Abstract
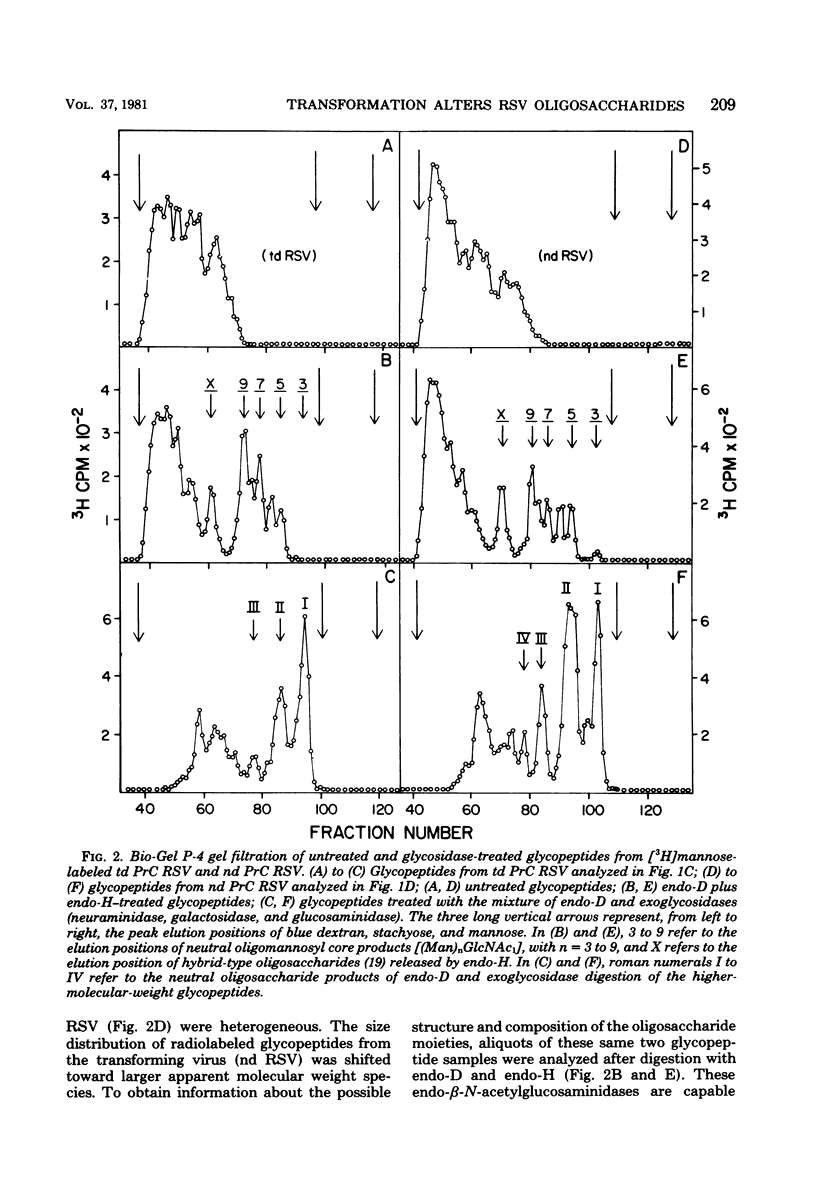
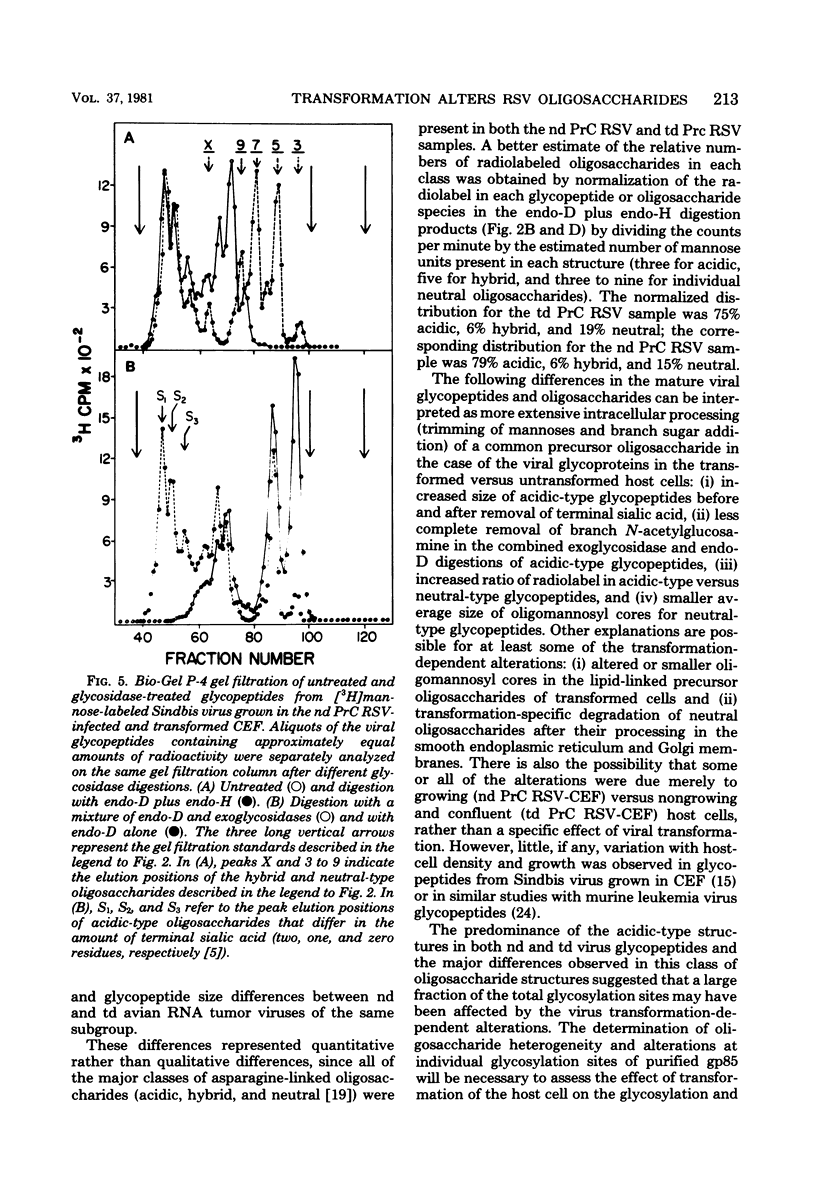
The influence of cell transformation on the glycosylation of viral envelope glycoproteins was examined by high-resolution gel filtration and specific glycosidase digestions of 3H-sugar-labeled glycopeptides from nondefective and transformation-defective Prague C strains of Rous sarcoma virus replicated in fibroblasts from the same chicken embryo. The major difference in glycosylation attributable to the viral transformation of the host cells was an increase in this relative amount of larger acidic-type oligosaccharides containing additional "branch" sugars (NeuNAc-Gal-GlcNAc-) compared with the smaller acidic-type and neutral-type oligosaccharides. There was also a shift in size distribution of neutral-type oligosaccharides toward smaller oligomannosyl cores in the transforming versus nontransforming virus glycopeptides. These alterations were consistent with a transformation-dependent increase in the extent of intracellular processing of a common precursor structure for the asparagine-linked oligosaccharides of Rous sarcoma virus.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- August J. T., Bolognesi D. P., Fleissner E., Gilden R. V., Nowinski R. C. A proposed nomenclature for the virion proteins of oncogenic RNA viruses. Virology. 1974 Aug;60(2):595–600. doi: 10.1016/0042-6822(74)90356-0. [DOI] [PubMed] [Google Scholar]
- Baenziger J. U., Fiete D. Structure of the complex oligosaccharides of fetuin. J Biol Chem. 1979 Feb 10;254(3):789–795. [PubMed] [Google Scholar]
- Bolognesi D. P., Bauer H., Gelderblom H., Hüper G. Polypeptides of avian RNA tumor viruses. IV. Components of the viral envelope. Virology. 1972 Mar;47(3):551–566. doi: 10.1016/0042-6822(72)90545-4. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Burke D., Keegstra K. Carbohydrate structure of Sindbis virus glycoprotein E2 from virus grown in hamster and chicken cells. J Virol. 1979 Feb;29(2):546–554. doi: 10.1128/jvi.29.2.546-554.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duesberg P. H., Martin G. S., Vogt P. K. Glycoprotein components of avian and murine RNA tumor viruses. Virology. 1970 Aug;41(4):631–646. doi: 10.1016/0042-6822(70)90428-9. [DOI] [PubMed] [Google Scholar]
- Etchison J. R., Robertson J. S., Summers D. F. Partial structural analysis of the oligosaccharide moieties of the vesicular stomatitis virus glycoprotein by sequential chemical and enzymatic degradation. Virology. 1977 May 15;78(2):375–392. doi: 10.1016/0042-6822(77)90115-5. [DOI] [PubMed] [Google Scholar]
- Glick M. C. Membrane glycopeptides from virus-transformed hamster fibroblasts and the normal counterpart. Biochemistry. 1979 Jun 12;18(12):2525–2532. doi: 10.1021/bi00579a015. [DOI] [PubMed] [Google Scholar]
- Hunt L. A. Altered synthesis and processing of oligosaccharides of vesicular stomatitis virus glycoprotein in different lectin-resistant Chinese hamster ovary cell lines. J Virol. 1980 Aug;35(2):362–370. doi: 10.1128/jvi.35.2.362-370.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A. Biosynthesis and maturation of cellular membrane glycoproteins. J Supramol Struct. 1979;12(2):209–226. doi: 10.1002/jss.400120207. [DOI] [PubMed] [Google Scholar]
- Hunt L. A., Etchison J. R., Summers D. F. Oligosaccharide chains are trimmed during synthesis of the envelope glycoprotein of vesicular stomatitis virus. Proc Natl Acad Sci U S A. 1978 Feb;75(2):754–758. doi: 10.1073/pnas.75.2.754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A., Summers D. F. Association of vesicular stomatitis virus proteins with HeLa cell membranes and released virus. J Virol. 1976 Dec;20(3):637–645. doi: 10.1128/jvi.20.3.637-645.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A., Summers D. F. Glycosylation of vesicular stomatitis virus glycoprotein in virus-infected HeLa cells. J Virol. 1976 Dec;20(3):646–657. doi: 10.1128/jvi.20.3.646-657.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A., Wright S. E., Etchison J. R., Summers D. F. Oligosaccharide chains of avian RNA tumor virus glycoproteins contain heterogeneous oligomannosyl cores. J Virol. 1979 Jan;29(1):336–343. doi: 10.1128/jvi.29.1.336-343.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keegstra K., Sefton B., Burke D. Sindbis virus glycoproteins: effect of the host cell on the oligosaccharides. J Virol. 1975 Sep;16(3):613–620. doi: 10.1128/jvi.16.3.613-620.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krantz M. J., Lee Y. C., Hung P. P. Characterization and comparison of the major glycoprotein from three strains of Rous sarcoma virus. Arch Biochem Biophys. 1976 May;174(1):66–73. doi: 10.1016/0003-9861(76)90324-6. [DOI] [PubMed] [Google Scholar]
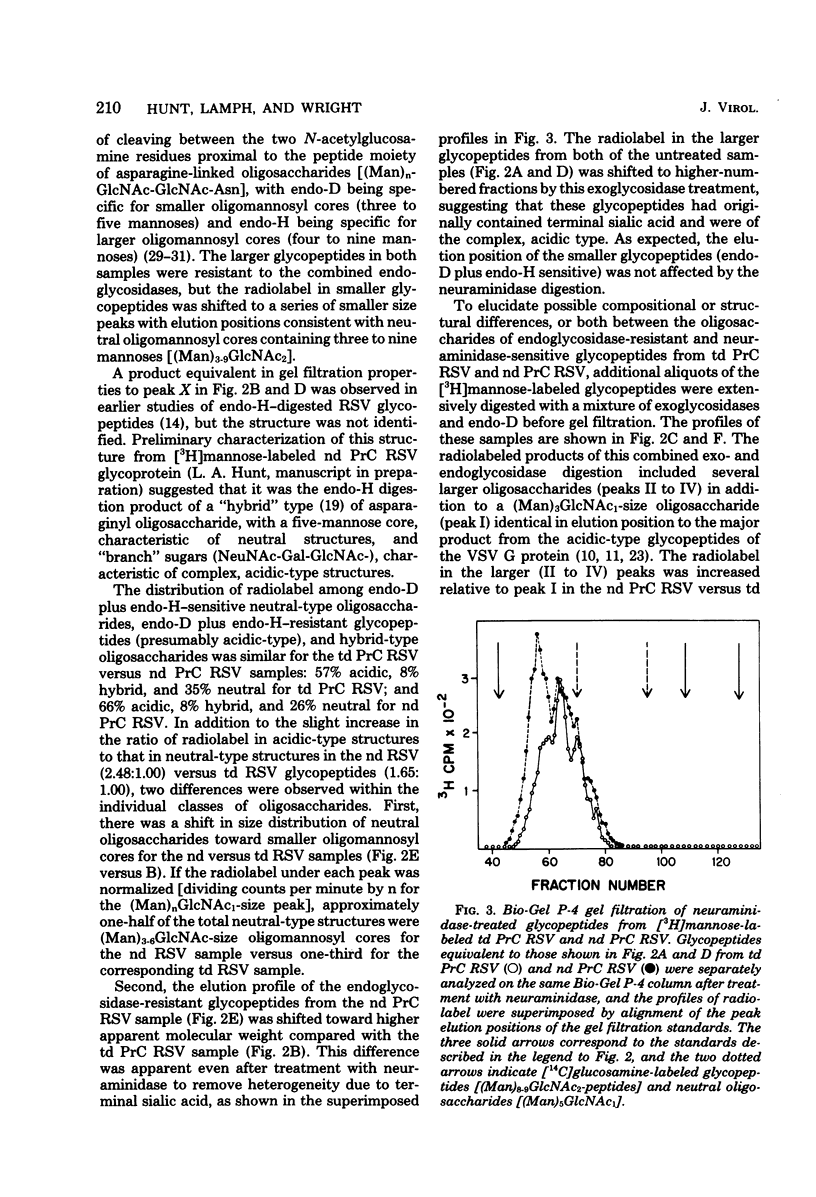
- Lai M. M., Duesberg P. H. Differences between the envelope glycoproteins and glycopeptides of avian tumor viruses released from transformed and from nontransformed cells. Virology. 1972 Nov;50(2):359–372. doi: 10.1016/0042-6822(72)90387-x. [DOI] [PubMed] [Google Scholar]
- Li E., Tabas I., Kornfeld S. The synthesis of complex-type oligosaccharides. I. Structure of the lipid-linked oligosaccharide precursor of the complex-type oligosaccharides of the vesicular stomatitis virus G protein. J Biol Chem. 1978 Nov 10;253(21):7762–7770. [PubMed] [Google Scholar]
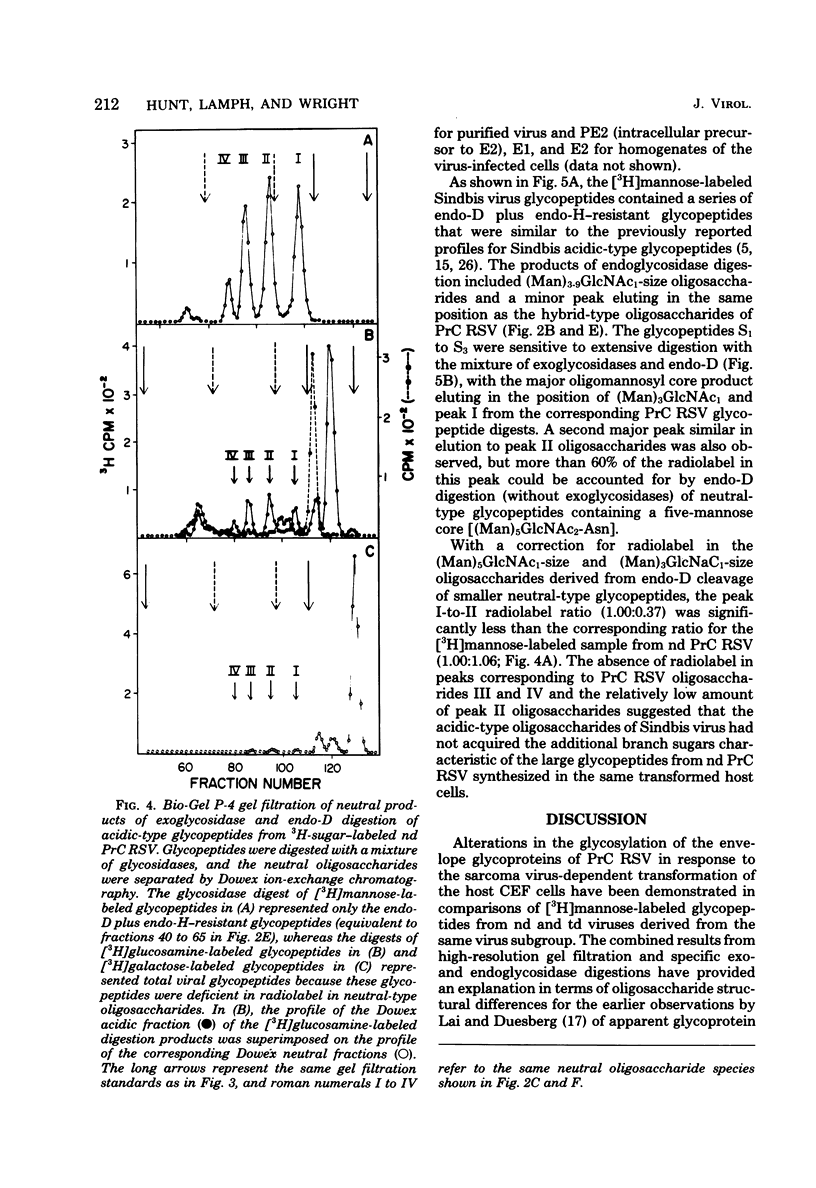
- Liang C. J., Yamashita K., Muellenberg C. G., Shichi H., Kobata A. Structure of the carbohydrate moieties of bovine rhodopsin. J Biol Chem. 1979 Jul 25;254(14):6414–6418. [PubMed] [Google Scholar]
- Love D. N., Weiss R. A. Pseudotypes of vesicular stomatitis virus determined by exogenous and endogenous avian RNA tumor viruses. Virology. 1974 Jan;57(1):271–278. doi: 10.1016/0042-6822(74)90127-5. [DOI] [PubMed] [Google Scholar]
- Robbins P. W., Hubbard S. C., Turco S. J., Wirth D. F. Proposal for a common oligosaccharide intermediate in the synthesis of membrane glycoproteins. Cell. 1977 Dec;12(4):893–900. doi: 10.1016/0092-8674(77)90153-2. [DOI] [PubMed] [Google Scholar]
- Robertson J. S., Etchison J. R., Summers D. F. Glycosylation sites of vesicular stomatitis virus glycoprotein. J Virol. 1976 Sep;19(3):871–878. doi: 10.1128/jvi.19.3.871-878.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson M. A., Etchison J. R., Robertson J. S., Summers D. F., Stanley P. Specific changes in the oligosaccharide moieties of VSV grown in different lectin-resistnat CHO cells. Cell. 1978 Mar;13(3):515–526. doi: 10.1016/0092-8674(78)90325-2. [DOI] [PubMed] [Google Scholar]
- Rosner M. R., Grinna L. S., Robbins P. W. Differences in glycosylation patterns of closely related murine leukemia viruses. Proc Natl Acad Sci U S A. 1980 Jan;77(1):67–71. doi: 10.1073/pnas.77.1.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santer U. V., Glick M. C. Partial structure of a membrane glycopeptide from virus-transformed hamster cells. Biochemistry. 1979 Jun 12;18(12):2533–2540. doi: 10.1021/bi00579a016. [DOI] [PubMed] [Google Scholar]
- Sefton B. M. Immediate glycosylation of Sindbis virus membrane proteins. Cell. 1977 Apr;10(4):659–668. doi: 10.1016/0092-8674(77)90099-x. [DOI] [PubMed] [Google Scholar]
- Sefton B. M. Virus-dependent glycosylation. J Virol. 1975 Jan;17(1):85–93. doi: 10.1128/jvi.17.1.85-93.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabas I., Schlesinger S., Kornfeld S. Processing of high mannose oligosaccharides to form complex type oligosaccharides on the newly synthesized polypeptides of the vesicular stomatitis virus G protein and the IgG heavy chain. J Biol Chem. 1978 Feb 10;253(3):716–722. [PubMed] [Google Scholar]
- Tai T., Yamashita K., Kobata A. The substrate specificities of endo-beta-N-acetylglucosaminidases CII and H. Biochem Biophys Res Commun. 1977 Sep 9;78(1):434–441. doi: 10.1016/0006-291x(77)91273-6. [DOI] [PubMed] [Google Scholar]
- Tai T., Yamashita K., Ogata-Arakawa M., Koide N., Muramatsu T., Iwashita S., Inoue Y., Kobata A. Structural studies of two ovalbumin glycopeptides in relation to the endo-beta-N-acetylglucosaminidase specificity. J Biol Chem. 1975 Nov 10;250(21):8569–8575. [PubMed] [Google Scholar]
- Trimble R. B., Tarentino A. L., Plummer T. H., Jr, Maley F. Asparaginyl glycopeptides with a low mannose content are hydrolyzed by endo-beta-N-acetylglucosaminidase H. J Biol Chem. 1978 Jul 10;253(13):4508–4511. [PubMed] [Google Scholar]
- Tuszynski G. P., Baker S. R., Fuhrer J. P., Buck C. A., Warren L. Glycopeptides derived from individual membrane glycoproteins from control and Rous sarcoma virus-transformed hamster fibroblasts. J Biol Chem. 1978 Sep 10;253(17):6092–6099. [PubMed] [Google Scholar]
- Wagner R. R., Prevec L., Brown F., Summers D. F., Sokol F., MacLeod R. Classification of rhabdovirus proteins: a proposal. J Virol. 1972 Dec;10(6):1228–1230. doi: 10.1128/jvi.10.6.1228-1230.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]