys | 1 | 0 | 0 | 0 |
Values denote numbers of animals from which disseminated cells were retrieved and cultured from the anatomical sources specified.
†Because all mouse M-4A4 CL16 tumors had metastases in the lungs, lung cultures were routinely positive and acted as a positive control for the retrieval and culture techniques. Numbers are not included because this is a study of efficiency of retrieval of lines from dormant cells in routinely metastasis-free organs.
To establish whether the retrieved cells were transiting through the organs after recent release from the primary tumor or were stable residents in the extravascular tissues of each organ, we inoculated each of the labeled parental metastatic (M-4A4 CL16) and nonmetastatic (NM-2C5) cell lines orthotopically into 20 additional mice, surgically resected the primary tumors when they reached 1 cm diameter, and kept the mice under observation for 1 to 4 more weeks, after which they were euthanized and autopsied. This was done to allow time for loose cells circulating in the blood to be eliminated. Blood cultures from all of these animals were uniformly negative for the presence of labeled cells, confirming the eradication of circulating tumor cells. Tumors formed by the metastatic cell line, M-4A4CL16, produced visible lung metastases in nine of nine mice, and 1 to 4 weeks after resection, we were able to retrieve DCCs from the lymph nodes, spleen, and bone marrow of several animals as shown (Table 2). From eight animals carrying primary tumors formed by the nonmetastatic cell line NM-2C5-GFP, we retrieved labeled cancer cells from the lungs, lymph nodes, spleen, and bone marrow (Table 2), but none of these mice had lung metastases. Thus, in summary, this experiment demonstrated that, after primary tumor resection, the DCCs were not found in the blood circulation and they preferred to grow into metastases only in specific tissues, namely the lungs and lymph nodes, although dormant tumor cells lay resting in the tissues of many other organs in several animals.
Tumorigenicity and “Metastatic Memory” of the DCCs
Although the tumor cells in various organs were alive, as shown by the data described above, it remained an open question as to whether growth did not occur there because the cells had lost their tumorigenic properties or because they could not express their tumorigenicity in that environment. To test whether the labeled M-4A4 CL16 cells retrieved from organs in which metastases did not occur were still capable of making tumors, we reinoculated the cell lines derived from the spleen and bone marrow (DCC lines), into the mammary fat pads of new batches of mice. We chose tumor cells that had been recovered from metastasis-carrying mice that had undergone resection of their primary tumors 1 week to 1 month earlier to ensure that the experiment was testing the tumorigenicity of residentlabeled cells in these organs and that transiting loose cells in the blood had been washed away. Within 14 days after inoculation into the mammary glands, we observed new primary mammary adenocarcinomas developing in all mice in the batches inoculated with each DCC-derived cell line (Figure 1, J–L; and Table 3), and these continued to grow until we terminated the experiment, when tumors reached ∼2 cm in diameter. This took approximately the same time (3 months) as for the tumors generated by the original parental cell line. Necropsies confirmed that all of these DCC-recipient mice had histopathologically confirmed metastases in the lungs (Figure 1I) or other organs (Table 3). Statistical analysis using Fisher’s exact test confirmed that the metastatic behavior of the dormant cells retrieved from the donor animal was not significantly different from that of the parental cell line (P = 1).
Table 3-6935.
Tumorigenicity and Metastatic Proficiencies of Lines Derived from Disseminated Cancer Cells (DCCs)
| Original cell line Derived cell line | M-4A4 CL16 |
NM-2C5 |
|||
|---|---|---|---|---|---|
| Derived from spleen DCC | Derived from bone marrow DCC | Derived from spleen DCC | Derived from bone marrow DCC | Derived from lymph node DCC | |
| Number of mammary-injected mice | 10 | 10 | 3 | 3 | 3 |
| Formation of primary tumor* | 10 | 10 | 3 | 3 | 3 |
| Formation of metastasis† | |||||
| Lung‡ | 9 | 10 | 0 | 0 | 0 |
| Lymph node§ | 9 | 9 | 0 | 0 | 0 |
| Pancreas | 0 | 1 | 0 | 0 | 0 |
| Ovary | 1 | 0 | 0 | 0 | 0 |
| Adrenal gland | 0 | 1 | 0 | 0 | 0 |
*Number of mice with ∼2 cm in diameter tumor.
†Number of mice with metastases in each site.
‡More than 50 metastases.
§Numerous thoracic and regional nodal metastases.
For the weakly metastatic line NM-2C5, we have now added to our previous observations16 by demonstrating that viable dormant cells can be recovered and grown from many different organs, in addition to the lungs (see above and Table 2), and that all of the DCC-derived lines of this phenotype were still tumorigenic in the mammary gland. None of the nine mice inoculated with these DCC-derived NM-2C5 lines in the current study had metastases in the lungs or other organs although, once again, disseminated solitary cells were seen in the lungs and other organs, as in the first generation animals from which the cells had been retrieved. Statistically there was no significant difference between the metastatic inefficiency of these retrieved cells and that of the parent NM2C5 line to the lungs (P = 0.17) and thoracic nodes (P = 1), although the weak colonization of lower abdominal nodes by the parent line was apparently diminished in the rescued DCCs (P = 0.0002). Animals inoculated simultaneously, as a positive control, with tumor cells rescued from the lungs of M-4A4 CL 16 tumor-bearing mice with lung metastases developed second generation lung metastases from the primary tumors that arose at the site of tumor cell inoculation, confirming that the results with the NM2C5-derived cells were not attributable to technical failure. Moreover, in our previous investigation we documented that animals inoculated orthotopically or intravenously with cells recovered from the lungs of mice with NM2C5 tumors did not develop any metastases. Combination of the data reported in the current report (Table 3) with those that we described previously16 demonstrates that in 15 of 15 animals inoculated with cells of the NM2C5 lineage, from various donor organs, there were no metastases in any organ of any recipient, even though the cells produced tumors at the local site of inoculation. This contrasts starkly with the metastatic performance of the disseminated cells of the M4A4 lineage recovered from spleen and bone marrow, which showed tumorigenicity and metastasis in 20 of 20 animals inoculated (Table 3). The difference in the metastatic behavior of rescued cells of the M-4A4 CL16 and NM-2C5 lineages is, therefore, striking (P < 0.0000001) and shows that the inherent tendency of each lineage is retained with no increase in metastatic capability of NM2C5 cells that had been retrieved and recycled.
Influence of the Organ Microenvironment
Having confirmed the viability and tumorigenic potential of the DCCs, we conducted tests to evaluate whether their differential proliferation in organs that permit metastasis versus those in which they do not grow might be regulated by diffusible molecules in different environments or by direct contact with host cells. We studied DCCs retrieved from the spleen, lymph nodes, bone marrow, blood, liver, and kidneys, all from animals carrying M-4A4 CL16 metastatic tumors. For controls, cells obtained from M-4A4 CL16 primary tumors as well as from lung metastases were used.
Culture with Organ-Conditioned Medium
Parallel aliquots of each type of DCCs were cultured in medium that had been conditioned by fragments of lung, spleen, liver, adrenals, kidney, and bone marrow or in serum-free medium that had not been conditioned at all. It was found that 3 days of culture with serum-free conditioned medium substantially reduced the survival (measured by MTT assays) and proliferation (measured by BrdU incorporation) of cell lines retrieved from all organs tested relative to cells grown in serum-free medium without conditioning (Supplemental Figure A, see http://ajp.amjpathol.org). Liver- and kidney-conditioned medium were the most inhibitory. Lung-conditioned medium was no more supportive than that of other organs, except in the case of tumor cells derived from lung metastases. These cells also showed diminished survival and proliferation, but to less extent than in medium conditioned by other organs.
Culture with Living Host Cells
Co-culture of the rescued cell lines from various organs with live normal mesenchymal cells from the lung, bone marrow, or spleen had a stimulatory effect on the survival and proliferation of the tumor cells (Supplemental Figure B, see http://ajp.amjpathol.org), regardless of the source from which the DCCs had been recovered. All cell lines derived from DCCs grew better than control DCCs without normal host cells. However, we detected no significant differences, using these methods, between data from co-culture experiments with cells from normal spleens, where metastases do not occur, and data from experiments involving culture with cells from the lungs, where they grow steadily and relentlessly. Similar results were obtained with normal marrow cells (data not shown).
Discussion
The reproducible recovery of genetically labeled, viable, tumorigenic cells from many metastasis-free organs of tumor-bearing host animals demonstrates that disseminated, solitary, human tumor cells frequently survive for prolonged periods in many organs. Moreover, the regular recovery of such dormant cells from routinely metastasis-free organs of mice that possessed numerous pulmonary metastases shows the importance of the local microenvironment in determining whether secondary tumors grow there. In such individuals, the metastatic potency of the primary tumor, in that same host is confirmed by the secondary tumor deposits in the lungs, yet cells from the identical tumor lie dormant in other organs. The fact that dormant cells can reactivate tumorigenic and metastatic properties, which were latent in that location in vivo, provides compelling direct evidence that the growth of even the most virulent tumor cells can be arrested by normal, nonimmune mechanisms in many organs. The clinical significance of these observations is that identification of the specific molecular mechanisms that subdue growth of DCCs in some organs or, conversely, the mechanisms that promote the growth of DCCs in other organs in the same animal would provide important information for the design of targeted metastasis-controlling drugs.
The fate of individual cells disseminating spontaneously (ie, without experimental intervention) from undisturbed primary tumors growing orthotopically has been difficult to study until recently. This was because it was not possible to track the tumor cells and identify them with certainty among the normal cells of the host, if the DCCs had not produced secondary tumors at the terminus of their journeys. However, the introduction of the technique of permanent cell labeling by transduction with genes encoding fluorescent proteins (GFP and RFP) advanced the field considerably and enabled investigators to identify disseminating tumor cells and metastases22–24 with confidence.
Early studies using this method,12,15 demonstrated that, after intravenous inoculation, viable and tumorigenic labeled cells could be rescued from downstream organs, where they had not made secondary colonies, for up to 30 days after injection. The study by Goldberg and colleagues15 was the first to show the viability and tumorigenicity of such experimentally disseminated cells recovered from the lungs and reinoculated orthotopically. Subsequently, we showed that labeled human breast cancer cells, spontaneously disseminated from undisturbed orthotopic primary tumors in nude mice, could be retrieved from their lungs for up to 6 months after removal of the primary tumor,16 which corresponds to a quarter of the life time of the mouse. Those primary tumors were generated by the NM-2C5 cell line, which rarely made any metastases in their hosts, and the study revealed that even nonmetastatic tumors can and do release many cells into the vascular compartment. The retrieved DCCs recapitulated tumorigenesis when reinoculated into the mammary fat pads of fresh recipients, but these second-generation tumors were also nonmetastatic although they did disseminate single cells again, which is confirmed in our current study (see also similar findings by Goldberg and colleagues15 for comparison). Even reinoculation of our retrieved cells intravenously did not generate pulmonary colonies.
The novelty of the current work is in showing, for the first time, that labeled, spontaneously-disseminated dormant cells, retrieved from metastasis-free extra-thoracic organs of animals harboring multiple actively growing pulmonary metastases, routinely reactivated proliferation, tumorigenicity, and spontaneous metastatic capability when reinoculated orthotopically. These DCCs from aggressive M-4A4 CL16 mammary tumors were recovered from the spleen and marrow, where they fail to make metastases. This establishes conclusively that the failure of the dormant DCCs in these organs to make metastases, whereas those in the lungs of the same animal did, was not attributable to lack of intrinsic metastatic capability. We have also previously shown that failure to colonize is not due to insufficient tumor cells being distributed to an organ,3,4 because massive doses infused directly into the blood supply do not necessarily establish growing colonies. It follows that metastasis formation is unsuccessful in many sites due to some property of the host organs in which the tumor cells came to rest.
A further novelty of the findings of this study is that tumors generated by the rescued DCCs of both M-4A4-CL16 and NM-2C5 lineages retained their metastatic memory. On orthotopic inoculation, they reiterated the high or low metastatic character and preferential organ colonization patterns of their ancestors, even after residency in ectopic organs. The period of residency of individual cells in distant organs is difficult to determine because we know from direct sequential observations on mice bearing labeled tumors (D. Tarin, unpublished observations) that malignant cells depart from the orthotopic primary tumor intermittently over time, and some have, therefore, been in the ectopic site longer than others. However, the experiments involving excision of the primary tumor show that in some of our experiments the dormant cells had been resident in the distant organs for at least a month, since the source of the disseminating cancer cells had been removed (also, in previous work,16 we found that viable tumorigenic cancer cells could still be recovered from distant organs up to 6 months after excision of the primary tumor).
The results currently presented demonstrate the roles of both i) intrinsic properties of the tumor cells and ii) the permissive or favorable influence of the mammary gland, lungs, and lymph nodes in determining the behavior of these lineages, which could not grow in the other organs from where they were retrieved. This work therefore confirms, by previously impossible direct observations on spontaneously disseminated cancer cells, the importance of tumor interactions with the local host cells, emphasized by several previous investigators1–3,5,24 in both the early (in the orthotopic organ) and late (in the ectopic organ) stages of the metastatic process.6,25
Clinical studies on metastasis formation in ambulatory patients with different types of cancers, treated with peritoneovenous shunts for intractable malignant ascites,4 yielded similar conclusions, indicating that the current experimental findings have potential therapeutic significance. Recently, the short-term culture of tumor cells from bone marrow of patients with no overt signs of metastases from primary cancers of the breast, prostate, colon, and kidney has been reported,26,27 and studies describing disseminated single cells with abnormal genomic profiles have been performed,28 illustrating the relevance of the findings in the present work to the spread of cancer in the human body. Further research directed at understanding how cancer cells in some organs fail to make metastases when cells from the same tumor succeed in other organs in the same host may lead to drugs or antibodies that can suppress formation of new deposits and growth of existing metastases.
Our studies in vitro to analyze the nature of the influence of the microenvironment on the tumor cells indicated that all of the media conditioned by normal organs are inhibitory to all of the DCC lines and that co-culture with either lung or spleen cells promoted their proliferation. There were no differences in patterns of inhibition by the conditioned media that might explain enhanced growth of tumor cells in the lungs and lymph nodes relative to other organs. Conversely, the presence of normal spleen or lung cells promoted proliferation of all lines derived from DCCs, but both types of normal cells stimulated proliferation of DCCs from all other organs. Therefore, the observations in vitro using these methods were not informative about the interactions in vivo, which decide dormancy or growth. This contrasts with earlier observations by ourselves20 and by Nicolson and colleagues29,30on primary tumor cells or cell lines (not DCCs) exposed to various normal organ-conditioned media or fragments in which differential inhibitory and stimulatory effects were noted, corresponding to the preferential colonization sites of the tumor cells in vivo. Accordingly, we consider that the complexity of the relationships between DCCs and the cells of the host organ will be best analyzed with emerging techniques for in vivo analysis, including transgenic animals, laser capture microdissection, and molecular imaging. Our ongoing work to date, using laser capture microdissection coupled with microarray, quantitative polymerase chain reaction, and proteomics analysis, has provided unequivocal evidence of evolving and reciprocal molecular communication between tumor cells and their nonneoplastic neighbors during tumor progression and metastasis in this xenogeneic model system31 and in human prostate cancer32 and confirms the feasibility of using such techniques to unravel how metastasis-competent cells fail to grow in some organs but thrive in others.
Supplementary Material
Acknowledgments
We thank Dr. Kersi Pestonjamasp for the technical advice in fluorescence microscopy and Ms. Linda Mellor for administrative help.
Footnotes
Address reprint requests to David Tarin, Rebecca and John Moores Comprehensive Cancer Center and Department of Pathology, University of California, San Diego, 3855 Health Sciences Dr. MC0803, La Jolla, CA 92093-0803. E-mail: dtarin@ucsd.edu.
Supported by the Loppicola Foundation (research fellowship awards to M.S. and V.M.).
Supplemental material for this article can be found on http://ajp.amjpathol.org.
References
- Paget S. The distribution of secondary growths in cancer of the breast. Lancet. 1889;i:571–573. [PubMed] [Google Scholar]
- Hart IR, Fidler IJ. Role of organ selectivity in the determination of metastatic patterns of B16 melanoma. Cancer Res. 1980;40:2281–2287. [PubMed] [Google Scholar]
- Tarin D, Price JE. Influence of microenvironment and vascular anatomy on “metastatic” colonization potential of mammary tumors. Cancer Res. 1981;41:3604–3609. [PubMed] [Google Scholar]
- Tarin D, Price JE, Kettlewell MG, Souter RG, Vass AC, Crossley B. Mechanisms of human tumor metastasis studied in patients with peritoneovenous shunts. Cancer Res. 1984;44:3584–3592. [PubMed] [Google Scholar]
- Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411:375–379. doi: 10.1038/35077241. [DOI] [PubMed] [Google Scholar]
- Fidler IJ. The organ microenvironment and cancer metastasis. Differentiation. 2002;70:498–505. doi: 10.1046/j.1432-0436.2002.700904.x. [DOI] [PubMed] [Google Scholar]
- Fidler IJ. Metastasis: quantitative analysis of distribution and fate of tumor emboli labeled with 125 I-5-iodo-2′-deoxyuridine. J Natl Cancer Inst. 1970;45:773–782. [PubMed] [Google Scholar]
- Potter KM, Juacaba SF, Price JE, Tarin D. Observations on organ distribution of fluorescein-labelled tumour cells released intravascularly. Invasion Metastasis. 1983;3:221–233. [PubMed] [Google Scholar]
- Juacaba SF, Horak E, Price JE, Tarin D. Tumor cell dissemination patterns and metastasis of murine mammary carcinoma. Cancer Res. 1989;49:570–575. [PubMed] [Google Scholar]
- Wasserman L, Dreilinger A, Easter D, Wallace A. A seminested RT-PCR assay for HER2/neu: initial validation of a new method for the detection of disseminated breast cancer cells. Mol Diagn. 1999;4:21–28. doi: 10.1016/s1084-8592(99)80046-0. [DOI] [PubMed] [Google Scholar]
- Lee Y-T. Breast carcinoma: patterns of metastasis at autopsy. J Surg Oncol. 1983;23:173–180. doi: 10.1002/jso.2930230311. [DOI] [PubMed] [Google Scholar]
- Naumov GN, MacDonald IC, Weinmeister PM, Kerkvliet N, Nadkarni KV, Wilson SM, Morris VL, Groom AC, Chambers AF. Persistence of solitary mammary carcinoma cells in a secondary site: a possible contributor to dormancy. Cancer Res. 2002;62:2162–2168. [PubMed] [Google Scholar]
- Demicheli R. Tumour dormancy: findings and hypotheses from clinical research on breast cancer. Semin Cancer Biol. 2001;11:297–306. doi: 10.1006/scbi.2001.0385. [DOI] [PubMed] [Google Scholar]
- Meltzer A. Dormancy and breast cancer. J Surg Oncol. 1990;43:181–188. doi: 10.1002/jso.2930430312. [DOI] [PubMed] [Google Scholar]
- Goldberg SF, Harms JF, Quon K, Welch DR. Metastasis-suppressed C8161 melanoma cells arrest in lung but fail to proliferate. Clin Exp Metastasis. 1999;17:601–607. doi: 10.1023/a:1006718800891. [DOI] [PubMed] [Google Scholar]
- Goodison S, Kawai K, Hihara J, Jiang P, Yang M, Urquidi V, Hoffman RM, Tarin D. Prolonged dormancy and site-specific growth potential of cancer cells spontaneously disseminated from non-metastatic breast tumors, revealed by labeling with green fluorescent protein (GFP) Clin Cancer Res. 2003;9:3808–3814. [PubMed] [Google Scholar]
- Bao L, Pigott R, Matsumura Y, Baban D, Tarin D. Correlation of VLA-4 integrin expression with metastatic potential in various human tumour cell lines. Differentiation. 1993;52:239–246. doi: 10.1111/j.1432-0436.1993.tb00636.x. [DOI] [PubMed] [Google Scholar]
- Urquidi V, Sloan D, Kawai K, Agarwal D, Woodman AC, Tarin D, Goodison S. Contrasting expression of thrombospondin-1 and osteopontin correlates with absence or presence of metastatic phenotype in an isogenic model of spontaneous human breast cancer metastasis. Clin Cancer Res. 2002;8:61–74. [PubMed] [Google Scholar]
- Montel V, Huang T-Y, Mose E, Pestonjamasp K, Tarin D. Expression profiling of primary tumors and matched lung and lymph node metastases in a xenograft model of breast cancer. Am J Pathol. 2005;166:1565–1579. doi: 10.1016/S0002-9440(10)62372-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horak E, Darling DL, Tarin D. Analysis of organ-specific effects on metastatic tumor formation by studies in vitro. J Natl Cancer Inst. 1986;76:913–922. [PubMed] [Google Scholar]
- Hansen MB, Nielsen SE, Berg K. Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Methods. 1989;119:203–210. doi: 10.1016/0022-1759(89)90397-9. [DOI] [PubMed] [Google Scholar]
- Ahmed F, Wyckoff J, Lin EY, Wang W, Wang Y, Hennighausen L, Miyazaki J, Jones J, Pollard JW, Condeelis JS, Seegal JE. GFP expression in the mammary gland for imaging of mammary tumor cells in transgenic mice. Cancer Res. 2002;62:7166–7169. [PubMed] [Google Scholar]
- Wong CW, Song C, Grimes MM, Fu W, Dewhurst MW, Muschel RJ, Al-Mehdi AB. Intravascular location of breast cancer cells after spontaneous metastasis to the lung. Am J Pathol. 2002;161:749–753. doi: 10.1016/S0002-9440(10)64233-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman RM. Orthotopic transplant mouse models with green fluorescent protein-expressing cancer cells to visualize metastasis and angiogenesis. Cancer Metastasis Rev. 1999;17:271–277. doi: 10.1023/a:1006188412324. [DOI] [PubMed] [Google Scholar]
- Stephenson RA, Dinney CP, Gohji K, Ordonez NG, Killion JJ, Fidler IJ. Metastatic model for human prostate cancer using orthotopic implantation in nude mice. J Natl Cancer Inst. 1992;84:951–957. doi: 10.1093/jnci/84.12.951. [DOI] [PubMed] [Google Scholar]
- Solakoglu O, Maierhofer C, Lahr G, Breit E, Scheunemann P, Heumos I, Pichlmeier U, Schlimok G, Oberneder R, Kollermann MW, Kollermann J, Speicher MR, Pantel K. Heterogeneous proliferative potential of occult metastatic cells in bone marrow of patients with solid epithelial tumors. Proc Natl Acad Sci USA. 2002;99:2246–2251. doi: 10.1073/pnas.042372199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gangnus R, Langer S, Breit E, Pantel K, Speicher MR. Genomic profiling of viable and proliferative micrometastatic cells from early-stage breast cancer patients. Clin Cancer Res. 2004;10:3457–3464. doi: 10.1158/1078-0432.CCR-03-0818. [DOI] [PubMed] [Google Scholar]
- Klein CA, Schmidt-Kittler O, Schardt JA, Pantel K, Speicher MR, Riethmüller G. Comparative genomic hybridization, loss of heterozygosity, and DNA sequence analysis of single cells. Proc Natl Acad Sci USA. 1999;96:4494–4499. doi: 10.1073/pnas.96.8.4494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicolson GL, Dulski K, Basson C, Welch DR. Preferential organ attachment and invasion in vitro by B16 melanoma cells selected for differing metastatic colonization and invasive properties. Invasion Metastasis. 1985;5:144–158. [PubMed] [Google Scholar]
- Nicolson GL, Dulski K. Organ specificity of metastatic tumor colonization is related to organ-selective growth properties of malignant cells. Int J Cancer. 1986;38:289–294. doi: 10.1002/ijc.2910380221. [DOI] [PubMed] [Google Scholar]
- Montel V, Mose E, Tarin D. Tumor-stromal interactions reciprocally modulate gene expression patterns during carcinogenesis and metastasis. Int J Cancer. 2006;119:251–263. doi: 10.1002/ijc.21757. [DOI] [PubMed] [Google Scholar]
- Stuart RO, Wachsman W, Berry CC, Wang-Rodriguez J, Wasserman L, Klacansky I, Masys D, Arden K, Goodison S, McClelland M, Wang Y, Sawyers A, Kalcheva I, Tarin D, Mercola D. In silico dissection of cell-type-associated patterns of gene expression in prostate cancer. Proc Natl Acad Sci USA. 2004;101:615–620. doi: 10.1073/pnas.2536479100. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.