Abstract
Two strains of Enterobacter cloacae were isolated from a patient before (strain MHN1) and during (strain MHN2) treatment with moxalactam and gentamicin. Strain MHN1 exhibited inducible ampC cephalosporinase production. In contrast, strain MHN2 expressed the enzyme constitutively at a 3,000-fold higher level. With the Escherichia coli ampC gene as a hybridization probe it was shown that the genomic arrangement of the ampC region was the same in both strains. To gain more insight into regulatory phenomena, the ampC genes were cloned, and their expression was studied in E. coli K-12. The ampC gene from MHN1 behaved normally and conferred inducible beta-lactam resistance. A regulatory region of at least 800 base pairs involved in controlling repression-induction was located immediately upstream of ampC. Surprisingly, when present in E. coli the ampC gene from MHN2 no longer overproduced the cephalosporinase, and inducible expression was observed. This indicates that in MHN2 stable cephalosporinase overproduction is controlled by another factor which is not linked to the ampC gene.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benton W. D., Davis R. W. Screening lambdagt recombinant clones by hybridization to single plaques in situ. Science. 1977 Apr 8;196(4286):180–182. doi: 10.1126/science.322279. [DOI] [PubMed] [Google Scholar]
- Bergström S., Lindberg F. P., Olsson O., Normark S. Comparison of the overlapping frd and ampC operons of Escherichia coli with the corresponding DNA sequences in other gram-negative bacteria. J Bacteriol. 1983 Sep;155(3):1297–1305. doi: 10.1128/jb.155.3.1297-1305.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergström S., Normark S. Beta-lactam resistance in clinical isolates of Escherichia coli caused by elevated production of the ampC-mediated chromosomal beta-lactamase. Antimicrob Agents Chemother. 1979 Oct;16(4):427–433. doi: 10.1128/aac.16.4.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang A. C., Cohen S. N. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J Bacteriol. 1978 Jun;134(3):1141–1156. doi: 10.1128/jb.134.3.1141-1156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole S. T. Nucleotide sequence coding for the flavoprotein subunit of the fumarate reductase of Escherichia coli. Eur J Biochem. 1982 Mar 1;122(3):479–484. doi: 10.1111/j.1432-1033.1982.tb06462.x. [DOI] [PubMed] [Google Scholar]
- Cole S. T., Sonntag I., Henning U. Cloning and expression in Escherichia coli K-12 of the genes for major outer membrane protein OmpA from Shigella dysenteriae, Enterobacter aerogenes, and Serratia marcescens. J Bacteriol. 1982 Jan;149(1):145–150. doi: 10.1128/jb.149.1.145-150.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edlund T., Grundström T., Normark S. Isolation and characterization of DNA repetitions carrying the chromosomal beta-lactamase gene of Escherichia coli K-12. Mol Gen Genet. 1979 Jun 7;173(2):115–125. doi: 10.1007/BF00330301. [DOI] [PubMed] [Google Scholar]
- Findell C. M., Sherris J. C. Susceptibility of Enterobacter to cefamandole: evidence for a high mutation rate to resistance. Antimicrob Agents Chemother. 1976 Jun;9(6):970–974. doi: 10.1128/aac.9.6.970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gootz T. D., Sanders C. C. Characterization of beta-lactamase induction in Enterobacter cloacae. Antimicrob Agents Chemother. 1983 Jan;23(1):91–97. doi: 10.1128/aac.23.1.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grundström T., Jaurin B. Overlap between ampC and frd operons on the Escherichia coli chromosome. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1111–1115. doi: 10.1073/pnas.79.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennessey T. D. Inducible beta-lactamase in Enterobacter. J Gen Microbiol. 1967 Nov;49(2):277–285. doi: 10.1099/00221287-49-2-277. [DOI] [PubMed] [Google Scholar]
- Jarlier V., Bismuth R., Nicolas M. H., Nguyen J., Truffot C., Grosset J. Survey of the phenotypes of susceptibility to beta-lactams in Enterobacteriaceae at the Pitie-Salpetriere Hospital. J Antimicrob Chemother. 1984 Sep;14 (Suppl B):59–65. doi: 10.1093/jac/14.suppl_b.59. [DOI] [PubMed] [Google Scholar]
- Jarlier V., Philippon A., Nicolas M. H., Bismuth R., Paul G., Fusciardi J. Enterobacter cloacae. Emergence in vivo d'un variant résistant aux nouvelles beta-lactamines lors d'un traitement par lamoxactam-gentamicine. Pathol Biol (Paris) 1984 May;32(5):399–403. [PubMed] [Google Scholar]
- Jaurin B., Grundström T. ampC cephalosporinase of Escherichia coli K-12 has a different evolutionary origin from that of beta-lactamases of the penicillinase type. Proc Natl Acad Sci U S A. 1981 Aug;78(8):4897–4901. doi: 10.1073/pnas.78.8.4897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaurin B., Normark S. Insertion of IS2 creates a novel ampC promoter in Escherichia coli. Cell. 1983 Mar;32(3):809–816. doi: 10.1016/0092-8674(83)90067-3. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Labia R., Andrillon J., Le Goffic F. Computerized microacidimetric determination of beta lactamase Michaelis-Menten constants. FEBS Lett. 1973 Jun 15;33(1):42–44. doi: 10.1016/0014-5793(73)80154-1. [DOI] [PubMed] [Google Scholar]
- Lampe M. F., Allan B. J., Minshew B. H., Sherris J. C. Mutational enzymatic resistance of Enterobacter species to beta-lactam antibiotics. Antimicrob Agents Chemother. 1982 Apr;21(4):655–660. doi: 10.1128/aac.21.4.655. [DOI] [PMC free article] [PubMed] [Google Scholar]
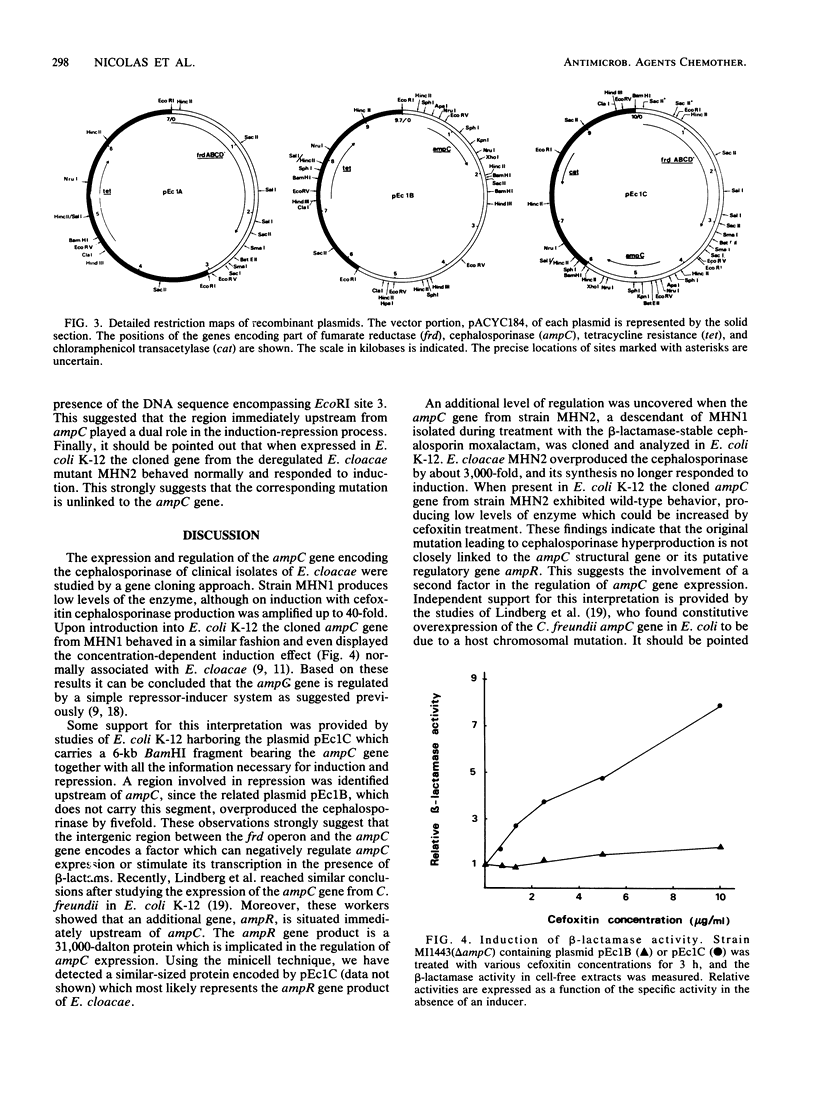
- Lindberg F., Westman L., Normark S. Regulatory components in Citrobacter freundii ampC beta-lactamase induction. Proc Natl Acad Sci U S A. 1985 Jul;82(14):4620–4624. doi: 10.1073/pnas.82.14.4620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loenen W. A., Brammar W. J. A bacteriophage lambda vector for cloning large DNA fragments made with several restriction enzymes. Gene. 1980 Aug;10(3):249–259. doi: 10.1016/0378-1119(80)90054-2. [DOI] [PubMed] [Google Scholar]
- Mathew A., Harris A. M., Marshall M. J., Ross G. W. The use of analytical isoelectric focusing for detection and identification of beta-lactamases. J Gen Microbiol. 1975 May;88(1):169–178. doi: 10.1099/00221287-88-1-169. [DOI] [PubMed] [Google Scholar]
- Medeiros A. A., Hare R., Papa E., Adam C., Miller G. H. Gram-negative bacilli resistant to third-generation cephalosporins: beta-lactamase characterization and susceptibility to Sch 34343. J Antimicrob Chemother. 1985 Jun;15 (Suppl 100):119–132. doi: 10.1093/jac/15.suppl_c.119. [DOI] [PubMed] [Google Scholar]
- Normark S., Bergström S., Edlund T., Grundström T., Jaurin B., Lindberg F. P., Olsson O. Overlapping genes. Annu Rev Genet. 1983;17:499–525. doi: 10.1146/annurev.ge.17.120183.002435. [DOI] [PubMed] [Google Scholar]
- Normark S., Edlund T., Grundström T., Bergström S., Wolf-Watz H. Escherichia coli K-12 mutants hyperproducing chromosomal beta-lactamase by gene repetitions. J Bacteriol. 1977 Dec;132(3):912–922. doi: 10.1128/jb.132.3.912-922.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olson B., Weinstein R. A., Nathan C., Kabins S. A. Broad-spectrum beta-lactam resistance in Enterobacter: emergence during treatment and mechanisms of resistance. J Antimicrob Chemother. 1983 Apr;11(4):299–310. doi: 10.1093/jac/11.4.299. [DOI] [PubMed] [Google Scholar]
- Olsson O., Bergström S., Normark S. Identification of a novel ampC beta-lactamase promoter in a clinical isolate of Escherichia coli. EMBO J. 1982;1(11):1411–1416. doi: 10.1002/j.1460-2075.1982.tb01331.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders C. C., Sanders W. E., Jr Emergence of resistance during therapy with the newer beta-lactam antibiotics: role of inducible beta-lactamases and implications for the future. Rev Infect Dis. 1983 Jul-Aug;5(4):639–648. doi: 10.1093/clinids/5.4.639. [DOI] [PubMed] [Google Scholar]
- Sykes R. B., Matthew M. The beta-lactamases of gram-negative bacteria and their role in resistance to beta-lactam antibiotics. J Antimicrob Chemother. 1976 Jun;2(2):115–157. doi: 10.1093/jac/2.2.115. [DOI] [PubMed] [Google Scholar]
- Tancrede C. H., Andremont A. O., Leonard F. C. Epidemiology of enterobacteria resistant to cefotaxime in hospital. J Antimicrob Chemother. 1984 Sep;14 (Suppl B):53–57. doi: 10.1093/jac/14.suppl_b.53. [DOI] [PubMed] [Google Scholar]
- Then R. L., Angehrn P. Trapping of nonhydrolyzable cephalosporins by cephalosporinases in Enterobacter cloacae and Pseudomonas aeruginosa as a possible resistance mechanism. Antimicrob Agents Chemother. 1982 May;21(5):711–717. doi: 10.1128/aac.21.5.711. [DOI] [PMC free article] [PubMed] [Google Scholar]