Abstract
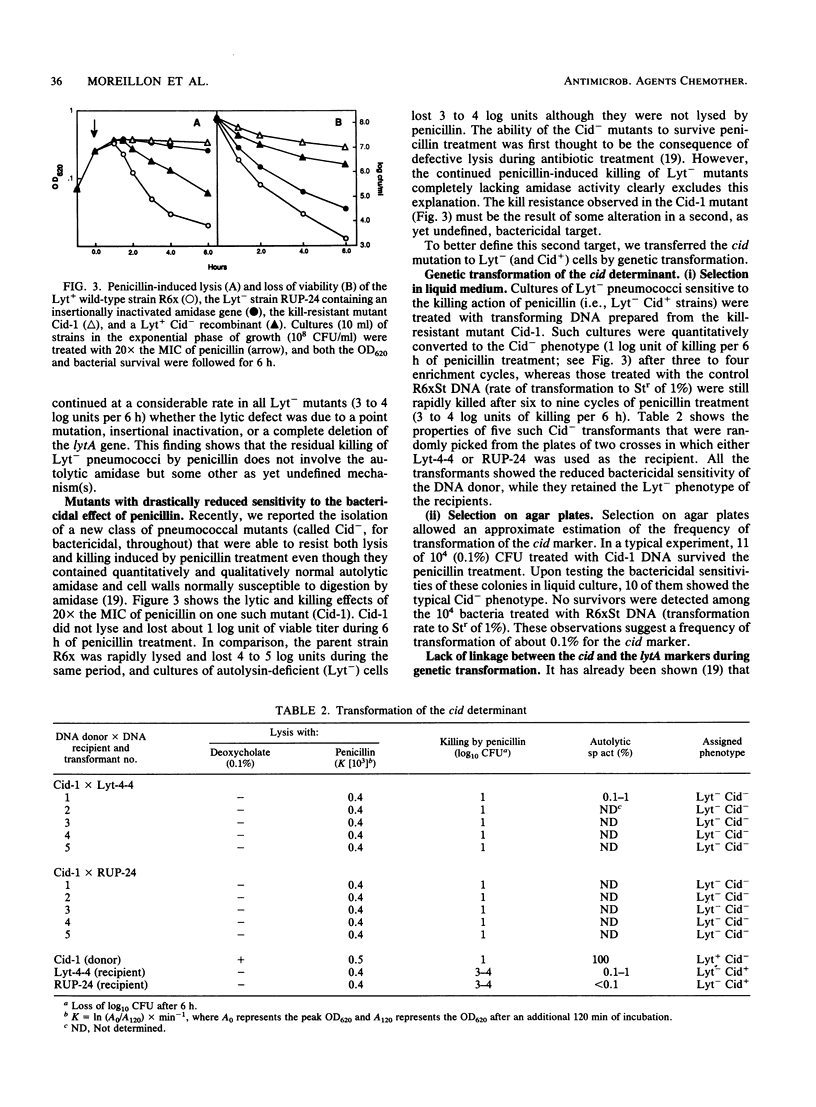
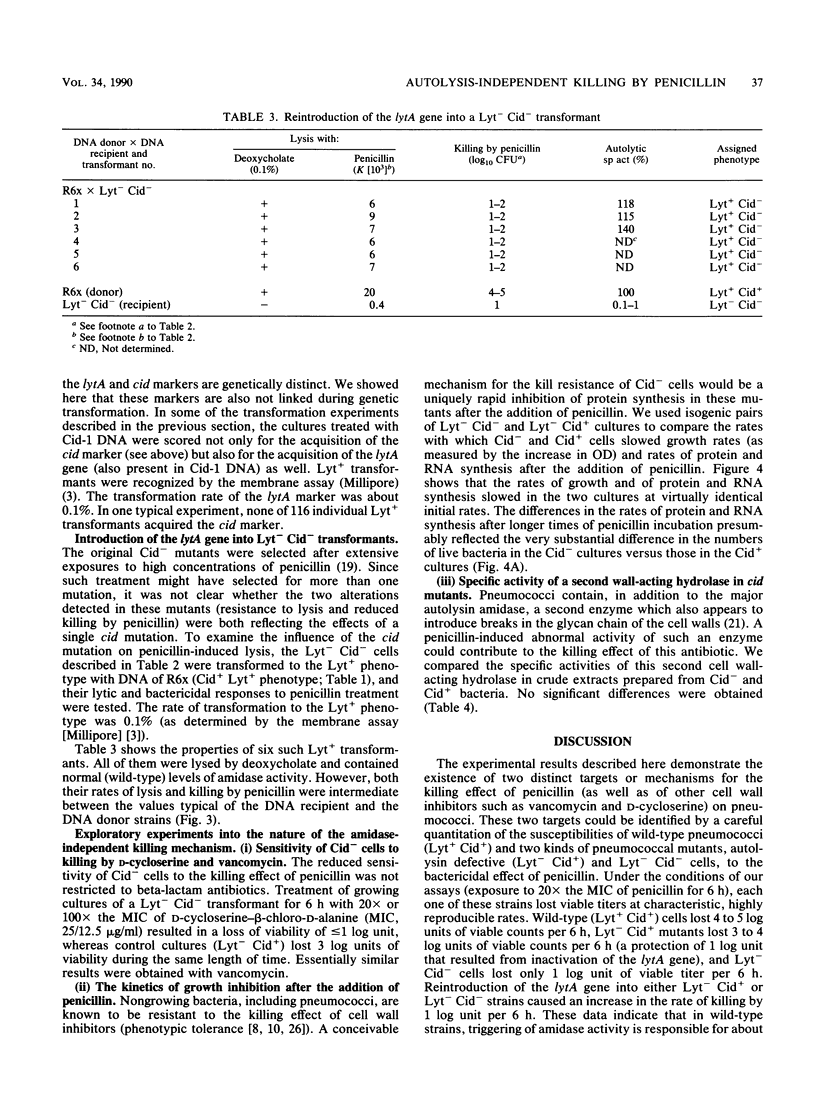
It has been assumed that penicillin (and also other cell wall inhibitors) kill pneumococci predominantly by triggering their major autolytic enzyme (an N-acetylmuramoyl-L-alanine amidase; referred to as amidase), resulting in massive cell wall degradation. Three types of experiments suggest that only part of this killing is due to cell lysis by amidase. (i) Suppression of penicillin-induced lysis by specific inhibitors of amidase protected pneumococci only marginally from killing in spite of prolonged exposure to concentrations of penicillin that were 10x, 20x, or 100x greater than the MIC. (ii) Mutants from which the amidase was completely eliminated by plasmid insertion or deletion (Lyt-) were still killed, albeit at a slower rate than the wild-type Lyt+ strains (3 to 4 log units instead of 4 to 5 log units per 6 h, i.e., about 1 log unit slower than the wild type; P less than 0.001). (iii) A new mutation (cid), which was not related to the amidase gene, further reduced killing of mutants lacking amidase to 1 log unit per 6 h (Lyt- Cid- phenotype). Reintroduction of the amidase gene into Lyt- Cid- cells partially restored penicillin-induced lysis but increased only slightly the rate of killing (from 1 log unit per 6 h in Lyt- Cid- cells to 2 log units per 6 h in Lyt+ Cid- cells). We conclude that penicillin kills pneumococci by two distinct mechanisms: one that involves the triggering of the amidase (about 1 log unit of killing per 6 h) and another, amidase-independent mechanism that is responsible for 3 to 4 log units of killing per 6 h. Triggering of the amidase activity in situ in growing bacteria was significantly reduced in Lyt+ Cid- cells, indicating that there is some regulatory interaction between the cid gene product and the amidase.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Garcia-Bustos J. F., Chait B. T., Tomasz A. Structure of the peptide network of pneumococcal peptidoglycan. J Biol Chem. 1987 Nov 15;262(32):15400–15405. [PubMed] [Google Scholar]
- García J. L., Sánchez-Puelles J. M., García P., López R., Ronda C., García E. Molecular characterization of an autolysin-defective mutant of Streptococcus pneumoniae. Biochem Biophys Res Commun. 1986 Jun 13;137(2):614–619. doi: 10.1016/0006-291x(86)91122-8. [DOI] [PubMed] [Google Scholar]
- Garrett J., Fusselman R., Hise J., Chiou L., Smith-Grillo D., Schulz J., Young R. Cell lysis by induction of cloned lambda lysis genes. Mol Gen Genet. 1981;182(2):326–331. doi: 10.1007/BF00269678. [DOI] [PubMed] [Google Scholar]
- Giudicelli S., Tomasz A. Attachment of pneumococcal autolysin to wall teichoic acids, an essential step in enzymatic wall degradation. J Bacteriol. 1984 Jun;158(3):1188–1190. doi: 10.1128/jb.158.3.1188-1190.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goessens W. H., Driessen A. J., Wilschut J., van Duin J. A synthetic peptide corresponding to the C-terminal 25 residues of phage MS2 coded lysis protein dissipates the protonmotive force in Escherichia coli membrane vesicles by generating hydrophilic pores. EMBO J. 1988 Mar;7(3):867–873. doi: 10.1002/j.1460-2075.1988.tb02886.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodell W., Tomasz A. Alteration of Escherichia coli murein during amino acid starvation. J Bacteriol. 1980 Dec;144(3):1009–1016. doi: 10.1128/jb.144.3.1009-1016.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Handwerger S., Tomasz A. Antibiotic tolerance among clinical isolates of bacteria. Rev Infect Dis. 1985 May-Jun;7(3):368–386. doi: 10.1093/clinids/7.3.368. [DOI] [PubMed] [Google Scholar]
- Henrich B., Lubitz W., Plapp R. Lysis of Escherichia coli by induction of cloned phi X174 genes. Mol Gen Genet. 1982;185(3):493–497. doi: 10.1007/BF00334146. [DOI] [PubMed] [Google Scholar]
- Horne D., Tomasz A. Tolerant response of Streptococcus sanguis to beta-lactams and other cell wall inhibitors. Antimicrob Agents Chemother. 1977 May;11(5):888–896. doi: 10.1128/aac.11.5.888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LACKS S., HOTCHKISS R. D. A study of the genetic material determining an enzyme in Pneumococcus. Biochim Biophys Acta. 1960 Apr 22;39:508–518. doi: 10.1016/0006-3002(60)90205-5. [DOI] [PubMed] [Google Scholar]
- Liu H. H., Tomasz A. Penicillin tolerance in multiply drug-resistant natural isolates of Streptococcus pneumoniae. J Infect Dis. 1985 Aug;152(2):365–372. doi: 10.1093/infdis/152.2.365. [DOI] [PubMed] [Google Scholar]
- Lopez R., Ronda-Lain C., Tapia A., Waks S. B., Tomasz A. Suppression of the lytic and bactericidal effects of cell wallinhibitory antibiotics. Antimicrob Agents Chemother. 1976 Oct;10(4):697–706. doi: 10.1128/aac.10.4.697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDowell T. D., Lemanski C. L. Absence of autolytic activity (peptidoglycan nicking) in penicillin-induced nonlytic death in a group A streptococcus. J Bacteriol. 1988 Apr;170(4):1783–1788. doi: 10.1128/jb.170.4.1783-1788.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreillon P., Tomasz A. Penicillin resistance and defective lysis in clinical isolates of pneumococci: evidence for two kinds of antibiotic pressure operating in the clinical environment. J Infect Dis. 1988 Jun;157(6):1150–1157. doi: 10.1093/infdis/157.6.1150. [DOI] [PubMed] [Google Scholar]
- Sanchez-Puelles J. M., Ronda C., Garcia J. L., Garcia P., Lopez R., Garcia E. Searching for autolysin functions. Characterization of a pneumococcal mutant deleted in the lytA gene. Eur J Biochem. 1986 Jul 15;158(2):289–293. doi: 10.1111/j.1432-1033.1986.tb09749.x. [DOI] [PubMed] [Google Scholar]
- Tiraby J. G., Fox M. S. Marker discrimination in transformation and mutation of pneumococcus. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3541–3545. doi: 10.1073/pnas.70.12.3541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasz A. Model for the mechanism controlling the expression of competent state in Pneumococcus cultures. J Bacteriol. 1966 Mar;91(3):1050–1061. doi: 10.1128/jb.91.3.1050-1061.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasz A., Moreillon P., Pozzi G. Insertional inactivation of the major autolysin gene of Streptococcus pneumoniae. J Bacteriol. 1988 Dec;170(12):5931–5934. doi: 10.1128/jb.170.12.5931-5934.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasz A., Waks S. Mechanism of action of penicillin: triggering of the pneumococcal autolytic enzyme by inhibitors of cell wall synthesis. Proc Natl Acad Sci U S A. 1975 Oct;72(10):4162–4166. doi: 10.1073/pnas.72.10.4162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuomanen E. Phenotypic tolerance: the search for beta-lactam antibiotics that kill nongrowing bacteria. Rev Infect Dis. 1986 Jul-Aug;8 (Suppl 3):S279–S291. doi: 10.1093/clinids/8.supplement_3.s279. [DOI] [PubMed] [Google Scholar]


