Abstract
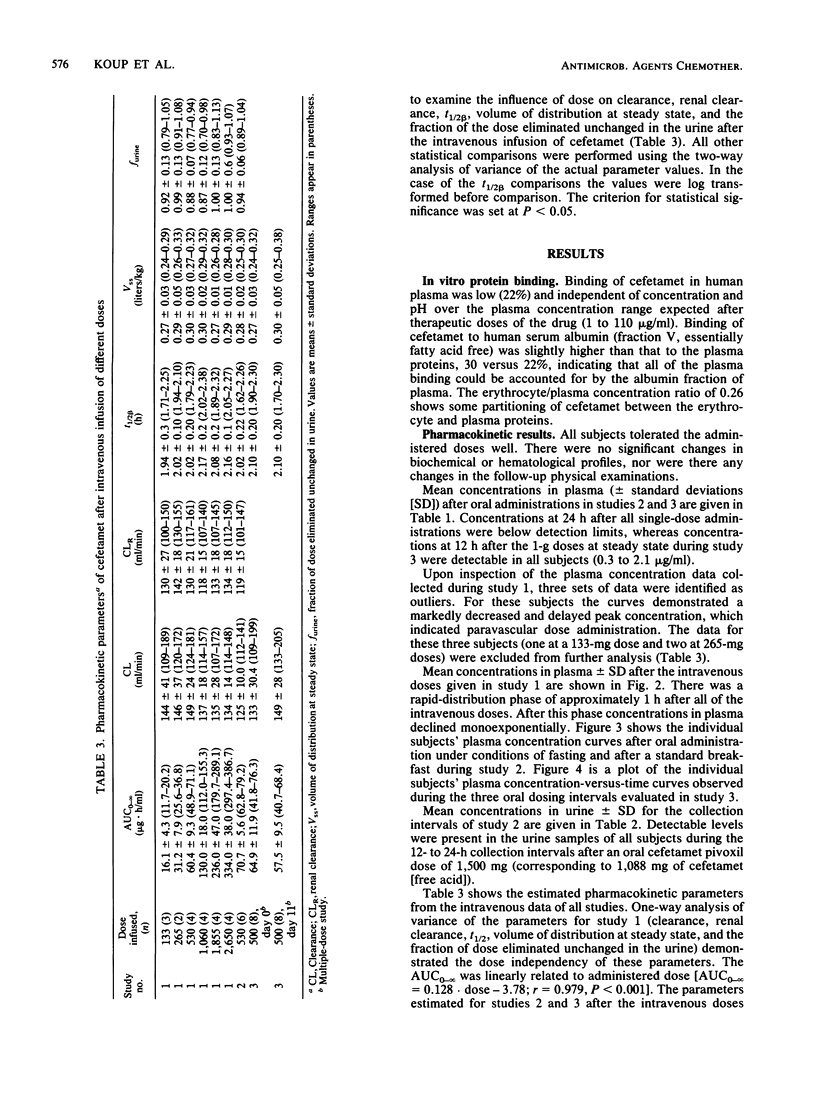
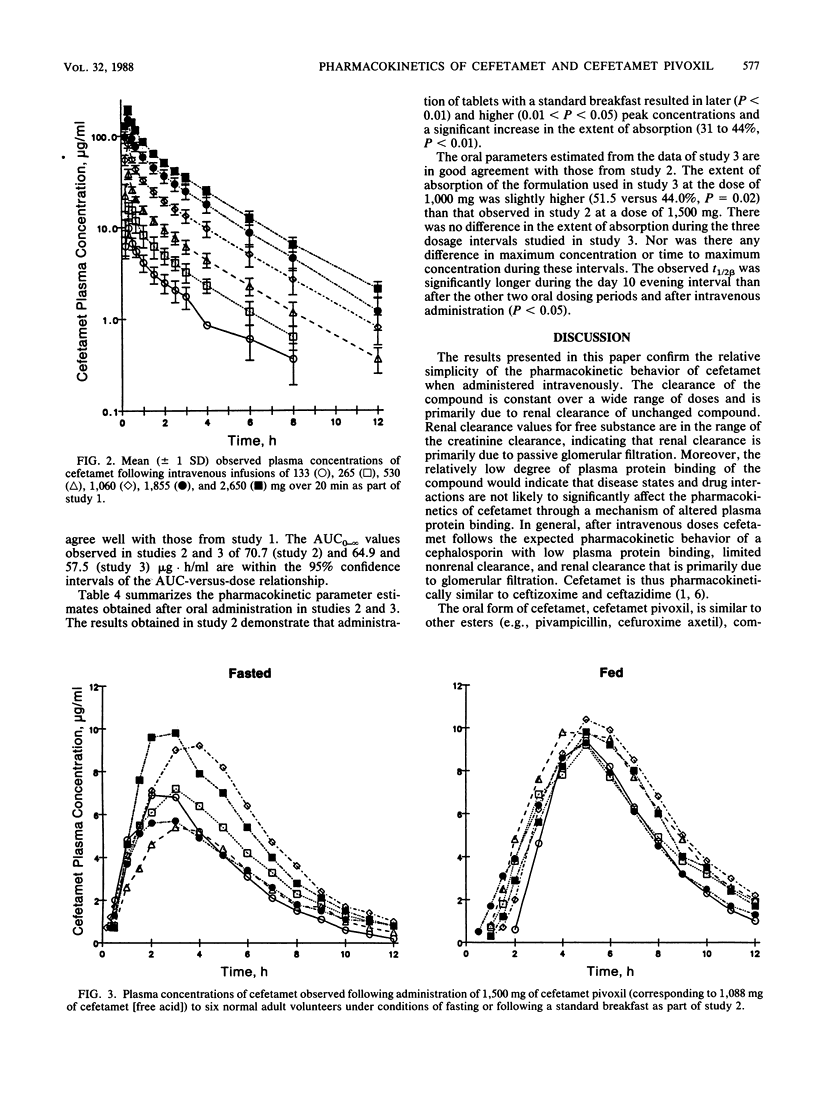
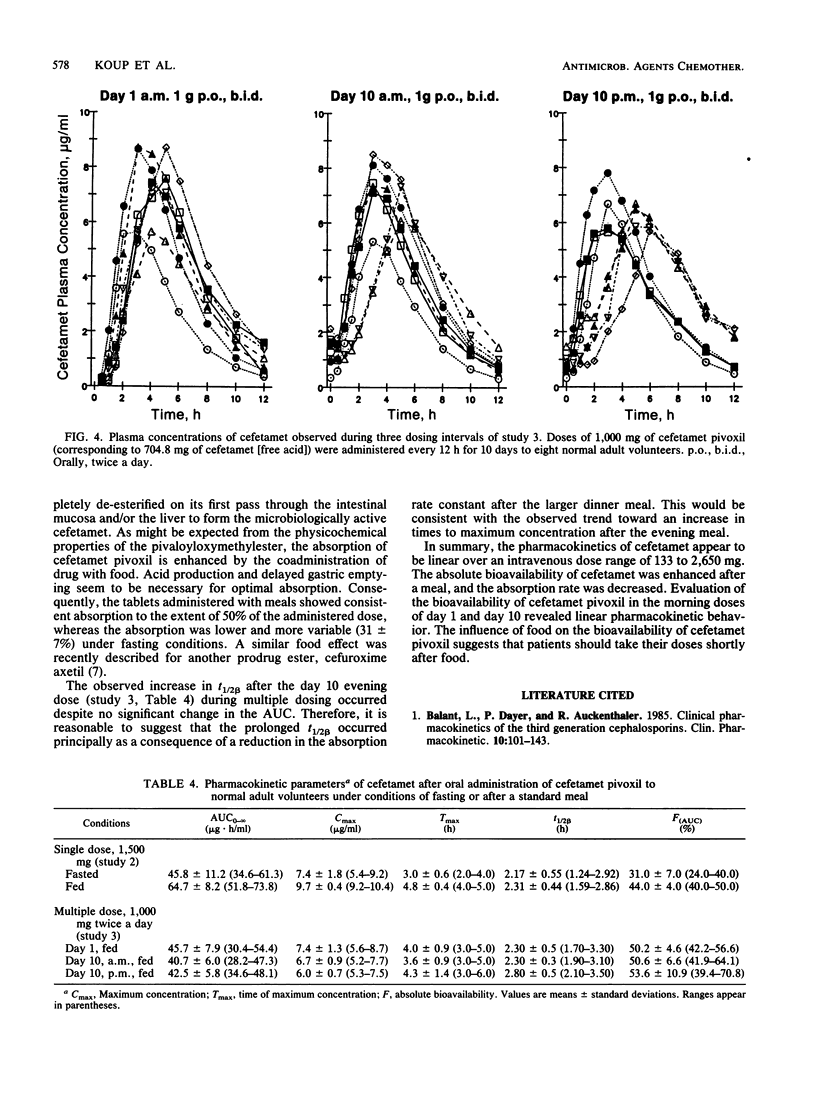
This report summarizes the results of three pharmacokinetic studies of cefetamet and cefetamet pivoxil conducted in normal adult male volunteers. In the first study the pharmacokinetics of cefetamet were evaluated after intravenous infusion of doses ranging from 133 to 2,650 mg. Over this dose range, the pharmacokinetics were linear. A dose-proportional increase in the area under the curve from zero to infinity was observed, whereas total clearance (140.3 +/- 23.6 ml/min), renal clearance (130.3 +/- 18.2 ml/min), volume of distribution at steady state (0.288 +/- 0.023 liter/kg), fraction excreted unchanged in the urine (94 +/- 11%), and elimination half-life (2.07 +/- 0.18 h) were independent of dose. In a second study the absolute bioavailability of single 1,500-mg doses of a tablet formulation of the pivaloyloxymethylester of cefetamet was evaluated under conditions of fasting and after a standard breakfast. Administration with food increased the extent of absorption (from 31 +/- 7 to 44 +/- 4%) while decreasing the rate of absorption (time to maximum concentration of drug in plasma increased from 3.0 +/- 0.6 to 4.8 +/- 0.4 h). The third study consisted of multiple oral administration of 1,000 mg of a similar oral tablet formulation twice daily for 10 days. This regimen was preceded and followed by intravenous administration of a 500-mg bolus dose of cefetamet. Oral doses were administered with breakfast and dinner. The absolute bioavailability of the tablet formulation was assessed after the first dose and after both the morning and the evening doses on day 10 of oral therapy. The compound was consistently absorbed to the extent of approximately 50% with no significant differences observed between the morning and evening doses on day 10.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balant L., Dayer P., Auckenthaler R. Clinical pharmacokinetics of the third generation cephalosporins. Clin Pharmacokinet. 1985 Mar-Apr;10(2):101–143. doi: 10.2165/00003088-198510020-00001. [DOI] [PubMed] [Google Scholar]
- Bauer L. A., Gibaldi M. Computation of model-independent pharmacokinetic parameters during multiple dosing. J Pharm Sci. 1983 Aug;72(8):978–979. doi: 10.1002/jps.2600720843. [DOI] [PubMed] [Google Scholar]
- Benet L. Z., Galeazzi R. L. Noncompartmental determination of the steady-state volume of distribution. J Pharm Sci. 1979 Aug;68(8):1071–1074. doi: 10.1002/jps.2600680845. [DOI] [PubMed] [Google Scholar]
- Cockcroft D. W., Gault M. H. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31–41. doi: 10.1159/000180580. [DOI] [PubMed] [Google Scholar]
- Williams P. E., Harding S. M. The absolute bioavailability of oral cefuroxime axetil in male and female volunteers after fasting and after food. J Antimicrob Chemother. 1984 Feb;13(2):191–196. doi: 10.1093/jac/13.2.191. [DOI] [PubMed] [Google Scholar]
- Wise R., Andrews J. M., Piddock L. J. In vitro activity of Ro 15-8074 and Ro 19-5247, two orally administered cephalosporin metabolites. Antimicrob Agents Chemother. 1986 Jun;29(6):1067–1072. doi: 10.1128/aac.29.6.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]


