Abstract
AIMS—To evaluate the scanning retinal thickness analyser (RTA), a novel non-invasive imaging instrument, in diagnosing and quantitatively characterising diabetic macular oedema, and to investigate the relation between central macula thickness measured by RTA and other clinical examinations. METHODS—Central macular thickness was measured using the RTA in 40 normal subjects and 60 patients with diabetic retinopathy. The reproducibility of the retinal thickness measurements was evaluated by calculating the mean of the inter- and intrasession variations. Central macular thickness was correlated with the results of visual acuity measurements, biomicroscopy, and fluorescein angiography. RESULTS—Intra- and intersession reproducibility of the RTA in normal subjects was plus or minus 5.2% (16 µm) and plus or minus 6.1% (19 µm), respectively. The mean central macular thickness was 182 (SD 16) µm in normal subjects, 283 (116) µm in diabetic eyes without clinically significant macular oedema (CSMO), and 564 (168) µm in diabetic eyes with CSMO. Central macular thickness was significantly greater (p<0.001) in eyes with diabetic retinopathy than in normal subjects, even when macular thickening did not meet the standard for CSMO (p=0.019) measured by biomicroscopy. Although greater fluorescein leakage at the macula results in greater central macular thickness, only eyes with diffuse leakage had statistically significant macular thickening compared with normal subjects (p=0.022). Central macular thickness measured with the RTA was significantly correlated with the logarithmic converted visual acuity (r2= 0.76) in diabetic eyes. CONCLUSION—Scanning RTA, which has good reproducibility, might be useful to quantitatively detect and monitor macular thickening in diabetic retinopathy. Central macular thickness was highly correlated with logarithmic converted visual acuity in diabetic macular oedema. Keywords: scanning retinal thickness analyser; macular thickness; diabetic retinopathy; macular oedema
Full Text
The Full Text of this article is available as a PDF (214.5 KB).
Figure 1 .
(A) Fundus photograph of a normal left eye in the retinal thickness scan. Nine fixation targets are shown on the posterior fundus. The foveal fixation target is indicated by a black arrowhead, and the scanned retinal area is indicated by the square. (B) A composite of 10 optical section images in a normal left eye covering a retinal area of 2 × 2 mm2 including the fovea. The left and right arrows indicate the vitreoretinal and chorioretinal interfaces, respectively, at the area of the assumed foveal depression corresponding to the foveal target position. The thin vertical arrows indicate the chorioretinal interfaces. (C) A scan profile of the densitometric reading curve at a spot near the fovea. The distance between the peaks on both sides represent the slit image width that is proportional to the retinal thickness at this point. (D) Numeric grid of the scanned area covering 2 × 2 mm2. The square indicates the 3 × 3 points, which correspond to a 600 × 600 µm2 area, used for the central macular thickness calculation. RPE = retinal pigment epithelium; ILM = internal limiting membrane; Thk = retinal thickness in arbitrary units.
Figure 2 .
Scattergrams of central macular thickness in normal eyes plotted against the age of the subjects. The central macular thickness did not vary significantly with age (p>0.99), and it was similar for right and left eyes. The solid line represents the mean value that corresponds to 182 µm, and the two broken lines above and below the solid line indicate the range of 2 SD. The shaded area represents the normal variation of the central macular thickness measured by the scanning retinal thickness analyser.
Figure 3 .
(A) Fundus photograph obtained from the right eye of a patient with localised diabetic macular oedema. Optical sectioning was performed within the area bound by the square. The black arrowhead indicates the fixation target for the central scan, presumably corresponding to the centre of the fovea. (B) A composite of optical section images shows the presence of intraretinal cysts (arrowheads) at the macula that cause elevation of the vitreoretinal interface and loss of a normal foveal depression. The chorioretinal interface (vertical black arrows) is partly obscured because of intraretinal fluid accumulation, but is visualised in the superior and inferior retinal area (vertical white arrows).
Figure 4 .
(A) Fundus photograph obtained from the left eye of a patient with diffuse diabetic macular oedema. The square delineates the scanned region demonstrated in the next optical section image. The black arrowhead indicates the fixation target for the central scan, presumably corresponding to the centre of the fovea. (B) Multiple cross sectional scanning image shows the relative elevation of the interretinal surface with gross foveal distortion. The vitreoretinal interface at the centre of the fovea (white arrow) showed an absent foveal depression.
Figure 5 .
Box plots of the central macular thickness in the three groups determined by using the scanning retinal thickness analyser. The macular thickness is significantly greater (p=0.008) in the eyes with clinically significant macular oedema (CSMO) than in the eyes without CSMO. The retinal thicknesses in the eyes with non-CSMO were significantly (p=0.019) greater than in the normal control eyes. The boxes include 50% of the measured values (25% and 75% percentiles) and demonstrate the position of the median (horizontal line). The shaded area indicates the thickness range in which CSMO and non-CSMO diagnoses overlapped (range 320-420 µm). CSMO = clinically significant macular oedema.
Figure 6 .

Box plots of central macular thickness determined using the scanning retinal thickness analyser in four subsets grading by fluorescein angiography. The macular thickness is significantly (p=0.022) greater in eyes with diffuse fluorescein leakage than in eyes with focal fluorescein leakage. However, no significant differences in macular thickness are seen between eyes with focal fluorescein leakage and each of the other two subsets as a result of a wide range of thickness values in each subgroup. The positions of the medians (solid line within the boxes) illustrate the increasing macular thickness in eyes with increasing degrees of fluorescein leakage.
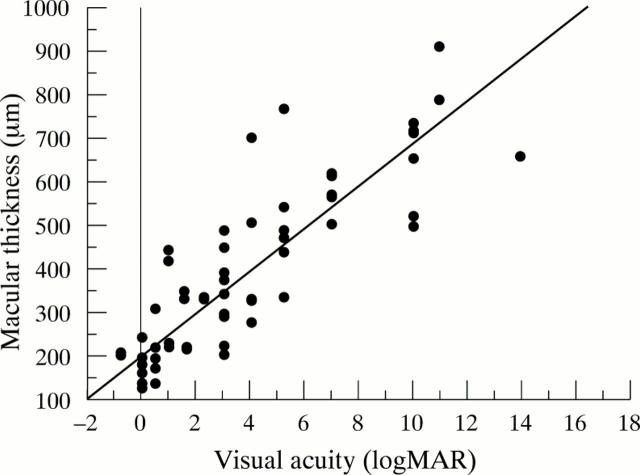
Figure 7 .
The relation between central macular thickness and visual acuity in eyes with diabetic retinopathy. The visual acuity is reported as the logarithm of the minimum angle of resolution (logMAR). Linear regression demonstrates a good correlation (n=60; r2=0.76; p<0.001) between the macular thickness and visual acuity.
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arend O., Remky A., Elsner A. E., Bertram B., Reim M., Wolf S. Quantification of cystoid changes in diabetic maculopathy. Invest Ophthalmol Vis Sci. 1995 Mar;36(3):608–613. [PubMed] [Google Scholar]
- Asrani S., Zeimer R., Goldberg M. F., Zou S. Application of rapid scanning retinal thickness analysis in retinal diseases. Ophthalmology. 1997 Jul;104(7):1145–1151. doi: 10.1016/s0161-6420(97)30170-5. [DOI] [PubMed] [Google Scholar]
- Ferris F. L., 3rd, Patz A. Macular edema. A complication of diabetic retinopathy. Surv Ophthalmol. 1984 May;28 (Suppl):452–461. doi: 10.1016/0039-6257(84)90227-3. [DOI] [PubMed] [Google Scholar]
- Hee M. R., Izatt J. A., Swanson E. A., Huang D., Schuman J. S., Lin C. P., Puliafito C. A., Fujimoto J. G. Optical coherence tomography of the human retina. Arch Ophthalmol. 1995 Mar;113(3):325–332. doi: 10.1001/archopht.1995.01100030081025. [DOI] [PubMed] [Google Scholar]
- Hee M. R., Puliafito C. A., Duker J. S., Reichel E., Coker J. G., Wilkins J. R., Schuman J. S., Swanson E. A., Fujimoto J. G. Topography of diabetic macular edema with optical coherence tomography. Ophthalmology. 1998 Feb;105(2):360–370. doi: 10.1016/s0161-6420(98)93601-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson C., Flanagan J. G., Turner G. S., McLeod D. Scanning laser tomography Z profile signal width as an objective index of macular retinal thickening. Br J Ophthalmol. 1998 Feb;82(2):121–130. doi: 10.1136/bjo.82.2.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinyoun J., Barton F., Fisher M., Hubbard L., Aiello L., Ferris F., 3rd Detection of diabetic macular edema. Ophthalmoscopy versus photography--Early Treatment Diabetic Retinopathy Study Report Number 5. The ETDRS Research Group. Ophthalmology. 1989 Jun;96(6):746–751. doi: 10.1016/s0161-6420(89)32814-4. [DOI] [PubMed] [Google Scholar]
- Kiri A., Dyer D. S., Bressler N. M., Bressler S. B., Schachat A. P. Detection of diabetic macular edema: Nidek 3Dx stereophotography compared with fundus biomicroscopy. Am J Ophthalmol. 1996 Nov;122(5):654–662. doi: 10.1016/s0002-9394(14)70483-0. [DOI] [PubMed] [Google Scholar]
- Landau D., Schneidman E. M., Jacobovitz T., Rozenman Y. Quantitative in vivo retinal thickness measurements in healthy subjects. Ophthalmology. 1997 Apr;104(4):639–642. doi: 10.1016/s0161-6420(97)30259-0. [DOI] [PubMed] [Google Scholar]
- Moss S. E., Klein R., Kessler S. D., Richie K. A. Comparison between ophthalmoscopy and fundus photography in determining severity of diabetic retinopathy. Ophthalmology. 1985 Jan;92(1):62–67. doi: 10.1016/s0161-6420(85)34082-4. [DOI] [PubMed] [Google Scholar]
- Ohnishi Y., Fujisawa K., Ishibashi T., Kojima H. Capillary blood flow velocity measurements in cystoid macular edema with the scanning laser ophthalmoscope. Am J Ophthalmol. 1994 Jan 15;117(1):24–29. doi: 10.1016/s0002-9394(14)73011-9. [DOI] [PubMed] [Google Scholar]
- Shahidi M., Ogura Y., Blair N. P., Rusin M. M., Zeimer R. Retinal thickness analysis for quantitative assessment of diabetic macular edema. Arch Ophthalmol. 1991 Aug;109(8):1115–1119. doi: 10.1001/archopht.1991.01080080075032. [DOI] [PubMed] [Google Scholar]
- Shahidi M., Zeimer R. C., Mori M. Topography of the retinal thickness in normal subjects. Ophthalmology. 1990 Sep;97(9):1120–1124. doi: 10.1016/s0161-6420(90)32457-0. [DOI] [PubMed] [Google Scholar]
- Smith R. T., Lee C. M., Charles H. C., Farber M., Cunha-Vaz J. G. Quantification of diabetic macular edema. Arch Ophthalmol. 1987 Feb;105(2):218–222. doi: 10.1001/archopht.1987.01060020072032. [DOI] [PubMed] [Google Scholar]
- Wilkins J. R., Puliafito C. A., Hee M. R., Duker J. S., Reichel E., Coker J. G., Schuman J. S., Swanson E. A., Fujimoto J. G. Characterization of epiretinal membranes using optical coherence tomography. Ophthalmology. 1996 Dec;103(12):2142–2151. doi: 10.1016/s0161-6420(96)30377-1. [DOI] [PubMed] [Google Scholar]
- Zambarakji H. J., Amoaku W. M., Vernon S. A. Volumetric analysis of early macular edema with the Heidelberg Retina Tomograph in diabetic retinopathy. Ophthalmology. 1998 Jun;105(6):1051–1059. doi: 10.1016/S0161-6420(98)96007-9. [DOI] [PubMed] [Google Scholar]
- Zeimer R. C., Mori M. T., Khoobehi B. Feasibility test of a new method to measure retinal thickness noninvasively. Invest Ophthalmol Vis Sci. 1989 Oct;30(10):2099–2105. [PubMed] [Google Scholar]
- Zeimer R., Shahidi M., Mori M., Zou S., Asrani S. A new method for rapid mapping of the retinal thickness at the posterior pole. Invest Ophthalmol Vis Sci. 1996 Sep;37(10):1994–2001. [PubMed] [Google Scholar]