Abstract
The development of bleomycin-induced lung injury, which is a model of pulmonary fibrosis, results from inflammatory cell infiltration, a process highly regulated by the expression of multiple adhesion molecules. Therefore, bleomycin-induced lung fibrosis was examined in E-selectin−/− mice, P-selectin−/− mice, and E-selectin−/− mice treated with anti-P-selectin monoclonal antibody (mAb) in comparison of wild-type mice. E-selectin−/− mice treated with anti-P-selectin mAb exhibited augmented lung fibrosis histologically, increased lung collagen deposition, and increased mortality compared to wild-type mice. Furthermore, lung interferon-γ mRNA expression decreased in E-selectin−/− mice treated with anti-P-selectin mAb relative to wild-type mice, while tumor necrosis factor-α and interleukin-6 mRNA expression increased in these mice. Similar changes were observed in E-selectin−/− mice, albeit to a lesser extent than those treated with anti-P-selectin mAb. Remarkably, flow cytometric analysis revealed that the frequency of interferon-γ-producing natural killer T (NKT) cells in the bronchoalveolar lavage was decreased in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb compared with wild-type mice. Moreover, the majority of NKT cells expressed high levels of CXCR3, suggesting that NKT cell infiltration is also dependent on CXCR3 expression. These results suggest that E- and P-selectins synergistically inhibit lung fibrosis by promoting the recruitment of NKT cells.
Idiopathic pulmonary fibrosis is a chronic and often fatal disorder with a 5-year survival rate of ∼50%.1 In addition, pulmonary fibrosis is frequently associated with certain connective tissue diseases, especially systemic sclerosis, dermatomyositis, and polymyositis. It comprises a diverse group of diseases characterized by inflammatory infiltrates, disruption of alveolar structure, and excessive synthesis and deposition of extracellular matrix.1 Active pulmonary fibrosis is accompanied by increased number of leukocytes in the bronchoalveolar lavage (BAL) fluid.1,2 Although the pathogenesis of pulmonary fibrosis remains unknown, these findings suggest that leukocyte infiltration regulates subsequent fibrosis in the lung. Bleomycin-induced lung fibrosis is widely used as an established animal model of pulmonary fibrosis. Intratracheal administration of bleomycin induces acute alveolitis and interstitial inflammation, characterized by the sequential recruitment of leukocytes in the first week.3 Subsequent to these inflammatory responses, fibrotic responses characterized by fibroblast proliferation and extracellular matrix synthesis occur in the second week.3 It is generally assumed that leukocytes infiltrating into the lung are involved in the evolution of pulmonary fibrosis by secreting reactive oxygen species, fibrogenic cytokines, and growth factors.1
Leukocyte recruitment into inflammatory sites involves adhesive interactions between leukocytes and the vascular endothelium. The first of these interactions is mediated predominantly by the selectin family of adhesion molecules, which are specialized at capturing freeflowing leukocytes from the blood. The selectin family consists of three cell-surface molecules expressed by leukocytes (L-selectin), vascular endothelium (E- and P-selectins), and platelets (P-selectin).4 While P-selectin is rapidly mobilized to the surface of activated endothelium or platelets, E-selectin expression is induced within several hours after activation with inflammatory cytokines.4 Inhibition or loss of P- or E-selectins lead to a significant reduction in neutrophil rolling and acute emigration in many inflammatory models, such as the Arthus reaction, dermal inflammation, and peritonitis models.5–7 By contrast, some studies have suggested that loss of P-selectin expression exacerbates inflammatory responses, such as experimental glomerulonephritis and collagen-induced arthritis models.8,9 Thus, E- and P-selectins regulate inflammatory responses either positively or negatively, according to the tissue site and the nature of the inflammatory stimuli.
We previously reported that the deficiency of L-selectin and/or ICAM-1 inhibited bleomycin-induced pulmonary fibrosis,3 suggesting that adhesion molecules regulate the fibrotic process in this model. A previous study showed that bleomycin-induced pulmonary fibrosis was inhibited in transgenic mice overexpressing soluble E-selectin that inhibits the binding of leukocytes to E-selectin on the endothelium.10 However, studies investigating the contribution of E- and P-selectins to this model are limited. The results of the present study indicate that E- and P-selectins synergistically inhibit bleomycin-induced lung fibrosis by promoting the recruitment of NK1.1+ T lymphocytes.
Materials and Methods
Animals
E-selectin−/− and P-selectin−/− mice were obtained from The Jackson Laboratory (Bar Harbor, ME). All mice were backcrossed 10 generations onto the C57BL/6 genetic background. All mice were healthy, fertile, and did not display evidence of infection or disease. Mice used for experiments were 12 to 16 weeks old. Age-matched wild-type (C57BL/6) mice (Jackson Laboratory) were used as controls. Both body and lung size was similar for mutant and wild-type mice (data not shown). All mice were housed in a specific pathogen-free barrier facility and screened regularly for pathogens. All studies and procedures were approved by the Committee on Animal Experimentation of Kanazawa University Graduate School of Medical Science.
Bleomycin Administration
Bleomycin sulfate (Nippon Kayaku, Tokyo, Japan) was administered to mice anesthetized by inhalation of diethyl ether. Using aseptic techniques, a single incision was made at the neck and the muscle covering the trachea was snipped to expose the tracheal rings. A single intratracheal instillation of bleomycin sulfate (8 mg/kg) in 250 μl of sterile saline was performed. For a blocking study, monoclonal antibodies (mAbs) to P-selectin (RB40.34, rat IgG1, 30 μg per mouse; BD PharMingen, San Jose, CA) were injected intravenously 30 minutes before bleomycin challenge and after challenge three times per week. The mAb concentration used in this study was that required to inhibit P-selectin-dependent leukocyte rolling in vivo as previously described.11
Preparation of BAL
BAL cells were prepared as described elsewhere.12 Briefly, at 2, 4, 8, 12, and 16 days after instillation, the mice were sacrificed and both lungs were excised. BAL fluid was collected as follows: 1 ml of saline was instilled three times and withdrawn from the lungs via an intratracheal cannula. A 500-μl aliquot of the recovered BAL fluid was analyzed for total and differential leukocyte counts after lysis of erythrocytes. Total leukocyte counts were performed using a hemocytometer in the presence of trypan blue. Cell differential counts were determined after cytospin centrifugation with May-Giemsa staining. A total of 200 cells were counted from randomly chosen high-power microscopic fields for each sample.
Histological Examination
The same mice were used for histological evaluation of fibrosis and measurement of hydroxyproline content, whereas separate mice were used for analysis of BAL components. After the right lung of each mouse was removed for hydroxyproline assay, the left lung was inflated and fixed with 3.5% paraformaldehyde and then paraffin-embedded. Six-μm sections were stained with hematoxylin and eosin (H&E) to evaluate alveolitis and with Azan-Mallory stain to identify collagen deposition in the lung. Five sections of the entire lung stained with H&E were chosen randomly from each mouse. The severity of lung inflammation was determined by semiquantitative scoring system as previously described.13 The pathological scores were defined as follows: 0, no lung abnormality; 1, presence of inflammation and fibrosis involving <25% of the lung; 2, lesions involving 25 to 50% of the lung; and 3, lesions involving >50% of the lung. The mean of the pathological scores for five sections was determined for individual mice.
Hydroxyproline Assay
Hydroxyproline is a modified amino acid uniquely found at a high percentage in collagen. Therefore, the tissue hydroxyproline content of lungs was measured as a quantitative measure of collagen deposition as previously described.14 Briefly, lungs were harvested 16 days after bleomycin administration and homogenized in 2 ml of phosphate-buffered saline, pH 7.4, with a Tissue Tearor (Iuchi, Osaka, Japan). Each sample (0.5 ml) was desiccated overnight at 110°C and then digested in 1 ml of 6 N HCl for 8 hours at 120°C. Samples were again desiccated for 6 hours at 120°C. Fifty μl of citrate/acetate buffer (5% citric acid, 7.24% sodium acetate, 3.4% NaOH, 1.2% glacial acetic acid, pH 6.0) and 1 ml of chloramine T solution (1.13 g of chloramine T, 8 ml of 1-propanol, 8 ml of H2O, 64 ml of citrate/acetate buffer) were added to each sample and the samples were left at room temperature for 20 minutes. Then, 1 ml of Ehrlich’s solution [10.13 g of p-(dimethylamino)benzaldehyde, 41.85 ml of 1-propanol, 17.55 ml of 70% perchloric acid] was added and incubated for 15 minutes at 65°C. Samples were cooled for 10 minutes, spun at 3100 × g for 5 minutes, and read at 550 nm on a spectrophotometer. A hydroxyproline standard solution of 0 to 4 mg/ml was used to generate a standard curve.
Flow Cytometric Analysis
BAL cells were stained with anti-CD3-PerCP Ab (clone 145-2C11; BD PharMingen) and either anti-NK1.1-phycoerythrine antibody (Ab) (clone PK136; BD PharMingen) or anti-CXCR3-phycoerythrin Ab (clone 220803; R&D Systems, Minneapolis, MN). Cells were washed and analyzed on a FACScan flow cytometer (BD PharMingen). Positive and negative populations of cells were determined using unreactive isotype-matched mAbs (Beckman-Coulter, Fullerton, CA) as controls for background staining.
Intracellular Cytokine Analysis
Interferon (IFN)-γ production in BAL lymphocytes was determined by flow cytometric intracellular cytokine analysis, as previously described.15 Briefly, cells were suspended at 106/ml in RPMI 1640 containing 2 mmol/L l-glutamine and incubated with 10 μg of brefeldin A for 2 hours at 37°C. Samples were stained for cell surface markers, NK1.1 and CD3, for 30 minutes at 4°C. After permeabilizing with FACS permeabilizing solution according to the manufacturer’s instructions (BD PharMingen), the cells were then stained for intracellular IFN-γ. Cells were washed and analyzed on a FACScan flow cytometer (BD PharMingen). Positive and negative populations of cells were determined using unreactive isotype-matched mAbs (Beckman-Coulter) as controls for background staining.
Real-Time Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Lungs were harvested 8 days after bleomycin administration, and total RNA was isolated from frozen lung specimens using RNeasy spin columns (Qiagen Ltd., Crawley, UK) and digested with DNaseI (Qiagen Ltd.) to remove chromosomal DNA in accordance with the manufacturer’s protocols. Total RNA was reverse-transcribed to cDNA using a reverse transcription system with random hexamers (Promega, Madison, WI). Expression of cytokines was analyzed by using real-time PCR quantification method, according to the manufacturer’s instructions (Applied Biosystems, Foster City, CA). Sequence-specific primers and probes were designed by predeveloped TaqMan assay reagents (Applied Biosystems). Real-time PCR (40 cycles of denaturation at 92°C for 15 seconds and annealing at 60°C for 60 seconds) was performed on an ABI Prism 7000 sequence detector (Applied Biosystems). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used to normalize mRNA. Relative expression of real-time PCR products was determined by using the ΔΔCt method16 to compare target gene and GAPDH mRNA expression. One of the control samples was chosen as a calibrator sample.
In Vivo Depletion of NK1.1+ Cells
Anti-NK1.1 mAb (PK136, 200 μg/mouse; BD PharMingen) was injected intraperitoneally at 4 and 2 days before bleomycin instillation and weekly thereafter. These were the mAb concentrations required to deplete NK1.1+ cells in vivo as previously described.17 Lungs were harvest 16 days after bleomycin administration. Depletion of NK1.1+ cells was confirmed by two-color flow cytometric analysis. The pathological scores, hydroxyproline assay, and BAL cell counts were performed and evaluated as described above.
Statistical Analysis
The Mann-Whitney U-test was used for determining the level of significance of differences in sample means and Bonferroni’s test was used for multiple comparisons.
Results
Pulmonary Fibrosis Is Augmented by Loss of E-Selectin Expression with or without P-Selectin Blockade
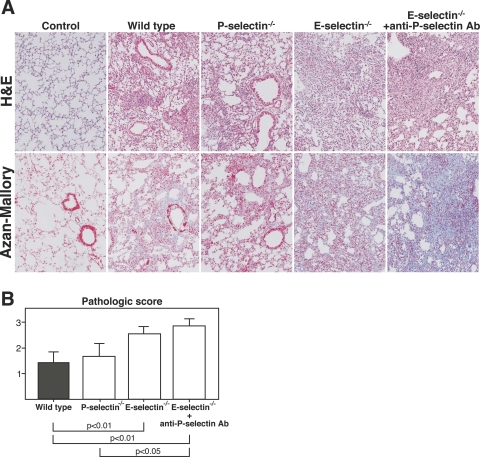
In bleomycin-induced pulmonary fibrosis, leukocytes accumulate mainly during the first week with subsequent fibrotic responses occurring in the second week. To estimate the fibrotic changes in the lung, H&E and Azan-Mallory stainings were performed 16 days after bleomycin administration. E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb exhibited an increase in lung parenchyma consolidation with disruption of the alveolar architecture and increased inflammatory cell infiltration compared with wild-type and P-selectin−/− mice (Figure 1A). The pathological scores of lung sections were significantly increased in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb compared with wild-type mice (P < 0.01 and P < 0.01, respectively; Figure 1B). Lung sections from saline-treated mutant mice showed no significant pulmonary consolidation or fibrosis (data not shown). Collagen deposition assessed by Azan-Mallory staining was similar in wild-type and P-selectin−/− mice (Figure 1A). In contrast, E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb showed augmented collagen deposition.
Figure 1.
Bleomycin-induced lung fibrosis was augmented in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin. A: Representative histological sections of lungs from wild-type, P-selectin−/−, E-selectin−/−, and E-selectin−/− mice treated with anti-P-selectin mAb 16 days after intratracheal bleomycin administration. Wild-type mice that received intratracheal saline injection served as controls (control). The lung sections were stained with H&E to evaluate alveolitis and with Azan-Mallory to identify collagen deposition. B: Lung fibrosis was evaluated according to the criteria described in Materials and Methods. The results are indicated as a mean ± SEM of eight mice in each group. Original magnifications, ×100.
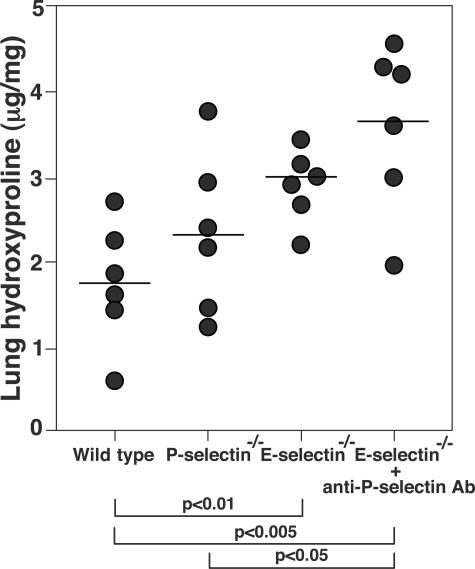
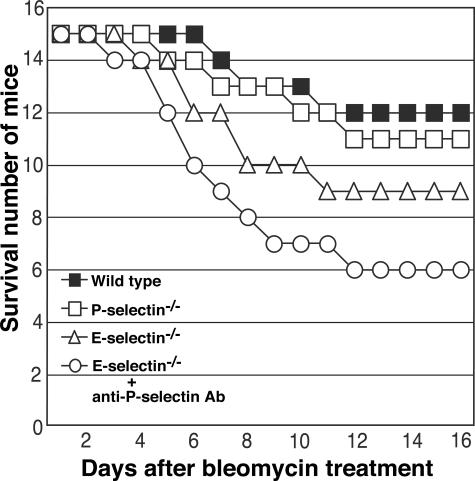
Pulmonary fibrosis was further assessed by quantitatively measuring the hydroxyproline content in the lungs. Sixteen days after bleomycin administration, the hydroxyproline content of lungs from E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb were significantly increased compared with wild-type mice (P < 0.01 and P < 0.005, respectively; Figure 2), whereas there was no significant difference between wild-type and P-selectin−/− mice. Consistent with the histological and quantitative evaluation, E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb suffered an increase in mortality relative to wild-type mice (20 and 40% increase, respectively; Figure 3). Thus, loss of E-selectin alone or concurrent loss of P- and E-selectin function resulted in a severe exacerbation of bleomycin-induced lung fibrosis.
Figure 2.
Hydroxyproline content of lungs in mutant and wild-type mice 16 days after intratracheal bleomycin administration. Symbols represent results from individual mice with the horizontal line indicating the mean value for each group of mice.
Figure 3.
Time course of survival in mutant and wild-type mice after intratracheal bleomycin administration (15 mice in each group).
Leukocyte Infiltration Is Increased by Loss of E-Selectin Expression with or without P-Selectin Blockade
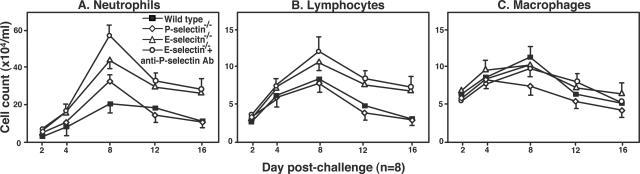
The effects of cell adhesion molecule blockade on leukocyte infiltration into the lung were evaluated by counting the inflammatory cells in the BAL at 2, 4, 8, 12, and 16 days after bleomycin challenge. The number of neutrophils in the BAL reached a maximum 8 days after bleomycin challenge and then gradually decreased in wild-type mice (Figure 4A). The neutrophil influx at day 8 after challenge was significantly increased in P-selectin−/− mice (P < 0.01), E-selectin−/− mice (P < 0.05), and E-selectin−/− mice treated with P-selectin mAb (P < 0.001) compared with wild-type mice (Figure 4A). There were no significant increases in neutrophil numbers in saline-treated mutant and wild-type mice (data not shown). Similar to neutrophil numbers, lymphocyte numbers peaked in the BAL 8 days after bleomycin administration (Figure 4B). At this time point, there was a significant increase in lymphocyte numbers in E-selectin−/− mice and E-selectin−/− mice with P-selectin blockade relative to wild-type mice (P < 0.05 and P < 0.05, respectively), whereas there was no significant difference in lymphocyte numbers between P-selectin−/− and wild-type mice. Macrophage numbers were similar between all groups at all time points (Figure 4C). Thus, E-selectin deficiency with or without the additional P-selectin blockade increased neutrophil and lymphocyte accumulation in the lung.
Figure 4.
Time course of neutrophil (A), lymphocyte (B), and macrophage (C) influx into the BAL from mutant and wild-type mice. BAL was collected at 2, 4, 8, 12, and 16 days after intratracheal bleomycin administration. Saline-treated mutant mice or wild-type mice showed no significant increase in leukocyte numbers (data not shown). All values represent the mean ± SEM of results obtained using eight mice in each group. Statistical analysis is presented in the Results section.
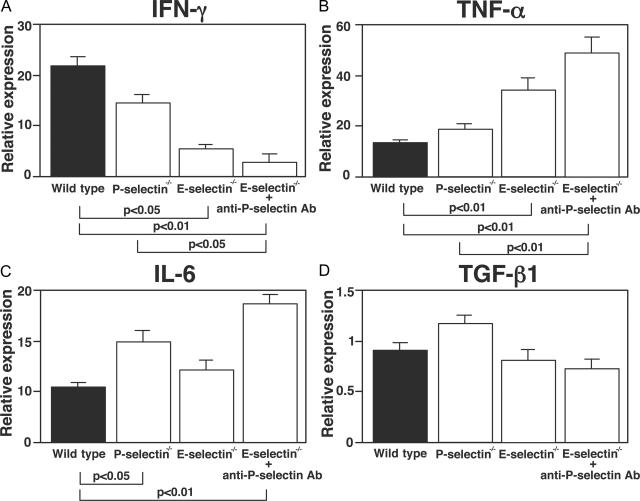
Effect of E- or P-Selectin Loss or Blockade on Cytokine and Growth Factor Production
A variety of cytokines and growth factors are implicated in the development of bleomycin-induced pulmonary fibrosis, including tumor necrosis factor (TNF)-α, IFN-γ, interleukin (IL)-6, and transforming growth factor (TGF)-β1.18–21 These cytokines are generated and released, in part, by inflammatory cells infiltrating into the lungs. To assess the effects of P- and E-selectin blockade on production of these effector cytokines, their mRNA expression level in the lung was examined 8 days after bleomycin challenge by real-time reverse transcriptase-polymerase chain reaction (RT-PCR). E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb showed a significant decrease in IFN-γ mRNA levels compared with wild-type mice (Figure 5A). In contrast, TNF-α mRNA expression level was significantly increased in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb relative to wild-type mice (Figure 5B). P-selectin−/− mice showed similar expression levels of IFN-γ and TNF-α relative to wild-type levels. There was no significant difference in IFN-γ and TNF-α mRNA expression levels between saline-treated mutant mice and controls (data not shown). IL-6 mRNA expression levels were significantly increased in E-selectin−/− mice with P-selectin blockade and P-selectin−/− mice but not in E-selectin−/− mice (Figure 5C). There was no significant difference between any groups of mice in TGF-β mRNA expression level (Figure 5D). Thus, the combined blockade of P- and E-selectins decreased IFN-γ expression but increased TNF-α and IL-6 expression.
Figure 5.
mRNA expression of IFN-γ (A), TNF-α (B), IL-6 (C), and TGF-β1 (D) in the lungs of mutant and wild-type mice 8 days after intratracheal bleomycin administration. mRNA expression was assessed by real-time RT-PCR. Values represent the mean ± SEM of results from seven mice in each group.
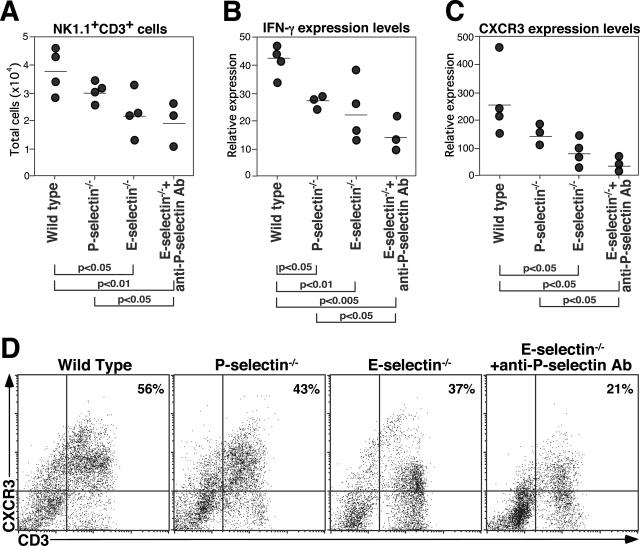
E-Selectin−/− Mice and E-Selectin−/− Mice Treated with Anti-P-Selectin mAb Exhibit Defective Natural Killer T (NKT) Cell Recruitment to the Lungs
To further define the lymphocyte influx into the lung, expression levels of CD3, NK1.1, and intracellular IFN-γ in BAL lymphocytes from wild-type, P-selectin−/−, and E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb were assessed by flow cytometry 4 days after bleomycin instillation. E-selectin−/− mice and E-selectin−/− mice treated with antiP-selectin mAb exhibited significantly reduced NK1.1+CD3+ cell number in BAL compared with wild-type mice (P < 0.05 and P < 0.01, respectively; Figure 6A), but not in homogenate lung parenchyma (data not shown). There was no significant difference in NK1.1+CD3+ cell number between wild-type mice and P-selectin−/− mice (Figure 6A). No significant difference was found in NK1.1+CD3+ cell numbers between wild-type mice and E-selectin−/− mice in peripheral blood and spleen before or after bleomycin instillation (data not shown). Furthermore, IFN-γ expression levelof CD3+ lymphocytes was significantly reduced in P-selectin−/− mice (P < 0.05), E-selectin−/− mice (P < 0.01), and E-selectin−/− mice treated with anti-P-selectin mAb (P < 0.005) compared with wild-type mice (Figure 6B). Thus, E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb exhibited defective NKT cell recruitment to the lungs with reduced intracellular IFN-γ expression.
Figure 6.
The reduced NK1.1+CD3+ cell numbers, decreased IFN-γ and CXCR3 expression levels in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb. BAL fluid was analyzed by flow cytometry 4 days after bleomycin administration. Total number of NK1.1+CD3+ cells (A), intracellular IFN-γ expression level of CD3+ cells (B), CXCR3 expression levels of CD3+ cells (C), and representative dot plots of CXCR3+CD3+ cell frequency (D). Percentages of CXCR3+CD3+ cells are shown in the upper right quadrants. These data are representative of three independent experiments.
Reduced CXCR3 Expression Levels on BAL Lymphocytes from E-Selectin−/− Mice and E-Selectin−/− Mice Treated with Anti-P-Selectin Ab
CXCR3 is a chemokine receptor expressed preferentially on NKT cells22 as well as Th1 and NK cells.23 To determine CXCR3 expression on BAL lymphocytes, BAL lymphocytes from wild-type and E-selectin mice 4 days after bleomycin administration were stained with anti-CD3 and anti-CXCR3 mAbs. BAL lymphocytes from E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb exhibited significantly lower CXCR3 expression levels compared with wild-type mice (P < 0.05 and P < 0.05, respectively; Figure 6C). Furthermore, the frequency of CXCR3+CD3+ cells was significantly reduced in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb compared with wild-type mice (Figure 6D). Therefore, loss of E-selectin expression decreased the expression level and frequency of CXCR3+CD3+ cells in BAL with more extensive decreases when treated with anti-P-selectin mAb.
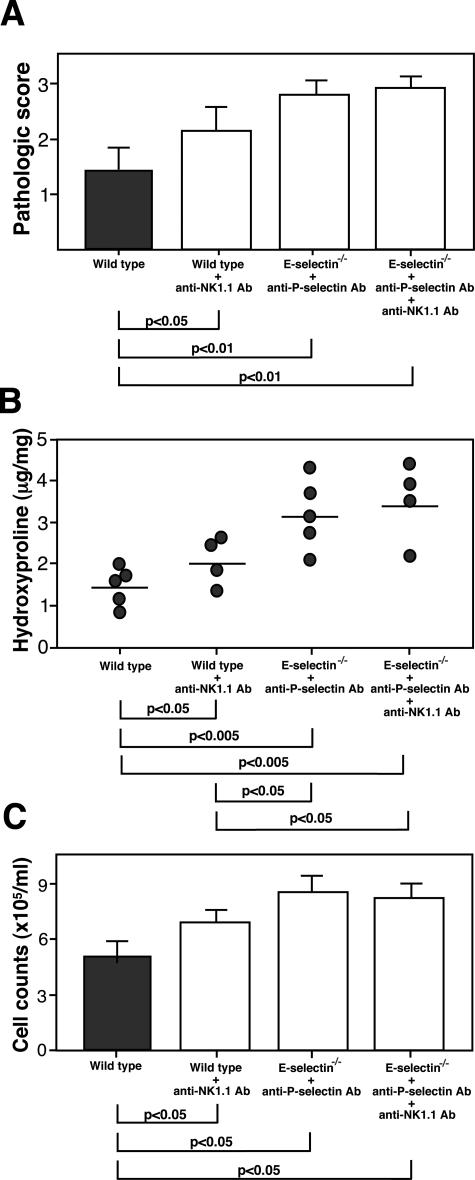
Depletion of NK1.1+ Cells Augmented Bleomycin-Induced Lung Fibrosis
To confirm a role of NK1.1+ cells in bleomycin-induced lung fibrosis, depletion of NK1.1+ cells by mAbs was performed. Wild-type mice were treated with mAbs to NK1.1 intraperitoneally at 4 and 2 days before bleomycin administration and weekly thereafter. Wild-type mice injected with anti-NK1.1 mAbs exhibited an increase in lung parenchyma consolidation with disruption of the alveolar architecture and increased inflammatory cell infiltration compared with untreated wild-type mice. Accordingly, the pathological scores of lung sections were significantly increased in wild-type mice treated with anti-NK1.1 mAb compared with untreated wild-type mice (P < 0.05, Figure 7A). Additionally, lung hydroxyproline content from wild-type mice treated with anti-NK1.1 mAb was significantly increased compared with untreated wild-type mice (P < 0.05, Figure 7B). Total leukocyte count from BAL fluid was significantly increased in wild-type mice treated with anti-NK1.1 mAb relative to untreated wild-type mice (P < 0.05, Figure 7C). Thus, depletion of NK1.1+ cells results in aggravation of pulmonary fibrosis in this model.
Figure 7.
Lack of NK1.1+ cells augmented bleomycin-induced lung fibrosis. The mean pathological score (A), lung hydroxyproline content (B), and total leukocyte counts from BAL fluid (C) of wild-type mice, wild-type mice injected with anti NK1.1 mAb, E-selectin−/− mice injected with anti-P-selectin mAb, and E-selectin−/− mice injected with anti-P-selectin mAb and anti-NK1.1 mAb. Lungs were harvest 16 days after bleomycin administration. The pathological scores, hydroxyproline assay, and BAL cell counts were as described in the Materials and Methods section. The results were obtained from four mice in each group.
When wild-type mice treated with anti-NK1.1 mAb were compared with E-selectin−/− mice treated with anti-P-selectin mAb, pathological scores, hydroxyproline content, and total leukocyte count were milder in wild-type mice treated with anti-NK1.1 mAb (Figure 7), although only hydroxyproline content reached statistical difference (P < 0.05). These results suggest that, although defective NKT cell recruitment is critical in the exacerbation of bleomycin-induced lung fibrosis by E- and P-selectin blockade, other components also participate in the development of lung fibrosis. Nonetheless, in E-selectin−/− mice treated with anti-P-selectin mAb, depletion of NK1.1+ cells by anti-NK1.1 mAb did not further augment pathological scores, hydroxyproline content, or total leukocyte count (Figure 7). Thus, defective recruitment of NKT cells by the absence of E- and P-selectins appeared to have a major role in aggravation of pulmonary fibrosis in this model.
Discussion
Cell adhesion molecules are thought to play critical roles in the development of several inflammatory diseases. Inhibition or loss of cell adhesion molecules attenuates inflammatory response in vivo in many experimental models. In this study, we investigated a role for P- and E-selectins in the development of pulmonary fibrosis. Unexpectedly, E-selectin deficiency exacerbated bleomycin-induced pulmonary fibrosis resulting in increased mortality, more severe histological changes, and increases in lung collagen deposition. Furthermore, treatment with anti-P-selectin mAb in E-selectin−/− mice generally exacerbated pulmonary fibrosis beyond that produced by loss of E-selectin alone, indicating an overlapping function of P- and E-selectins in this model. Consistent with this, the cutaneous contact hypersensitivity response and TNF-α-induced leukocyte rolling are almost completely eliminated by loss of both E- and P-selectins, whereas loss of each molecule alone results in little inhibition, if any.24–26 Therefore, the results of the present study reveal an inhibitory and cooperative role of P- and E-selectins in the development of bleomycin-induced pulmonary fibrosis.
Although it is assumed that leukocytes and cytokines produced by leukocytes participate in the induction of pulmonary fibrosis, it is still controversial as to which subtype of leukocyte plays an essential role. Some evidence suggests that neutrophils play a central role in pulmonary fibrosis. Neutrophils release mediators that stimulate fibroblasts to proliferate and produce excess collagen.27 Bleomycin-induced fibrotic regions are not detected in mice treated with a neutrophil elastase inhibitor28 and neutrophil elastase-deficient mice are resistant to bleomycin-induced fibrosis.29 Moreover, patients with idiopathic pulmonary fibrosis generally demonstrate neutrophil-predominant alveolitis, and the frequency of neutrophils in the BAL is a clinical indicator for active pulmonary fibrosis associated with systemic sclerosis.1,2 In the present study, mice lacking P- and/or E-selectin function exhibited increased neutrophil numbers after bleomycin treatment. These results suggest that other adhesion pathways or mechanical forces, which do not require P- or E-selectins, primarily mediate neutrophil recruitment in bleomycin-induced lung inflammation. Consistent with this, mice lacking both L-selectin and ICAM-1 exhibited almost complete elimination of neutrophil entry into the alveolar space after bleomycin treatment.3
It is possible that loss of P- and/or E-selectin function selectively alters the trafficking patterns of specific lymphocyte subpopulations to the lung. This may result in differential production of cytokines, which may then directly or indirectly influence the development of lung fibrosis. Previous studies have demonstrated anti-fibrotic effects of IFN-γ on bleomycin-induced pulmonary fibrosis.15,30–32 These studies suggest that the mechanisms by which IFN-γ inhibits tissue fibrosis could be through the inhibition of TGF-β1 signaling. Two studies have demonstrated that neutralizing IFN-γ by administration of anti-IFN-γ mAb before bleomycin treatment augmented lung fibrosis with increased lung collagen content.31,32 By contrast, other studies have shown that IFN-γ production is increased in bleomycin-induced pulmonary fibrosis, suggesting a pathogenic role for IFN-γ.21,33 Furthermore, bleomycin-induced lung fibrosis was attenuated in IFN-γ-deficient mice.21 Results of this study showed that lack of E-selectin, with or without P-selectin blockade, reduced IFN-γ expression and augmented lung fibrosis, suggesting a protective effect of IFN-γ. However, exogenous IFN-γ treatment did not affect the severity of lung fibrosis or mortality rate in E-selectin−/− mice (data not shown), although Jiang and colleagues32 have demonstrated that treatment with exogenous IFN-γ in CXCR3−/− mice could reverse the fibrotic phenotype and significantly attenuate the development of lung fibrosis. In addition, loss of E-selectin alone or with P-selectin blockade increased TNF-α mRNA expression. Several studies have shown that TNF-α plays a key role among various cytokines in bleomycin-induced lung fibrosis.18 High levels of TNF-α have also been demonstrated in the lungs of patients with idiopathic pulmonary fibrosis.34 Furthermore, loss of E-selectin with P-selectin blockade increased expression of IL-6 that induces the production of collagen and glycosaminoglycans by fibroblasts.35 These results indicate that one of the major mechanisms by which loss of P- and E-selectin function exacerbated the fibrotic process is by regulating cytokine balance.
Our flow cytometric analysis revealed that NKT cells in the BAL were markedly reduced in E-selectin−/− mice and E-selectin−/− mice treated with anti-P-selectin mAb (Figure 6A). Indeed, depletion of NKT cells in wild-type mice augmented lung fibrosis with increased BAL cell count (Figure 7). NKT cells are innate memory cells that play important roles in the initiation and regulation of the immune response because of their ability to produce large amounts of cytokines by stimulation with their glycolipid ligand, α-galactosylceramide (α-GalCer). On stimulation with α-GalCer, NKT cells rapidly produce cytokines, including IFN-γ.15 Consistent with our results, a recent study showed that treatment with α-GalCer attenuates the development of bleomycin-induced pulmonary fibrosis.15 Another study showed that mice lacking NKT cells aggravated pulmonary fibrosis and adoptive transfer of NKT cells into these mice attenuates pulmonary fibrosis.31 The present study is the first to reveal that NKT cell infiltration into the lung is dependent on E- and P-selectin expression. Furthermore, in E-selectin−/− mice treated with anti-P-selectin mAb, depletion of NK1.1+ cells did not further augment the severity of lung fibrosis (Figure 7), suggesting that defective recruitment of NKT cells plays a major role in exacerbating lung fibrosis by the blockade of E- and P-selectins. Because it has been shown that P- and E-selectins preferentially mediate rolling and emigration of Th1 cells over Th2 cells,36,37 E- and P-selectins may mediate the recruitment of NKT cells into the lung. In this study, CD3+ lymphocytes in BAL from wild-type mice expressed high levels of CXCR3 (Figure 6C), suggesting that lymphocytes accumulating in the lungs are Th1-type cells rather than Th2-type cells whereas CXCR3 expression level and frequency were reduced in E-selectin−/− mice and E-selectin−/− mice with P-selectin blockade (Figure 6, C and D). Furthermore, these mice exhibited significantly reduced NKT cell number in BAL compared to wild-type mice. A previous study showed that NKT cells expressed high levels of CXCR338,39 and that CXCR3 plays a significant role in recruiting NKT cells to inflammatory sites in human.38 Moreover, CXCR3−/− mice exhibit augmented fibrosis in the bleomycin-induced pulmonary fibrosis model.32 Collectively, these results suggest that E- and P-selectins selectively and synergistically recruit NKT cells into the lungs in collaboration with CXCR3, although the precise mechanisms underlying this molecular interaction require further study.
To date, there have been few reports directly addressing an in vivo role of selectins in bleomycin-induced pulmonary fibrosis. The present study indicates that P- and E-selectins both contribute to pulmonary fibrosis by mediating the accumulation of type 1 cells, especially NKT cells that may have a protective effect. Furthermore, functional loss of these molecules resulted in increased production of TNF-α that may initiate early lung inflammation by mediating the accumulation of neutrophils and exacerbating fibrosis. These results provide additional clues to understanding the complexity of the pathogenesis of pulmonary fibrosis.
Acknowledgments
We thank Ms. Y. Yamada and Ms. M. Matsubara for their technical assistance.
Footnotes
Address reprint requests to Manabu Fujimoto, M.D., Department of Dermatology, Kanazawa University Graduate School of Medical Science, 13-1 Takaramachi, Kanazawa, Ishikawa 920-8641, Japan. E-mail: fujimoto-m@umin.ac.jp.
Supported by the grant-in-aid from the Ministry of Education, Science, and Culture of Japan.
References
- Paine R, III, Ward PA. Cell adhesion molecules and pulmonary fibrosis. Am J Med. 1999;107:268–279. doi: 10.1016/s0002-9343(99)00226-0. [DOI] [PubMed] [Google Scholar]
- Silver RM, Miller KS, Kinsella MB, Smith EA, Schabel SI. Evaluation and management of scleroderma lung disease using bronchoalveolar lavage. Am J Med. 1990;88:470–476. doi: 10.1016/0002-9343(90)90425-d. [DOI] [PubMed] [Google Scholar]
- Hamaguchi Y, Nishizawa Y, Yasui M, Hasegawa M, Kaburagi Y, Komura K, Nagaoka T, Saito E, Shimada Y, Takehara K, Kadono T, Steeber DA, Tedder TF, Sato S. Intercellular adhesion molecule-1 and L-selectin regulate bleomycin-induced lung fibrosis. Am J Pathol. 2002;161:1607–1618. doi: 10.1016/S0002-9440(10)64439-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tedder TF, Li X, Steeber DA. The selectins and their ligands: adhesion molecules of the vasculature. Adv Mol Cell Biol. 1999;28:65–111. [Google Scholar]
- Yanaba K, Kaburagi Y, Takehara K, Steeber DA, Tedder TF, Sato S. Relative contributions of selectins and intercellular adhesion molecule-1 to tissue injury induced by immune complex deposition. Am J Pathol. 2003;162:1463–1473. doi: 10.1016/S0002-9440(10)64279-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Issekutz AC, Issekutz TB. The role of E-selectin, P-selectin, and very late activation antigen-4 in T lymphocyte migration to dermal inflammation. J Immunol. 2002;168:1934–1939. doi: 10.4049/jimmunol.168.4.1934. [DOI] [PubMed] [Google Scholar]
- Bullard DC, Qin L, Lorenzo I, Quinlin WM, Doyle NA, Bosse R, Vestweber D, Doerschuk CM, Beaudet AL. P-selectin/ICAM-1 double mutant mice: acute emigration of neutrophils into the peritoneum is completely absent but is normal into pulmonary alveoli. J Clin Invest. 1995;95:1782–1788. doi: 10.1172/JCI117856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullard DC, Mobley JM, Justen JM, Sly LM, Chosay JG, Dunn CJ, Lindsey JR, Beaudet AL, Staite ND. Acceleration and increased severity of collagen-induced arthritis in P-selectin mutant mice. J Immunol. 1999;163:2844–2849. [PubMed] [Google Scholar]
- Rosenkranz AR, Mendrick DL, Cotran RS, Mayadas TN. P-selectin deficiency exacerbates experimental glomerulonephritis: a protective role for endothelial P-selectin in inflammation. J Clin Invest. 1999;103:649–659. doi: 10.1172/JCI5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azuma A, Takahashi S, Nose M, Araki K, Araki M, Takahashi T, Hirose M, Kawashima H, Miyasaka M, Kudoh S. Role of E-selectin in bleomycin induced lung fibrosis in mice. Thorax. 2000;55:147–152. doi: 10.1136/thorax.55.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel EJ, Jung U, Bullard DC, Norman KE, Wolitzky BA, Vestweber D, Beaudet AL, Ley K. Absence of trauma-induced leukocyte rolling in mice deficient in both P-selectin and intercellular adhesion molecule-1 (ICAM-1). J Exp Med. 1996;183:57–65. doi: 10.1084/jem.183.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore BB, Coffey MJ, Christensen P, Sitterding S, Ngan R, Wilke CA, McDonald R, Phare SM, Peters-Golden M, Paine R, III, Toews GB. GM-CSF regulates bleomycin-induced pulmonary fibrosis via a prostaglandin-dependent mechanism. J Immunol. 2000;165:4032–4039. doi: 10.4049/jimmunol.165.7.4032. [DOI] [PubMed] [Google Scholar]
- Kuwano K, Hagimoto N, Kawasaki M, Yatomi T, Nakamura N, Nagata S, Suda T, Kunitake R, Maeyama T, Miyazaki H, Hara N. Essential roles of the Fas-Fas ligand pathway in the development of pulmonary fibrosis. J Clin Invest. 1999;104:13–19. doi: 10.1172/JCI5628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrier DJ, Phan SH, McGarry BM. The effects of the nude (nu/nu) mutation on bleomycin-induced pulmonary fibrosis. A biochemical evaluation. Am Rev Respir Dis. 1983;127:614–617. doi: 10.1164/arrd.1983.127.5.614. [DOI] [PubMed] [Google Scholar]
- Kimura T, Ishii Y, Morishima Y, Shibuya A, Shibuya K, Taniguchi M, Mochizuki M, Hegab AE, Sakamoto T, Nomura A, Sekizawa K. Treatment with alpha-galactosylceramide attenuates the development of bleomycin-induced pulmonary fibrosis. J Immunol. 2004;172:5782–5789. doi: 10.4049/jimmunol.172.9.5782. [DOI] [PubMed] [Google Scholar]
- Meijerink J, Mandigers C, van de Locht L, Tonnissen E, Goodsaid F, Raemaekers J. A novel method to compensate for different amplification efficiencies between patient DNA samples in quantitative real-time PCR. J Mol Diagn. 2001;3:55–61. doi: 10.1016/S1525-1578(10)60652-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerwenka A, Baron JL, Lanier LL. Ectopic expression of retinoic acid early inducible-1 gene (RAE-1) permits natural killer cell-mediated rejection of a MHC class I-bearing tumor in vivo. Proc Natl Acad Sci USA. 2001;98:11521–11526. doi: 10.1073/pnas.201238598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piguet PF, Collart MA, Grau GE, Kapanci Y, Vassalli P. Tumor necrosis factor/cachectin plays a key role in bleomycin-induced pneumopathy and fibrosis. J Exp Med. 1989;170:655–663. doi: 10.1084/jem.170.3.655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang K, Gharaee-Kermani M, McGarry B, Remick D, Phan SH. TNF-α-mediated lung cytokine networking and eosinophil recruitment in pulmonary fibrosis. J Immunol. 1997;158:954–959. [PubMed] [Google Scholar]
- Daniels CE, Wilkes MC, Edens M, Kottom TJ, Murphy SJ, Limper AH, Leof EB. Imatinib mesylate inhibits the profibrogenic activity of TGF-beta and prevents bleomycin-mediated lung fibrosis. J Clin Invest. 2004;114:1308–1316. doi: 10.1172/JCI19603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segel MJ, Izbicki G, Cohen PY, Or R, Christensen TG, Wallach-Dayan SB, Breuer R. Role of interferon-gamma in the evolution of murine bleomycin lung fibrosis. Am J Physiol. 2003;285:L1255–L1262. doi: 10.1152/ajplung.00303.2002. [DOI] [PubMed] [Google Scholar]
- Kim CH, Johnston B, Butcher EC. Trafficking machinery of NKT cells: shared and differential chemokine receptor expression among V alpha 24(+)V beta 11(+) NKT cell subsets with distinct cytokine-producing capacity. Blood. 2002;100:11–16. doi: 10.1182/blood-2001-12-0196. [DOI] [PubMed] [Google Scholar]
- Luster AD, Ravetch JV. Biochemical characterization of a gamma interferon-inducible cytokine (IP-10). J Exp Med. 1987;166:1084–1097. doi: 10.1084/jem.166.4.1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staite ND, Justen JM, Sly LM, Beaudet AL, Bullard DC. Inhibition of delayed-type contact hypersensitivity in mice deficient in both E-selectin and P-selectin. Blood. 1996;88:2973–2979. [PubMed] [Google Scholar]
- Frenette PS, Mayadas TN, Rayburn H, Hynes RO, Wagner DD. Susceptibility to infection and altered hematopoiesis in mice deficient in both P- and E-selectins. Cell. 1996;84:563–574. doi: 10.1016/s0092-8674(00)81032-6. [DOI] [PubMed] [Google Scholar]
- Bullard DC, Kunkel EJ, Kubo H, Hicks MJ, Lorenzo I, Doyle NA, Koerschuk CM, Ley K, Beaudet AL. Infectious susceptibility and severe deficiency of leukocyte rolling and recruitment in E-selectin and P-selectin double mutant mice. J Exp Med. 1996;183:2329–2336. doi: 10.1084/jem.183.5.2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coker RK, Laurent GJ, Shahzeidi S, Lympany PA, du Bois RM, Jeffery PK, McAnulty RJ. Transforming growth factors-β1, -β2, and -β3 stimulate fibroblast procollagen production in vitro but are differentially expressed during bleomycin-induced lung fibrosis. Am J Pathol. 1997;150:981–991. [PMC free article] [PubMed] [Google Scholar]
- Taooka Y, Maeda A, Hiyama K, Ishioka S, Yamakido M. Effects of neutrophil elastase inhibitor on bleomycin-induced pulmonary fibrosis in mice. Am J Respir Crit Care Med. 1997;156:260–265. doi: 10.1164/ajrccm.156.1.9612077. [DOI] [PubMed] [Google Scholar]
- Dunsmore SE, Roes J, Chua FJ, Segal AW, Mutsaers SE, Laurent GJ. Evidence that neutrophil elastase-deficient mice are resistant to bleomycin-induced fibrosis. Chest. 2001;120:35S–36S. doi: 10.1378/chest.120.1_suppl.s35. [DOI] [PubMed] [Google Scholar]
- Gurujeyalakshmi G, Giri SN. Molecular mechanisms of antifibrotic effect of interferon gamma in bleomycin-mouse model of lung fibrosis: downregulation of TGF-beta and procollagen I and III gene expression. Exp Lung Res. 1995;21:791–808. doi: 10.3109/01902149509050842. [DOI] [PubMed] [Google Scholar]
- Kim JH, Kim HY, Kim S, Chung JH, Park WS, Chung DH. Natural killer T (NKT) cells attenuate bleomycin-induced pulmonary fibrosis by producing interferon-gamma. Am J Pathol. 2005;167:1231–1241. doi: 10.1016/s0002-9440(10)61211-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang D, Liang J, Hodge J, Lu B, Zhu Z, Yu S, Fan J, Gao Y, Yin Z, Homer R, Gerard C, Noble PW. Regulation of pulmonary fibrosis by chemokine receptor CXCR3. J Clin Invest. 2004;114:291–299. doi: 10.1172/JCI16861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gur I, Or R, Segel MJ, Shriki M, Izbicki G, Breuer R. Lymphokines in bleomycin-induced lung injury in bleomycin-sensitive C57BL/6 and -resistant BALB/c mice. Exp Lung Res. 2000;26:521–534. doi: 10.1080/019021400750048072. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Theodore C, Guillemin B, Yu M, Rom WN. Enhanced IL-1β and tumor necrosis factor-α release and messenger RNA expression in macrophages from idiopathic pulmonary fibrosis or after asbestos exposure. J Immunol. 1993;150:4188–4196. [PubMed] [Google Scholar]
- Duncan MR, Berman B. Stimulation of collagen and glycosaminoglycan production in cultured human adult dermal fibroblasts by recombinant human interleukin 6. J Invest Dermatol. 1991;97:686–692. doi: 10.1111/1523-1747.ep12483971. [DOI] [PubMed] [Google Scholar]
- Xu H, Manivannan A, Jiang HR, Liversidge J, Sharp PF, Forrester JV, Crane IJ. Recruitment of IFN-gamma-producing (Th1-like) cells into the inflamed retina in vivo is preferentially regulated by P-selectin glycoprotein ligand 1:P/E-selectin interactions. J Immunol. 2004;172:3215–3224. doi: 10.4049/jimmunol.172.5.3215. [DOI] [PubMed] [Google Scholar]
- Austrup F, Vestweber D, Borges E, Lohning M, Brauer R, Herz U, Renz H, Hallmann R, Scheffold A, Radbruch A, Hamann A. P- and E-selectin mediate recruitment of T-helper-1 but not T-helper-2 cells into inflammed tissues. Nature. 1997;385:81–83. doi: 10.1038/385081a0. [DOI] [PubMed] [Google Scholar]
- Thomas SY, Hou R, Boyson JE, Means TK, Hess C, Olson DP, Strominger JL, Brenner MB, Gumperz JE, Wilson SB, Luster AD. CD1d-restricted NKT cells express a chemokine receptor profile indicative of Th1-type inflammatory homing cells. J Immunol. 2003;171:2571–2580. doi: 10.4049/jimmunol.171.5.2571. [DOI] [PubMed] [Google Scholar]
- Johnston B, Kim CH, Soler D, Emoto M, Butcher EC. Differential chemokine responses and homing patterns of murine TCR alpha beta NKT cell subsets. J Immunol. 2003;171:2960–2969. doi: 10.4049/jimmunol.171.6.2960. [DOI] [PubMed] [Google Scholar]