Abstract
Peroxisomal proliferator-activated receptor (PPAR)-α is a ligand-activated transcriptional factor that regulates genes involved in lipid metabolism and energy homeostasis. PPAR-α activators, including fibrates, have been used to treat dyslipidemia for several decades. In contrast to their known effects on lipids, the pharmacological consequences of PPAR-α activation on cardiac metabolism and function are not well understood. Therefore, we evaluated the role that PPAR-α receptors play in the heart. Our studies demonstrate that activation of PPAR-α receptors using a selective PPAR-α ligand results in cardiomyocyte necrosis in mice. Studies in PPAR-α-deficient mice demonstrated that cardiomyocyte necrosis is a consequence of the activation of PPAR-α receptors. Cardiac fatty acyl-CoA oxidase mRNA levels increased at doses in which cardiac damage was observed and temporally preceded cardiomyocyte degeneration, suggesting that peroxisomal β-oxidation correlates with the appearance of microscopic injury and cardiac injury biomarkers. Increased myocardial oxidative stress was evident in mice treated with the PPAR-α agonists coinciding with increased peroxisomal biomarkers of fatty acid oxidation. These findings suggest that activation of PPAR-α leads to increased cardiac fatty acid oxidation and subsequent accumulation of oxidative stress intermediates resulting in cardiomyocyte necrosis.
Peroxisomal proliferator-activated receptors (PPARs) are ligand-activated transcription factors that belong to the superfamily of nuclear receptors.1 Several studies have demonstrated a cross talk between PPAR signaling and other nuclear receptor pathways. This convergence and interaction of different pathways can occur at multiple levels, including competition between PPARs and other nuclear receptors.2 This complexity could explain some of the diverse biological effects of PPAR ligands in tissues and across species.
To date, three PPAR subtypes have been identified: α, β, and γ. They perform different physiological functions based on their divergent patterns of tissue expression, ligand-binding specificities, and physiological functions when activated. PPAR-α was originally cloned from a mouse liver cDNA library3 and subsequently cloned from frogs,4 rats,5 guinea pigs,6 and humans.7,8 PPAR-α is considered a therapeutic target for dyslipidemia with a promising future given the existing literature and clinical experience with fibrates throughout several decades of clinic use. Fibrates have been used to treat dyslipidemia because of their triglyceride lowering and high-density lipoprotein-elevating effects. More recent research demonstrates the anti-inflammatory properties of PPAR-α agonists at the vessel wall, which could contribute to the reduction of atherosclerosis observed in animal models and humans.9,10
PPAR-α is predominantly expressed in tissues exhibiting high rates of β-oxidation such as brown adipose tissue, liver, heart, diaphragm, kidney, and skeletal muscle.11 PPAR-α is a key transcriptional regulator of fatty acid oxidation (FAO), acting by controlling the expression of a comprehensive set of genes that regulate most aspects of fatty acid uptake and metabolism.12 Quantitatively, the major sites of FAO at rest and during exercise are the cardiac and slow twitch type I skeletal muscle fibers.13 In these tissues, the oxidation of fatty acids meets the high ATP demands. Under resting conditions in vivo, the mouse heart derives ∼70% of its energy from the oxidation of fatty acids, and the remainder primarily from glycolysis and glucose oxidation.14 The relative contribution of FAO to energy production increases in fasting, diabetes, and in experimental conditions in which the expression of PPAR-α receptors in the heart is augmented. This leads to the activation of genes involved in the uptake and degradation of fatty acids to provide sufficient energy to the myocardium. In contrast to the clearly defined effects of PPAR-α agonists on lipoprotein metabolism and cholesterol homeostasis, the net effects of PPAR-α activation on cardiac metabolism and function in normal and disease states and the pathological consequences are not well understood. Several studies conducted in conditions of pressure-induced cardiac hypertrophy and diabetes suggests that the rate of FAO is a major determinant of glucose and lactate utilization.15 In contrast to these findings, more recent data by Finck and colleagues16 demonstrated that cardiac overexpression of PPAR-α receptors increased FAO, decreased glucose utilization, and induced a diabetic-like cardiomyopathy, suggesting that substrate availability may not be a key determinant of PPAR-α function in the heart. These studies, however, were conducted in nondiabetic mice with cardiac-restricted overexpression of the human PPAR-α transgene at ∼80-fold the levels found in control littermates. In addition, PPAR-α receptors are down-regulated in animal models of diabetes17 and in failing human hearts in diabetic patients.18 This raises the question as to whether cardiac metabolism and function are also impaired when wild-type mice undergo a pharmacological activation of PPAR-α receptors, emphasizing the need to test pharmacological agents that will identify the role of PPAR-α nuclear receptors in cardiac function and homeostasis. Based on the known functions attributed to PPAR-α receptors, we hypothesized that the ligand activation of PPAR-α receptors plays a critical role in cardiac muscle energy homeostasis in rodents. To test this hypothesis, we used a potent and selective PPAR-α agonist to demonstrate that sustained activation of PPAR-α results in cardiomyocyte necrosis that correlated with an increase in peroxisomal β-oxidation and oxidative stress injury. These effects were shown to be directly PPAR-α-driven and support the mechanistic link between the activation of peroxisomal fatty acid β-oxidation, perturbations in cardiac energy metabolism, generation of reactive oxygen species, and cardiomyocyte necrosis.
Materials and Methods
Animals and Diets
PPAR-α wild-type and null mice 9 to 12 weeks of age on an SV 129 background were used in these studies.19 PPAR-α-deficient (−/−) mice were originally obtained from Dr. F. Gonzalez, National Cancer Institute, National Institutes of Health (Bethesda, MD) and bred at Charles River Laboratories (Wilmington, MA) to generate PPAR-α-deficient and control littermates. Animals arrived at an age of 8 to 10 weeks and were housed at 23 ± 1°C on a 12-hour light/dark cycle. They were allowed ad libitum access to food (Purine Prolab RMH 3000 rodent diet; WF Fisher and Sons Inc., Somerville, NJ) and water and were acclimated for at least a week before experimentation.
Treatment
At the age of 10 weeks, animals were randomly divided in groups of five to six animals per cage and orally dosed once a day with either vehicle (water) or a PPAR-α fibric acid derivative (IC50 α/β/γ in nmol/L: 47/>10,000/1520). This synthetic analog is potent (47 nmol/L) and has more than 212-fold selectivity over the PPAR-β and 32-fold selectivity over the PPAR-γ receptor subtype in the scintillation proximity assay-binding assay. No significant binding activity was detected when this compound was tested at 1 μmol/L against 38 distinct nuclear and transmembrane receptors and enzymes. Body weight was measured twice a week. The study protocol was approved by the institutional animal care and use committee. All animals received humane treatment according to the criteria stated by the National Academy of Sciences National Research Council publication 86-23, 1985. At the end of the dosing period, all animals were euthanized and necropsied.
Serum Chemistry and Metabolites Analysis
Blood samples were taken from the vena cava on the day of euthanasia before excision of cardiac and skeletal muscles. Samples were kept on ice, centrifuged at 14,000 rpm for 10 minutes at 4°C, and subsequently used to measure circulating levels of triglycerides and total creatine kinase (CK) using the Roche Hitachi 917 chemistry analyzer according to the manufacturer’s instructions (Roche Diagnostics, Indianapolis, IN). Skeletal (CK-MM) and cardiac (CK-MB) isoenzymes were determined using commercially available kits (Helena Rapid Electrophoresis System; Helena Laboratories, Beaumont, TX). Troponin I levels were measured by a two-site sandwich chemiluminescent immunoassay using the Bayer ACS:180 according to the manufacturer’s instructions (Bayer Health Care, Diagnostic Division, Tarrytown, NY).
Histopathology and Immunochemistry
Cardiac and gastrocnemius (including soleus and plantaris) muscles were collected from all animals and processed for microscopic evaluation using a routine hematoxylin and eosin (H&E) stain or immunohistochemistry. The heart and gastrocnemius/soleus were fixed in 10% neutral buffered formalin and embedded in paraffin. Serial 5-μm sections were cut, deparaffinized, rehydrated, stained routinely with H&E, and examined by light microscopy. Identification of slow twitch type I and fast twitch type 2 myofibers was performed using immunohistochemistry with anti-mouse MHCs and MHCf antibodies purchased from Novocastra (Newcastle, UK). In brief, gastrocnemius/soleus muscles were snap-frozen in liquid nitrogen, and 8-μm sections were fixed in ethanol and acetone and incubated with 0.3% hydrogen peroxide at room temperature for 5 minutes using a biotin blocking system (DakoCytomation, Carpinteria, CA) according to manufacturer’s directions. All rinses were done three times for 5 minutes in wash buffer (Biogenex, San Ramon, CA). Nonspecific binding to mouse primary antibodies was prevented by using an animal research kit (DakoCytomation) according to the manufacturer’s directions. Mouse MHCs and MHCf antibodies and matching irrelevant mouse IgG1 were biotinylated using a biotinylated goat anti-mouse IgG Fab reagent before the application to the skeletal muscle section. Any excess goat anti-mouse IgG Fab was adsorbed with normal mouse serum. Both primary antibodies were detected using DakoCytomation streptavidin-horseradish peroxidase for 30 minutes at room temperature followed by DakoCytomation liquid DAB+ for 5 minutes at room temperature.
Determination of Peroxisomal β-Oxidation and Fatty Acyl-CoA Oxidase (AOX) mRNA Levels
Peroxisomal β-oxidation was determined in tissue homogenates by the method of Lazarow.20 The activity of the cyanide-insensitive fatty acyl CoA oxidizing system was determined by measuring palmitoyl-CoA-dependent reduction of NAD at 340 nm. AOX mRNA levels were determined by Quantigene (Genospectra Inc., Fremont, CA). Flash-frozen samples were homogenized in tissue preparation buffer containing 50 mmol/L HEPES, 2% lithium lauryl sulfate, 0.5% Micro-O-Protect (Roche Diagnostic), 8 mmol/L ethylenediaminetetraacetic acid, 500 mmol/L lithium chloride, and 3.8 mg/ml proteinase K (EM Science). Tissue homogenate (6 to 24 μl) was applied to 100 μl of Quantigene lysis buffer containing the probe sets for mouse AOX, PDK4, or UCP3 in a 96-well plate (Costar, Cambridge, MA). Subsequently, 90 μl was applied to each well of the capture plate containing 100 μl of the tissue preparation buffer. After an overnight incubation at 53°C, the plates were developed according to the manufacturer’s protocol. In brief, plates were allowed to sit at room temperature for 15 minutes, washed twice with Quantigene wash buffer, and 100 μl of amplifier reagent was added to each well. After a 1-hour incubation at 53°C, plates were allowed to sit at room temperature for 15 minutes, washed twice with wash buffer, and 100 μl of label probe reagent was applied to each well. After 1 hour of incubation at 53°C, plates were washed six times with wash buffer, and 100 μl of substrate was added. The plates were incubated for 30 minutes at 37°C and immediately read on a Dynex luminometer. The capture plates and all reagents were purchased from Genospectra Inc. or prepared as described in the Quantigene manufacturer’s protocol.
PPAR-α-Induced Oxidative Stress
Biomarkers of aerobic metabolism and oxidative stress were determined in heart samples collected from mice orally dosed with either vehicle or PPAR-α agonist. In brief, 100 mg of tissue were homogenized on ice for 10 seconds at 30,000 rpm (PowerGen 125; Fisher Scientific, Loughborough, UK) in 9 vol of buffer containing 20 mmol/L Tris-HCl, pH 7.4. Ice-cold 5% metaphosphoric acid buffer was used as buffer when the glutathione was assayed. Heart homogenates were centrifuged at 3000 × g for 10 minutes at 4°C and the supernatant was collected and assayed within 40 minutes. Assays were performed using an automated chemistry analyzer (Advia 1650 chemistry system; Bayer PLC, Newbury, UK) with its associated analytical kits, according to the manufacturer’s instructions for CK, alanine aminotransferase, lactate dehydrogenase, alkaline phosphatase, glucose, and lactate. Assay kits were obtained from an alternative vendor (Randox Laboratories Ltd., Crumlin, UK) for glutamate dehydrogenase, glucose-6-phosphate dehydrogenase, glutathione reductase, and hydroxy butyrate dehydrogenase. Glutathione was determined similarly (GSH; Bio-Stat Ltd., Stockport, UK). Phosphofructokinase (PFK), carnitine palmitoyl transferase-I, β-hydroxyacyl CoA dehydrogenase, and glycogen phosphorylase were determined using conditions and reagents as described in the literature.21–24
Statistics
Results are expressed as means ± SD. Statistical significance was determined by Student’s t-test. Results are considered significant with P < 0.05.
Results
Activation of PPAR-α Receptors Causes Cardiomyocyte Necrosis
To determine the effect of ligand-dependent activation of PPAR-α receptors in cardiac muscle, a high-affinity ligand for the murine PPAR-α receptor (IC50 = 47 nmol/L) and >100-fold selective over the PPAR-β/γ subtypes was orally administered to wild-type mice once a day for 12 consecutive days. At the end of the dosing period, all animals were euthanized and necropsied. Hearts were collected and processed for histopathological evaluation. Minimal to moderate multifocal cardiomyocyte necrosis was observed in both ventricles with a predisposition for subendocardial and subepicardial areas of the left ventricle (Figure 1) in mice orally administered the PPAR-α agonist. Cardiomyocyte necrosis was characterized by cytoplasmic hypereosinophilia and vacuolation, myofiber fragmentation followed by mononuclear cell infiltration and fibrosis (Figure 1).
Figure 1.
Cardiomyocyte necrosis induced by PPAR-α agonists. a: Right ventricle. Multifocal moderate cardiomyocyte necrosis involving the entire wall of the right ventricle (transmural). b: Left ventricle. Cardiomyocyte necrosis is often observed in the subepicardial areas of the left ventricle. The finding is characterized by myofiber hypereosinophilia, loss of cross striations, cytoplasmic vacuolation, and infiltration of variable number of mononuclear cells and neutrophils. The PPAR-α agonist was administered orally at 150 mg/kg/day for 12 consecutive days. Scale bars = 50 μm (a); 20 μm (b).
To investigate whether the cardiac degeneration observed in rodents was attributable to the activation of PPAR-α receptors, PPAR-α-deficient mice and control littermates were orally dosed with either vehicle or a synthetic PPAR-α agonist for 12 consecutive days. In control mice, activation of PPAR-α receptors increased the expression of AOX, PDK4, and UCP-3, consistent with the proposed role of PPAR-α in peroxisomal proliferation, control of pyruvate dehydrogenase activity, and fatty acid transport to mitochondria. In contrast, no gene activation was observed in PPAR-α-deficient mice (Figure 2). Increased peroxisomal β-oxidation in control mice receiving the PPAR-α agonist was evidenced by changes in peroxisomal activity measured by the generation of NADPH (108 ± 15 μmol/L/minute/mg and 16 ± 4 μmol/L/minute/mg of heart homogenates for agonist and vehicle-treated mice, respectively, n = 6; P < 0.001) consistent with the correlation between the generation of NADPH, synthesis of peroxisomal membrane proteins, and induction of AOX mRNA documented in the literature.25 In addition, a decrease in the utilization of glucose as metabolic substrate was evident in mice orally administered with the PPAR-α agonist as demonstrated by the significant changes in the tissue level of glucose and the activity of key enzymes involved in the control of cellular glucose homeostasis (Figure 3).
Figure 2.
UCP3, AOX, and PDK4 mRNA levels in wild-type and PPAR-α-deficient mice orally dosed for 12 consecutive days with 150 mg/kg/day of a synthetic PPAR-α ligand. Cardiac mRNA levels for UCP3, AOX, and PDK4 were determined by Quantigene following the manufacturer’s protocol using specific probe sets for murine UCP3, AOX, and PDK4. Data are shown as mean ± SD (n = 4 to 6 mice/group). *P < 0.001.
Figure 3.
Cardiac PFK (A), hydroxy butyrate dehydrogenase (B), glycogen phosphorylase activities (C), and glucose (D) levels in wild-type mice orally dosed with 100 mg/kg/day of a synthetic PPAR-α ligand for 12 consecutive days. PFK, hydroxy butyrate dehydrogenase, glycogen phosphorylase activities, and glucose concentrations were determined using an Advia 1650 chemistry system as described under Materials and Methods. Data are expressed as mean ± SD from 10 to 13 mice per group. Asterisks show statistical significance. *P < 0.001.
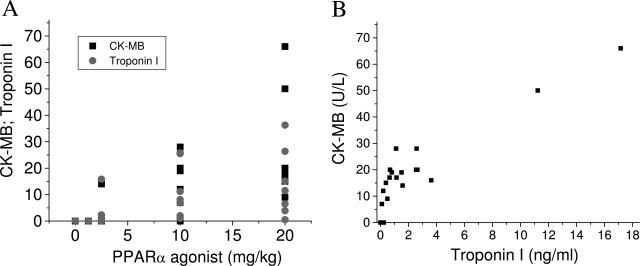
Cardiomyocyte necrosis occurred in seven of eight treated wild-type mice, and the severity of the microscopic findings was proportional to increases in plasma CK and its cardiac isoenzyme CK-MB (Table 1). Because the low circulating levels of CK-MB could indicate skeletal muscle release rather than cardiac injury, an additional experiment was performed to evaluate whether changes in plasma CK-MB are accompanied by increases in troponin I, a more selective and sensitive biomarker of cardiac injury. For this purpose, the synthetic PPAR-α agonist was orally administered to mice for 12 days at doses ranging from 1.25 mg/kg/day to 20 mg/kg/day. At the end of the dosing period, all animals were euthanized and serum was collected for troponin I and CK-MB determinations. As shown in Figure 4A increases in plasma CK-MB and troponin I levels were noted in mice treated with PPAR-α agonists and correlated with the dose administered. More importantly, there was a strong correlation (r = 0.88, P < 0.0001) between increases in plasma levels of CK-MB and troponin I (Figure 4B). There was no case in which a change in CK-MB was not accompanied by an increase in troponin I. Taken together, these findings support the observation that the increases in plasma CK-MB observed in our studies are indicative of cardiac injury. No changes in serum chemistry parameters or microscopic evidence of cardiac damage were observed in treated PPAR-α-deficient mice, demonstrating that the activation of PPAR-α receptors is required to induce cardiomyocyte necrosis in rodents.
Table 1.
Serum Chemistry and Incidence of Cardiomyocyte Degeneration in Wild-Type and PPAR-α Knockout Mice Orally Dosed with 150 mg/kg/Day of a PPAR-α Agonist for 12 Consecutive Days
| Mice | Treatment | Necrosis (incidence/group) | CK (U/L) | CK-MB (U/L) |
|---|---|---|---|---|
| Wild type (n = 8) | Vehicle | 0/8 | 112 ± 40 | 0 |
| Wild type (n = 8) | Agonist | 7/8 | 4250 ± 5785 | 127 ± 85 |
| PPAR-α KO (n = 8) | Vehicle | 0/4 | 70 ± 9 | 0 |
| PPAR-α KO (n = 8) | Agonist | 0/4 | 63 ± 7 | 0 |
At the end of the dosing period, the animals were euthanized and necropsied. Values for total plasma CK and its cardiac CK-MB isoenzyme were measured and expressed as mean ± SD. Histopathology was conducted in all animals as described in Materials and Methods.
Figure 4.
Serum CK-MB and troponin I (A) levels in wild-type mice orally dosed for 12 consecutive days with a synthetic PPAR-α agonist at doses ranging from 1.25 mg/kg/day to 20 mg/kg/day. Relationship between increases in plasma levels of CK-MB and troponin I (B). CK-MB and troponin I levels were determined as described under Materials and Methods.
We also examined whether the pharmacological activation of PPAR-α receptors lead to microscopic damage in tissues in which FAO is the primary source of cellular energy. In the heart, which derives 70% of its energy from FAO, myofiber degeneration occurred mainly in subendocardial and subepicardial areas of the left ventricle where energy demand and oxygen requirement is augmented during systolic contraction and blood perfusion and oxygen supply is the lowest.26 In the right ventricle, degeneration was transmural, consistent with a similar energy requirement and blood flow from the subepicardial toward the subendocardial areas.26 The pharmacological activation of PPAR-α receptors in skeletal muscles also demonstrates an association between FAO and microscopic damage in this tissue. Because skeletal muscle is composed of type I and type II fibers, and type I fibers are known to depend on oxidative metabolism and FAO as a source of energy,27 we used immunohistochemistry to determine whether the activation of PPAR-α receptors results in aerobic, glucose-dependent type II myofiber degeneration and/or involves type I fibers with a high dependency on oxidative metabolism. As shown in Figure 5, H&E staining and immunohistochemistry in skeletal muscle indicates that myofiber degeneration was mainly observed in type I fibers (abundant in soleus muscle), which, like cardiac myofibers, rely on fatty acid metabolism as energy source.
Figure 5.
Serial frozen sections of gastrocnemius and soleus. Sections were stained with H&E (a, b), MHCs antibodies (c, d), and MHCf antibodies (e, f). Arrows point toward the same myofiber in each figure. PPAR-α-mediated myofiber degeneration/necrosis is primarily found in the soleus (a), a muscle rich in type 1 (slow) myofibers (c). e: The adjacent gastrocnemius is not affected and is mainly composed of type 2 (fast) myofibers. b: Acutely injured myofibers are pale eosinophilic and infiltrated by a variable number of neutrophils and mononuclear cells (arrow). These myofibers are stained with the MHCs antibody (d, arrow) but not the MHCf antibody (f, arrow). Scale bars = 200 μm (a, c, e); 50 μm (b, d, f).
PPAR-α-Mediated Cardiomyocyte Necrosis Correlates with Increases in Peroxisomal β-Oxidation
To test the hypothesis that PPAR-α-mediated cardiomyocyte necrosis correlates with increases in peroxisomal β-oxidation in the heart, mice were orally dosed with either vehicle or various doses of a synthetic PPAR-α agonist once daily for 12 consecutive days. At the end of the dosing period, histopathology, serum chemistry, and AOX mRNA levels were evaluated. Cardiomyocyte necrosis occurred at doses ≥10 mg/kg and correlated with elevations in serum enzymes indicating heart injury (Table 2). Although AOX mRNA levels in the liver were increased at lower doses, cardiac AOX mRNA was only increased at the same doses at which cardiac damage was observed (Table 2), showing that the elevations in cardiac AOX mRNA levels correlate with the appearance of microscopic injury and serum biomarkers for cardiac injury. An additional experiment was performed to examine the temporal relationship between cardiac AOX mRNA levels and the appearance of plasma biomarkers of cardiac injury. As shown in Table 3, cardiac AOX mRNA levels increased significantly (P < 0.01) 5 days after the activation of PPAR-α receptors whereas changes in plasma levels of CK-MB were noticeable only at day 8. This indicates that the increase in myocardial β-oxidation precedes cardiomyocyte degeneration and suggests that a sustained increase in peroxisomal β-oxidation is necessary to induce cardiac damage.
Table 2.
Plasma CK and AOX mRNA Levels in Liver and Heart of Mice after Oral Administration of a PPAR-α Agonist
| Treatment (mg/kg/day) | Total CK (U/L) | CK-MB (U/L) | Liver AOX (RLU) | Heart AOX (RLU) | Histological incidence of necrosis |
|---|---|---|---|---|---|
| 0 | 77 ± 14 | 0 ± 0 | 578 ± 44 | 18 ± 1.3 | 0/5 |
| 0.05 | 70 ± 7 | 0 ± 0 | 1075 ± 196 | 17 ± 1.3 | 0/4 |
| 0.1 | 106 ± 94 | 0 ± 0 | 1447 ± 237 | 17 ± 1.9 | 0/5 |
| 1 | 121 ± 20* | 0 ± 0 | 1876 ± 79 | 17 ± 2.1 | 0/4 |
| 10 | 505 ± 143* | 19 ± 10* | 2207 ± 267* | 34 ± 4.7* | 3/5 |
| 100 | 1027 ± 317* | 129 ± 24* | 1907 ± 81* | 29 ± 1.6* | 5/5 |
Mice were orally dosed with vehicle or selective PPAR-α agonist for 12 consecutive days at doses escalating from 0.05 to 100 mg/kg/day. At the end of the dosing period, animals were euthanized and necropsied. Liver and cardiac AOX mRNA, plasma CK, and the heart CK-MB levels were determined as described in Materials and Methods. Significant elevations are indicated with an asterisk (P < 0.01); n = 4 to 5 mice/group/dose.
Table 3.
Cardiac AOX mRNA and Plasma CK-MB Levels in Mice Orally Dosed with a PPAR-α Agonist at 10 mg/kg/Day for 12 Consecutive Days
| Days | 0 | 3 | 5 | 8 | 12 |
|---|---|---|---|---|---|
| AOX | 21 ± 4 | 19 ± 2 | 34 ± 5* | 37 ± 4* | 43 ± 2* |
| CK-MB | 0 ± 0 | 0 ± 0 | 0 ± 0 | 22 ± 8* | 49 ± 25* |
| CK | 109 ± 20 | 90 ± 14 | 183 ± 57* | 291 ± 71* | 804 ± 285* |
AOX mRNA and plasma CK-MB isoenzyme levels were determined as described in Materials and Methods. AOX mRNA is expressed as RLU, whereas CK-MB levels are expressed as U/L. Data are expressed as mean ± SD (n = 5 mice/group).
P < 0.05.
Increased Myocardial Oxidative Stress in Mice Treated with PPAR-α Agonists
Key enzymes of the fatty acid β-oxidation pathways in peroxisomes and mitochondria as well as the ω-hydroxylation pathway in the endoplasmic reticulum are transcriptionally regulated by PPAR-α agonists.28–30 Our studies using a synthetic ligand for PPAR-α shows significant increases in cardiac mRNA levels of UCP3 and AOX, enzymes involved in the transfer and utilization of fatty acids, in response to PPAR-α agonism (Figure 2). Because many of these oxidases are important intracellular sources of reactive oxygen species, we determined whether sustained PPAR-α-mediated increases in FAO leads to oxidative stress injury in cardiac muscles. Parameters of tissue viability, tissue redox status, serum chemistry, and histopathology were evaluated in mice orally dosed with a PPAR-α agonist for 12 consecutive days. The doses of 1 mg/kg and 100 mg/kg were chosen to induce either no or severe cardiac injury, respectively. Histopathological evidence of cardiomyocyte necrosis in the heart was observed in mice orally dosed with 100 mg/kg and correlated with elevations in serum CK (443 ± 215 U/L versus 35 ± 7.0 U/L, P < 0.0001, n = 5 mice/group, for treated and control animals, respectively) and CK-MB (80 ± 31 U/L versus 0 ± 0 U/L, n = 5 mice/group, for treated and control, respectively).
Changes in cardiac muscle metabolic homeostasis were observed at all doses. Increased cardiac glutamate dehydrogenase (30% at 1 mg/kg and 42% at 100 mg/kg) and alkaline phosphatase (78% at 1 mg/kg and 210% at 100 mg/kg) were indicative of changes in mitochondria31 and vascular endothelia,32 respectively. Lactate concentration was decreased by 39 and 49%, respectively, in cardiac tissue and serum from mice dosed at 100 mg/kg (Figure 6). These findings together with the decrease in PFK and glycogen phosphorylase activities, and increased glucose levels in heart homogenates prepared from mice treated with the PPAR-α agonist (Figure 3) strongly suggest a shift from anaerobic to aerobic metabolism, consistent with a compound-related increase in FAO. Evidence for treatment-related increased oxidative stress in mice dosed at 100 mg/kg was demonstrated by the elevations in glutathione reductase (77%, n = 10, P < 0.01), GSH (13%, n = 10, P < 0.05), and glucose-6-phosphate dehydrogenase (56%, n = 10, P < 0.01) (Figure 7). No changes in oxidative stress biomarkers and serum levels of CK-MB were observed in mice receiving 1 mg/kg. Taken together, these studies strongly suggest that the ligand-dependent activation of PPAR-α receptors leads to sustained increase in peroxisomal β-oxidation and subsequent accumulation of oxidative stress intermediates resulting in cardiomyocyte necrosis in the heart and myofiber degeneration in skeletal muscle. In addition to the activation of peroxisomal β-oxidation, increases in cardiac mitochondrial FAO as a result of PPAR-α activation would also lead to the formation of intracellular reactive oxygen species. We did not measure mitochondrial FAO directly, but the activities of two mitochondrial enzymes involved in the uptake and degradation of fatty acids: carnitine palmitoyl transferase-I [102,571 ± 17,515 U/L (n = 12) and 105,828 ± 17,108 U/L (n = 13) for agonist- and vehicle-treated groups, respectively] and β-hydroxyacyl CoA dehydrogenase [665,824 ± 232,918 U/L (n = 11) and 447,089 ± 141,099 U/L (n = 13) for agonist- and vehicle-treated groups, respectively] did not differ. Increased β-hydroxyacyl CoA dehydrogenase activity in response to PPAR-α activation was observed in liver homogenates (data not shown).
Figure 6.
Glutamate dehydrogenase, alkaline phosphatase, and alanine aminotransferase activities and lactate concentrations in heart homogenates. Mice were orally administered either vehicle or a synthetic PPAR-α agonist at 1 or 100 mg/kg/day for 12 consecutive days. At the end of the study, animals were euthanized, and hearts were collected and homogenized for further analysis. Glutamate dehydrogenase (A), alkaline phosphatase (B), and alanine aminotransferase (C) activities and lactate (D) concentrations were determined using an Advia 1650 chemistry system as described under Materials and Methods. Data are expressed as mean ± SD from 10 mice per group. Asterisks show statistical significance. *P < 0.05.
Figure 7.
Cardiac glucose-6-phosphate dehydrogenase and glutathione reductase activities and GSH concentrations in either vehicle- or compound-treated mice. Glucose-6-phosphate dehydrogenase (A) and glutathione reductase (B) activities and GSH (C) concentrations were determined in heart homogenates from mice as described under Materials and Methods. Data are expressed as mean ± SD (n = 10/group); *P < 0.05.
Discussion
The present study demonstrated that cardiomyocyte necrosis observed in mice after treatment with a potent PPAR-α agonist is a direct consequence of activation of PPAR-α receptors because no cardiac damage was observed in PPAR-α-deficient mice orally administered with a PPAR-α agonist. These findings suggest that the dramatic decrease in FAO and increase in glucose oxidation observed in PPAR-α-deficient mice33 is sufficient to prevent the development of PPAR-α-mediated cardiomyocyte degeneration and necrosis. These studies also support a mechanistic link between cardiac PPAR-α signaling, peroxisomal β-oxidation, and perturbations in cardiac energy metabolism resulting in the generation of reactive oxygen species and myofiber degeneration. Several potent and selective PPAR-α agonists were tested, and they all cause cardiomyocyte degeneration in rodents suggesting that these findings are not specific to a particular agent but rather a class effect.
In contrast to the well-defined effects of PPAR-α on lipoprotein metabolism, the role of this nuclear receptor on cardiac metabolism in normal and pathological states is less clear. PPAR-α regulates a broad array of genes involved in cellular fatty acid uptake and oxidation in tissues that use fatty acids for ATP production. Changes in PPAR-α receptor levels or activation by either endogenous or exogenous ligands determine the tissue-specific substrate preferences observed under various developmental, physiological, and pathological conditions.34 PPAR-α levels are decreased in rodent models of ventricular pressure overload and in failing human heart35–38 leading to an increased utilization of glucose and lactate. In contrast, the utilization of glucose is impaired in the diabetic heart39 resulting in an almost exclusive use of FAO as energy source to the myocardium. Studies in transgenic mice with cardiac overexpression of PPAR-α receptors40 demonstrated that PPAR-α receptors control FAO and glucose utilization, resulting in a diabetic-like cardiomyopathy. These studies, however, were conducted using mice with cardiac-restricted overexpression of the human PPAR-α transgene at ∼80-fold the levels found in control littermates raising the question as to whether cardiac metabolism and function are also impaired when cardiac PPAR-α receptor levels remain unchanged and its activity is induced by sustained treatment with a PPAR-α agonist. Our studies demonstrate that chronic activation of cardiac PPAR-α receptors in mice leads to the development of cardiomyocyte necrosis in the heart and myofiber degeneration in type I skeletal muscle fibers. Necrosis/degeneration was therefore observed in tissues that mainly depend on oxidative metabolism and FAO as a source of energy suggesting an increased utilization of fatty acids. Consistent with this hypothesis, PDK4 and AOX; two genes involved in substrate switching and peroxisomal β-oxidation were significantly increased in the hearts of mice treated with the PPAR-α agonist. Tissue levels of triglycerides and fatty acids were decreased by treatment with a PPAR-α agonist (data not shown). In addition, the increase in the tissue level concentration of glucose and PFK; a key enzyme involved in glycolysis; further support the substrate switching and a shift from anaerobic to aerobic metabolism.
An increase in peroxisomal β-oxidation in the heart correlates with the appearance of compound-related microscopic injury and elevations in serum biomarkers for cardiac injury. Therefore, cardiomyocyte necrosis is hypothesized to be a consequence of sustained increases in peroxisomal FAO and the accompanying oxidative stress damage. This hypothesis is based on early observations41 and our own findings that sustained exposure to PPAR-α agonists leads to a persistent increase in oxidative stress. This hypothesis is also supported by studies in PPAR-α-deficient mice,29 cardiac-specific overexpression of PPAR-α,16 and the control of cardiac malonyl CoA decarboxylase by synthetic PPAR-α agonists.42 Increases in hydrogen peroxide and reactive oxygen species were detected in mice overexpressing PPAR-α in the heart and were exacerbated when the animals were fed a diet enriched in triglyceride containing long-chain fatty acids16 consistent with a prominent role of peroxisomal β-oxidation in this process. Our studies also provided additional evidence for cellular oxidative stress as documented by the up-regulation of the glutathione system and enzymes that detoxify reactive oxygen species. In addition to the activation in peroxisomal β-oxidation, increases in mitochondrial FAO as a result of PPAR-α activation could be an important source of intracellular reactive oxygen species and may be involved in the pathogenesis of PPAR-α-mediated cardiomyopathy observed in rodents. No evidence of increased cardiac mitochondrial FAO was observed in mice treated with the PPAR-α agonist when compared with vehicle-treated animals as demonstrated by the activity of two key enzymes involved in the transport and degradation of fatty acids in mitochondria. These findings are intriguing given the role that PPAR-α receptors play in the transcriptional control of genes involved in mitochondrial FAO.14,15 Nevertheless, the regulation of enzyme protein levels and activity and fatty acid flux through the mitochondrial pathway in response to PPAR-α activation is less known. Interestingly, only a twofold increase in carnitine palmitoyl transferase-1 activity was observed in transgenic mice expressing high levels of PPAR-α in the heart (∼80-fold the levels found in control littermates) dosed with WY-14,643, a selective and synthetic PPAR-α agonist.16 Similarly, the oxidation of palmitate in hearts isolated from the same transgenic mice was only ∼50% higher than in wild-type mice43 suggesting that high levels of PPAR-α expression are required to induce cardiac mitochondrial β-oxidation with synthetic ligands. Additional studies, however, will need to be performed to clearly delineate whether the flux of fatty acids through the mitochondrial pathway is affected in the hearts of wild-type mice treated with potent and synthetic PPAR-α selective agents.
The relevance of these findings to nonrodent species and to humans in particular remains unknown. Studies in the literature44,45 indicate that higher levels of lipid peroxidation can be induced in the rodent myocardium compared with humans, making the rodent a more sensitive species to oxidative stress injury. Mitochondrial and cellular membranes of rodent cardiomyocytes are composed of greater proportions of polyunsaturated phospholipids than the cardiomyocytes of primates and humans, and the levels of unsaturation correlate with proportionately greater free radical damage and lipid peroxidation of macromolecules and DNA leading to cell death. In addition, species differences in response to PPAR-α agonists are also noted. For example, there is a marked species difference in the hepatic response to PPAR-α agonism that has been described in the literature.46–48 Rats and mice are extremely sensitive, hamsters and dogs exhibit an intermediate response, and guinea pigs, monkeys, and humans are relatively nonresponsive at dose levels that produce a marked hepatic response in rodents. It is possible therefore that, based on the lack of adverse cardiac effects in humans chronically treated with fibrates, there is a difference in response to PPAR-α agonists across species similar to the continuum observed for liver peroxisomal proliferation and hepatocarcinogenesis. Additional studies will need to be conducted in various species to further explore the significance of these findings to nonrodent species and in particular to humans.
Acknowledgments
We thank Kim Kowsz, Karen Steerer, Mary Payette, Sandy Summers, Phil Cleall, Peter Towell, and Andrew Moody for their assistance and technical expertise.
Footnotes
Address reprint requests to Omar L. Francone, Pfizer Global Research and Development, Department of Cardiovascular and Metabolic Diseases, Eastern Point Rd., Groton, CT 06340. E-mail: omar.l.francone@pfizer.com.
References
- Wilson TM, Brown PJ, Sternbach DD, Henke BR. The PPARs: from orphan receptors to drug discovery. J Med Chem. 2000;43:527–550. doi: 10.1021/jm990554g. [DOI] [PubMed] [Google Scholar]
- Corton JC, Anderson SP, Stauber A. Central role of peroxisome proliferators-activated receptor in the actions of peroxisome proliferators. Annu Rev Pharmacol Toxicol. 2000;40:491–518. doi: 10.1146/annurev.pharmtox.40.1.491. [DOI] [PubMed] [Google Scholar]
- Issemann I, Green S. Activation of a member of the steroid hormone receptor superfamily by peroxisome proliferators. Nature. 1990;347:645–650. doi: 10.1038/347645a0. [DOI] [PubMed] [Google Scholar]
- Dreyer C, Krey G, Keller H, Givel F, Helftenbein G, Wahli W. Control of the peroxisomal β-oxidation pathway by a novel family of nuclear hormone receptors. Cell. 1992;68:879–887. doi: 10.1016/0092-8674(92)90031-7. [DOI] [PubMed] [Google Scholar]
- Göttlicher M, Widmark E, Li Q, Gustafsson JA. Fatty acids activate a chimera of the clofibric acid-activated receptor and the glucocorticoid receptor. Proc Natl Acad Sci USA. 1992;89:4653–4657. doi: 10.1073/pnas.89.10.4653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell AR, Savory R, Horley NJ, Choudhury AI, Dickins M, Gray TJB, Salter AM, Bell DR. Molecular basis of nonresponsiveness to peroxisome proliferators: the guinea-pig PPARα is functional and mediates peroxisome proliferators-induced hypolipidemia. Biochem J. 1998;332:689–693. doi: 10.1042/bj3320689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher T, Yi HF, McBride OW, Gonzalez FJ. cDNA cloning, chromosomal mapping, and functional characterization of the human peroxisome proliferators activated receptor. Biochemistry. 1993;32:5598–5604. doi: 10.1021/bi00072a015. [DOI] [PubMed] [Google Scholar]
- Mukherjee R, Jow L, Noonan D, McDonnell DP. Human and rat peroxisome proliferators activated receptors (PPARs) demonstrate similar tissue distribution but different responsiveness to PPAR activators. J Steroid Biochem Mol Biol. 1994;51:157–166. doi: 10.1016/0960-0760(94)90089-2. [DOI] [PubMed] [Google Scholar]
- Li AC, Binder CJ, Gutierrez A, Brown KK, Plotkin CR, Pattison JW, Valledor AF, Davis RA, Willson TM, Witztum JL, Palinski W, Glass CK. Differential inhibition of macrophage foam-cell formation and atherosclerosis in mice by PPAR α, β/δ, and γ. J Clin Invest. 2004;114:1564–1576. doi: 10.1172/JCI18730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubins HB, Robins SJ, Collins D, Fye CL, Anderson JW, Elam MB, Faas FH, Linares E, Schaeffer EJ, Schectman G, Wilt TJ, Wittes J. Gemfibrozyl for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. N Engl J Med. 1999;341:410–418. doi: 10.1056/NEJM199908053410604. [DOI] [PubMed] [Google Scholar]
- Auboeuf D, Rieusset J, Fajas L, Valier P, Frering V, Riou JP, Staels B, Auwerx J, Laville M, Vidal H. Tissue distribution and quantification of the expression of mRNAs of peroxisome proliferators-activated receptors and liver X receptor-α in humans. Diabetes. 1997;46:1319–1327. doi: 10.2337/diab.46.8.1319. [DOI] [PubMed] [Google Scholar]
- Mandard S, Müller M, Kersten S. Peroxisome proliferators-activated receptor α target genes. Cell Mol Sci. 2004;61:393–416. doi: 10.1007/s00018-003-3216-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeukendrup AE. Regulation of fat metabolism in skeletal muscle. Ann NY Acad Sci. 2002;967:217–235. doi: 10.1111/j.1749-6632.2002.tb04278.x. [DOI] [PubMed] [Google Scholar]
- Taegtmeyer H. Energy metabolism of the heart: from basic concepts to clinical applications. Curr Prob Cardiol. 1994;19:61–86. doi: 10.1016/0146-2806(94)90008-6. [DOI] [PubMed] [Google Scholar]
- Lehman JJ, Kelly DP. Transcriptional activation of energy metabolic switches in the developing and hypertrophied heart. Clin Exp Pharmacol Physiol. 2002;29:339–345. doi: 10.1046/j.1440-1681.2002.03655.x. [DOI] [PubMed] [Google Scholar]
- Finck BN, Han X, Courtois M, Aimond F, Nerbonne JM, Kovacs A, Gross RW, Kelly DP. A critical role for PPARα-mediated lipotoxicity in the pathogenesis of diabetic cardiomyopathy: modulation by dietary fat content. Proc Natl Acad Sci USA. 2003;100:1226–1231. doi: 10.1073/pnas.0336724100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depre C, Young ME, Ying J, Ahuja HS, Han Q, Garza N, Davies PJA, Taegtmeyer H. Streptozotocin-induced changes in cardiac gene-expression in the absence of severe contractile dysfunction. J Mol Cell Cardiol. 2000;32:985–996. doi: 10.1006/jmcc.2000.1139. [DOI] [PubMed] [Google Scholar]
- Razeghi P, Young ME, Cockrill TC, Frazier OH, Taegtmeyer H. Downregulation of myocardial myocyte enhancer Factor 2C and myocyte enhancer factor 2C-regulate gene expression in diabetic patients with nonischemic heart failure. Circulation. 2002;106:407–411. doi: 10.1161/01.cir.0000026392.80723.dc. [DOI] [PubMed] [Google Scholar]
- Lee SS, Pineau T, Drago J, Lee EJ, Owens JW, Kroetz DL, Fernandez-Salguero PM, Westphal H, Gonzalez FJ. Targeted disruption of the α isoform of the peroxisomal proliferators-activated receptor in mice results in abolishment of the pleiotropic effects of peroxisome proliferators. Mol Cell Biol. 1995;15:3012–3022. doi: 10.1128/mcb.15.6.3012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazarow PB. Assay of peroxisomal β-oxidation of fatty acids. Methods Enzymol. 1981;72:315–319. doi: 10.1016/s0076-6879(81)72021-4. [DOI] [PubMed] [Google Scholar]
- Shonk CE, Boxer GE. Enzyme patterns in human tissue. I. Method for determination of glycolytic enzymes. Cancer Res. 1964;24:709–721. [PubMed] [Google Scholar]
- Yates DW, Garland PB. Carnitine palmitoyl transferase activities of rat liver mitochondria. Biochem J. 1970;119:547–552. doi: 10.1042/bj1190547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Binstock JF, Schulz H. Fatty acid oxidation complex from Escherichia coli. Methods Enzymol. 1981;71:403–411. doi: 10.1016/0076-6879(81)71051-6. [DOI] [PubMed] [Google Scholar]
- Helmreich E, Cori CF. The effects of pH and temperature on the kinetics of the phosphorylase reaction. Proc Natl Acad Sci USA. 1964;52:647–654. doi: 10.1073/pnas.52.3.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruimboom-Brees IM, Brees DJ, Shen AC, Keener M, Francone O, Amacher DE, Loy JK, Kerlin RL. Using laser scanning cytometry to measure PPAR-mediated peroxisome proliferation and β oxidation. Toxicol Pathol. 2005;33:86–91. doi: 10.1080/01926230590881817. [DOI] [PubMed] [Google Scholar]
- Ohzono K, Koyanagi S, Urabe Y, Harasawa Y, Tomoike H, Nakamura M. Transmural distribution of myocardial infarction: difference between the right and left ventricles in a canine model. Circ Res. 1986;59:63–73. doi: 10.1161/01.res.59.1.63. [DOI] [PubMed] [Google Scholar]
- Spangenburg EE, Booth FW. Molecular regulation of individual skeletal muscle fiber type. Acta Physiol Scand. 2003;178:413–424. doi: 10.1046/j.1365-201X.2003.01158.x. [DOI] [PubMed] [Google Scholar]
- Reddy JK, Goel SK, Nemali MR, Carrino JJ, Laffler TG, Reddy MK, Sperbeck SJ, Osumi T, Hashimoto T, Lalwani ND, Sabasiva Rao M. Transcriptional regulation of peroxisomal fatty acid-CoA oxidase and enoyl-CoA hydratase/3-hydroxyacyl-CoA dehydrogenase in rat liver by peroxisome proliferators. Proc Natl Acad Sci USA. 1986;83:1747–1751. doi: 10.1073/pnas.83.6.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardwick JP, Song BJ, Huberman E, Gonzalez FJ. Isolation, complementary DNA sequence, and regulation of rat hepatic lauric acid w-hydroxylase (cytochrome P450Law): identification of new cytochrome P-450 gene family. J Biol Chem. 1987;262:801–810. [PubMed] [Google Scholar]
- Johnson EF, Palmer C, Hsu M. The peroxisome proliferators-activated receptor: transcriptional activation of the CYP4A6 gene. Ann NY Acad Sci. 1996;804:373–386. doi: 10.1111/j.1749-6632.1996.tb18629.x. [DOI] [PubMed] [Google Scholar]
- Mastorodemos V, Zaganas I, Spanaki C, Bessa M, Plaitakis A. Molecular basis of human glutamate dehydrogenase regulation under changing energy demands. J Neurosci Res. 2005;79:65–73. doi: 10.1002/jnr.20353. [DOI] [PubMed] [Google Scholar]
- Koyama T, Xie Z, Gao M, Suzuki J, Batra S. Adaptive changes in the capillary network in the left ventricle of rat heart. Jpn J Physiol. 1998;48:229–241. doi: 10.2170/jjphysiol.48.229. [DOI] [PubMed] [Google Scholar]
- Campbell FM, Kozak R, Wagner A, Altarejos JY, Dyck JR, Belke DD, Severson DL, Kelly DP, Lopaschuk GD. A role of PPARα in the control of cardiac malonyl-CoA levels: reduced fatty acid oxidation rates and increased glucose oxidation rates in the hearts of mice lacking PPARα are associated with higher concentrations of malonyl-CoA and reduced expression of malonyl-CoA decarboxylase. J Biol Chem. 2002;277:4098–4103. doi: 10.1074/jbc.M106054200. [DOI] [PubMed] [Google Scholar]
- Barger PM, Kelly DP. PPAR signaling in the control of cardiac energy metabolism. Trends Cardiovasc Med. 2000;10:238–245. doi: 10.1016/s1050-1738(00)00077-3. [DOI] [PubMed] [Google Scholar]
- Barger PM, Brandt JM, Leone TC, Weinheimer CJ, Kelly DP. Deactivation of peroxisome proliferators-activated receptor-α during cardiac hypertrophic growth. J Clin Invest. 2000;105:1723–1730. doi: 10.1172/JCI9056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depre C, Shipley GL, Chen W, Han Q, Doenst T, Moore ML, Stepkowski S, Davies PJ, Taegtmeyer H. Unloaded heart in vivo replicates fetal gene expression of cardiac hypertrophy. Nat Med. 1998;4:1269–1275. doi: 10.1038/3253. [DOI] [PubMed] [Google Scholar]
- Sack MN, Rader TA, Park S, Bastin J, McCune SA, Kelly DP. Fatty acid oxidation enzyme gene expression is downregulated in the failing heart. Circulation. 1996;94:2837–2842. doi: 10.1161/01.cir.94.11.2837. [DOI] [PubMed] [Google Scholar]
- Sack MN, Dish DL, Rockman HA, Kelly DP. A role for Sp and nuclear receptor transcription factors in a cardiac hypertrophic growth program. Proc Natl Acad Sci USA. 1997;94:6438–6443. doi: 10.1073/pnas.94.12.6438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley WC, Lopaschuk GD, McCormack JG. Regulation of energy substrate metabolism in the diabetic heart. Cardiovasc Res. 2002;34:25–33. doi: 10.1016/s0008-6363(97)00047-3. [DOI] [PubMed] [Google Scholar]
- Finck BN, Lehman JJ, Leone TC, Welch MJ, Bennett MJ, Kovacs A, Han X, Gross RW, Kozak R, Lopaschuk GD, Kelly DP. The cardiac phenotype induced by PPARalpha overexpression mimics that caused by diabetes mellitus. J Clin Invest. 2002;109:121–130. doi: 10.1172/JCI14080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reddy JK, Rao MS. Peroxisome proliferators and cancer: mechanisms and implications. Trends Pharmacol Sci. 1986;7:438–443. [Google Scholar]
- Young ME, Goodwin GW, Ying J, Guthrie P, Wilson CR, Laws FA, Taegtmeyer H. Regulation of cardiac and skeletal muscle malonyl-CoA decarboxylase by fatty acids. Am J Physiol. 2001;280:E471–E479. doi: 10.1152/ajpendo.2001.280.3.E471. [DOI] [PubMed] [Google Scholar]
- Hopkins TA, Sugden MC, Holness MJ, Kozak R, Dyck JR, Lopaschuk GD. Control of cardiac pyruvate dehydrogenase activity in peroxisome proliferators-activated receptor-α transgenic mice. Am J Physiol. 2003;285:H270–H276. doi: 10.1152/ajpheart.00852.2002. [DOI] [PubMed] [Google Scholar]
- Pamplona R, Prat J, Cadenas S, Rojas C, Perez-Campo R, Lopez Torres M, Barja G. Low fatty acid unsaturation protects against lipid peroxidation in liver mitochondria from longevous species: the pigeon and human case. Mech Ageing Dev. 1996;86:53–66. doi: 10.1016/0047-6374(95)01673-2. [DOI] [PubMed] [Google Scholar]
- Pamplona R, Portero-Otin M, Ruiz C, Gredilla R, Herrero A, Barja G. Double bond content of phospholipids and lipid peroxidation correlate with maximum longevity in the heart of mammals. Mech Ageing Dev. 1999;112:169–183. doi: 10.1016/s0047-6374(99)00045-7. [DOI] [PubMed] [Google Scholar]
- Ashby J, Brady A, Elcombe CR, Elliott BM, Ishmael J, Odum J, Tugwood JD, Kettle S, Purchase IF. Mechanistically based human hazard assessment of peroxisome proliferator-induced hepatocarcinogenesis. Hum Exp Toxicol. 1994;13(Suppl 2):S1–S117. doi: 10.1177/096032719401300201. [DOI] [PubMed] [Google Scholar]
- Bentley P, Calder I, Elcombe C, Grasso P, Stringer D, Wiegand HJ. Hepatic peroxisome proliferation in rodents and its significance for humans. Fd Chem Tox. 2003;31:857–907. doi: 10.1016/0278-6915(93)90225-n. [DOI] [PubMed] [Google Scholar]
- Cattley RC, DeLuca J, Elcombe C, Fenner-Crisp P, Lake BG, Marsman DS, Pastoor TA, Popp JA, Robinson DE, Schwetz B, Tugwood J, Wahli W. Do peroxisome proliferating compounds pose a hepatocarcinogenic risk to humans? Regul Toxic Pharmacol. 1998;27:47–60. doi: 10.1006/rtph.1997.1163. [DOI] [PubMed] [Google Scholar]