Abstract
Species of the virilis group of Drosophila differ by multiple inversions and chromosome fusions that probably accompanied, or led to, speciation. Drosophila virilis has the primitive karyotype for the group, and natural populations are exceptional in having no chromosomal polymorphisms. We report that the genomic locations of Penelope and Ulysses transposons are nonrandomly distributed in 12 strains of D. virilis. Furthermore, Penelope and Ulysses insertion sites in D. virilis show a statistically significant association with the breakpoints of inversions found in other species of the virilis group. Sixteen newly induced chromosomal rearrangements were isolated from the progeny of D. virilis hybrid dysgenic crosses, including 12 inversions, 2 translocations, and 2 deletions. Penelope and Ulysses were associated with the breakpoints of over half of these new rearrangements. Many rearrangement breakpoints also coincide with the chromosomal locations of Penelope and Ulysses insertions in the parental strains and with breakpoints of inversions previously established for other species of the group. Analysis of homologous sequences from D. virilis and Drosophila lummei indicated that Penelope insertion sites were closely, but not identically, located at the nucleotide sequence level. Overall, these results indicate that Penelope and Ulysses insert in a limited number of genomic locations and are consistent with the possibility that these elements play an important role in the evolution of the virilis species group.
Besides inducing many types of small mutations, such as insertions and deletions, transposable elements (TEs) are well known to promote the formation of inversions and other large and small chromosomal rearrangements (e.g., ref. 1). A growing body of evidence suggests that TEs mediate genome restructuring in natural populations of a wide variety of species. For example, in hominids, a Y chromosome inversion was mediated by recombination between LINE-1 elements before the radiation of extant human populations (2), and TEs appear to have played a role in mediating some of the major restructuring of the human sex chromosomes that has taken place during the last 300 million years (3). Also, the five families of Ty retrotransposons have been important in restructuring the Saccharomyces cerevisiae genome (4). In wild populations of Drosophila melanogaster, the hobo element has been implicated in the origin of endemic inversions (5) and in Drosophila buzzatii, the breakpoints (BPs) of a cosmopolitan inversion contain large insertions corresponding to a TE (6). The frequency and relative importance of TE-induced rearrangements in natural populations have, however, been difficult to establish in any satisfactory quantitative way. One likely reason is that instability and rapid divergence of TEs make their identification at rearrangement BPs increasingly difficult with the passage of time (7).
In contrast to the rich karyotypic variation found in most of the 12 species of the D. virilis species group, one member, D. virilis, is exceptional in having a monomorphic, relatively primitive, karyotype (8). No spontaneous inversions have previously been reported in geographical strains of D. virilis, despite extensive searches (9–12).
Contrary to the normal mode of independent activation, at least five structurally different families of TEs are simultaneously activated by interstrain hybridization (hybrid dysgenesis) in D. virilis. These families include Ulysses, Helena, Paris, Telemac, and Penelope (13–15). The Penelope family of elements has been identified as playing a critical role in D. virilis hybrid dysgenesis (15, 16). This family is also exceptional in being absent from a number of D. virilis strains but is present and active in others. This patchy distribution has been explained by a recent Penelope invasion of the species (17).
Full-sized and at least potentially functional copies of Penelope and Ulysses were previously found in most species of the virilis group (18). Exceptionally, the Penelope element was restricted to the chromocenter in Drosophila kanekoi, Drosophila lummei, and some strains of D. virilis (15), suggesting the presence of only inactive elements. The chromosomal distributions of Penelope and Ulysses in other species of the virilis group showed highly significant deviations from independence (18). Surprisingly, a statistically significant coincidence was also observed earlier between the chromosomal subsections in which these elements were inserted and the subsections that contained inversion BPs previously established in species of the virilis group (18). Both insertion site preference and selection might have contributed to these nonrandom distribution patterns.
We here report the occurrence of multiple chromosomal rearrangements in the progeny of Penelope-activated hybrid dysgenic crosses in D. virilis. There is strong statistical support for nonrandom distribution of the insertion sites of Penelope and Ulysses throughout the D. virilis genome. These insertion sites are coincident both with hot spots for BPs of new hybrid dysgenesis-induced rearrangements and BPs of established inversions in other species of the virilis group.
Materials and Methods
Drosophila Strains.
D. virilis strains 1051.51 (Chile), 1051.49 (Argentina), 1051.48 (Mexico), 1051.47 (China), 1051.8 (Truckee, CA), and 1051.9 (Sendai, Japan) were obtained from the Drosophila Species Center, Bowling Green, OH.
D. virilis strains 2003 (Uman, Ukraine); 2005 (Magarach, Crimea); 9 (Batumi, Georgia); 160 (b, gp, cd, pe, gl); 110 (tb, gp; st); 140 (va, eb); and 142 (st, es) were obtained from the Stock Center of the Institute of Developmental Biology, Moscow.
Flies stocks were maintained on standard medium at 25°C.
Cytological Analysis.
Larvae were grown at 18°C in cultures supplemented with live yeast solution 2 days before dissection. Salivary glands from third instar larvae were dissected in 45% acetic acid and squashed according to procedures developed by Lim (19). Procedures and DNA probes for in situ hybridization studies were as described in ref. 18.
DNA Manipulations.
Genomic libraries from D. virilis strain 160 and D. lummei strain 201 (a strain caught in the Moscow region in 1970) were prepared by partial Sau3A digestion with subsequent ligation into the BamHI site of Lambda Dash phage arms (Stratagene). A Sequenase I kit (Stratagene) was used to Sequence Penelope-containing clones.
Statistical Methods.
For the χ2 test for goodness of fit of observed TE distributions with those expected from a Poisson distribution, the rare classes with five and six elements were pooled. We used the coefficient of dispersion, defined as the ratio of the variance and the mean of a Poisson distribution (e.g., ref. 20). The Poisson parameter, Ëobs, is computed as the ratio of the observed number of elements to that of chromosomal subsections. For a positive Poisson distribution (truncated by omission of the zero class), we estimated the expected Poisson distribution parameter, Ëexp, by the method described in ref. 21, equations 4.79 and 4.82. We then estimated the effective distribution coefficient, de = Ëobs/Ëexp, and Se= Sde, where S is the total number of sites (L.A.Z., unpublished work). The parameters de and Se are interpreted as, respectively, the fraction and the effective absolute number of subsections that were potentially available for insertion (S = 1,359 for chromosomal subsections of D. virilis). The coincidence of mobile element insertion sites and chromosomal BPs was tested by the statistical method used in ref. 18.
Results
The Chromosomal Distribution of Penelope and Ulysses in D. virilis.
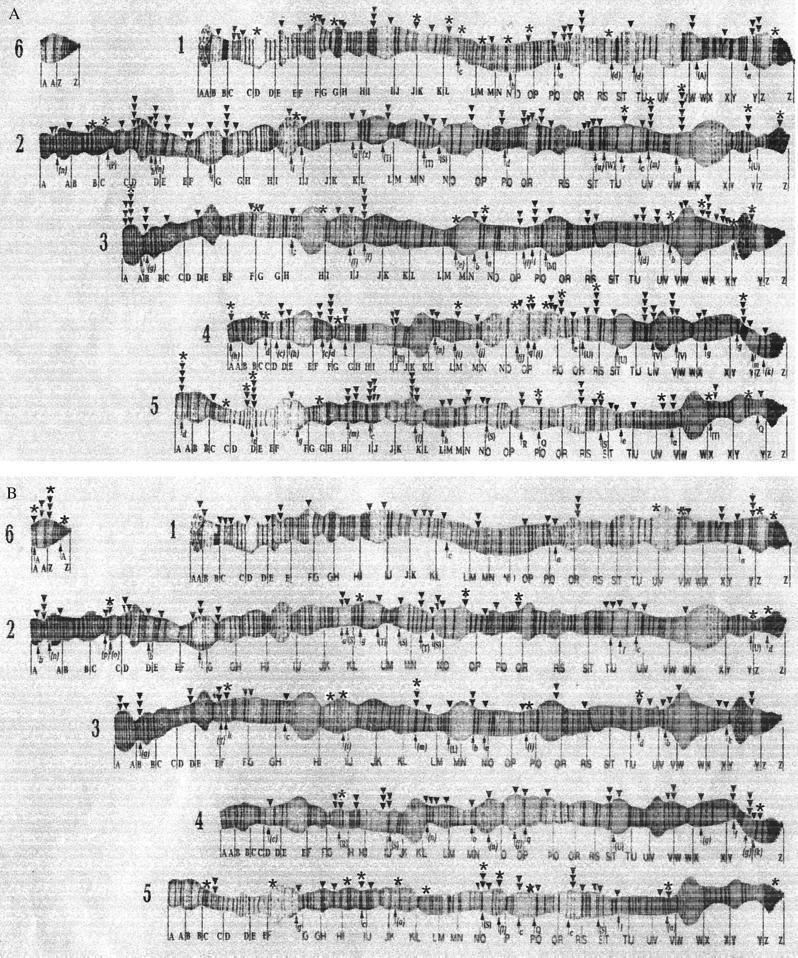
We examined the chromosomal locations of Penelope and Ulysses in 12 D. virilis strains previously shown to carry these elements (17). Each of the 181 Penelope and 144 Ulysses euchromatic insertions identified was localized to one of the 1,359 chromosomal subsections of the D. virilis genome (11) (Fig. 1 A and B and Table 1).
Figure 1.
The distributions of Penelope (A) and Ulysses (B) within the D. virilis genome as revealed by in situ hybridization. The numbers at the left of the figures identify the six chromosomes. The triangles above the chromosomes represent the insertion sites of Penelope or Ulysses in the 12 D. virilis strains studied. The stars above the chromosomes represent the subset of these insertion sites identified in hybrid dysgenesis reference strains 160 and 9 (a total of 47 Penelope sites in strain 160 and a total of 32 Ulysses sites in strains 160 and 9 combined). The arrows below the chromosomes indicate the positions of inversion BPs previously identified in other species of the virilis group. Individual inversions are identified by lowercase letters according to the classification system of refs. 8 and 28. Unique inversions that are polymorphic in individual species of the virilis group are shown in parentheses.
Table 1.
Distribution of insertion sites, and total numbers of elements inserted (in parentheses), for Penelope and Ulysses, in the six chromosomes of D. virilis
| Chromosome No. | Number of insertions at a single
site
|
Total | ||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 5 | 6 | ||
| Penelope | ||||||||
| 1 | 211 (0) | 23 (23) | 9 (18) | 2 (6) | 0 | 0 | 0 | 34 (47) |
| 2 | 271 (0) | 28 (28) | 9 (18) | 2 (6) | 3 (12) | 0 | 1 (6) | 43 (70) |
| 3 | 241 (0) | 20 (20) | 8 (16) | 4 (12) | 1 (4) | 1 (5) | 0 | 34 (57) |
| 4 | 208 (0) | 20 (20) | 10 (20) | 2 (6) | 4 (16) | 0 | 0 | 36 (62) |
| 5 | 227 (0) | 17 (17) | 10 (20) | 4 (12) | 1 (4) | 2 (10) | 0 | 34 (63) |
| 6 | 20 (0) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 1178 (0) | 108 (108) | 46 (92) | 14 (42) | 9 (36) | 3 (15) | 1 (6) | 181 (299) |
| Ulysses | ||||||||
| 1 | 217 (0) | 23 (23) | 3 (6) | 2 (6) | 0 | 0 | 0 | 28 (35) |
| 2 | 281 (0) | 23 (23) | 9 (18) | 1 (3) | 0 | 0 | 0 | 33 (44) |
| 3 | 246 (0) | 19 (19) | 8 (16) | 2 (6) | 0 | 0 | 0 | 29 (41) |
| 4 | 220 (0) | 14 (14) | 8 (16) | 2 (6) | 0 | 0 | 0 | 24 (36) |
| 5 | 235 (0) | 19 (19) | 5 (10) | 2 (6) | 0 | 0 | 0 | 26 (35) |
| 6 | 16 (0) | 1 (1) | 2 (4) | 0 | 1 (4) | 0 | 0 | 4 (9) |
| Total | 1215 (0) | 99 (99) | 35 (70) | 9 (27) | 1 (4) | 0 | 0 | 144 (200) |
We tested whether the distribution of subsections with 1, 2, 3, etc., Penelope or Ulysses insertions varied among chromosomes. The heterogeneity among chromosomes was found not to be statistically significant (P = 0.64 and 0.44 for Penelope and Ulysses, respectively) by using the permutation method (10,000 runs) and Fisher's exact test. Analysis of the pooled chromosomal distributions was, therefore, justified and revealed large deviations from a Poisson distribution. A χ2 test for goodness of fit gave values of 1,761 and 853 with df = 4 for Penelope and Ulysses, respectively. The observed number of subsections with no elements (the zero class in Table 1) exceeded that expected from a Poisson distribution. The resulting values for the coefficient of dispersion were much larger than 1 (Table 2), and values of the Poisson parameter were smaller than those expected from a positive Poisson distribution.
Table 2.
Distribution parameters of Penelope and Ulysses calculated from the data in Table 1
| Family | Coefficient of dispersion | Poisson
parameter
|
Effective distribution, de, % | Effective number of sites, Se | |
|---|---|---|---|---|---|
| Ëobs | Ëexp | ||||
| Penelope | 2.03 | 0.220 | 1.10 ± 0.20 | 19.9 | 271 |
| Ulysses | 1.53 | 0.147 | 0.70 ± 0.21 | 21.1 | 287 |
In contrast, the data of Table 1 fitted a positive Poisson distribution almost perfectly: The corresponding χ2 values are 10.6 and 0.45 (P = 0.014 and 0.93, for Penelope and Ulysses, respectively). However, the combined test (χ2 = 11.1 with df = 6) was insignificant (P = 0.09). Therefore, an excess in the zero class causes the deviation of the observed distribution of mobile elements. The values of the effective distribution coefficient, de, are fairly similar in both families of mobile elements (Table 2). These statistical results suggest that only about one-fifth of the chromosomal subsections are actually available for insertion by these elements. The remaining subsections appear effectively unavailable for insertion. The similarity of the distributions of Penelope (or Ulysses) among different chromosomes, as well as that between the distributions of the two families themselves, suggests that a common mechanism underlies this phenomenon.
Comparison of Fig. 1 A and B suggests several “hot spots” for Penelope and Ulysses insertion in all of the large chromosomes. We failed to find any correlations between these hot spots and known cytogenetic landmarks such as weak points or 5S genes in D. virilis chromosomes (12). However, when the locations of Penelope and Ulysses insertion sites were compared with the BPs of inversions previously described in other species of the virilis group (8, 11), statistically significant associations were observed (Table 3). The chromosomal subsections of 80 Penelope, and 57 Ulysses insertion sites coincide with inversion BPs previously described. Furthermore, Penelope and Ulysses had 82 insertion sites in common in the D. virilis strains examined. Similar results were previously observed for the distributions of Penelope and Ulysses in other species of the virilis group (18).
Table 3.
Nonrandom occurrence of mobile elements Penelope (Pen) and Ulysses (Uly) at inversion BP in the genome of D. virilis
| Comparison type | Cobs* | Cexp† | Significance, P | Coincidence, P |
|---|---|---|---|---|
| Pen vs. BP | 80 | 18.78 | ≪0.001 | 0.50 |
| Uly vs. BP | 57 | 14.94 | ≪0.001 | 0.33 |
| Pen vs. Uly | 82 | 19.18 | ≪0.001 | 0.50 |
| Pen and Uly vs. BP | 56 | 8.51 | ≪0.001 | 0.65 |
Cobs is the observed number of common subsections.
Cexp is the expected number of common subsections, given a total of 1,359 subsections in the D. virilis chromosome map. The total number of inversion BPs compared in other species of the virilis group was 141, and the numbers of Penelope and Ulysses insertions sites observed in D. virilis were 181 and 144, respectively.
Chromosomal Rearrangements in the Progeny of Dysgenic Crosses.
We made dysgenic crosses between females of D. virilis strain 9, which had no active Penelope copies, and males of strain 160, which carried multiple Penelope copies (18). F1 hybrid females and males were mated inter se, and progeny with obvious morphological abnormalities, such as misshapen wings and abdomens, were selected in the F2 and F3 generations. When possible, these individuals were used to propagate lines. Each of these individuals was crossed with five males or females of strain 9, and the salivary glands of at least 10 larval progeny per cross were checked for the presence of chromosomal rearrangements. From the progeny of these phenotypically abnormal flies, we isolated 16 new chromosomal rearrangements, including 12 inversions, 2 translocations, and 2 deletions (Table 4; Fig. 2). Although our method did not allow estimation of the frequency of newly occurring rearrangements, abnormal phenotypes served as useful indicators of transpositional activity and increased the probability of rearrangement detection.
Table 4.
Characterization of chromosomal rearrangements induced in the progeny of D. virilis dysgenic crosses
| Type of rearrangement | Location of distal BP | Location of proximal BP | Coincidence of new
rearrangement BPs with
|
||
|---|---|---|---|---|---|
| Penelope in strain 160 | Ulysses in strains 160 or 9 | Inversion BPs in other species* | |||
| Inversions | XDk | XIe | |||
| XIa | XTf | XTf (N,T,A) | |||
| XRc | XXa | ||||
| 2Bj | 2Gd | 2Bj | |||
| 2Ij | 2Za | 2Ij, 2Za | 2Ij (Mo, La, Ezo, Lu); 2Za (Mo, La, Li, Ezo, Ka) | ||
| 2Si/Ta | 2Zc | 2Si/Ta (Ezo, Lu); 2Zc (Ka) | |||
| 3FeS | 3Z | 3Fe, 3Z | 3Fe (Li); 3Z (N, A, Mo, La) | ||
| 3Hk | 3Wf* | 3Hk, 3Wf | 3Hk (Mo, La) | ||
| 3Nhi | 3Ri | 3Nhi (A, N) | |||
| 4Mhi | 4Sa | 4Mhi, 4Sa | |||
| 4Od | 4Sa | 4Sa | 4Od (Mo) | ||
| 5Iab | 5Pn | 5Iab, 5Pn | 5Pn (Ezo, Ka) | ||
| Translocations | XHki | 5Ma | |||
| 2Eh | 4Ga | 2Eh, 4Ga | 4Ga (Li) | ||
| Deletions | 5Ab | 5Ci | (5Ci) | ||
| 2Eh | 2Fa | ||||
| Total observed | 16 | 16 | 10 | 4 | 12 |
| Total expected | 1.11 | 0.75 | 3.29 | ||
| Significance, P† | 3.3 × 10−8 | 5.7 × 10−3 | 3.6 × 10−5 | ||
| Coefficient of coincidence† | 0.29 | 0.10 | 0.30 | ||
141 BPs were observed in 1,359 genomic subsections in 10 species. N, Drosophila novamexicana; T, Drosophila texana; A, Drosophila americana; Mo, Drosophila montana; La, Drosophila lacicola; Ezo, Drosophila ezoana; Lu, Drosophila lummei; Li, Drosophila littoralis; Ka, Drosophila kanekoi.
Obtained by following the statistical procedure described by ref. 18.
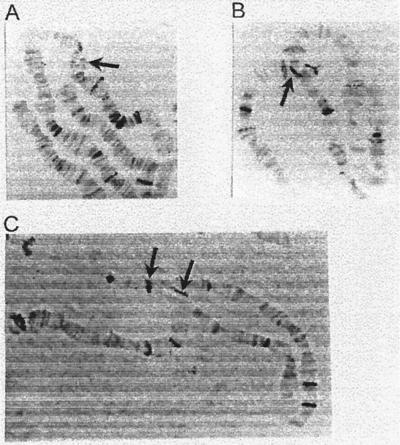
Figure 2.
Examples of chromosomal rearrangements isolated from the progeny of dysgenic crosses. (A) Deletion of the distal end of chromosome 5. The arrow indicates the deletion BP. The Penelope element present in the 5Ci subsection of strain 160 was probably lost in the process of deletion formation. (B) Inversion in chromosome 2. An arrow indicates the inversion BP located in band 2Ij, in which a Penelope element is also inserted. (C) Inversion in chromosome 3. Penelope elements located in both inversion BPs (3Hk and 3Wf) are indicated by arrows.
In situ hybridization studies revealed that a significant fraction of the BPs of newly induced rearrangements were associated with Penelope or Ulysses (Table 4). Of a total of 32 BPs, 16 proximal and 16 distal, 10 were associated with Penelope and 4 with Ulysses. The expected numbers of TE/BP associations are 1.11 for Penelope and 0.75 for Ulysses, both showing highly significant deviations from those observed (P = 3.3 × 10−8 and 5.7 × 10−3, respectively). These 14 associations were divided equally between distal and proximal BPs.
The locations of the BPs of the newly induced rearrangements were also compared with the locations of Penelope and Ulysses in the 2 parental strains, 160 and 9, in which a total of 47 Penelope and 32 Ulysses insertions were located (Fig. 1 A and B, respectively). Eleven rearrangement BPs coincided with Penelope sites present in parental strain 160. Of these, 10 retained their Penelope elements and 1 (BP 5Ci) no longer retained its element (Table 4, column 4). Four inversion BPs coincided with Ulysses sites present in either parental strain (Table 4, column 5), and all four BPs retained their Ulysses elements.
Finally we compared the location of the BPs of newly induced rearrangements with the locations of inversion BPs previously observed in other species of the virilis group (8, 11). A total of 12 coincidences between the subsections of new and old BPs were observed (Table 4, column 6), compared with an expected number of 3.29. The coincidence coefficients were highly significant in every case (Table 4).
Structure of Penelope Insertion Sites in D. virilis and D. lummei.
The frequent association between the insertion sites of Penelope and Ulysses and between these sites and inversion BPs described in species of the virilis group, including D. virilis, suggests possible hot spots for TE integration. To investigate how precisely the insertion sites are related, a selected homologous chromosomal region of D. virilis and D. lummei was cloned from each species. This region included an intron of the eyeless gene, a cluster of Pdv elements (22), and a Penelope element insertion. In situ hybridization, by using the flanking regions of both genomic clones as probes, enabled us to localize the region to the 49F subsection of chromosome 4.
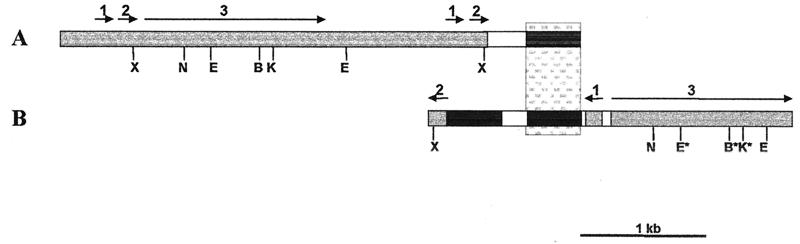
Fig. 3 illustrates the structure of this homologous region isolated from genomic libraries of D. virilis strain 160 and D. lummei strain 203. Both sequences contain Penelope elements as well as portions of the same genomic region. However, the two Penelope elements are inserted in opposite orientation with respect to the flanking sequence and, in the case of D. lummei, only discontinuous fragments of a Penelope element are present. Although the two Penelope elements apparently inserted in the same general chromosomal region in the two species, they did not do so at identical sites at the nucleotide level.
Figure 3.
Schematic representation of structures of Penelope elements isolated from genomic libraries. (A) A Penelope-containing genomic clone of D. virilis. (B) A Penelope-containing genomic clone of D. lummei. Gray boxes indicate Penelope sequences; black boxes indicate sequences of the eyeless gene. The stippled box displays homologous regions of the eyeless gene in the two species. White (unshaded) boxes represent genomic sequences of unknown origin. The symbols X, N, E, B, and K represent the restriction sites XhoI, NdeI, EcoRI, BamHI, and KpnI, respectively. Asterisks indicate restriction sites that are damaged in D. lummei. Arrows above the boxes indicate the sizes and positions of fragments within a full-length Penelope element.
Alignment of the D. lummei Penelope element with a consensus copy of D. virilis (p6) indicated many base substitutions, deletions, and insertions in the D. lummei sequence (data not shown). The overall nucleotide similarity between the two Penelope elements was 82%, ignoring gaps. The D. lummei element appears to be a highly diverged defective one that is unable to transpose.
Discussion
It is well known that most mobile elements exhibit some level of insertion site preference (23). However, the degree of target site specificity can vary widely, depending on a number of factors. In this paper, we have shown highly significant deviations from randomness of the distributions of Penelope and Ulysses insertion sites in the D. virilis genome. An excess of subsections in the zero (unoccupied) class accounts for the deviations observed. Statistical analysis indicates that only about one-fifth of the chromosomal subsections is effectively available for occupation by these elements. Statistically significant coincidence coefficients were also observed between the locations of Penelope and Ulysses insertion sites in D. virilis and BPs of chromosomal rearrangements in other species of the group. It seems likely that a common mechanism underlies these nonrandom associations. Because Penelope plays a key role in hybrid dysgenesis in D. virilis (15), the activity of a Penelope-encoded endonuclease seems a likely candidate for determining this insertional specificity.
Penelope is a member of the non-long-terminal repeat (LTR) retroelements (15). Sequence profile analysis suggests that the Penelope polyprotein is an active endonuclease that could function as an integrase. It is related to intron-encoded endonucleases and a bacterial repair endonuclease (M.B.E., unpublished results). This type of non-LTR retroposon transposes by cleavage of the DNA to produce a priming site for reverse transcriptase (23). In some retroposons, such as the R1 and R2 families in insects, the high target-site specificity reflects the high specificity of the element-encoded endonuclease (20). Although the limited data suggest that Penelope insertion site preference is considerably less specific than that of the R1 and R2 retroposons, it appears high enough to produce a strong pattern of nonrandomness. The activity of a Penelope-encoded nuclease may also be responsible for breaks in ectopically paired elements that result in rearrangements through the subsequent random rejoining of fragments. This might account for the high coincidence of BPs of newly induced rearrangements with Penelope and Ulysses insertion sites present in the parental strains.
Genetic and molecular studies indicate that all three systems of hybrid dysgenesis (24) in D. melanogaster (P-M, I-R and hobo) are accompanied by multiple rearrangements because of recombination between insertion sequences of a single TE family (25–27). However, in contrast to the D. melanogaster systems, induced rearrangements in D. virilis may be associated with at least two unrelated families of transposons at their BPs. It should be noted that we did not attempt to check whether other TE families that are activated in D. virilis hybrid dysgenesis are associated with the BPs of those rearrangements not associated with Penelope or Ulysses. Such a possibility is currently being investigated (M.B.E., unpublished results).
Results of the present D. virilis study show that the chromosomal distributions of Penelope and Ulysses exhibit significant correlations with the subsections of inversion BPs known for other species of the group. Similar correlations with inversion BPs were reported earlier with respect to the chromosomal distributions of Penelope and Ulysses in other species of the virilis group (18). Because these elements are old components of their genomes, species of the virilis group seem likely to share a common pattern of site specificity for Penelope and Ulysses insertion.
A possible scenario to explain the observed results is as follows. A Penelope invasion appears to have taken place at least once early in the history of the virilis species group (18). The remnants of that invasion appear to be represented by inactive diverged Penelope copies in contemporary species of the virilis group (including D. virilis), often located in the centric heterochromatin (ref. 18; M.B.E., unpublished results). This earlier Penelope invasion is postulated to have activated multiple elements during hybrid dysgenesis and to have induced numerous gross chromosomal rearrangements that led to, or accompanied, speciation in the group. Our recent finding of ancient highly diverged copies of Penelope in some strains of D. virilis, and in a closely related species, D. lummei, strongly supports the sequence of events proposed (M.B.E., unpublished results).
The absence of chromosomal rearrangements in contemporary natural populations of D. virilis might be explained by the early divergence of the lineage that gave rise to this species from the rest of the species group. Indeed, it has been postulated that D. virilis evolved in southeast Asia, isolated from the remaining species of the group until relatively recently (8). According to our scenario, this divergence would be required to have occurred after the initial Penelope invasion but before the massive chromosomal restructuring that occurred in the other species of the group (8). The documented contemporary invasion of D. virilis (17) has only recently started to provide the potential for Penelope-induced rearrangements in natural populations of this species. It will be interesting to see whether such rearrangements start to appear in natural populations, as they have done in the laboratory.
Acknowledgments
We thank William Heed, Andrew Holyoake, and John McDonald for critically reading the manuscript. This work was supported by Russian Ministry of Sciences Grant no. 970450066 for fundamental science (to M.B.E.), by Russian Foundation of Basic Research Grant no. 98–04-49292, by National Institutes of Health Grant 1R03 TW00491–01 (to L.A.Z.), by National Science Foundation (NSF) Grant no. DEB 9815754, and by an international supplement to NSF Grant no. DEB-9119349A (to M.G.K.).
Abbreviations
- TE
transposable element
- BP
breakpoint
Footnotes
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.210386297.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.210386297
References
- 1.Lim J K, Simmons M J. BioEssays. 1994;16:269–275. doi: 10.1002/bies.950160410. [DOI] [PubMed] [Google Scholar]
- 2.Schwartz A, Chan D C, Brown L G, Alagappan R, Pettay D, Disteche C, McGillivray B, de la Chapelle A, Page D C. Hum Mol Genet. 1998;7:1–11. doi: 10.1093/hmg/7.1.1. [DOI] [PubMed] [Google Scholar]
- 3.Lahn B T, Page D C. Science. 1999;286:964–967. doi: 10.1126/science.286.5441.964. [DOI] [PubMed] [Google Scholar]
- 4.Kim J M, Vanguri S, Boeke J D, Gabriel A, Voytas D F. Genome Res. 1998;8:464–478. doi: 10.1101/gr.8.5.464. [DOI] [PubMed] [Google Scholar]
- 5.Lyttle T W, Haymer D S. Genetica. 1992;86:113–126. doi: 10.1007/BF00133715. [DOI] [PubMed] [Google Scholar]
- 6.Caceres M, Ranz J M, Barbadilla A, Long M, Ruiz A. Science. 1999;285:415–418. doi: 10.1126/science.285.5426.415. [DOI] [PubMed] [Google Scholar]
- 7.Mathiopoulos K D, della Torre A, Predazzi V, Petrarca V, Coluzzi M. Proc Natl Acad Sci USA. 1998;95:12444–12449. doi: 10.1073/pnas.95.21.12444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Patterson J T, Stone W S. Evolution in the Genus Drosophila. New York: Macmillan; 1952. pp. 437–501. [Google Scholar]
- 9.Patterson J T, Stone W, Griffin A B. Univ Texas Pub. 1940;4032:218–250. [Google Scholar]
- 10.Fujii S. Cytologia. 1942;12:435–459. [Google Scholar]
- 11.Hsu T C. Univ Texas Pub. 1952;5204:35–72. [Google Scholar]
- 12.Gubenko I S, Evgen'ev M B. Genetica. 1984;65:127–139. [Google Scholar]
- 13.Lozovskaya E R, Scheinker V S, Evgen'ev M B. Genetics. 1990;126:619–623. doi: 10.1093/genetics/126.3.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Petrov D A, Schutzman J L, Hartl D L, Lozovskaya E R. Proc Natl Acad Sci USA. 1995;92:8050–8054. doi: 10.1073/pnas.92.17.8050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evgen'ev M B, Zelentsova H, Shostak N, Kozitsina M, Barsky V, Lankenau D-H, Corces V G. Proc Natl Acad Sci USA. 1997;94:196–201. doi: 10.1073/pnas.94.1.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vieira J, Vieira C P, Hartl D L, Lozovskaya E R. Genet Res. 1998;71:109–117. doi: 10.1017/s001667239800322x. [DOI] [PubMed] [Google Scholar]
- 17.Evgen'ev, M., Zelentsova, H., Mnjoian, L., Poluectova, H. & Kidwell, M. G. (2000) Chromosoma, in press. [DOI] [PubMed]
- 18.Zelentsova H, Poluectova H, Mnjoian L, Lyozin G, Veleikodvorskaja V, Zhivotovsky L, Kidwell M G, Evgen'ev M B. Chromosoma. 1999;108:443–456. doi: 10.1007/s004120050396. [DOI] [PubMed] [Google Scholar]
- 19.Lim J K. Drosophila Inf Serv. 1993;72:73–77. [Google Scholar]
- 20.Sokal R R, Rohlf F J. Biometry: The Principles and Practice of Statistics in Biological Research. 2nd Ed. San Francisco, CA.: Freeman; 1981. p. 87. [Google Scholar]
- 21.Johnson N L, Kotz S, Kemp A W. Univariate Discrete Distributions. New York: Wiley; 1992. p. 182. [Google Scholar]
- 22.Zelentsova E S, Vashakidze R P, Kraev A S, Evgen'ev M B. Chromosoma. 1986;93:469–476. [Google Scholar]
- 23.Craig N L. Annu Rev Biochem. 1997;66:437–474. doi: 10.1146/annurev.biochem.66.1.437. [DOI] [PubMed] [Google Scholar]
- 24.Kidwell M G, Kidwell J F, Sved J A. Genetics. 1977;36:813–833. doi: 10.1093/genetics/86.4.813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Engels W R, Preston C R. Genetics. 1984;107:657–678. doi: 10.1093/genetics/107.4.657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Busseau I A, Pelisson A, Bucheton A. Mol Gen Genet. 1989;218:222–228. doi: 10.1007/BF00331272. [DOI] [PubMed] [Google Scholar]
- 27.Lim J K. Proc Natl Acad Sci USA. 1988;85:9153–9157. doi: 10.1073/pnas.85.23.9153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Poluektova E V, Zhdanov M I, Grigor'eva G A, Mitrofanov V G. Genetika (Moscow) 1994;30:72–76. [PubMed] [Google Scholar]