Abstract
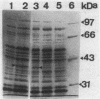
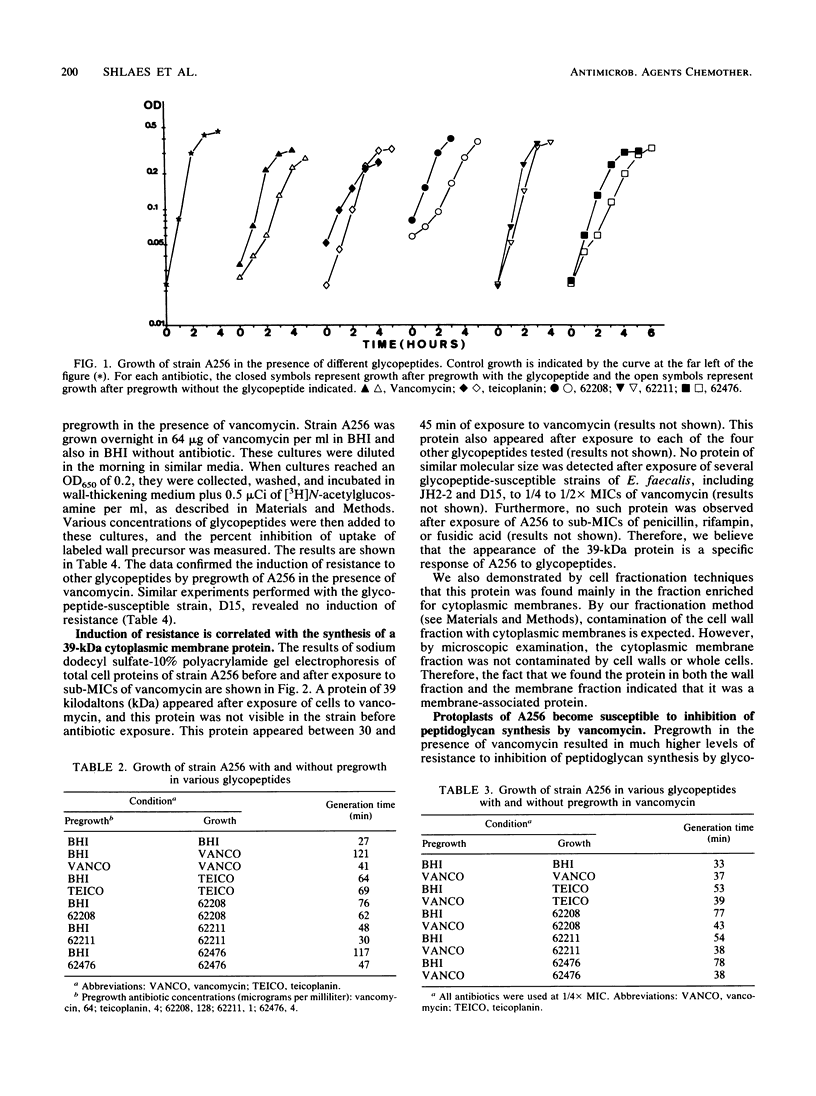
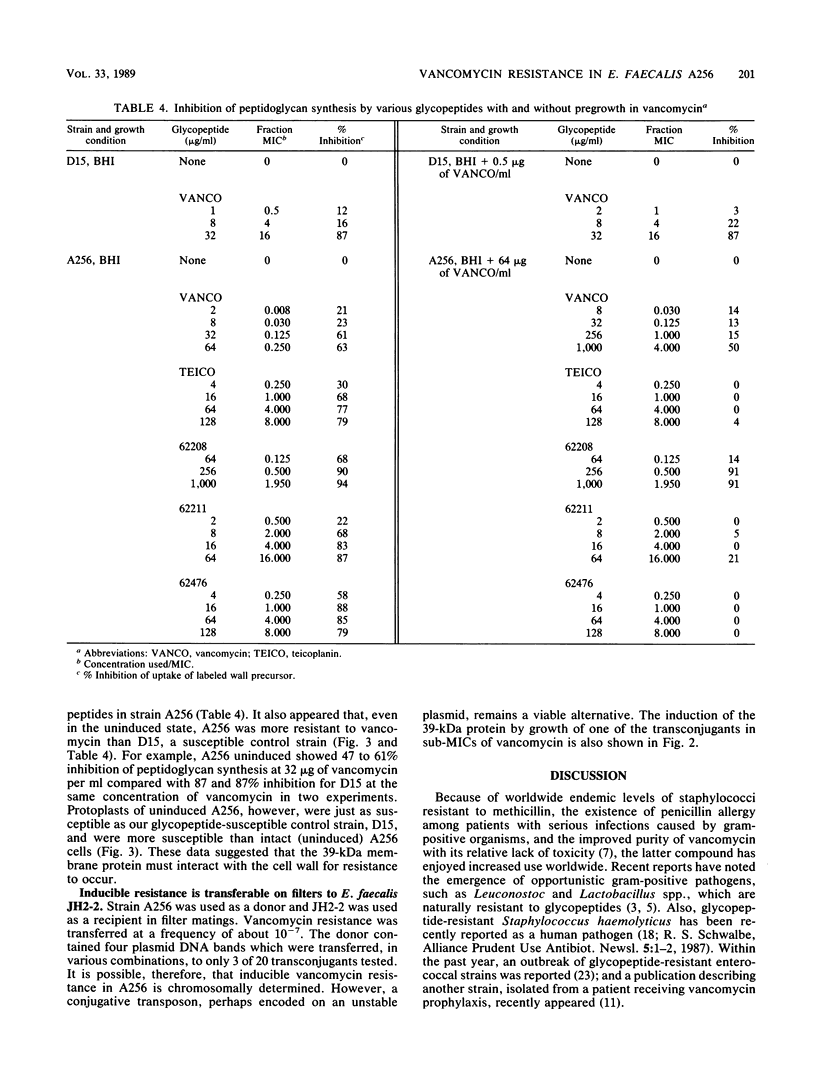
A strain of Enterococcus faecalis (A256) was isolated from the urine of a patient with urinary sepsis and was found to exhibit susceptibilities (micrograms per milliliter) to various glycopeptides as follows: vancomycin, 256; teicoplanin, 16; 62208, 512; 62211, 4; and 62476, 16. As judged by growth rates before and after exposure to sub-MICs of glycopeptides, vancomycin and 62476 induced self-resistance, 62208 and 62211 induced slight self-resistance, and teicoplanin did not induce self-resistance. Vancomycin induced cross-resistance to all other glycopeptides tested, as judged both in growth experiments and by direct measurement of inhibition of peptidoglycan synthesis in cells exposed to sub-MICs of vancomycin. Thus, the spectra of activity of the glycopeptides were not correlated with their patterns of induction. There was a correlation between the increased synthesis of a 39-kilodalton (kDa) protein located in the cytoplasmic membrane and the induction of resistance. Protoplasts of A256 were susceptible to inhibition of peptidoglycan synthesis by vancomycin at levels similar to those for susceptible strains. Vancomycin resistance was transferable on filters from the parent strain to E. faecalis JH2-2 at a frequency of about 10(-7), and the 39-kDa protein was also inducible by glycopeptides in these transconjugants. We conclude that A256 is resistant to glycopeptides by virtue of the synthesis of a 39-kDa cytoplasmic membrane protein, that this protein is probably involved in preventing access of the glycopeptides to their peptidoglycan targets, and that this resistance is transferable, probably by conjugation.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barrett J. F., Schramm V. L., Shockman G. D. Hydrolysis of soluble, linear, un-cross-linked peptidoglycans by endogenous bacterial N-acetylmuramoylhydrolases. J Bacteriol. 1984 Aug;159(2):520–526. doi: 10.1128/jb.159.2.520-526.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beck W. D., Berger-Bächi B., Kayser F. H. Additional DNA in methicillin-resistant Staphylococcus aureus and molecular cloning of mec-specific DNA. J Bacteriol. 1986 Feb;165(2):373–378. doi: 10.1128/jb.165.2.373-378.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buu-Hoï A., Branger C., Acar J. F. Vancomycin-resistant streptococci or Leuconostoc sp. Antimicrob Agents Chemother. 1985 Sep;28(3):458–460. doi: 10.1128/aac.28.3.458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambers H. F. Coagulase-negative staphylococci resistant to beta-lactam antibiotics in vivo produce penicillin-binding protein 2a. Antimicrob Agents Chemother. 1987 Dec;31(12):1919–1924. doi: 10.1128/aac.31.12.1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colman G., Efstratiou A. Vancomycin-resistant leuconostocs, lactobacilli and now pediococci. J Hosp Infect. 1987 Jul;10(1):1–3. doi: 10.1016/0195-6701(87)90025-9. [DOI] [PubMed] [Google Scholar]
- Facklam R. R. Recognition of group D streptococcal species of human origin by biochemical and physiological tests. Appl Microbiol. 1972 Jun;23(6):1131–1139. doi: 10.1128/am.23.6.1131-1139.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geraci J. E., Hermans P. E. Vancomycin. Mayo Clin Proc. 1983 Feb;58(2):88–91. [PubMed] [Google Scholar]
- Goldstein B. P., Selva E., Gastaldo L., Berti M., Pallanza R., Ripamonti F., Ferrari P., Denaro M., Arioli V., Cassani G. A40926, a new glycopeptide antibiotic with anti-Neisseria activity. Antimicrob Agents Chemother. 1987 Dec;31(12):1961–1966. doi: 10.1128/aac.31.12.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacob A. E., Douglas G. J., Hobbs S. J. Self-transferable plasmids determining the hemolysin and bacteriocin of Streptococcus faecalis var. zymogenes. J Bacteriol. 1975 Mar;121(3):863–872. doi: 10.1128/jb.121.3.863-872.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph R., Shockman G. D. Autolytic formation of protoplasts (autoplasts) of Streptococcus faecalis 9790: release of cell wall, autolysin, and formation of stable autoplasts. J Bacteriol. 1974 May;118(2):735–746. doi: 10.1128/jb.118.2.735-746.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan A. H., Gilligan P. H., Facklam R. R. Recovery of resistant enterococci during vancomycin prophylaxis. J Clin Microbiol. 1988 Jun;26(6):1216–1218. doi: 10.1128/jcm.26.6.1216-1218.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Bouguenec C., Horodniceanu T. Conjugative R plasmids in Streptococcus faecium (group D). Antimicrob Agents Chemother. 1982 May;21(5):698–705. doi: 10.1128/aac.21.5.698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leclercq R., Derlot E., Duval J., Courvalin P. Plasmid-mediated resistance to vancomycin and teicoplanin in Enterococcus faecium. N Engl J Med. 1988 Jul 21;319(3):157–161. doi: 10.1056/NEJM198807213190307. [DOI] [PubMed] [Google Scholar]
- Lewis P., Garaud J. J., Parenti F. A multicentre open clinical trial of teicoplanin in infections caused by gram-positive bacteria. J Antimicrob Chemother. 1988 Jan;21 (Suppl A):61–67. doi: 10.1093/jac/21.suppl_a.61. [DOI] [PubMed] [Google Scholar]
- Moellering R. C., Jr, Korzeniowski O. M., Sande M. A., Wennersten C. B. Species-specific resistance to antimocrobial synergism in Streptococcus faecium and Streptococcus faecalis. J Infect Dis. 1979 Aug;140(2):203–208. doi: 10.1093/infdis/140.2.203. [DOI] [PubMed] [Google Scholar]
- Perkins H. R., Nieto M. The chemical basis for the action of the vancomycin group of antibiotics. Ann N Y Acad Sci. 1974 May 10;235(0):348–363. doi: 10.1111/j.1749-6632.1974.tb43276.x. [DOI] [PubMed] [Google Scholar]
- Schwalbe R. S., Stapleton J. T., Gilligan P. H. Emergence of vancomycin resistance in coagulase-negative staphylococci. N Engl J Med. 1987 Apr 9;316(15):927–931. doi: 10.1056/NEJM198704093161507. [DOI] [PubMed] [Google Scholar]
- Shlaes D. M., Levy J., Wolinsky E. Enterococcal bacteremia without endocarditis. Arch Intern Med. 1981 Apr;141(5):578–581. [PubMed] [Google Scholar]
- Tynecka Z., Ward J. B. Peptidoglycan synthesis in Bacillus licheniformis. The inhibition of cross-linking by benzylpenicillin and cephaloridine in vivo accompanied by the formation of soluble peptidoglycan. Biochem J. 1975 Jan;146(1):253–267. doi: 10.1042/bj1460253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ubukata K., Yamashita N., Konno M. Occurrence of a beta-lactam-inducible penicillin-binding protein in methicillin-resistant staphylococci. Antimicrob Agents Chemother. 1985 May;27(5):851–857. doi: 10.1128/aac.27.5.851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uttley A. H., Collins C. H., Naidoo J., George R. C. Vancomycin-resistant enterococci. Lancet. 1988 Jan 2;1(8575-6):57–58. doi: 10.1016/s0140-6736(88)91037-9. [DOI] [PubMed] [Google Scholar]
- Williamson R., Gutmann L., Horaud T., Delbos F., Acar J. F. Use of penicillin-binding proteins for the identification of enterococci. J Gen Microbiol. 1986 Jul;132(7):1929–1937. doi: 10.1099/00221287-132-7-1929. [DOI] [PubMed] [Google Scholar]