Abstract
BACKGROUND—Radiofrequency ablation of tissues in pulmonary veins can eliminate paroxysmal atrial fibrillation. OBJECTIVE—To explore the characteristics of normal pulmonary veins so as to provide more information relevant to radiofrequency ablation. METHODS—20 structurally normal heart specimens were examined grossly. Histological sections were made from 65 pulmonary veins. RESULTS—The longest myocardial sleeves were found in the superior veins. The sleeves were thickest at the venoatrial junction in the left superior pulmonary veins. For the superior veins, the sleeves were thickest along the inferior walls and thinnest superiorly. The sleeves were composed mainly of circularly or spirally oriented bundles of myocytes with additional bundles that were longitudinally or obliquely oriented, sometimes forming mesh-like arrangements. Fibrotic changes estimated at between 5% and 70% across three transverse sections were seen in 17 veins that were from individuals aged 30 to 72 years. CONCLUSIONS—The myocardial architecture in normal pulmonary veins is highly variable. The complex arrangement, stretch, and increase in fibrosis may produce greater non-uniform anisotropic properties. Keywords: arrhythmias; catheter ablation; fibrillation; cardiac veins
Full Text
The Full Text of this article is available as a PDF (243.8 KB).
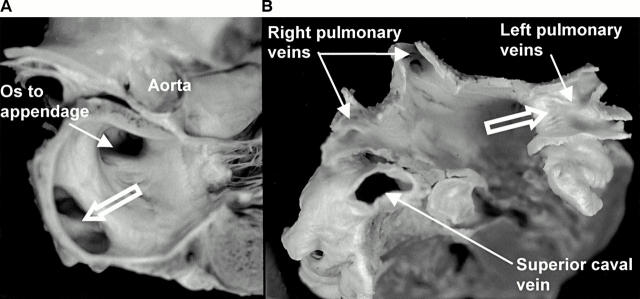
Figure 1 .
(A) Longitudinal section through the left atrium showing a common orifice (open arrows) to the left pulmonary veins. (B) Another heart sectioned transversely and viewed from above to show the entrances of the four pulmonary veins. Note the common venous orifice (open arrow) on the left and the relation of the superior caval vein to the right superior pulmonary vein.
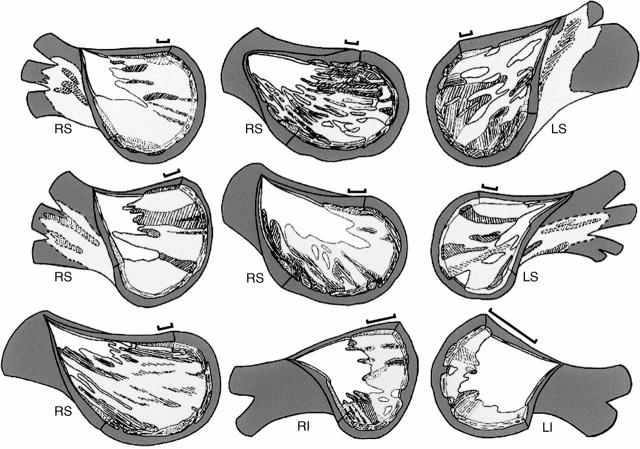
Figure 2 .
Graphic reconstructions of nine pulmonary veins with the venous wall (white) removed from the endothelial aspect to show the extensions of the muscular sleeves (pale grey). The bars represent 2 mm along the length of each vein. The unhatched areas of pale grey represent circularly or spirally arranged myocytes; hatched areas represent longitudinal or oblique myocytes. Dark grey areas represent the fibro-fatty tissues of the adventitia, the layer that is external to the sleeves. White areas among pale grey areas represent gaps in the venous wall that were without the outer myocardial sleeve. LI, left inferior pulmonary vein; LS, left superior pulmonary vein; RI, right inferior pulmonary vein; RS, right superior pulmonary vein.
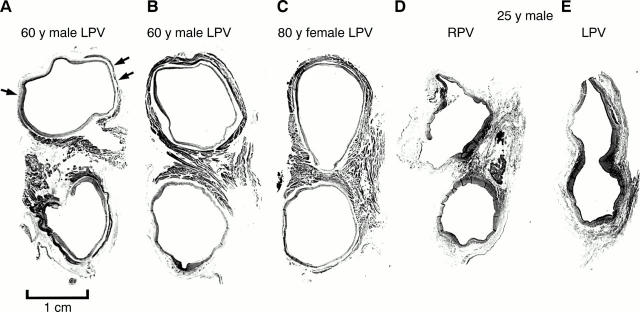
Figure 3 .
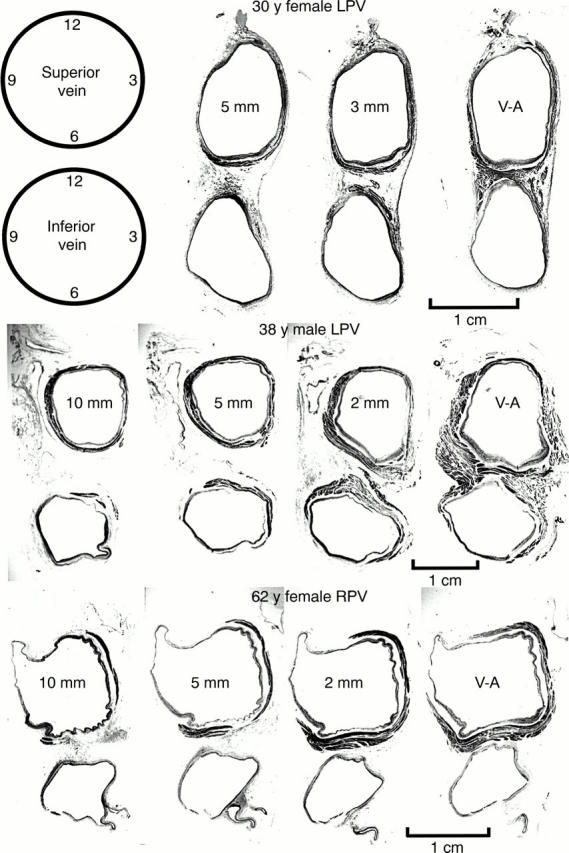
Transverse sections through three sets of left pulmonary veins (LPV) showing the variations in circumferential arrangement of the myocardial sleeves. Myocardium is shown as dark grey and the venous wall is shown as pale grey. Note the myocardial sleeve completely surrounding the superior vein and extending beyond 10 mm from the venoatrial junction in the 38 year old patient. In contrast, there is incomplete encirclement of the sleeves in the 30 year old and 62 year old patients. The inferior veins have less extensive sleeves. 2 mm, 3 mm, 5 mm, 10 mm are the distances of the sections from the venoatrial junction (V-A). Trichrome stain.
Figure 4 .
Transverse sections showing variations in widths of the muscular isthmus between superior and inferior veins at the venoatrial junction in four hearts. The myocardial sleeves (dark grey) are a complex arrangement of circular and longitudinal bundles of myocytes. The myocardial sleeves in (A) show extensive fibrotic replacement (pale grey; small arrows) compared with another case of the same age shown in (B). The isthmus is narrow in (C) and fibrotic changes are insignificant. The case shown in (D) and (E) has hardly any sleeves. The left veins share an orifice. LPV, left pulmonary veins; RPV, right pulmonary veins. Trichrome stain.
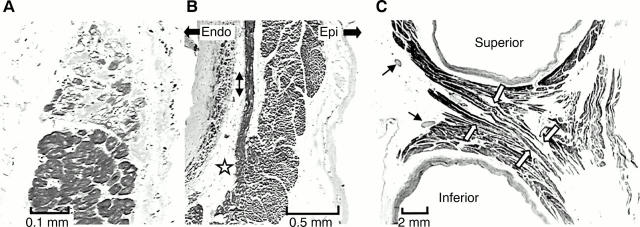
Figure 5 .
(A) and (B) are longitudinal sections of the myocardial sleeve of the right superior pulmonary vein from a 47 year old man. (A) Degenerative changes with fibrous replacement. (B) Circularly arranged myocytes occupy nearly the full thickness. A fascicle of longitudinally arranged myocytes (double arrows) arises from the circularly arranged myocytes (star) as a separate bundle surrounded by fibrous tissue and fat. The bundle terminates within circularly oriented myocytes when followed distally (not shown). (C) is an enlargement of the isthmus between superior and inferior veins shown in fig 4B. The white arrows indicate the crossover of muscle bundles linking superior and inferior veins. There are abundant nerve bundles (thin arrows) in the vicinity. Trichrome stain. Epi and Endo mark the orientations for epicardial and endothelial surfaces, respectively, for panel (B).
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BURCH G. E., ROMNEY R. B. Functional anatomy and throttle valve action on the pulmonary veins. Am Heart J. 1954 Jan;47(1):58–66. doi: 10.1016/0002-8703(54)90211-2. [DOI] [PubMed] [Google Scholar]
- Chen Y. J., Tai C. T., Hsieh M. H., Tsai C. F., Lin W. S., Chen S. A. Dependence of electrogram duration in right posteroseptal atrium and atrium-pulmonary vein junction on pacing site: mechanism and implications regarding atrioventricular nodal reentrant tachycardia and paroxysmal atrial fibrillation. J Cardiovasc Electrophysiol. 2000 May;11(5):506–515. doi: 10.1111/j.1540-8167.2000.tb00003.x. [DOI] [PubMed] [Google Scholar]
- Haïssaguerre M., Jaïs P., Shah D. C., Arentz T., Kalusche D., Takahashi A., Garrigue S., Hocini M., Peng J. T., Clémenty J. Catheter ablation of chronic atrial fibrillation targeting the reinitiating triggers. J Cardiovasc Electrophysiol. 2000 Jan;11(1):2–10. doi: 10.1111/j.1540-8167.2000.tb00727.x. [DOI] [PubMed] [Google Scholar]
- Haïssaguerre M., Jaïs P., Shah D. C., Takahashi A., Hocini M., Quiniou G., Garrigue S., Le Mouroux A., Le Métayer P., Clémenty J. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med. 1998 Sep 3;339(10):659–666. doi: 10.1056/NEJM199809033391003. [DOI] [PubMed] [Google Scholar]
- Ho S. Y., Sanchez-Quintana D., Cabrera J. A., Anderson R. H. Anatomy of the left atrium: implications for radiofrequency ablation of atrial fibrillation. J Cardiovasc Electrophysiol. 1999 Nov;10(11):1525–1533. doi: 10.1111/j.1540-8167.1999.tb00211.x. [DOI] [PubMed] [Google Scholar]
- Hsieh M. H., Chen S. A., Tai C. T., Tsai C. F., Prakash V. S., Yu W. C., Liu C. C., Ding Y. A., Chang M. S. Double multielectrode mapping catheters facilitate radiofrequency catheter ablation of focal atrial fibrillation originating from pulmonary veins. J Cardiovasc Electrophysiol. 1999 Feb;10(2):136–144. doi: 10.1111/j.1540-8167.1999.tb00654.x. [DOI] [PubMed] [Google Scholar]
- Jaïs P., Haïssaguerre M., Shah D. C., Chouairi S., Gencel L., Hocini M., Clémenty J. A focal source of atrial fibrillation treated by discrete radiofrequency ablation. Circulation. 1997 Feb 4;95(3):572–576. doi: 10.1161/01.cir.95.3.572. [DOI] [PubMed] [Google Scholar]
- Jaïs P., Shah D. C., Haïssaguerre M., Hocini M., Garrigue S., Clémenty J. Atrial fibrillation: role of arrhythmogenic foci. J Interv Card Electrophysiol. 2000 Jan;4 (Suppl 1):29–37. doi: 10.1023/a:1009857926782. [DOI] [PubMed] [Google Scholar]
- Lin W. S., Prakash V. S., Tai C. T., Hsieh M. H., Tsai C. F., Yu W. C., Lin Y. K., Ding Y. A., Chang M. S., Chen S. A. Pulmonary vein morphology in patients with paroxysmal atrial fibrillation initiated by ectopic beats originating from the pulmonary veins: implications for catheter ablation. Circulation. 2000 Mar 21;101(11):1274–1281. doi: 10.1161/01.cir.101.11.1274. [DOI] [PubMed] [Google Scholar]
- Masani F. Node-like cells in the myocardial layer of the pulmonary vein of rats: an ultrastructural study. J Anat. 1986 Apr;145:133–142. [PMC free article] [PubMed] [Google Scholar]
- Nathan H., Eliakim M. The junction between the left atrium and the pulmonary veins. An anatomic study of human hearts. Circulation. 1966 Sep;34(3):412–422. doi: 10.1161/01.cir.34.3.412. [DOI] [PubMed] [Google Scholar]
- Nathan H., Gloobe H. Myocardial atrio-venous junctions and extensions (sleeves) over the pulmonary and caval veins. Anatomical observations in various mammals. Thorax. 1970 May;25(3):317–324. doi: 10.1136/thx.25.3.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robbins I. M., Colvin E. V., Doyle T. P., Kemp W. E., Loyd J. E., McMahon W. S., Kay G. N. Pulmonary vein stenosis after catheter ablation of atrial fibrillation. Circulation. 1998 Oct 27;98(17):1769–1775. doi: 10.1161/01.cir.98.17.1769. [DOI] [PubMed] [Google Scholar]
- Saito T., Waki K., Becker A. E. Left atrial myocardial extension onto pulmonary veins in humans: anatomic observations relevant for atrial arrhythmias. J Cardiovasc Electrophysiol. 2000 Aug;11(8):888–894. doi: 10.1111/j.1540-8167.2000.tb00068.x. [DOI] [PubMed] [Google Scholar]
- Satoh T., Zipes D. P. Unequal atrial stretch in dogs increases dispersion of refractoriness conducive to developing atrial fibrillation. J Cardiovasc Electrophysiol. 1996 Sep;7(9):833–842. doi: 10.1111/j.1540-8167.1996.tb00596.x. [DOI] [PubMed] [Google Scholar]
- Spach M. S., Dolber P. C. Relating extracellular potentials and their derivatives to anisotropic propagation at a microscopic level in human cardiac muscle. Evidence for electrical uncoupling of side-to-side fiber connections with increasing age. Circ Res. 1986 Mar;58(3):356–371. doi: 10.1161/01.res.58.3.356. [DOI] [PubMed] [Google Scholar]
- Tse H. F., Lau C. P., Kou W., Pelosi F., Oral H., Kim M., Michaud G. F., Knight B. P., Moscucci M., Strickberger S. A. Prevalence and significance of exit block during arrhythmias arising in pulmonary veins. J Cardiovasc Electrophysiol. 2000 Apr;11(4):379–386. doi: 10.1111/j.1540-8167.2000.tb00332.x. [DOI] [PubMed] [Google Scholar]