Abstract
Phospholipid translocases (PLTs) have been implicated in the generation of phospholipid asymmetry in membrane bilayers. In budding yeast, putative PLTs are encoded by the DRS2 gene family of type 4 P-type ATPases. The homologous proteins Cdc50p, Lem3p, and Crf1p are potential noncatalytic subunits of Drs2p, Dnf1p and Dnf2p, and Dnf3p, respectively; these putative heteromeric PLTs share an essential function for cell growth. We constructed temperature-sensitive mutants of CDC50 in the lem3Δ crf1Δ background (cdc50-ts mutants). Screening for multicopy suppressors of cdc50-ts identified YPT31/32, two genes that encode Rab family small GTPases that are involved in both the exocytic and endocytic recycling pathways. The cdc50-ts mutants did not exhibit major defects in the exocytic pathways, but they did exhibit those in endocytic recycling; large membranous structures containing the vesicle-soluble N-ethylmaleimide-sensitive factor attachment protein receptor Snc1p intracellularly accumulated in these mutants. Genetic results suggested that the YPT31/32 effector RCY1 and CDC50 function in the same signaling pathway, and simultaneous overexpression of CDC50, DRS2, and GFP-SNC1 restored growth as well as the plasma membrane localization of GFP-Snc1p in the rcy1Δ mutant. In addition, Rcy1p coimmunoprecipitated with Cdc50p-Drs2p. We propose that the Ypt31p/32p–Rcy1p pathway regulates putative phospholipid translocases to promote formation of vesicles destined for the trans-Golgi network from early endosomes.
INTRODUCTION
Most eukaryotic cells display an asymmetric distribution of phospholipids in the plasma membrane. In general, the aminophospholipids phosphatidylserine (PS) and phosphatidylethanolamine (PE) are enriched in the inner leaflet facing the cytoplasm, whereas phosphatidylcholine (PC), sphingomyelin, and glycolipids are predominantly found in the outer leaflet of the plasma membrane. This lipid asymmetry is generated and maintained by ATP-driven lipid transporters or translocases. The type 4 subfamily of P-type ATPases is implicated in the translocation of phospholipids from the external to the cytosolic leaflet (Graham, 2004; Pomorski et al., 2004; Holthuis and Levine, 2005). Five members of this subfamily (Drs2p, Neo1p, Dnf1p, Dnf2p, and Dnf3p) are encoded by the genome of the yeast Saccharomyces cerevisiae (Catty et al., 1997). Dnf1p and Dnf2p are localized to the plasma membrane, and loss of Dnf1p and Dnf2p abolishes ATP-dependent transport of fluorescently 7-nitrobenz-2-oxa-1,3-diazol-4-yl (NBD)–labeled analogues of PE, PS, and PC from the outer to the inner plasma membrane leaflet (Pomorski et al., 2003). Drs2p is localized to endosomes and the trans-Golgi network (TGN) (Chen et al., 1999; Hua et al., 2002; Pomorski et al., 2003; Saito et al., 2004), suggesting that Drs2p regulates phospholipid asymmetry in these membranes. Moreover, Golgi membranes isolated from a temperature-sensitive drs2 mutant lacking DNF1, DNF2, and DNF3 exhibited defects in the ATP-dependent transport of an NBD-labeled analogue of PS (Natarajan et al., 2004). Alder-Baerens et al. (2006) also demonstrated that post-Golgi secretory vesicles contained Drs2p- and Dnf3p-dependent NBD-labeled phospholipid translocase activity and that the asymmetric PE arrangement in these vesicles was disrupted in the drs2Δ dnf3Δ mutant. The drs2Δ mutant exhibits TGN defects comparable with those exhibited by strains with clathrin mutations and is defective in the formation of clathrin-coated vesicles (Chen et al., 1999; Gall et al., 2002). Drs2p is also implicated in endocytic recycling because the exocytic vesicle-soluble N-ethylmaleimide-sensitive factor attachment protein receptor (v-SNARE) Snc1p accumulated intracellularly in the drs2Δ mutant (Hua et al., 2002; Saito et al., 2004).
Cdc50p, a conserved integral membrane protein, was identified as a protein required for polarized growth (Misu et al., 2003). Cdc50p colocalizes with Drs2p at endosomal and TGN membranes, and it associates with Drs2p as a potential noncatalytic subunit (Saito et al., 2004). In the absence of Cdc50p, Drs2p is retained in the endoplasmic reticulum (ER) and vice versa. In addition to Cdc50p, two Cdc50p-related proteins, Lem3p/Ros3p and Crf1p, are encoded by the yeast genome. Similar to Cdc50p, Lem3p is a potential noncatalytic subunit that associates with Dnf1p (Saito et al., 2004) and Dnf2p (see Results), and Crf1p is a potential noncatalytic subunit for Dnf3p (see Results). These heteromeric putative phospholipid translocases (PLTs) constitute an essential family for cell growth in which Cdc50p-Drs2p, Lem3p-Dnf1p/2p, and Crf1p-Dnf3p play major, intermediate, and minor roles, respectively (Hua et al., 2002; Saito et al., 2004). The dnf1Δ dnf2Δ drs2Δ mutant exhibits a defect in endocytic internalization at 15°C as assayed by uptake of the endocytic tracer dye N-(3-triethylammoniumpropyl)-4-(p-diethylaminophenylhexatrienyl) pyridinium dibromide (FM4-64) (see Materials and Methods) (Pomorski et al., 2003). The drs2Δ dnf1Δ mutant exhibits a substantial defect in the transport of alkaline phosphatase to the vacuole (Hua et al., 2002). The dnf1Δ dnf2Δ dnf3Δ mutant intracellularly accumulates Snc1p due to defects in endocytic recycling (Hua et al., 2002). The cellular functions shared by these heteromeric putative PLTs, however, need to be further explored.
In this study, we constructed temperature-sensitive (ts) cdc50 mutations in a strain lacking LEM3 and CRF1. Screening for multicopy suppressors of the ts growth phenotype identified YPT32 encoding a Rab family small GTPase, which has been implicated in the formation of exocytic vesicles from the TGN along with its close homologue Ypt31p (Benli et al., 1996; Jedd et al., 1997). The cdc50-ts lem3Δ crf1Δ mutants, however, did not exhibit major defects in the formation of exocytic vesicles, but instead they exhibited severe defects in endocytic recycling. Interestingly, during the course of this study, it was reported that Ypt31p/32p also regulate endocytic recycling through its effector Rcy1p (Chen et al., 2005). The F-box protein Rcy1p (recycling 1) is involved in recycling out of early endosomes (Wiederkehr et al., 2000). Rcy1p associates with Skp1p, a component of the Skp1p-cullin-F-box protein (SCF) complex but not with other components required for ubiquitin ligase activity, suggesting that Rcy1p regulates endocytic recycling independently of ubiquitination (Galan et al., 2001). The rcy1Δ mutant accumulates large membranous structures that seem to be swollen early endosomes (Wiederkehr et al., 2000) and similar structures accumulated in the cdc50-ts lem3Δ crf1Δ mutants. Simultaneous overexpression of Cdc50p-Drs2p and GFP-Snc1p suppressed the defects in endocytic recycling of the rcy1Δ mutant, and Rcy1p was coimmunoprecipitated with Cdc50p and Drs2p. We propose that heteromeric putative PLTs cooperate with Ypt31p/32p-Rcy1p in endocytic recycling.
MATERIALS AND METHODS
Media and Genetic Techniques
Unless otherwise specified, strains were grown in rich medium (YPDA: 1% yeast extract [Difco, Detroit, MI], 2% bacto-peptone [Difco], 2% glucose, and 0.01% adenine). Strains carrying plasmids were selected in synthetic medium (SD) containing the required nutritional supplements (Rose et al., 1990). When indicated, 0.5% casamino acids (Difco) were added to the SD media without uracil (SDA-Ura). Phosphate-depleted rich medium contained 1% yeast extract, 2% bacto-peptone, and 4% glucose, and it was depleted of phosphate as described by Rubin (1973). The synthetic minimal (MV) media used for cell labeling were prepared as described previously (Rothblatt and Schekman, 1989). Standard genetic manipulations of yeast were performed as described previously (Guthrie and Fink, 1991). Yeast transformations were performed by the lithium acetate method (Elble, 1992; Gietz and Woods, 2002). Escherichia coli strains DH5α and XL1-Blue were used for construction and amplification of plasmids.
Strains and Plasmids
Yeast strains used in this study are listed in Table 1. The cdc50-ts strains were constructed as follows. First, random mutations in CDC50 were introduced by a polymerase chain reaction (PCR)-based method as described previously (Toi et al., 2003) by using the template pKT1262 (YCplac111-CDC50) and the primers CDC50-5′ (5′-AAGTGACGAATGGAATGATC-3′) corresponding to the nucleotide positions −613 to −594 of CDC50 and CDC50-3′100R (5′-GTCGCACTATTTTCCAAGCG-3′) complementary to the nucleotide positions 110–129 downstream of the CDC50 stop codon, to generate the ∼1.9-kilobase DNA fragment CDC50*. The marker fragment His3MX6 cassette flanked by sequences around the 130 base pairs downstream of the CDC50 stop codon were generated by PCR by using the template pFA6a-His3MX6 (Longtine et al., 1998) and the primers CDC50-3′100F1 (5′-CGCTTGGAAAATAGTGCGACCGGATCCCCGGGTTAATTAA-3′) and CDC50-3′100R1 (5′-TTCTATCATTTCATCATCTAAATGGGAATAGCAAACCCTGGGAGTTCTTTGAATTCGAGCTCGTTTAAAC-3′) to generate CDC50-3′-His3MX6. The underlined sequence within CDC50-3′100F1 corresponds to the nucleotide positions 110-129 downstream of the CDC50 stop codon, and is complementary to the sequence of CDC50-3′100R; the underlined sequence within CDC50-3′100R1 is complementary to the nucleotide positions 130–179 downstream of the CDC50 stop codon. Then, a second PCR was performed to connect the marker fragment to the randomly mutagenized CDC50 fragment, by using CDC50* and CDC50-3′-His3MX6 as templates and CDC50-5′ and CDC50-3′100R1 as primers. The amplified DNA fragment was introduced into the genome of YKT496 (MATα lem3Δ::TRP1), and His+ transformants were selected at 25°C. Of these transformants, 418 clones were streaked on two YPDA plates, one plate of which was incubated at 25°C and the other plate was incubated at 37°C. Fourteen clones, which exhibited mild ts growth phenotypes, were crossed with YKM48 (MATa GAL1p-HA-CDC50::KanMX6 crf1Δ::hphMX3), and the resulting diploids were sporulated and dissected. The obtained spore clones whose genotypes were cdc50*::His3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 were tested for growth at 25 and 37°C. The two clones (cdc50-11::His3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 and cdc50-162::His3MX6 lem3Δ::TRP1 crf1Δ::hphMX3) that exhibited the tightest ts phenotype were chosen for further analyses.
Table 1.
S. cerevisiae strains used in this study
| Straina | Genotype | Reference or source |
|---|---|---|
| ANS2-3A | MATαura3-52 leu2-3, 112 his3 his4 sec2-56 | Gift from A. Nakano |
| ANS6-2D | MATα ura3-52 leu2-3, 112 trp1-289 his3 his4 sec6-4 | Gift from A. Nakano |
| AKY15 | MATaade2-101 lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 Δpho8::PHO8Δ60 vps30Δ::LEU2 | Kihara et al. (2001) |
| YEF473 | MATa/α lys2-801/lys2-801 ura3-52/ura3-52 his3Δ-200/his3Δ-200 trp1Δ-63/trp1Δ-63 leu2Δ-1/leu2Δ-1 | Longtine et al. (1998) |
| YKT38 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 | Misu et al. (2003) |
| YKT259 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 CDC50-EGFP::kanMX6 | Saito et al. (2004) |
| YKT496 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 lem3Δ::TRP1 | This study |
| YKT760 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DNF1–13Myc::TRP1 | This study |
| YKT768 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DRS2-EGFP::kanMX6 | Saito et al. (2004) |
| YKT792 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DRS2–13Myc::TRP1 | This study |
| YKT871 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DRS2-mRFP1::TRP1 | This study |
| YKT902 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 lem3Δ::TRP1 crf1Δ::hphMX3 | This study |
| YKT903 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 KEX2-EGFP::kanMX6 | This study |
| YKT921 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DNF2-EGFP::kanMX6 | This study |
| YKT923 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 lem3Δ::TRP1 DNF2-EGFP::kanMX6 | This study |
| YKT925 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DNF3-EGFP::kanMX6 | This study |
| YKT928 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 crf1Δ::hphMX3 DNF3-EGFP::kanMX6 | This study |
| YKT942 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-162::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 | This study |
| YKT949 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 dnf1Δ::hphMX3 dnf2Δ::kanMX6 dnf3Δ::HIS3MX6 | This study |
| YKT951 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 rcy1Δ::kanMX4 | This study |
| YKT957 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 VPS10-EGFP::kanMX6 | This study |
| YKT993 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 | This study |
| YKT994 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 DRS2-EGFP::kanMX6 | This study |
| YKT1000 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 KEX2-EGFP::kanMX6 | This study |
| YKT1001 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-162::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 KEX2-EGFP::kanMX6 | This study |
| YKT1004 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::kanMX4 crf1Δ::hphMX3 SEC7-mRFP1::TRP1 | This study |
| YKT1008b | MATα ura3 his3 trp1 leu2 cdc50-11::HIS3MX6 lem3Δ::TRP1 sec6-4 | This study |
| YKT1009b | MATaura3 his3 trp1 leu2 his4? lem3Δ::TRP1 crf1Δ::hphMX3 sec6-4 | This study |
| YKT1010b | MATα ura3 his3 trp1 leu2 his4? sec6-4 | This study |
| YKT1011b | MATaura3 his3 trp1 leu2 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 sec6-4 | This study |
| YKT1062 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 DNF2-13Myc::TRP1 | This study |
| YKT1086 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 VPS10-EGFP::kanMX6 | This study |
| YKT1088 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-162::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3 VPS10-EGFP::kanMX6 | This study |
| YKT1098 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 CRF1–3HA::HIS3MX6 | This study |
| YKT1099 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 CRF1-3HA::HIS3MX6 DNF2-13Myc::TRP1 | This study |
| YKT1100 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 CRF1-3HA::HIS3MX6 DNF3-13Myc::TRP1 | This study |
| YKT1101 | MATα lys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 CDC50-13Myc::HIS3MX6 | This study |
| YKT1102 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 rcy1Δ::hphMX4 CDC50-EGFP::kanMX6 | This study |
| YKT1310 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 lem3Δ::TRP1 crf1Δ::hphMX3 KEX2-EGFP::kanMX6 | This study |
| YNF61 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 lem3Δ::TRP1 crf1Δ::hphMX3 [GFP-SNC1 URA3 CEN] | Transformant of YKT902 with pRS416 GFP-SNC1 |
| YNF63 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1[GFP-SNC1 URA3 CEN] | Transformant of YKT38 with pRS416 GFP-SNC1 |
| YNF65 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3[GFP-SNC1 URA3 CEN] | Transformant of YKT993 with pRS416 GFP-SNC1 |
| YNF67 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-162::HIS3MX6 lem3Δ::TRP1 crf1Δ::hphMX3[GFP-SNC1 URA3 CEN] | Transformant of YKT942 with pRS416 GFP-SNC1 |
| YNF153 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 cdc50-11::HIS3MX6 lem3Δ::kanMX4 crf1Δ::hphMX3 SEC7-mRFP1::TRP1[GFP-SNC1 URA3 CEN] | Transformant of YKT1004 with pRS416 GFP-SNC1 |
| YNF784 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 dnf1Δ::hphMX3 dnf2Δ::kanMX6 dnf3Δ::HIS3MX6[GFP-SNC1 URA3 CEN] | Transformant of YKT949 with pRS416 GFP-SNC1 |
| KKT276 | MATalys2Δ0 ura3Δ0 his3Δ1 leu2Δ0 vps1Δ::kanMX4 | Gift from C. Boone |
| KKT277 | MATalys2Δ0 ura3Δ0 his3Δ1 leu2Δ0 vps4Δ::kanMX4 | Gift from C. Boone |
| YKM48 | MATalys2-801 ura3-52 his3Δ-200 trp1Δ-63 leu2Δ-1 GAL1p-HA-CDC50::kanMX6 crf1Δ::hphMX3 | This study |
a YKT, YNF, and YKM strains are isogenic derivatives of YEF473 except for YKT1008, YKT1009, YKT1010, and YKT1011.
b YKT1008 is segregant from a cross of ANS6-2D with YKT993, YKT1009 is segregant from a cross of ANS6-2D with YKT942, YKT1010 is segregant from a cross of ANS6-2D with YKT902, and YKT1011 is segregant from a cross of YKT1008 with YKT1009.
Yeast strains carrying a complete gene deletion (CRF1), enhanced green fluorescent protein (EGFP)-tagged genes (KEX2, VPS10, DNF2, and DNF3), 13 x Myc-tagged genes (CDC50, DNF2, and DNF3), the 3 x hemagglutinin (HA)-tagged CRF1 gene, or the monomeric red fluorescent protein 1 (mRFP1)-tagged DRS2 gene were constructed by PCR-based procedures as described previously (Longtine et al., 1998). DNF2-EGFP was functional, because the drs2Δ dnf3Δ DNF2-EGFP mutant grew at the same rate as the drs2Δ dnf3Δ DNF2 mutant at 28°C, at which the drs2Δ dnf3Δ dnf2Δ mutant exhibited a synthetic growth defect (our unpublished data). DRS2-mRFP1 was functional, because cells harboring the DRS2-mRFP1 allele instead of the DRS2 grew normally at 18°C, at which the drs2Δ mutant was lethal (our unpublished data). The rcy1 or lem3 disruption mutants were constructed on our strain background as follows. The regions containing the disruption marker and the flanking sequences were PCR amplified using genomic DNA derived from the rcy1Δ::KanMX4 or lem3Δ::KanMX4 strain (a gift from C. Boone, University of Toronto, Toronto, Ontario, Canada) as a template. The amplified DNA fragments were then introduced into the appropriate strains. All constructs made by the PCR-based procedure were verified by colony-PCR amplification to confirm that replacement had occurred at the expected loci. The rcy1Δ::hphMX4 strain was constructed by replacing the KanMX4 cassette of YKT951 (rcy1Δ::KanMX4) with the hphMX4 cassette prepared from pAG32 (Goldstein and McCusker, 1999). The vps1Δ and vps4Δ deletion mutants were gifts from C. Boone, and the vps30Δ strain was a gift from Y. Ohsumi (National Institute for Basic Biology, Okazaki, Japan).
The plasmids used in this study are listed in Table 2. The YPT32Q72L and YPT32S27N alleles were made by using a QuikChange site-directed mutagenesis kit (Stratagene, La Jolla, CA) with the plasmid pKT1555 (YEplac181-YPT32). The mutagenic oligonucleotides were YPT32-Q72L-1 (GAT TTG GGA CAC GGC AGG TCT AGA ACG TTA CAG GGC CAT CAC G) and its complement YPT32-Q72L-2 for YPT32Q72L and YPT32-S27N-1 (GAC TCC GGT GTG GGT AAA AAT AAT CTG TTG TCG AGA TTT ACA AC) and its complement YPT32-S27N-2 for YPT32S27N. We sequenced the entire open reading frame of YPT32 in each construct to verify that only the desired substitution was introduced. Epitope-tagged Rcy1 proteins were functional, because both pRS416-GFP-RCY1 and YEplac195-HA-BS-RCY1 plasmids suppressed the growth defect of the rcy1Δ mutant at 18°C (our unpublished data). Schemes detailing the construction of plasmids are available upon request.
Table 2.
Plasmids used in this study
| Plasmid | Characteristics | Reference or source |
|---|---|---|
| pKT1259 [YEplac181 CDC50] | CDC50 LEU2 2 μm | Misu et al. (2003) |
| pRS416 GFP-SNC1 | PTPI1-GFP-SNC1 URA3 CEN | Lewis et al. (2000) |
| pRS416 GFP-SNC1 pm | PTPI1-GFP-SNC1pm URA3 CEN | Lewis et al. (2000) |
| pKT1262 [YCplac111 CDC50] | CDC50 LEU2 CEN | This study |
| pKT1472 [YEplac195 DRS2 CDC50] | DRS2 CDC50 URA3 2 μm | Saito et al. (2004) |
| pKT1486 [PACT1- SUC2] | PACT1-SUC2 URA3 CEN | This study |
| pKT1487 [pRS416 GFP-PEP12] | PTPI1-GFP-PEP12 URA3 CEN | This study |
| pKT1490 [pRS315 GFP-SNC1] | PTPI1-GFP-SNC1 LEU2 CEN | This study |
| pKT1554 [YEplac181 YPT31] | YPT31 LEU2 2 μm | This study |
| pKT1555 [YEplac181 YPT32] | YPT32 LEU2 2 μm | This study |
| pKT1560 [YEplac181 GFP-RCY1] | GFP-RCY1 LEU2 2 μm | This study |
| pKT1563 [pRS416 mRFP-SNC1] | PTPI1-mRFP1-SNC1 URA3 CEN | This study |
| pKT1564 [pRS416 HA-SNC1] | PTPI1-HA-SNC1 URA3 CEN | This study |
| pKT1566 [YEplac181 GFP-TLG1] | GFP-TLG1 LEU2 2 μm | This study |
| pKT1578 [YEplac181 YPT32Q72L] | YPT32Q72LLEU2 2 μm | This study |
| pKT1579 [YEplac181 YPT32S27N] | YPT32S27NLEU2 2 μm | This study |
| pKT1626 [YEplac195 HA-BS-RYC1] | HA-RCY1 URA3 2 μm | This study |
| YEplac181 | LEU2 2 μm | Gietz and Sugino (1988) |
| YEplac195 | URA3 2 μm | Gietz and Sugino (1988) |
| pRS315 | LEU2 CEN | Sikorski and Hieter (1989) |
Isolation of Multicopy Suppressors of the cdc50-ts lem3Δ crf1Δ Mutants
The cdc50-11 lem3Δ crf1Δ (YKT993) and cdc50-162 lem3Δ crf1Δ (YKT942) strains were transformed with a yeast genomic DNA library constructed in the multicopy plasmid YEp13 (kindly provided by Y. Ohya, University of Tokyo, Tokyo, Japan). After transformation, cells were incubated at 25°C for 24 h to allow recovery and then were incubated at 37°C for 2 d on SD-Leu plates. Approximately 63,000 YKT993 transformants and 42,000 YKT942 transformants were screened, and 59 and 54 transformants that reproducibly grew at 37°C were obtained, respectively. The transformants that contained CDC50 or CRF1 on the plasmid were identified by colony-PCRs and were eliminated. The strains that contained LEM3 on the plasmid were identified by the PCR by using total DNA prepared from the rest of the candidate transformants and were eliminated. Plasmids from the remaining eight and four transformants of YKT993 and YKT942, respectively, were further analyzed. Based on restriction mapping, nine of the 12 plasmids were shown to be identical, whereas the other three plasmids were distinct from each other. The four different plasmids were retransformed into YKT993 and YKT942 to confirm their ability to suppress the ts growth phenotypes. Two of the four plasmids exhibited weaker suppression activities and were not further analyzed. The genes contained within the remaining two plasmids were identified by sequencing both ends of the inserts. The two plasmids contained overlapping genomic DNA fragments, and subcloning analysis revealed that the YPT32 gene was responsible for the suppression activity.
Purification of Secretory Vesicles and Enzyme Assays
Purification of secretory vesicles was performed as described (Harsay and Bretscher, 1995) with minor modifications. Briefly, cells were grown at 25°C to the early to mid-logarithmic phase (OD600 of 0.5–0.7) in 1 liter of synthetic medium, harvested by centrifugation, and resuspended in phosphate-depleted rich medium. Growth was continued at 25°C for 90 min to induce acid phosphatase secretion. Cells were then incubated at 37°C for 2 h before harvesting. Cells were converted to spheroplasts and lysed using 20 strokes in a Dounce glass homogenizer with a tight pestle (Wheaton Industries, Millville, NJ) in lysis buffer containing 0.8 M sorbitol, with 10 mM triethanolamine and 1 mM EDTA, adjusted to pH 7.2 with acetic acid (TEA), and protease inhibitors (1 μg/ml aprotinin, 1 μg/ml leupeptin, 1 μg/ml pepstatin, 2 mM benzamidine, and 1 mM phenylmethylsulfonyl fluoride). A 700 × g spin for 10 min generated the pellet (P1) and supernatant (S1) fractions. The S1 fraction was spun at 13,000 × g for 20 min to generate P2 and S2. The S2 fraction was centrifuged at 100,000 × g for 1 h in an SW28 rotor (Beckman Coulter, Fullerton, CA) to generate membrane pellets (P3). P3 membrane pellets were overlaid with 300 μl of 0.8 M sorbitol/TEA and placed on ice for 2–3 h to allow easy resuspension. For gradient fractionation, a 12-ml 15–30% continuous Nycodenz gradient in 0.8 M sorbitol/TEA was created. The P3 membranes were adjusted to 35% Nycodenz in a total volume of 1 ml and loaded into the bottom of the gradient using a 10-cm needle. Gradients were centrifuged in a P40ST rotor (Hitachi, Tokyo, Japan) at 100,000 × g for 19 h, and 0.8-ml fractions were manually collected from the top of the samples.
Enzymatic assays for acid phosphatase, exoglucanase, and invertase activities were performed as described previously (Harsay and Bretscher, 1995), except that fractions were not diluted when the invertase activity was measured.
Antibodies
The rabbit anti-Pma1p polyclonal antibodies were gifts from R. Serrano (Polytechnic University of Valencia, Valencia, Spain). The rabbit anti-carboxypeptidase Y (CPY) and anti-invertase polyclonal antibodies were gifts from A. Nakano (RIKEN, Wako, Japan). Rabbit anti-Lem3p polyclonal antibodies were gifts from M. Umeda (Kyoto University, Kyoto, Japan). The mouse anti-HA (HA.11) and anti-myc (9E10) monoclonal antibodies were purchased from BAbCO (Richmond, CA) and Sigma-Aldrich (St. Louis, MO), respectively. The horseradish peroxidase-conjugated secondary antibodies (sheep anti-mouse IgG and donkey anti-rabbit IgG) used for immunoblotting were purchased from GE Healthcare (Little Chalfont, Buckinghamshire, United Kingdom).
Immunoprecipitation Analysis
Immunoprecipitation analysis of Lem3p-Dnf2p and Crf1p-Dnf3p was performed as described previously (Saito et al., 2004). Coimmunoprecipitation analysis of Rcy1p with Cdc50p, Drs2p, Dnf1p, and Dnf2p was similarly performed. Briefly, cells were grown at 30°C to a cell density of 0.5 OD600/ml in SDA-Ura medium. Cells collected from 400 ml of the cultures were washed twice with ice-cold water and resuspended at 200 OD600/ml in lysis buffer (10 mM Tris-HCl, pH 7.5, 0.3 M sorbitol, 0.1 M NaCl, and 5 mM MgCl2) containing protease inhibitors (1 μg/ml aprotinin, 1 μg/ml leupeptin, 1 μg/ml pepstatin, 2 mM benzamidine, and 1 mM phenylmethylsulfonyl fluoride). The cells were lysed by Multi-beads shocker (Yasui Kikai, Osaka, Japan). Cell lysates were centrifuged at 400 × g for 5 min, and the resulting supernatant was centrifuged at 100,000 × g for 1 h at 4°C. For immunoprecipitation, the pellets were solubilized in 0.8 ml of immunoprecipitation buffer (10 mM Tris-HCl, pH 7.5, 150 mM NaCl, 2 mM EDTA, and 1% 3-[(3-cholamidopropyl)dimethylammonio]propanesulfonate [CHAPS]) containing the above-mentioned protease inhibitors. Insoluble material was removed by centrifugation at 20,630 × g for 5 min at 4°C. The cleared lysates were incubated with 5 μg of anti-Myc antibody for 1 h at 4°C. We then rotated the samples with 20 μl of protein G-Sepharose 4 Fast Flow (GE Healthcare) for 1 h at 4°C. The protein G-Sepharose beads were pelleted and washed three times with immunoprecipitation buffer in the absence of detergents before suspending them in SDS-PAGE sample buffer.
Immunoblotting Analysis
Immunoblotting analysis was performed as described previously (Misu et al., 2003). For SDS-PAGE of Drs2p-Myc, Dnf1p-Myc, Dnf2p-Myc, and Dnf3p-Myc, samples were heated at 37°C for 20 min before loading. For SDS-PAGE of Pma1p, 10-μl aliquots of each fraction from the Nycodenz density gradient fractionation were mixed with 10 μl of 2X SDS-PAGE sample buffer, followed by heating at 37°C for 15 min before loading. Anti-HA, anti-Myc, and anti-Lem3p antibodies were used at a 1:1000 dilution. Anti-Pma1p antibodies were used at a 1:5000 dilution. For the colony blot assay, anti-CPY antibodies were used at a 1:1000 dilution.
Microscopic Observations
Cells were observed using a Nikon ECLIPSE E800 microscope (Nikon Instec, Tokyo, Japan) equipped with an HB-10103AF superhigh-pressure mercury lamp and a 1.4 numerical aperture 100× Plan Apo oil immersion objective lens (Nikon Instec) with appropriate fluorescence filter sets (Nikon Instec) or differential interference contrast optics. Images were acquired using a cooled digital charge-coupled device camera (C4742-95-12NR; Hamamatsu Photonics, Hamamatsu, Japan) and AQUACOSMOS software (Hamamatsu Photonics). Observations are based on the characterization of >100 cells.
To assess the endocytic pathway using the lipophilic styryl dye FM4-64, cells were grown to late-logarithmic phase in YPDA medium at 25°C or 37°C for 3 h. Four OD600 units of cells were labeled with 32 μM FM4-64 (Invitrogen, Carlsbad, CA) in 100 μl of YPDA medium for 15 min at 25 or 37°C. Cells were harvested by centrifugation, resuspended in 200 μl of fresh YPDA medium, and chased at 25 or 37°C for 30 min. Cells were washed twice with 100 μl of SD medium and immediately observed using a G-2A filter set. To mark early endosomes with FM4-64, cells were grown to early logarithmic phase in SDA-Ura medium at 25°C and then shifted to 37°C for 3 h. Four OD600 units of cells were incubated with 32 μM FM4-64 in 100 μl of YPDA medium on ice for 30 min. Cells were harvested by centrifugation, resuspended in 200 μl of fresh YPDA medium and chased at 37°C for 10 min. After chase, cells were harvested by centrifugation, resuspended in ice-cold SD medium, and immediately observed. To visualize the vacuole lumen, cells were stained with CellTracker Blue CMAC (Invitrogen) according to the manufacturer's protocol and observed using a UV-1A filter set.
Most GFP- or mRFP1-tagged proteins were observed in living cells, which were grown to early to mid-logarithmic phase, harvested, and resuspended in SD medium. Cells were immediately observed using a GFP bandpass (for GFP) or a G2-A (for mRFP1) filter set. Localization of GFP- or mRFP1-tagged proteins was also examined in fixed cells. Fixation was performed for 5 min at 37°C by direct addition of a commercial 37% formaldehyde stock (Wako Pure Chemical Industries, Osaka, Japan) to a final concentration of 0.5% in the medium, followed by a 10-min incubation at 25°C. After fixation, cells were washed twice with phosphate-buffered saline and examined. The cell fixation protocol resulted in images of the GFP- and mRFP1-fusion proteins that were similar to those obtained with living cells (our unpublished observation).
Electron Microscopy (EM)
Ultrastructural observation of cells by conventional EM was performed using the glutaraldehyde-permanganate fixation technique (Kaiser and Schekman, 1990). Cells were embedded in Spurr's resin (Nissin EM, Tokyo, Japan). Thin sections (50–60 nm) were cut on an Ultracut microtome (Leica, Wetzlar, Germany) equipped with a Sumiknife (Sumitomo Electric Industries, Osaka, Japan), stained with 3% uranyl acetate and Reynold's lead citrate, and viewed using an H-7100 electron microscope (Hitachi) at 75 kV. Immuno-EM was performed using the aldehyde fixation/metaperiodate permeabilization method (Mulholland and Botstein, 2002), except that glutaraldehyde was not included in the fixative. Cells were embedded in LR White resin (medium grade; London Resin Company, Berkshire, United Kingdom) and sectioned as described above. Mouse anti-HA antibody (HA.11) was used as a primary antibody at a 1:500 dilution. Ten-nanometer gold-conjugated anti-mouse IgG antibodies (BBInternational, Cardiff, United Kingdom) were preadsorbed with fixed wild-type cells and used as secondary antibodies at a 1:100 dilution. Samples were poststained with uranyl acetate and viewed as described above.
35S Pulse-Chase and Immunoprecipitation Experiments
Metabolic labeling of yeast cells, preparation of cell extracts, and immunoprecipitation were performed essentially as described previously (Rothblatt and Schekman, 1989; Gaynor et al., 1994; Yahara et al., 2001). To visualize target proteins, total proteins or immunoprecipitated proteins were resolved by SDS-PAGE followed by analysis using a FLA3000 fluorescent image analyzer (Fuji Photo Film, Tokyo, Japan).
To assay internal and external invertase, cells were grown to mid-logarithmic phase in MV-low sulfate medium containing 5% glucose at 37°C for 2 h. Three (labeled for 7 min) or five (labeled for 2 min) OD600 units of cells were collected, converted to spheroplasts with Zymolyase 100T (25 U/ml cell suspension; Seikagaku Kogyo, Tokyo, Japan), and incubated at 37°C for 30 min. Spheroplasts were resuspended in MV-no sulfate medium containing 1 M sorbitol and 0.1% glucose, grown at 37°C for 30 min, and labeled with 50 μCi of Tran35S-label (PerkinElmer Life and Analytical Sciences, Boston, MA) per OD600 unit of cells for 7 or 2 min. A chase was initiated by adding a chase solution (MV-no sulfate medium with 4 mM ammonium sulfate, 0.012% l-cysteine, and 0.016% l-methionine) containing 0.1% glucose and 1 M sorbitol to an equal volume of the samples. After the pulse-chase, ice-cold sodium azide was added to spheroplast suspensions to a final concentration of 10 mM, and the suspensions were separated by centrifugation into spheroplasts and media as the intracellular and extracellular fractions, respectively. Both fractions were subjected to immunoprecipitation using rabbit anti-invertase antibodies.
To assay total proteins secreted into the medium, cells were grown to mid-logarithmic phase in MV-low sulfate medium containing 5% glucose at 37°C for 2 h. Three OD600 units of cells were collected, resuspended in MV-no sulfate medium containing 2% glucose and 0.03% bovine serum albumin, and grown at 37°C for 30 min. The cells were labeled with 100 μCi of Tran35S-label per OD600 unit of cells at 37°C for 15 min. A chase was initiated by adding the chase solution containing 2% glucose and 0.5% casamino acids to an equal volume of the samples. After a 45-min chase, cell suspensions were treated with sodium azide and separated into intracellular and extracellular fractions as described above. Trichloroacetic acid (TCA) was added to both fractions to a final concentration of 10%, and the fractions were kept on ice for 15 min, followed by centrifugation at 20,630 × g for 15 min. After one wash with 10% TCA and two washes with ice-cold acetone, pellets were solubilized in SDS-PAGE sample buffer.
To examine the sorting pathway for CPY, cells were pulse chased as described above. After a chase, cells were suspended in spheroplast stop solution (1 M sorbitol, 25 mM Tris-HCl, pH 7.5, 20 mM NaN3, 10 mM dithiothreitol, and 20 mM NaF) and treated with 2.5 U/ml Zymolyase 100T. Intracellular and extracellular fractions were prepared as in the assay for invertase and subjected to immunoprecipitation by using rabbit anti-CPY antibodies.
RESULTS
Cdc50 Family Proteins Form Complexes with Drs2 Family Proteins
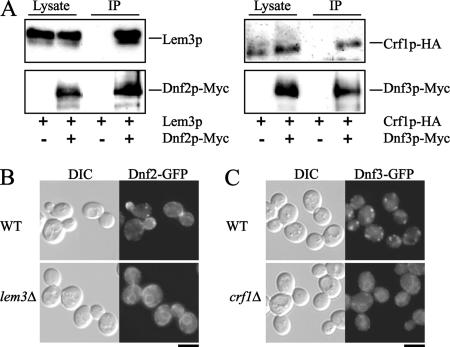
In a previous study, we demonstrated that Cdc50p and Lem3p associate with Drs2p and Dnf1p, respectively (Saito et al., 2004). Dnf1p and its close homologue Dnf2p (83% similar and 69% identical at the amino acid sequence level) exhibit redundant activities in phospholipid translocation across the plasma membrane (Pomorski et al., 2003; our unpublished data). Therefore, we examined whether Lem3p physically associates with Dnf2p in coimmunoprecipitation experiments. We constructed a gene that encoded a C-terminally Myc-tagged version of Dnf2p and introduced it into cells. Dnf2p-Myc was immunoprecipitated with an anti-Myc antibody from membrane protein extracts prepared by solubilization in 1% CHAPS. Lem3p was coimmunoprecipitated with Dnf2p-Myc, whereas Lem3p was not detected in immunoprecipitates from cells lacking DNF2-Myc (Figure 1A). In addition, Dnf2p-EGFP as well as Dnf1p-EGFP was localized in the ER in the lem3Δ mutant (Figure 1B). Neither Cdc50p nor Crf1p-HA was coimmunoprecipitated with Dnf2p-Myc, and Dnf2p-EGFP was localized to the plasma membrane in cdc50Δ and crf1Δ mutants (our unpublished data). Both Crf1p and Dnf3p colocalize with markers for early/late endosomes or the TGN, and deletion of either CRF1 or DNF3 resulted in no discernible phenotypes (Hua et al., 2002; Pomorski et al., 2003; our unpublished observation). We examined whether Crf1p binds to Dnf3p in coimmunoprecipitation experiments. We created cells expressing both a C-terminally Myc-tagged version of DNF3 and an HA-tagged version of CRF1. Crf1p-HA was coimmunoprecipitated with Dnf3p-Myc, whereas Crf1p-HA was not detected in immunoprecipitates from cells lacking DNF3-Myc (Figure 1A). In addition, Dnf3p-EGFP was localized in the ER of the crf1Δ mutant (Figure 1C). Neither Cdc50p nor Lem3p was coimmunoprecipitated with Dnf3p-Myc, and the localization patterns of Dnf3p-EGFP in cdc50Δ and lem3Δ mutants were identical to that in wild-type cells (our unpublished data). In addition, neither Drs2p-Myc nor Dnf1p-Myc coimmunoprecipitated with Crf1p-HA, and the localization patterns of Drs2p-EGFP and Dnf1p-EGFP in the crf1Δ mutant were identical to that in wild-type cells (our unpublished data). These results, together with our previous study (Saito et al., 2004), demonstrate that Cdc50 family proteins (Cdc50p, Lem3p, and Crf1p) form complexes with Drs2 family proteins (Drs2p, Dnf1p and Dnf2p, and Dnf3p, respectively) in vivo.
Figure 1.
Cdc50 family proteins form complexes with Drs2 family proteins. (A) Coimmunoprecipitation of Lem3p and Crf1p with Dnf2p and Dnf3p, respectively. Cells were grown at 25°C to a cell density of 0.5 OD600/ml in YPDA medium. Membrane extracts were then prepared as described in Materials and Methods. Myc-tagged Dnf2p or Dnf3p was immunoprecipitated with an anti-Myc antibody from these extracts. Immunoprecipitates were subjected to SDS-PAGE, followed by immunoblot analysis using antibodies against Lem3p or HA (top) and Myc (bottom). The results shown are representative of several experiments. The strains used were as follows: YKT1099 (DNF2-Myc LEM3) and YKT1098 (DNF2 LEM3) (left) and YKT1100 (DNF3-Myc CRF1-HA) and YKT1098 (DNF3 CRF1-HA) (right). (B) Localization of Dnf2p-EGFP. Wild-type (YKT921) and lem3Δ (YKT923) cells containing the DNF2-EGFP construct in the genome were grown to early to mid-logarithmic phase in YPDA medium at 30°C and immediately observed by fluorescence microscopy. (C) Localization of Dnf3p-EGFP. Wild-type (YKT925) and crf1Δ (YKT928) cells containing the DNF3-EGFP construct in the genome were grown and observed as described in (B). Bars, 5 μm.
Isolation of YPT32 as a Multicopy Suppressor of a cdc50-ts Mutation
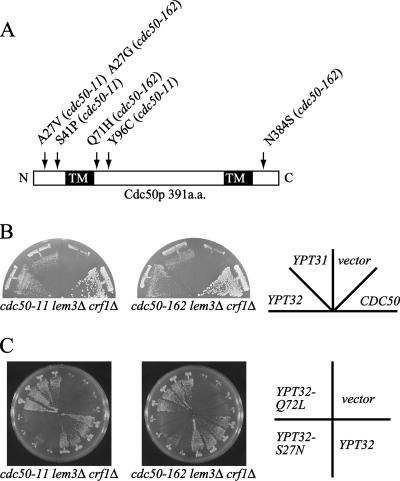
To facilitate studies of the essential functions of putative PLTs composed of members of the Cdc50p and Drs2p families except for Neo1p, we constructed ts mutants of CDC50 in the lem3Δ crf1Δ background (hereafter referred to as the cdc50-ts mutants). We obtained two mutants, cdc50-11 lem3Δ crf1Δ and cdc50-162 lem3Δ crf1Δ (hereafter referred to as the cdc50-11 and cdc50-162 mutants, respectively), that grew well at 25°C, but exhibited a tight ts growth defect at 37°C. DNA sequencing of the two mutant alleles revealed that each allele contained three mutations that resulted in amino acid substitutions (A27V, S41P, and Y96C in cdc50-11 and A27G, Q71H, and N384S in cdc50-162) (Figure 2A). All substitution sites except for N384S were located in the N-terminal quarter of the protein. The alanine residue at position 27, a residue that was mutated in both alleles, is conserved among Cdc50p homologues in Caenorhabditis elegans, Drosophila melanogaster, and Homo sapiens.
Figure 2.
Overexpression of YPT32 or YPT31 suppresses the temperature-sensitive growth defect of the cdc50-ts mutants. (A) The domain structure of Cdc50p and the amino acid substitutions in Cdc50-11p and Cdc50-162p. The black boxes indicate potential transmembrane domains. (B) Suppression of the cdc50-ts mutations by multicopy YPT32 or YPT31. cdc50-11 (YKT993) and cdc50-162 (YKT942) cells were transformed with pKT1555 (YEplac181-YPT32), pKT1554 (YEplac181-YPT31), pKT1259 (YEplac181-CDC50), or a control vector (YEplac181). Transformants were streaked onto a YPDA plate, followed by incubation at 37°C for 2 d. (C) The GTP-bound form, but not the GDP-bound form, of Ypt32p suppresses the ts growth defect of cdc50-ts mutants. cdc50-11 (YKT993) and cdc50-162 (YKT942) mutant cells were transformed with pKT1578 (YEplac181-YPT32Q72L), pKT1579 (YEplac181-YPT32S27N), pKT1555 (YEplac181-YPT32), or a control vector (YEplac181). For each plasmid, four independent transformants were streaked onto a YPDA plate, followed by incubation at 37°C for 3 d.
We examined the growth profiles of the cdc50-ts mutants after a shift to 37°C. We counted the number of cells at 1, 2, 3, and 4 h after the shift and found that cdc50-ts mutants rapidly stopped growing at 37°C (Supplemental Figure S1A). Because Drs2p-EGFP was localized in the ER in the cdc50Δ mutant (Saito et al., 2004), we next examined the localization of Drs2p-EGFP in the cdc50-11 mutant after the shift to 37°C. At 25°C (0 min), Drs2p-EGFP was observed in punctate structures scattered throughout the cell and partially localized to the ER, although the fluorescence of Drs2p-EGFP was much less clear in the cdc50-11 mutant than in wild-type cells (Supplemental Figure S1B). When incubated at 37°C for 1 h, there was a substantial decrease of Drs2p-EGFP observed in punctate structures and in the ER in the cdc50-11 mutant. After a 2-h incubation at 37°C, fluorescence of Drs2p-EGFP was barely detectable, although the very faint fluorescence was localized to the ER and a few large compartments located in the bud or near the bud neck (Supplemental Figure S1B). These results suggested that the Cdc50p–Drs2p complex was rapidly inactivated or degraded at 37°C. We analyzed phenotypes of the cdc50-ts mutants after 2- to 3-h incubation at 37°C in further experiments.
To obtain clues to essential functions of the CDC50 and DRS2 families, we screened for multicopy suppressors of the growth defect of the cdc50-ts mutants at 37°C. We found that overexpression of YPT32 suppressed, albeit weakly, the growth defect of the cdc50-ts mutants (Figure 2B). Ypt32p, a Rab family small GTPase, shares a high degree of sequence similarity (81% identity and 90% similarity) with Ypt31p, overproduction of which also weakly suppressed the growth defect of the cdc50-ts mutants (Figure 2B). Overexpression of YPT32Q72L, which mimics a GTP-bound form of the protein, but not of YPT32S27N, which mimics a GDP-bound form, suppressed the ts growth phenotype of the cdc50-ts mutants to the same extent as overexpression of YPT32 (Figure 2C), suggesting that YPT32 suppressed the cdc50-ts mutations by hyperactivation of its downstream pathway through its effectors. Previous studies have reported that YPT31/32 are involved in intra-Golgi transport, the formation of transport vesicles at the TGN (Benli et al., 1996; Jedd et al., 1997), and the recruitment of Sec2p, the guanine nucleotide exchange factor for Sec4p, to secretory vesicles (Ortiz et al., 2002). Therefore, we investigated the vesicular trafficking phenotypes in the cdc50-ts mutants.
The Secretory Pathway Seems Normal in a cdc50-ts Mutant
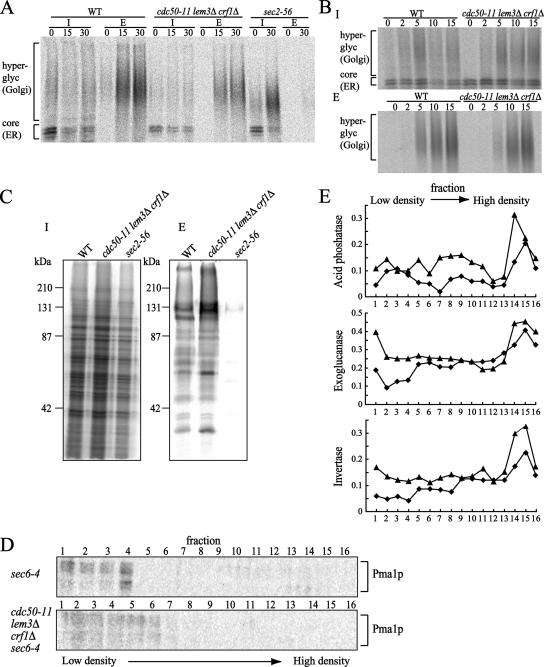
Because YPT31/32 have been shown to be required for exit from the TGN in the exocytic pathway (Benli et al., 1996; Jedd et al., 1997) and the drs2Δ mutant exhibits defects in Golgi function (Chen et al., 1999; Hua et al., 2002), we investigated defects in the secretory pathways of the cdc50-11 mutant. We first tested for the secretion of the periplasmic enzyme invertase. Because invertase acquires N-linked oligosaccharide chains, which are heterogeneously modified in the Golgi, it is secreted as a highly glycosylated protein and thus occurs as a high-molecular-mass smear after SDS-PAGE. In secretory mutants, invertase secretion is inhibited and the enzyme accumulates in the lumen of secretory compartments. Cells were incubated at 37°C for 2.5 h, induced to produce invertase for 30 min, pulse labeled for 7 min with Tran35S-label, and chased at 37°C. After 15 min of chase, wild-type cells secreted most of the invertase as a fully glycosylated form. In the cdc50-11 mutant, invertase was also secreted in a fully glycosylated form with kinetics similar to those of wild-type cells, whereas with a control secretory mutant, sec2-56 (Novick et al., 1980), invertase remained intracellular even after a 30-min chase (Figure 3A). To examine a kinetic delay of invertase secretion more precisely, cells were pulse labeled for 2 min and chased for 2, 5, 10, and 15 min. In the cdc50-11 mutant, invertase was secreted in a fully glycosylated form with kinetics similar to that of wild-type cells after a 10-min chase, although a slight delay was observed in the external appearance of invertase after a 5-min chase (Figure 3B).
Figure 3.
Secretion and formation of secretory vesicles are nearly normal in a cdc50-ts mutant. (A and B) Pulse-chase experiments of invertase secretion. Wild-type (YKT38), cdc50-11 (YKT993), and sec2-56 (ANS2-3A) cells (A) or wild-type (YKT38) and cdc50-11 (YKT993) cells (B) were grown for 2.5 h at 37°C, induced to produce invertase for 30 min, pulse labeled with Tran35S-label for 7 min (A) or 2 min (B), and chased for the indicated time at 37°C. Samples were separated by centrifugation into internal (I) and external (E) fractions. Invertase was recovered by immunoprecipitation and visualized by SDS-PAGE and a phosphorimager system. (C) General secretion by the cdc50-11 mutant. Wild-type (YKT38), cdc50-11 (YKT993), and sec2-56 (ANS2-3A) cells were grown for 2.5 h at 37°C, pulse labeled with Tran35S-label for 15 min, and chased for 45 min. Samples were separated by centrifugation into I and E fractions. Proteins in each fraction were precipitated with TCA and visualized by SDS-PAGE and a phosphorimager system. (D) Western blots for Pma1p, a marker for low-density vesicles, in Nycodenz gradient fractions. sec6-4 (YKT1010) and cdc50-11 sec6-4 (YKT1011) cells transformed with pKT1486 (PACT1-SUC2) were incubated for 2 h at 37°C. Secretory vesicles were prepared and fractionated as described in Materials and Methods. Numbered fractions (as indicated at the top) were heated in sample buffer for 15 min at 37°C, separated by SDS-PAGE, and probed with antibodies against Pma1p. (E) Activities of marker enzymes for high-density vesicles in Nycodenz gradient fractions. Fractions were prepared from sec6-4 (diamonds) and cdc50-11 sec6-4 (triangles) cells as described in D. Hydrolyzing enzyme activities are expressed in arbitrary units based on the absorbance measured at 415 nm (acid phosphatase, top; exoglucanase, middle) or at 540 nm (invertase, bottom).
To test whether the cdc50-ts mutants exhibited a cargo-specific defect in protein transport, we next examined general secretion in the cdc50-11 mutant. Cells were incubated at 37°C for 2.5 h, pulse labeled with Tran35S-label for 15 min, and chased for 45 min. Cells and media were separated by centrifugation into the intracellular and extracellular fractions, respectively. Judging from the band intensities in the intracellular fractions, the overall level of protein synthesis was similar among wild-type, cdc50-11, and sec2-56 cells. In the extracellular fractions, wild-type and cdc50-11 mutant cells exhibited similar levels of protein secretion into the medium, whereas the sec2-56 mutant secreted almost no proteins (Figure 3C). These results suggest that secretion from the cdc50-11 mutant was nearly normal.
Harsay and Bretscher (1995) used the ts late-acting sec mutant sec6-4 to demonstrate that newly synthesized secretory proteins are exported via at least two different classes of secretory vesicles, which have similar diameters, but differ in their densities. The high-density vesicles (HDSVs) contain the soluble, secreted enzymes invertase and acid phosphatases together with most of the exoglucanase activity, whereas the low-density vesicles (LDSVs) contain the plasma membrane H+-ATPase Pma1p (Harsay and Bretscher, 1995; David et al., 1998). Disruption of a gene encoding a dynamin-related protein (VPS1) or the clathrin heavy chain (CHC1) abolishes the production of HDSVs, yielding LDSVs that contain all the secreted cargo (Gurunathan et al., 2002). Because DRS2 was implicated in the formation of clathrin-coated vesicles (Chen et al., 1999; Gall et al., 2002), we examined whether the production of these two types of secretory vesicles was affected in the cdc50-11 mutant. We created a quadruple cdc50-11 lem3Δ crf1Δ sec6-4 mutant (cdc50-11 sec6-4 mutant) and purified secretory vesicles by Nycodenz density gradient centrifugation of membranes prepared from temperature-shifted cdc50-11 sec6-4, or sec6-4 mutant cells. Western blots of the gradient fractions indicated that Pma1p levels were highest in the lower density membranes from the cdc50-11 sec6-4 mutant as well as from the sec6-4 mutant (Figure 3D), whereas the activities of invertase, acid phosphatase, and exoglucanase were found in the higher density fractions from both the cdc50-11 sec6-4 and sec6-4 mutants (Figure 3E). We also carried out the density gradient purification of membranes from sec6-4 and cdc50-11 sec6-4 cells grown at the permissive temperature (25°C). Pma1p was not detected by Western blots of any gradient fractions from sec6-4 and cdc50-11 sec6-4 cells (our unpublished data). Similarly, activities of invertase, acid phosphatase, and exoglucanase were not found in any fractions from both the sec6-4 and sec6-4 cdc50-11 mutants (our unpublished data). These results suggested that the activities of invertase, acid phosphatase, and exoglucanase and the Pma1p levels observed in the density gradient fractions from both strains incubated at 37°C reflected the high- and low-density classes of exocytic vesicles, respectively. These results suggest that the cdc50-11 mutant did not exhibit major defects in production of these two types of secretory vesicles.
The Endocytic Pathway to the Vacuole and the Vacuolar Protein Sorting Pathway Seems Normal in the cdc50-ts Mutants
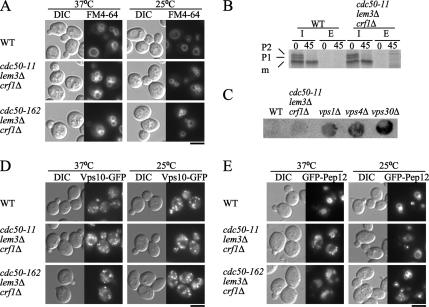
Endocytic transport from the plasma membrane to the vacuole was previously examined in dnf and drs2 mutants. Defective endocytic internalization of the lipophilic dye FM4-64 and α-mating factor was demonstrated in the dnf1Δ dnf2Δ drs2Δ mutant (Pomorski et al., 2003), whereas the drs2Δ, cdc50Δ, and dnf1Δ dnf2Δ dnf3Δ mutants were shown to be defective in a certain step in the postinternalization transport to the vacuole (Chen et al., 1999; Hua et al., 2002; Misu et al., 2003). All these assays were conducted at lower temperatures such as 15 or 18°C. The cdc50-ts mutants, however, internalized and delivered FM4-64 to the vacuole at 37°C during a 30-min incubation (Figure 4A). Although the sizes of the vacuoles visualized by FM4-64 staining were somewhat smaller in the cdc50-ts mutants, this phenotype was not affected by the incubation temperature. These results suggest that the essential function of the putative PLTs at higher temperatures is not in endocytic transport from the plasma membrane to the vacuole.
Figure 4.
Normal endocytic and VPS pathways in the cdc50-ts mutants. (A) Internalization and transport of FM4-64 to the vacuole. Wild-type (YKT38), cdc50-11 (YKT993), and cdc50-162 (YKT942) cells grown for 3 h were stained with FM4-64 for 15 min in YPDA medium, and chased for 30 min in fresh YPDA medium, at the indicated temperature. Bar, 5 μm. (B) Pulse-chase experiments of CPY intracellular transport. Wild-type (YKT38) and cdc50-11 (YKT993) cells were grown for 2.5 h at 37°C, pulse labeled with Tran35S-label for 15 min, and chased for 45 min at 37°C. CPY was immunoprecipitated from I and E fractions, resolved by SDS-PAGE, and visualized using a phosphorimager system. (C) Secretion of CPY. Wild-type (YKT38), cdc50-11 (YKT993), vps1Δ (KKT276), vps4Δ (KKT277), and vps30Δ (AKY15) cells were grown for 3 h at 37°C in contact with a nitrocellulose filter, and secreted CPY was detected by probing with antibodies against CPY. (D) Localization of Vps10p-EGFP. Wild-type (YKT957), cdc50- 11 (YKT1086), and cdc50-162 (YKT1088) cells containing the VPS10-EGFP construct in the genome were grown for 3 h at 25 or 37°C in SD medium. Bar, 5 μm. (E) Localization of GFP-Pep12p. Wild-type (YKT38), cdc50-11 (YKT993), and cdc50-162 (YKT942) cells transformed with pKT1487 (pRS416-GFP-PEP12) were grown for 3 h at 25°C or 37°C in SDA-Ura medium. Bar, 5 μm.
Next, we assessed the vacuolar protein sorting (VPS) pathway by monitoring the maturation of a vacuolar soluble protein, CPY. Previous studies showed that drs2Δ, cdc50Δ, and dnf1Δ drs2Δ mutants exhibited a kinetic defect in CPY transport to the vacuole at lower temperatures (Chen et al., 1999; Hua et al., 2002; Misu et al., 2003). Wild-type and cdc50-11 mutant cells were preincubated for 2.5 h, labeled with Tran35S-label for 15 min, and chased for 45 min at 37°C. After a 15-min pulse, wild-type cells contained two precursor forms of CPY: a 67-kDa species (P1) in the ER and a fully glycosylated 69-kDa form (P2) in the Golgi. After a 45-min chase, precursors were cleaved to the 65-kDa mature form (m) after delivery to the vacuole by proteinase A (Stevens et al., 1982). Analysis of the cdc50-11 mutant showed that transport from the ER to the Golgi and from the Golgi to the vacuole occurred at normal rates (Figure 4B). Some class E vps mutants are capable of forming mature CPY at nearly normal rates despite a defect in the VPS pathway, because vacuolar proteases are mature in the class E compartment. The class E vps mutants, however, secrete a significant fraction of CPY into the medium (Raymond et al., 1992), whereas the cdc50-11 mutant did not secrete CPY into the extracellular space (Figure 4B). This result was confirmed by colony blot assays (Roberts et al., 1991; Wiederkehr et al., 2000), by which secreted CPY can be detected. In contrast to vps1Δ, vps30Δ (class A) and vps4Δ (class E), the cdc50-11 mutant did not secrete significant amounts of CPY (Figure 4C). These results suggest that the VPS pathway is not affected in the cdc50-11 mutant at higher temperatures.
The membrane-bound CPY-sorting receptor Vps10p has been reported to cycle between the TGN and late endosomes (Cereghino et al., 1995; Cooper and Stevens, 1996), and a complex called the “retromer,” which comprises five proteins, is required for the endosome-to-TGN retrieval of Vps10p (Seaman et al., 1997). Thus, the removal of components of the retromer results in missorting of Vps10p to the vacuole (Seaman et al., 1997). We created cells expressing a C-terminally EGFP-tagged version of VPS10 and observed the localization of Vps10p-EGFP. In wild-type cells, Vps10p-EGFP resulted in a punctate fluorescence pattern characteristic of TGN-localized yeast proteins (Figure 4D) as described previously (Cereghino et al., 1995; Cooper and Stevens, 1996). Similarly, Vps10p-EGFP created a punctate fluorescence pattern in the cdc50-11 and cdc50-162 mutants incubated at 37°C as well as 25°C (Figure 4D), suggesting that Vps10p-EGFP was normally retrieved from late endosomes in the cdc50-ts mutants. These results are consistent with our observation that CPY sorting was normal in the cdc50-11 mutant (Figure 4, B and C).
We also analyzed the localization of the endosomal syntaxin Pep12p. Whereas endogenous Pep12p typically occurs as scattered or perivacuolar puncta (Lewis et al., 2000), GFP-Pep12p, the expression of which was driven by the strong TPI1 promoter, reached the vacuolar outer membrane via late endosomes (Black and Pelham, 2000). We observed the localization of GFP-Pep12p expressed under the control of the TPI1 promoter in the cdc50-ts mutants. In wild-type cells and the cdc50-ts mutants, GFP-Pep12p was found in small punctate structures and vacuolar membranes at 25 and 37°C (Figure 4E). The small punctate structures seemed to be late endosomes, because they were much smaller than the vacuoles. These results suggest that in the cdc50-ts mutants, GFP-Pep12p was normally transported to late endosomes, and overexpressed GFP-Pep12p was further delivered to the vacuole.
Together, our results suggest that the cdc50-ts mutants do not have major defects in the endocytic pathway to the vacuole, the VPS pathway, or the recycling pathway between the TGN and late endosomes. Thus, PLTs composed of proteins from the Cdc50p and Drs2p families are unlikely to be essential for membrane trafficking via these pathways at higher temperatures.
cdc50-ts Mutants Show Mislocalization of Recycling Proteins
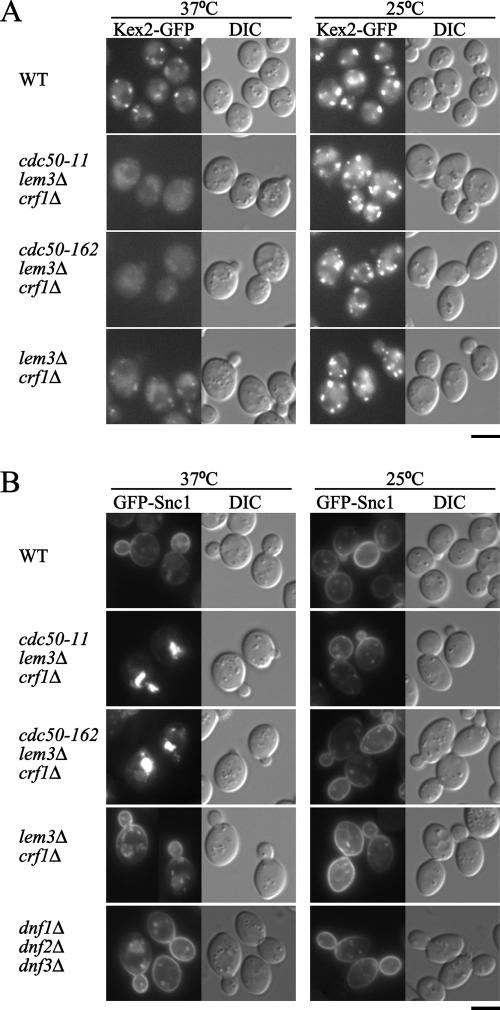
During the course of this study, Segev and colleagues reported that Ypt31p/32p are also involved in the retrieval of the furin homologue Kex2p and the v-SNARE Snc1p from endosomes to the TGN (Chen et al., 2005). Thus, we examined localization of GFP-tagged versions of these proteins in the cdc50-ts mutants. Kex2p is a TGN resident protein that is localized due to constant retrieval from late and early endosomes (Brickner and Fuller, 1997; Lewis et al., 2000). At 25°C, C-terminally EGFP-tagged Kex2p resulted in cytoplasmic fluorescent puncta typical of proteins that are localized in the yeast TGN (Figure 5A). We also observed weak fluorescent signals that seem to correspond to vacuoles as judged by colocalization with CellTracker Blue CMAC, a dye that stains the vacuolar lumen (Supplemental Figure S2). In lem3Δ crf1Δ mutant cells incubated at 37°C, punctate localization patterns of Kex2p-GFP were observed as in wild-type cells, although they were less clear. In contrast, in the cdc50-ts mutants incubated at 37°C, Kex2p-EGFP was primarily localized in vacuoles (Figure 5A and Supplemental Figure S2), as was demonstrated in the ypt31Δ ypt32-ts mutant (Chen et al., 2005). Similar to Vps10p, Kex2p mislocalizes to the vacuole when cycling between endosomes and the TGN is impaired (Wilcox et al., 1992; Cooper and Stevens, 1996; Conibear and Stevens, 2000). Because Vps10p exhibited the normal punctate localization in the cdc50-ts mutants (Figure 4D), our results suggest that the cdc50-ts mutants are defective in the retrieval pathway from early endosomes.
Figure 5.
Recycling marker proteins are mislocalized in the cdc50-ts mutants. (A) Localization of Kex2p-EGFP. Wild-type (YKT903), cdc50-11 (YKT1000), cdc50-162 (YKT1001), and lem3Δ crf1Δ (YKT1310) cells containing the KEX2-EGFP construct in the genome were grown for 3 h at 25 or 37°C in SD medium. (B) Localization of GFP-Snc1p. Wild-type (YNF63), cdc50-11 (YNF65), cdc50-162 (YNF67), lem3Δ crf1Δ (YNF61), and dnf1Δ dnf2Δ dnf3Δ (YNF784) cells carrying the pRS416-GFP-SNC1 plasmid were grown for 3 h at 25 or 37°C in SDA-Ura medium. Bars, 5 μm.
Snc1p is recycled from the plasma membrane via early endosomes to the TGN (Lewis et al., 2000). Previous studies reported that Snc1p was mislocalized in the cdc50Δ, drs2Δ, and dnf1Δ dnf2Δ dnf3Δ mutants (Hua et al., 2002; Saito et al., 2004). In wild-type, lem3Δ crf1Δ, and cdc50-ts mutant cells at 25°C, GFP-Snc1p was primarily localized to the plasma membrane at a polarized site, such as a growing bud or a cytokinesis site, with a small fraction of GFP-Snc1p observed in intracellular punctate structures (Figure 5B). In the lem3Δ crf1Δ mutants at 37°C, GFP-Snc1p was also localized to the plasma membrane at a polarized site, although GFP-Snc1p in internal structures slightly increased (Figure 5B). In contrast, in the cdc50-ts mutants at 37°C, GFP-Snc1p accumulated in a few large compartments located in the bud or near the bud neck, with a concomitant decrease in the amount of the protein observed at the plasma membrane (Figure 5B). These results suggested that the lem3Δ crf1Δ double mutations did not cause the mislocalization of GFP-Snc1p but the combination of the inactivation of Cdc50p and the double mutations did. We also examined the localization of GFP-Snc1p in the dnf1Δ dnf2Δ dnf3Δ mutant. The localization pattern of GFP-Snc1p in the dnf1Δ dnf2Δ dnf3Δ mutant was very similar to that in the lem3Δ crf1Δ mutant both at 25 and 37°C (Figure 5B). This result is consistent with our conclusion that Lem3p and Crf1p interact with Dnf1p/2p and Dnf3p, respectively, and that the lem3Δ crf1Δ double mutations inactivate the Dnf1p, Dnf2p, and Dnf3p activity. However, our result that the dnf1Δ dnf2Δ dnf3Δ mutant did not exhibit a major defect for the localization of GFP-Snc1p is inconsistent with a previous report by Hua et al. (2002), which showed that a substantial fraction of GFP-Snc1p was intracellularly localized. This contradiction may be due to differences of the strain background.
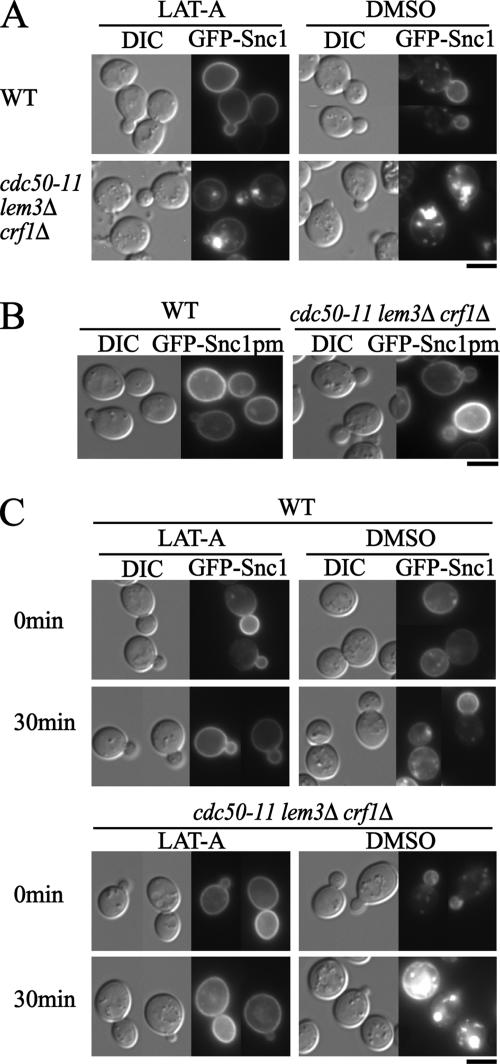
The atypical labeling of intracellular structures with GFP-Snc1p in the cdc50-ts mutants could result from defects in the transport pathway from the TGN to the plasma membrane or in that from the plasma membrane to the TGN via endosomes. When cdc50-11 cells preincubated at 37°C for 3 h were treated with 100 μM of the actin assembly inhibitor latrunculin A (LAT-A) for 10 min at 37°C to block endocytosis, GFP-Snc1p was observed to be localized to the plasma membrane in addition to intracellular large structures (Figure 6A); the percentages of cdc50-11 cells with GFP-Snc1p at the plasma membrane increased from 42% (n = 183) (dimethyl sulfoxide [DMSO] control) to 83% (n = 133) (LAT-A treatment). These results suggested that newly synthesized GFP-Snc1p reached the plasma membrane in the mutant cells. Consistent with this result, a mutant form of GFP-Snc1p, GFP-Snc1p-pm, which can reach the plasma membrane but cannot be internalized by endocytosis (Lewis et al., 2000), was exclusively localized to the plasma membrane in the cdc50-11 mutant as well as wild-type cells at 37°C (Figure 6B). These results also suggest that GFP-Snc1p needs to be endocytosed to be incorporated into the large intracellular structures. To confirm this with GFP-fused wild-type Snc1p, cells grown at 25°C were pretreated with 100 μM LAT-A for 10 min at 25°C (0 min), followed by incubation at 37°C for 30 min in the presence of 100 μM LAT-A (30 min) (Figure 6C). LAT-A treatment inhibited the intracellular accumulation of GFP-Snc1p in the cdc50-11 mutant. These results suggest that cdc50-11 mutant cells are defective in the plasma membrane-to-TGN transport pathway but not in the TGN-to-plasma membrane transport pathway.
Figure 6.
The cdc50-ts mutant is defective in plasma membrane-to-TGN transport. (A) Plasma membrane localization of GFP-Snc1p is restored in the cdc50-ts mutant by blocking endocytosis with LAT-A. Wild-type (YNF63) and cdc50-11 (YNF65) cells carrying the pRS416-GFP-SNC1 plasmid were preincubated at 37°C for 3 h and then treated with 100 μM LAT-A or DMSO (vehicle control) for 10 min at 37°C in SDA-Ura medium. (B) Localization of GFP-Snc1p-pm. Wild-type (YKT38) and cdc50-11 (YKT993) cells transformed with pRS416-GFP-SNC1 pm were grown to early logarithmic phase at 25°C and shifted to 37°C for 3 h in SDA-Ura medium. (C) Pretreatment with LAT-A inhibits abnormal accumulation of GFP-Snc1p in the cdc50-ts mutant. Wild-type (YNF63) and cdc50-11 (YNF65) cells carrying the pRS416-GFP-SNC1 plasmid were grown to early logarithmic phase at 25°C and were then pretreated with 100 μM LAT-A or DMSO (vehicle control) for 10 min at 25°C in SDA-Ura medium (0 min), followed by incubation at 37°C for 30 min in SDA-Ura medium in the presence (LAT-A) or absence (DMSO) of 100 μM LAT-A (30 min). Bars, 5 μm.
Together, our results implicate putative PLTs in the retrieval pathway from early endosomes to the TGN. Ypt31p/32p and putative PLTs may functionally interact in this pathway.
Given that Snc1p accumulates in intracellular compartments in ypt31Δ ypt32-ts mutant cells (Chen et al., 2005), we tested for the effects of overproduction of the GTP-bound (Q72L) or GDP-bound (S27N) form of Ypt32p (Figure 2C) on the localization of GFP-Snc1p. In cells overexpressing YPT32Q72L, GFP-Snc1p was primarily localized to the plasma membrane of the growing site as it was in wild-type cells. In contrast, GFP-Snc1p accumulated in abnormal compartments located in the bud or near the bud neck in ∼15% of the cells carrying YPT32S27N (n = 149) (Supplemental Figure S3), similar to the results observed with the cdc50-ts mutants. Because Ypt31p/32p were implicated in transport out of the TGN (Benli et al., 1996; Jedd et al., 1997), we examined whether GFP-Snc1p-pm reached the plasma membrane in the mutant strains. GFP-Snc1p-pm was exclusively localized to the plasma membrane in cells carrying YPT32S27N or YPT32Q72L (Supplemental Figure S3). These results also suggest that YPT32 is involved in endocytic recycling.
We next examined whether overexpression of YPT32 suppressed mislocalization of GFP-Snc1p in the cdc50-ts mutants. When YPT32 was overexpressed at 37°C, the percentages of cdc50-11 and cdc50-162 mutant cells with GFP-Snc1p at sites with polarized plasma membranes increased from 39% (n = 117) to 60% (n = 120) and from 43% (n = 170) to 55% (n = 141), respectively (our unpublished data). Similarly, the percentages of cdc50-11 and cdc50-162 mutant cells with GFP-Snc1p in large compartments at 37°C slightly decreased from 44% (n = 119) to 35% (n = 132) and from 38% (n = 120) to 28% (n = 140), respectively (our unpublished data). These results are consistent with a weak suppression of the growth defect in the cdc50-ts mutants by overexpression of YPT32 (Figure 2B). In addition, overexpression of YPT32 did not suppress the cold-sensitive growth phenotype of the cdc50Δ mutant at 18°C (our unpublished data). These results suggest that YPT32 is not a bypass suppressor of the cdc50 mutations and thus raise the possibility that YPT32 requires the residual PLT activity of Cdc50p-Drs2p for the suppression.
In the cdc50-11 Mutant, GFP-Snc1p and GFP-Tlg1p Seem to Accumulate in Early Endosome-derived Structures
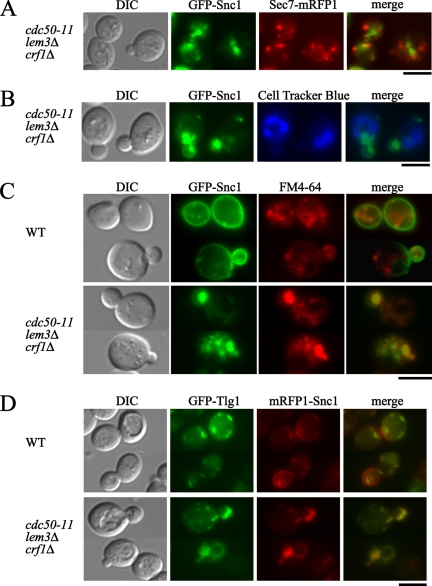
We morphologically examined the Snc1p-containing structures in the cdc50-11 mutant. To examine whether the large structures were associated with the TGN, the TGN was visualized with a TGN marker, C-terminally mRFP1-tagged Sec7p (Franzusoff et al., 1991), in cdc50-11 mutant cells expressing GFP-SNC1. Although Sec7p-mRFP1 was observed in punctate structures scattered throughout wild-type and cdc50-11 mutant cells at 37°C, few of the GFP-Snc1p–positive structures were colocalized with Sec7p-mRFP1 (Figure 7A), suggesting that the large GFP-Snc1p-labeled structures were not associated with the TGN. We next examined whether a recycling defect in the cdc50-ts mutants caused GFP-Snc1p to be missorted to the vacuole as with Kex2p-EGFP. Few of the GFP-Snc1p–positive structures, however, were also stained by CellTracker Blue CMAC (Figure 7B), suggesting that GFP-Snc1p did not accumulate in the vacuole. Thus, our results suggest that GFP-Snc1p is associated with endosomal structures.
Figure 7.
GFP-Snc1p and GFP-Tlg1p accumulate in early endosome-derived structures in the cdc50-ts mutant. (A) GFP-Snc1p and the TGN marker Sec7p-mRFP1 are not colocalized in the cdc50-11 mutant. cdc50-11 SEC7-mRFP1 (YNF153) cells carrying pRS416-GFP-SNC1 were incubated at 37°C for 3 h in SDA-Ura medium. Obtained images were merged to compare the two signal patterns. (B) GFP-Snc1p is not localized to vacuoles stained with CellTracker Blue CMAC in the cdc50-11 mutant. cdc50-11 (YNF65) cells carrying the pRS416-GFP-SNC1 plasmid were grown at 37°C for 3 h, followed by staining with 100 μM CellTracker Blue CMAC at 37°C for 15 min. (C) GFP-Snc1p was colocalized with FM4-64 after a short incubation in the cdc50-11 mutant. Wild-type (YNF63) and cdc50-11 (YNF65) cells carrying the pRS416-GFP-SNC1 plasmid were grown at 37°C for 3 h, stained with 32 μM FM4-64 on ice for 30 min, and chased in fresh medium at 37°C for 10 min. (D) mRFP1-Snc1p and GFP-Tlg1p are colocalized in the cdc50-11 mutant. Wild-type (YKT38) and cdc50-11 (YKT993) cells cotransformed with pKT1566 (YEplac181-GFP-TLG1) and pKT1563 (pRS416-mRFP1-SNC1) were incubated at 37°C for 3 h in SD-Leu-Ura medium, followed by microscopic examination after fixation with 0.5% formaldehyde. Bars, 5 μm.
RCY1 is involved in endocytosis and recycling out of early endosomes (Wiederkehr et al., 2000). Chen et al. (2005) have proposed that Rcy1p functions as an effector of Ypt31p/32p and that in the ypt31Δ ypt32-ts mutant, GFP-Snc1p is mostly localized to intracellular compartments as in RCY1 null mutants. In both cases, the GFP-Snc1p–labeled abnormal compartments were suggested to be derived from early endosomes. Because the large GFP-Snc1p–labeled structures in the cdc50-ts mutants were reminiscent of those in the rcy1Δ mutants reported by Wiederkehr et al. (2000), we examined whether GFP-Snc1p is localized to early endosomes in the cdc50-ts mutants. To identify early endosomes, we used FM4-64, which stains early endosomes after short incubation (Vida and Emr, 1995). Cells were incubated at 37°C for 3 h, labeled with FM4-64 on ice for 30 min, and chased at 37°C for 10 min. In wild-type cells, 71% of FM4-64–positive punctate structures were colocalized with GFP-Snc1p-positive structures (n = 183), whereas 84% of GFP–Snc1p structures were colocalized with FM4-64 labeling (n = 183) (Figure 7C). These results suggest that most of the GFP-Snc1p–positive structures correspond to early endosomes, consistent with a previous report in which internal GFP-Snc1p–positive punctate structures were shown to be early endosomes or the TGN (Lewis et al., 2000). In contrast, in cdc50-11 mutant cells, FM4-64 was observed to label abnormally large structures, and 73% of these FM4-64–positive structures were colocalized with the large GFP-Snc1p–positive structures (n = 191; Figure 7C). Conversely, 90% of the large GFP-Snc1p–positive structures were colocalized with FM4-64–positive structures (n = 182; Figure 7C). These results suggest that GFP-Snc1p accumulated in the abnormal early endosome-derived structures in the cdc50-11 mutant, as was observed in the ypt31Δ ypt32-ts and rcy1Δ mutants.
Because in the rcy1Δ mutant, the target membrane-associated (t)-SNARE Tlg1p seems to intracellularly accumulate in abnormal compartments (Wiederkehr et al., 2000) as was observed for Snc1p, we examined the localization of Tlg1p in cdc50-11 mutant cells. Tlg1p is recycled between the TGN and early endosomal membrane, and is consequently present in both membranes (Lewis et al., 2000). To examine the localization of Tlg1p in the cdc50-11 mutant, we constructed a multicopy plasmid containing GFP-TLG1 driven by its own promoter. In wild-type cells, GFP-Tlg1p was observed to be scattered throughout the cell (Figure 7D). In contrast, there was a substantial decrease of GFP-Tlg1p localized to punctate structures in cdc50-11 mutant cells, and GFP-Tlg1p was instead localized to large compartments (Figure 7D). To examine whether Tlg1p and Snc1p were localized in the same large compartments in cdc50-11 mutant cells, we constructed a single-copy plasmid containing the mRFP1-SNC1 gene driven by the TPI1 promoter and examined cells coexpressing GFP-TLG1 and mRFP1-SNC1. Quantitative analysis of individual spots revealed that in the cdc50-11 mutant cells, 96% of the mRFP1-Snc1p–positive large structures were also labeled with GFP-Tlg1p (n = 170), whereas 86% of the GFP-Tlg1–positive structures were also labeled with mRFP1-Snc1p (n = 163) (Figure 7D). These results suggest that the cdc50-11 mutant was defective in the retrieval pathway from early endosomes to the TGN and that Snc1p and Tlg1p accumulated in the abnormal structures derived from early endosomes in the cdc50-ts mutants.
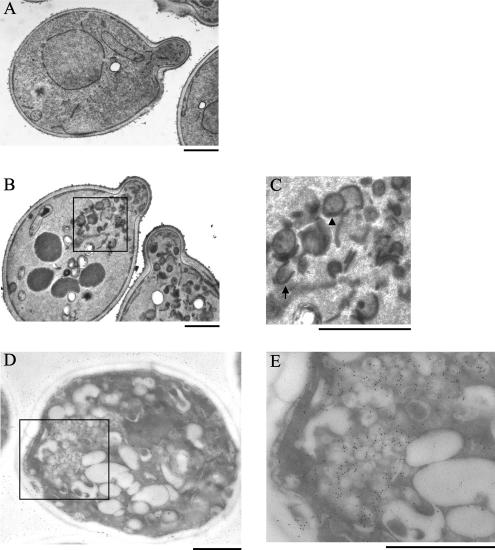
Electron Microscopic Examination of the Abnormal Structures Containing HA-Snc1p in the cdc50-11 Mutant
EM was performed to visualize and characterize the abnormally large compartments containing GFP-Snc1p in the cdc50-ts mutants by the glutaraldehyde-permanganate fixation technique. We found that these large structures were actually composed of a cluster of abnormal membranous structures. As shown in Figure 8, B and C, cdc50-11 mutant cells incubated at 37°C accumulated a number of double-membrane rings (arrowhead) and horseshoe-like structures (arrow) resembling Berkeley bodies (Novick et al., 1980) in or near the buds where GFP-Snc1p accumulated (Figure 5B). To examine whether these structures corresponded to the GFP-Snc1p–containing compartments observed by fluorescence microscopy, we performed immunoelectron microscopic analysis for HA-Snc1p in the cdc50-11 mutant incubated at 37°C with an anti-HA antibody by the aldehyde fixation/metaperiodate permeabilization method. We observed numerous immunogold-labeled abnormal membrane structures located in or near the bud in the cdc50-11 mutant expressing HA-tagged Snc1p (Figure 8, D and E) but not in mutant cells expressing GFP-tagged Snc1p (our unpublished data). HA-Snc1p labeling was mostly specific for these compartments and only a minor fraction of immunogold signals was observed on other membranes. Thus, we concluded that these immunogold-positive structures were likely the same GFP-Snc1p–positive structures observed by fluorescence microscopy.
Figure 8.
Electron microscopic examination of the abnormal structures containing HA-Snc1p in the cdc50-ts mutant. (A–C) The electron microscopic observation was performed using the glutaraldehyde-permanganate fixation technique. Wild-type (YKT38; A) and cdc50-11 mutant cells (YKT993; B and C) were grown at 25°C to early logarithmic phase, shifted to 37°C, and grown in YPDA medium for 3 h. Cells were prepared for EM as described in Materials and Methods. The boxed region in B is enlarged in C. An arrow and an arrowhead indicate a horseshoe-like structure and a double-membrane ring, respectively. Bars, 1 μm. (D and E) Immunoelectron microscopic observation of the cdc50-11 mutant was performed by the aldehyde fixation/metaperiodate permeabilization method. cdc50-11 mutant cells (YKT993) transformed with pKT1564 (pRS416-HA-SNC1) were grown at 25°C to early logarithmic phase, shifted to 37°C, and grown in SDA-Ura medium for 3 h. Cells were prepared for immuno-EM as described in Materials and Methods and labeled with an anti-HA antibody. The boxed region in D is enlarged in E. Bars, 1 μm.
The Cdc50p–Drs2p Complex Binds to Rcy1p
Rcy1p contains an amino-terminal F-box, which interacts with Skp1p (Bai et al., 1996) to form a non-SCF complex, and a CAAX-box motif at its C-terminus, which mediates its interaction with membranes (Zhang and Casey 1996; Galan et al., 2001). Because Chen et al. (2005) proposed that Rcy1p functions as an effector of Ypt31p/32p in the regulation of endocytic recycling, we examined the functional relationship between the Cdc50p–Drs2p complex and Rcy1p. The cdc50Δ mutation exhibited synthetic lethality with a wide range of endocytosis-related mutations, including the vrp1Δ, lem3Δ (Saito et al., 2004), rgp1Δ, ric1Δ (Kishimoto et al., 2005), and tlg2Δ mutations (our unpublished data). Vrp1p is required for the proper organization of cortical actin patches and the internalization step during endocytosis (Munn et al., 1995). The Ric1p–Rgp1p complex, which acts as a GTP exchange factor for Ypt6p, and the t-SNARE Tlg2p are required for efficient fusion of endosome-derived vesicles with the Golgi (Tsukada et al., 1999; Lewis et al., 2000; Siniossoglou et al., 2000). The rcy1Δ mutation also exhibited synthetic lethality with all the mutations listed above (our unpublished data). Importantly, the rcy1Δ mutation did not exhibit a synthetic growth defect in combination with cdc50Δ; the rcy1Δ cdc50Δ mutant exhibited the same cold-sensitive growth phenotype as the rcy1Δ and cdc50Δ single mutants (our unpublished data). These genetic results suggest that CDC50 and RCY1 function in the same pathway.
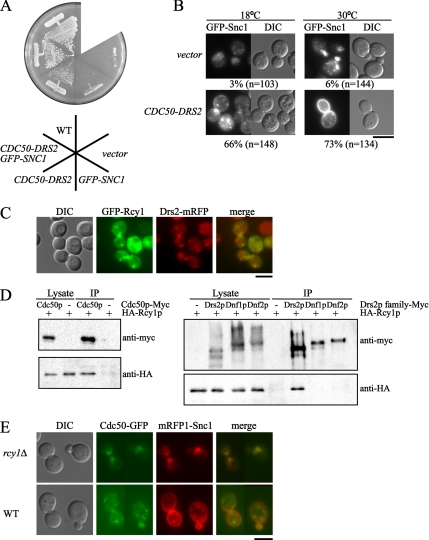
Cooverexpression of CDC50 and DRS2 weakly suppressed the growth defect of the rcy1Δ mutant at 18°C (Figure 9A). In contrast, overexpression of CDC50-DRS2 efficiently restored the plasma membrane localization of GFP-Snc1p in the rcy1Δ mutant. GFP-Snc1p was localized to the plasma membrane in only a few of the rcy1Δ cells (3 and 6% of the cells at 18 and 30°C, respectively), whereas when CDC50-DRS2 was overexpressed, GFP-Snc1p was localized to polarized plasma membrane sites of the rcy1Δ cells to an extent comparable to that in wild-type cells (66 and 73% of the mutant cells at 18 and 30°C, respectively) (Figure 9B). Interestingly, overexpression of GFP-SNC1 was responsible for this discrepancy; simultaneous overexpression of GFP-SNC1 and CDC50-DRS2 efficiently suppressed the growth defect of the rcy1Δ mutant, but overexpression of GFP-SNC1 alone did not (Figure 9A). These results suggest that Cdc50p-Drs2p function downstream of the Rcy1p pathway and that Snc1p is also involved in this pathway as a functional component rather than a cargo in the endocytic recycling route.
Figure 9.
The Cdc50p–Drs2p complex functionally and physically interacts with Rcy1p. (A) Suppression of the growth defect of the rcy1Δ mutant by simultaneous overexpression of CDC50, DRS2, and GFP-SNC1. rcy1Δ mutant cells (YKT951) were cotransformed with combinations of plasmids as follows: pKT1472 (YEplac195-DRS2-CDC50) and pKT1490 (pRS315-GFP-SNC1) for DRS2-CDC50 GFP-SNC1, pKT1472 (YEplac195-DRS2-CDC50) and pRS315 for DRS2-CDC50, YEplac195 and pKT1490 (pRS315-GFP-SNC1) for GFP-SNC1, and YEplac195 and pRS315 for vector. Transformants and wild-type cells (YKT38) were streaked onto a YPDA plate, followed by incubation at 18°C for 9 d. (B) Simultaneous overexpression of CDC50, DRS2, and GFP-SNC1 partially restored the plasma membrane localization of GFP-Snc1p in the rcy1Δ mutant. rcy1Δ mutant cells (YKT951) were cotransformed with pKT1490 (pRS315-GFP-SNC1) and a control vector (YEplac195; top) or pKT1472 (YEplac195-DRS2-CDC50; bottom). Cells were incubated at 18 or 30°C for 12 h in SD-Leu-Ura medium. Numbers indicate the percentages of cells in which GFP-Snc1p was localized to the plasma membrane. Bar, 5 μm. (C) GFP-Rcy1p partially colocalized with Drs2p-mRFP1. DRS2-mRFP1 (YKT871) cells transformed with pKT1560 (YEplac181-GFP-RCY1) were grown to early to mid-logarithmic phase at 30°C in SD-Leu-Ura medium. Obtained images were merged to compare the two signal patterns. Bar, 5 μm. (D) Coimmunoprecipitation of Rcy1p with Cdc50p, Drs2p, Dnf1p, and Dnf2p. Cells grown to mid-logarithmic phase at 30°C in SDA-Ura medium. Membrane extracts were then prepared as described in Materials and Methods. Myc-tagged Cdc50p, Drs2p, Dnf1p, or Dnf2p were immunoprecipitated with an anti-Myc antibody from membrane extracts. Immunoprecipitates were subjected to SDS-PAGE, followed by immunoblot analysis using antibodies against Myc (top) and HA (bottom). The results shown are representative of several experiments. The yeast strains used were as follows: YKT1101 (CDC50-Myc) and YKT38 (CDC50) (left); YKT792 (DRS2-Myc), YKT760 (DNF1-Myc), YKT1062 (DNF2-Myc), and YKT38 (DRS2 DNF1 DNF2) (right). All these strains carried pKT1626 (YEplac195-HA-BS-RCY1). (E) Colocalization of Cdc50p-EGFP with mRFP1-Snc1p in the rcy1Δ mutant. CDC50-EGFP (YKT259) and rcy1Δ CDC50-EGFP (YKT1102) cells transformed with pKT1563 (pRS416-mRFP1-SNC1) were grown to mid-logarithmic phase at 30°C in SDA-Ura medium. Bar, 5 μm.
The close functional relationship between Cdc50p-Drs2p and Rcy1p raises the possibility that these proteins are components of the same machinery. We examined whether Rcy1p colocalized with Drs2p. We constructed a strain that used a multicopy plasmid and the RCY1 promoter to express an N-terminally GFP-tagged version of RCY1 and the DRS2 genomic locus to express a C-terminally mRFP-tagged version of DRS2. Both Drs2p-mRFP1 and GFP-Rcy1p were observed in punctate structures scattered throughout the cell and partially localized to the mother-daughter neck of dividing cells (Figure 9C). Quantitative analysis of individual spots revealed that 74% of GFP-Rcy1p–positive structures were also Drs2p-mRFP1 positive (n = 270). Conversely, 49% of Drs2p-mRFP1–positive structures were also GFP-Rcy1p positive (n = 226) (Figure 9C).
To examine the physical association between Rcy1p and the Cdc50p–Drs2p complex, we performed coimmunoprecipitation experiments. We constructed a strain that used the CDC50 genomic locus to express a C-terminally Myc-tagged version of CDC50 and a multicopy plasmid with the own (RCY1) promoter to express an N-terminally HA-tagged version of RCY1. Cdc50p-Myc was immunoprecipitated with an anti-Myc antibody from membrane protein extracts prepared by solubilization in 1% CHAPS. The resulting immunoprecipitates were examined by immunoblot analysis with anti-Myc and anti-HA antibodies. HA-Rcy1p coimmunoprecipitated with Cdc50p-Myc (Figure 9D). HA-Rcy1p was not detected in control immunoprecipitates from cells lacking CDC50-Myc (Figure 9D). Similarly, we performed coimmunoprecipitation experiments using cells expressing a C-terminally Myc-tagged version of DRS2 from the DRS2 genomic locus and the same HA-tagged version of RCY1. HA-Rcy1p was detected in the Drs2p-Myc immunocomplex precipitated with an anti-Myc antibody but not in control immunoprecipitates from cells lacking DRS2-Myc (Figure 9D). In addition, we found that the C-terminal cytosolic region of Drs2p (amino acid positions 1216-1355) interacted with Rcy1p by the two-hybrid method (our unpublished data). In contrast, when we performed coimmunoprecipitation experiments using cells expressing a C-terminally Myc-tagged version of DNF1 or DNF2 from these genomic loci and the same HA-tagged version of RCY1, HA-Rcy1p could not be detected in the Dnf1p- and Dnf2p–Myc immunocomplexes precipitated with an anti-Myc antibody (Figure 9D). These results suggest that Rcy1p specifically interacts with the Cdc50p–Drs2p complex in vivo.
The recruitment of Rcy1p from the cytoplasm to endosomal membranes was suggested to be regulated by Ypt31p/32p (Chen et al., 2005), whereas like Snc1p, the Cdc50p–Drs2p complex seems to be recycled through the endocytic recycling pathway (Saito et al., 2004). The effect of Rcy1p depletion on the localization of Cdc50p-EGFP was examined by fluorescence microscopy. In wild-type cells at 30°C, Cdc50p-EGFP was observed as punctate structures scattered throughout the cell (Figure 9; E; Misu et al., 2003; Saito et al., 2004). In contrast, in the rcy1Δ mutant cells at 30°C, Cdc50p-EGFP was localized to large structures near the tip or neck of the bud, and the strength of the punctate signals concomitantly decreased (Figure 9E). These Cdc50p-EGFP–positive large structures were also labeled by mRFP1-Snc1p in the rcy1Δ mutant (Figure 9E). In the rcy1Δ mutant, 70% of mRFP1-Snc1p–positive large structures were colocalized with Cdc50p-EGFP–positive structures (n = 215), whereas 76% of the Cdc50p-EGFP–positive structures were colocalized with mRFP1-Snc1p–positive structures (n = 182). These results demonstrate that Cdc50p–Drs2p complexes accumulated in the early endosome-derived structures as Snc1p did in the rcy1Δ mutant (Chen et al., 2005). This suggests that Cdc50p-Drs2p needs to interact with Rcy1p to promote early endosome-to-TGN transport.
DISCUSSION
In the current study, we have identified YPT31/32 as multicopy suppressors of cdc50-ts mutants. Because the YPT31/32 genes were suggested to be involved in the endocytic recycling pathway as well as in the formation of post-Golgi exocytic vesicles, we examined the possible involvement of the putative PLTs in these processes. Our results suggested that the putative PLTs are involved in the endocytic recycling pathway in conjunction with Ypt31p/32p and Rcy1p, rather than in the post-Golgi exocytic pathway. Although we did not find major defects in production of high- and low-density classes of exocytic vesicles in the sec6-4 cdc50-11 mutant, Gall et al. (2002) reported that drs2 mutants exhibited defects in the formation of the high-density vesicles. This discrepancy may be attributed to the differences of methods in which high-density vesicles were detected. In Gall et al. (2002), the vesicles that were histochemically stained with acid phosphatase activity by EM were referred to as dense vesicles. Because the formation of these vesicles, which were induced by a sla2 mutation or LAT-A treatment, was suppressed by drs2-ts mutations, they argued that Drs2p is involved in the high-density vesicle formation. However, primary defects in the sla2 mutant cells are in endocytosis, not in exocytosis (Wendland et al., 1998; Engqvist-Goldstein and Drubin, 2003), and Lat-A also blocks endocytosis as well as exocytosis (Ayscough et al., 1997). Therefore, accumulation of acid phosphatase-containing vesicles in the sla2 mutant may be a consequence of secondary effects due to perturbation of the endocytic pathways, making the interpretation of the effects of drs2 mutations complicated.
Heteromeric Putative PLTs Are Required for Transport from Early Endosomes to the TGN
Similar to Cdc50p-Drs2p, Crf1p-Dnf3p may be localized to endosomal/TGN compartments, because Dnf3p-HA colocalizes with the endosome/TGN marker Kex2p (Hua et al., 2002) and Dnf3-HA cofractionated with endosomal/TGN membranes (Pomorski et al., 2003). In contrast, Lem3p-Dnf1p/2p is primarily localized to the plasma membrane, although it is also found in punctate structures that may correspond to endosomal/TGN compartments (Hua et al., 2002; Kato et al., 2002; Pomorski et al., 2003). We showed that Dnf1p-EGFP was primarily localized to these intracellular punctate structures in the cdc50Δ mutant and was exclusively localized on the plasma membrane when endocytosis was blocked, suggesting that Lem3p-Dnf1p/2p is recycled by the endocytic pathway (Saito et al., 2004). Lem3p-Dnf1p/2p may cooperate with Cdc50p-Drs2p at early endosomes for endocytic recycling. If this is the case, what is the function of Lem3p-Dnf1p/2p when it is localized at the plasma membrane? Interestingly, the lem3Δ and dnf1Δ dnf2Δ dnf3Δ mutants display elongated cell morphologies at lower temperatures (Hua et al., 2002; Saito, K., Fujimura-Kamada, K., and Tanaka, K., unpublished observations), suggesting that Lem3p-Dnf1p/2p may be involved in the negative regulation of polarized cell growth. In contrast to the depolarized organization of the actin cytoskeleton in the cdc50Δ mutant at 18°C (Misu et al., 2003), the cdc50-ts mutants exhibited polarized organization of the actin cytoskeleton at 37°C, possibly due to the effect of the lem3Δ mutation (our unpublished data).
The Snc1p-containing large membranous structures that accumulated in the cdc50-ts mutants seem to be of endocytic origin, suggesting that the cdc50-ts mutants are defective in the formation of vesicles from early endosomes. The Rab family small GTPase Ypt6p and its nucleotide exchange factor consisting of Ric1p and Rgp1p are also required for endocytic recycling (Siniossoglou et al., 2000; Siniossoglou and Pelham, 2001). These factors are localized to the TGN to assist fusion between endosomally derived vesicles and the TGN. Both Ric1p-EGFP and Rgp1p-EGFP exhibited normal punctate localization patterns in the cdc50-ts mutants (our unpublished data), suggesting that the fusion step of endocytic vesicles with the TGN is not severely affected in the cdc50-ts mutants. Synthetic growth defects observed with combinations of the cdc50Δ mutation and the ric1Δ, rgp1Δ, and ypt6 mutations (Kishimoto et al., 2005) may be due to multiple defects in different steps of the endocytic recycling pathway.
One possible explanation for accumulation of early endosomal structures is that proteins required for the formation of vesicles destined for the TGN are not normally transported from the TGN to early endosomes in the cdc50-ts mutants. Tlg1p, which is thought to be recycled between early endosomes and the TGN (Lewis et al., 2000), however, accumulated in the Snc1p-containing structures, suggesting that the TGN-to-early endosome pathway was not impaired. Mislocalization of Kex2p in drs2 mutants was explained by defective transport from the TGN (Chen et al., 1999), but it is also possible that Kex2p was transported to vacuoles by default due to defective retrieval from the early endosome to the TGN as suggested previously (Wilcox et al., 1992; Holthuis et al., 1998; Spelbrink and Nothwehr, 1999).
One intriguing possibility is that the putative PLTs are directly involved in vesicle formation at early endosomes. A proposed role for PLTs is that they locally generate phospholipid asymmetry to assist membrane deformation during vesicle budding or to recruit proteins that promote vesicle formation (Graham, 2004). It was previously suggested that Drs2p is involved in Arf1p- and clathrin-dependent vesicle formation; DRS2 was originally identified because a mutant allele was synthetically lethal with arf1Δ (Chen and Graham, 1998; Chen et al., 1999), and significantly fewer clathrin-coated vesicles were isolated from drs2Δ cells than from wild-type cells (Chen et al., 1999). Thus, the accumulation of early endosomal structures in the cdc50-ts mutants may be due to defective formation of clathrin-coated vesicles. Involvement of clathrin in the endocytic recycling pathway has been demonstrated in mammalian cells (van Dam and Stoorvogel, 2002) and suggested in yeast; clathrin, and clathrin adaptor complex 1 act to recycle chitin synthase III (Chs3p) from the early endosome to the TGN (Valdivia et al., 2002).
Functional Relationship between Putative PLTs and the Ypt31p/32p–Rcy1p Pathway
Results from our group and other groups suggest that RCY1 is functionally equivalent to CDC50. Immuno-EM suggested that the rcy1Δ mutant also accumulates large membranous structures that contain Tlg1p (Wiederkehr et al., 2000). In addition to defects in endocytic recycling, the cdc50Δ and rcy1Δ mutants share a number of other phenotypes, including cold sensitivity of growth, minimum defects in secretion, and vacuolar protein sorting (Wiederkehr et al., 2000), and intracellular assembly of cortical actin patches (Kishimoto et al., 2005; Kishimoto, K., Yamamoto, T., and Tanaka, K., unpublished observations). Growth defects of the cdc50Δ mutant were not exacerbated by rcy1Δ, whereas rcy1Δ as well as cdc50Δ resulted in synthetic growth defects when combined the lem3Δ mutation. These results, taken together with the coimmunoprecipitation of Rcy1p with Cdc50p as well as with Drs2p, suggest that Rcy1p plays an important role in regulation or function of the Cdc50p–Drs2p putative PLT. We favor an upstream regulatory role for Ypt31p/32p-Rcy1p based on the following observations. Simultaneous overexpression of CDC50, DRS2, and GFP-SNC1 efficiently restored growth and endocytic recycling of GFP-Snc1p in the rcy1Δ mutant, whereas overexpression of YPT31/32 only weakly suppressed defects in growth and endocytic recycling of GFP-Snc1p in the cdc50-ts mutants. In addition, overexpression of YPT31/32 did not suppress the cold-sensitive growth defect of the cdc50Δ mutant, suggesting that a residual activity of Cdc50p-Drs2p is required for the suppression.
Genetic studies suggested that transport protein particle (TRAPP) II, a large complex that mediates membrane traffic, functions upstream of YPT31 (Wang and Ferro-Novick, 2002; Yamamoto and Jigami, 2002; Sciorra et al., 2005). An allele of TRS130 encoding a subunit of TRAPP II was identified as a synthetic lethal mutation with arf1Δ (Zhang et al., 2002). This trs130-101 arf1Δ mutant did not exhibit defects in either CPY processing or invertase secretion, and overexpression of YPT31/32 suppressed the growth defects of the trs130-101 arf1Δ mutant. Interestingly, it was recently reported that mutants in another component of TRAPP II, TRS120, exhibited defects in the endocytic recycling of Snc1p, but not in general secretion (Cai et al., 2005). In the trs120 mutant, aberrant and large membrane structures accumulated, reminiscent of the membrane structures observed in the cdc50-ts mutants. Although it is controversial whether TRAPP II is a GDP/GTP exchange factor for Ypt31p/32p (Jones et al., 2000; Wang and Ferro-Novick, 2002), TRAPP II may activate Ypt31p/32p to promote vesicle formation from early endosomes.
Localization of Rcy1p to internal membranes was not affected by blockade of endocytic internalization (our unpublished data), whereas the ypt31Δ ypt32-ts mutations caused diffuse GFP–Rcy1p labeling in the cytoplasm (Chen et al., 2005). Thus, Ypt31p/32p may regulate recruitment of Rcy1p to endosomal membranes by stimulating the interaction of Rcy1p with Cdc50p-Drs2p on endosomal membranes. Cdc50p-Drs2p, however, is not likely to be the sole receptor for Rcy1p, because GFP-Rcy1p was still localized to internal punctate structures in the cdc50-ts mutants at 37°C (our unpublished observations). Because Rcy1p also binds to Snc1p (Chen et al., 2005), Ypt31p/32p-GTP-Rcy1p might recruit and couple putative PLTs with cargo proteins on early endosomal membranes. Interestingly, efficient suppression of defects in growth and endocytic recycling in the rcy1Δ mutant by overexpression of CDC50 and DRS2 also required the overexpression of GFP-SNC1. Formation of a functional complex consisting of Rcy1p, Snc1p, and Cdc50p-Drs2p may lead to utilization of the activity of putative PLTs to promote subsequent processes for vesicle budding.
Supplementary Material
ACKNOWLEDGMENTS
We thank Drs. Charles Boone, Michael Lewis, Hugh Pelham, Roger Tsien, Yoshinori Ohsumi, Yoshikazu Ohya, Masato Umeda, Akihiko Nakano, and Ramon Serrano for yeast strains, plasmids, and antibodies; and Dr. Masahiko Watanabe for use of the microtome. We thank our colleagues in the Tanaka laboratory for valuable discussions. We also thank Eriko Itoh for technical assistance. This work was supported by grants-in-aid for scientific research from the Japan Society for the Promotion of Science and the Ministry of Education, Culture, Sports, Science and Technology of Japan (to K.F.-K., K.S., T.Y., and K.T.).
Abbreviations used:
- EM
electron microscopy
- ER
endoplasmic reticulum
- GFP
green fluorescent protein
- LAT-A
latrunculin A
- mRFP1
monomeric red fluorescent protein 1
- PC
phosphatidylcholine
- PE
phosphatidylethanolamine
- PLT
phospholipid translocase
- PS
phosphatidylserine
- TGN
trans-Golgi network
- ts
temperature-sensitive
- VPS
vacuolar protein sorting.
Footnotes
 The online version of this article contains supplemental material at MBC Online (http://www.molbiolcell.org).
The online version of this article contains supplemental material at MBC Online (http://www.molbiolcell.org).
This article was published online ahead of print in MBC in Press (http://www.molbiolcell.org/cgi/doi/10.1091/mbc.E06-05-0461) on November 8, 2006.
REFERENCES
- Alder-Baerens N., Lisman Q., Luong L., Pomorski T., Holthuis J. C. Loss of P4 ATPases Drs2p and Dnf3p disrupts aminophospholipid transport and asymmetry in yeast post-Golgi secretory vesicles. Mol. Biol. Cell. 2006;17:1632–1642. doi: 10.1091/mbc.E05-10-0912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ayscough K. R., Stryker J., Pokala N., Sanders M., Crews P., Drubin D. G. High rates of actin filament turnover in budding yeast and roles for actin in establishment and maintenance of cell polarity revealed using the actin inhibitor latrunculin-A. J. Cell Biol. 1997;137:399–416. doi: 10.1083/jcb.137.2.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai C., Sen P., Hofmann K., Ma L., Goebl M., Harper J. W., Elledge S. J. SKP1 connects cell cycle regulators to the ubiquitin proteolysis machinery through a novel motif, the F-box. Cell. 1996;86:263–274. doi: 10.1016/s0092-8674(00)80098-7. [DOI] [PubMed] [Google Scholar]
- Benli M., Doring F., Robinson D. G., Yang X., Gallwitz D. Two GTPase isoforms, Ypt31p and Ypt32p, are essential for Golgi function in yeast. EMBO. J. 1996;15:6460–6475. [PMC free article] [PubMed] [Google Scholar]
- Black M. W., Pelham H. R. A selective transport route from Golgi to late endosomes that requires the yeast GGA proteins. J. Cell Biol. 2000;151:587–600. doi: 10.1083/jcb.151.3.587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brickner J. H., Fuller R. S. SOI1 encodes a novel, conserved protein that promotes TGN-endosomal cycling of Kex2p and other membrane proteins by modulating the function of two TGN localization signals. J. Cell Biol. 1997;139:23–36. doi: 10.1083/jcb.139.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai H., Zhang Y., Pypaert M., Walker L., Ferro-Novick S. Mutants in trs120 disrupt traffic from the early endosome to the late Golgi. J. Cell Biol. 2005;171:823–833. doi: 10.1083/jcb.200505145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catty P., de Kerchove d'Exaerde A., Goffeau A. The complete inventory of the yeast Saccharomyces cerevisiae P-type transport ATPases. FEBS Lett. 1997;409:325–332. doi: 10.1016/s0014-5793(97)00446-8. [DOI] [PubMed] [Google Scholar]
- Cereghino J. L., Marcusson E. G., Emr S. D. The cytoplasmic tail domain of the vacuolar protein sorting receptor Vps10p and a subset of VPS gene products regulate receptor stability, function, and localization. Mol. Biol. Cell. 1995;6:1089–1102. doi: 10.1091/mbc.6.9.1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C. Y., Graham T. R. An arf1Δ synthetic lethal screen identifies a new clathrin heavy chain conditional allele that perturbs vacuolar protein transport in Saccharomyces cerevisiae. Genetics. 1998;150:577–589. doi: 10.1093/genetics/150.2.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C. Y., Ingram M. F., Rosal P. H., Graham T. R. Role for Drs2p, a P-type ATPase and potential aminophospholipid translocase, in yeast late Golgi function. J. Cell Biol. 1999;147:1223–1236. doi: 10.1083/jcb.147.6.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S. H., Chen S., Tokarev A. A., Liu F., Jedd G., Segev N. Ypt31/32 GTPases and their novel F-box effector protein Rcy1 regulate protein recycling. Mol. Biol. Cell. 2005;16:178–192. doi: 10.1091/mbc.E04-03-0258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conibear E., Stevens T. H. Vps52p, Vps53p, and Vps54p form a novel multisubunit complex required for protein sorting at the yeast late Golgi. Mol. Biol. Cell. 2000;11:305–323. doi: 10.1091/mbc.11.1.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper A. A., Stevens T. H. Vps10p cycles between the late-Golgi and prevacuolar compartments in its function as the sorting receptor for multiple yeast vacuolar hydrolases. J. Cell Biol. 1996;133:529–541. doi: 10.1083/jcb.133.3.529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David D., Sundarababu S., Gerst J. E. Involvement of long chain fatty acid elongation in the trafficking of secretory vesicles in yeast. J. Cell Biol. 1998;143:1167–1182. doi: 10.1083/jcb.143.5.1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elble R. A simple and efficient procedure for transformation of yeasts. Biotechniques. 1992;13:18–20. [PubMed] [Google Scholar]
- Engqvist-Goldstein A. E., Drubin D. G. Actin assembly and endocytosis: from yeast to mammals. Annu. Rev. Cell Dev. Biol. 2003;19:287–332. doi: 10.1146/annurev.cellbio.19.111401.093127. [DOI] [PubMed] [Google Scholar]
- Franzusoff A., Redding K., Crosby J., Fuller R. S., Schekman R. Localization of components involved in protein transport and processing through the yeast Golgi apparatus. J. Cell Biol. 1991;112:27–37. doi: 10.1083/jcb.112.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galan J. M., Wiederkehr A., Seol J. H., Haguenauer-Tsapis R., Deshaies R. J., Riezman H., Peter M. Skp1p and the F-box protein Rcy1p form a non-SCF complex involved in recycling of the SNARE Snc1p in yeast. Mol. Cell. Biol. 2001;21:3105–3117. doi: 10.1128/MCB.21.9.3105-3117.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gall W. E., Geething N. C., Hua Z., Ingram M. F., Liu K., Chen S. I., Graham T. R. Drs2p-dependent formation of exocytic clathrin-coated vesicles in vivo. Curr. Biol. 2002;12:1623–1627. doi: 10.1016/s0960-9822(02)01148-x. [DOI] [PubMed] [Google Scholar]
- Gaynor E. C., te Heesen S., Graham T. R., Aebi M., Emr S. D. Signal-mediated retrieval of a membrane protein from the Golgi to the ER in yeast. J. Cell Biol. 1994;127:653–665. doi: 10.1083/jcb.127.3.653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gietz R. D., Sugino A. New yeast-Escherichia coli shuttle vectors constructed with in vitro mutagenized yeast genes lacking six-base pair restriction sites. Gene. 1988;74:527–534. doi: 10.1016/0378-1119(88)90185-0. [DOI] [PubMed] [Google Scholar]
- Gietz R. D., Woods R. A. Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Methods Enzymol. 2002;350:87–96. doi: 10.1016/s0076-6879(02)50957-5. [DOI] [PubMed] [Google Scholar]
- Goldstein A. L., McCusker J. H. Three new dominant drug resistance cassettes for gene disruption in Saccharomyces cerevisiae. Yeast. 1999;15:1541–1553. doi: 10.1002/(SICI)1097-0061(199910)15:14<1541::AID-YEA476>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- Graham T. R. Flippases and vesicle-mediated protein transport. Trends Cell Biol. 2004;14:670–677. doi: 10.1016/j.tcb.2004.10.008. [DOI] [PubMed] [Google Scholar]
- Gurunathan S., David D., Gerst J. E. Dynamin and clathrin are required for the biogenesis of a distinct class of secretory vesicles in yeast. EMBO J. 2002;21:602–614. doi: 10.1093/emboj/21.4.602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guthrie C., Fink G. R. San Diego, CA: Academic Press; 1991. Guide to Yeast Genetics and Molecular Biology. [Google Scholar]
- Harsay E., Bretscher A. Parallel secretory pathways to the cell surface in yeast. J. Cell Biol. 1995;131:297–310. doi: 10.1083/jcb.131.2.297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holthuis J. C., Nichols B. J., Dhruvakumar S., Pelham H. R. Two syntaxin homologues in the TGN/endosomal system of yeast. EMBO J. 1998;17:113–126. doi: 10.1093/emboj/17.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holthuis J. C., Levine T. P. Lipid traffic: floppy drives and a superhighway. Nat. Rev. Mol. Cell Biol. 2005;6:209–220. doi: 10.1038/nrm1591. [DOI] [PubMed] [Google Scholar]
- Hua Z., Fatheddin P., Graham T. R. An essential subfamily of Drs2p-related P-type ATPases is required for protein trafficking between Golgi complex and endosomal/vacuolar system. Mol. Biol. Cell. 2002;13:3162–3177. doi: 10.1091/mbc.E02-03-0172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jedd G., Mulholland J., Segev N. Two new Ypt GTPases are required for exit from the yeast trans-Golgi compartment. J. Cell Biol. 1997;137:563–580. doi: 10.1083/jcb.137.3.563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones S., Newman C., Liu F., Segev N. The TRAPP complex is a nucleotide exchanger for Ypt1 and Ypt31/32. Mol. Biol. Cell. 2000;11:4403–4411. doi: 10.1091/mbc.11.12.4403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaiser C. A., Schekman R. Distinct sets of SEC genes govern transport vesicle formation and fusion early in the secretory pathway. Cell. 1990;61:723–733. doi: 10.1016/0092-8674(90)90483-u. [DOI] [PubMed] [Google Scholar]
- Kato U., Emoto K., Fredriksson C., Nakamura H., Ohta A., Kobayashi T., Murakami-Murofushi K., Kobayashi T., Umeda M. A novel membrane protein, Ros3p, is required for phospholipid translocation across the plasma membrane in Saccharomyces cerevisiae. J. Biol. Chem. 2002;277:37855–37862. doi: 10.1074/jbc.M205564200. [DOI] [PubMed] [Google Scholar]
- Kihara A., Noda T., Ishihara N., Ohsumi Y. Two distinct Vps34 phosphatidylinositol3-kinase complexes function in autophagy and carboxypeptidase Y. sorting in Saccharomyces cerevisiae. J. Cell Biol. 2001;152:519–530. doi: 10.1083/jcb.152.3.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kishimoto T., Yamamoto T., Tanaka K. Defects in structural integrity of ergosterol and the Cdc50p-Drs2p putative phospholipid translocase cause accumulation of endocytic membranes, onto which actin patches are assembled in yeast. Mol. Biol. Cell. 2005;16:5592–5609. doi: 10.1091/mbc.E05-05-0452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis M. J., Nichols B. J., Prescianotto-Baschong C., Riezman H., Pelham H. R. Specific retrieval of the exocytic SNARE Snc1p from early yeast endosomes. Mol. Biol. Cell. 2000;11:23–38. doi: 10.1091/mbc.11.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longtine M. S., McKenzie A., 3rd, Demarini D. J., Shah N. G., Wach A., Brachat A., Philippsen P., Pringle J. R. Additional modules for versatile and economical PCR-based gene deletion and modification in Saccharomyces cerevisiae. Yeast. 1998;14:953–961. doi: 10.1002/(SICI)1097-0061(199807)14:10<953::AID-YEA293>3.0.CO;2-U. [DOI] [PubMed] [Google Scholar]
- Misu K., Fujimura-Kamada K., Ueda T., Nakano A., Katoh H., Tanaka K. Cdc50p, a conserved endosomal membrane protein, controls polarized growth in Saccharomyces cerevisiae. Mol. Biol. Cell. 2003;14:730–747. doi: 10.1091/mbc.E02-06-0314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulholland J., Botstein D. Immunoelectron microscopy of aldehyde-fixed yeast cells. Methods Enzymol. 2002;351:50–81. doi: 10.1016/s0076-6879(02)51841-3. [DOI] [PubMed] [Google Scholar]
- Munn A. L., Stevenson B. J., Geli M. I., Riezman H. end5, end6, and end7, mutations that cause actin delocalization and block the internalization step of endocytosis in Saccharomyces cerevisiae. Mol. Biol. Cell. 1995;6:1721–1742. doi: 10.1091/mbc.6.12.1721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natarajan P., Wang J., Hua Z., Graham T. R. Drs2p-coupled aminophospholipid translocase activity in yeast Golgi membranes and relationship to in vivo function. Proc. Natl. Acad. Sci. USA. 2004;101:10614–10619. doi: 10.1073/pnas.0404146101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novick P., Field C., Schekman R. Identification of 23 complementation groups required for post-translational events in the yeast secretory pathway. Cell. 1980;21:205–215. doi: 10.1016/0092-8674(80)90128-2. [DOI] [PubMed] [Google Scholar]
- Ortiz D., Medkova M., Walch-Solimena C., Novick P. Ypt32 recruits the Sec4p guanine nucleotide exchange factor, Sec2p, to secretory vesicles; evidence for a Rab cascade in yeast. J. Cell Biol. 2002;157:1005–1015. doi: 10.1083/jcb.200201003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pomorski T., Lombardi R., Riezman H., Devaux P. F., van Meer G., Holthuis J. C. Drs2p-related P-type ATPases Dnf1p and Dnf2p are required for phospholipid translocation across the yeast plasma membrane and serve a role in endocytosis. Mol. Biol. Cell. 2003;14:1240–1254. doi: 10.1091/mbc.E02-08-0501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pomorski T., Holthuis J. C., Herrmann A., van Meer G. Tracking down lipid flippases and their biological functions. J. Cell Sci. 2004;117:805–813. doi: 10.1242/jcs.01055. [DOI] [PubMed] [Google Scholar]
- Raymond C. K., Howald-Stevenson I., Vater C. A., Stevens T. H. Morphological classification of the yeast vacuolar protein sorting mutants: evidence for a prevacuolar compartment in class E vps mutants. Mol. Biol. Cell. 1992;3:1389–1402. doi: 10.1091/mbc.3.12.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts C. J., Raymond C. K., Yamashiro C. T., Stevens T. H. Methods for studying the yeast vacuole. Methods Enzymol. 1991;194:644–661. doi: 10.1016/0076-6879(91)94047-g. [DOI] [PubMed] [Google Scholar]
- Rose M. D., Winston F., Hieter P. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 1990. Methods in Yeast Genetics: A Laboratory Course Manual. [Google Scholar]
- Rothblatt J., Schekman R. A hitchhiker's guide to analysis of the secretory pathway in yeast. Methods Cell Biol. 1989;32:3–36. doi: 10.1016/s0091-679x(08)61165-6. [DOI] [PubMed] [Google Scholar]
- Rubin G. M. The nucleotide sequence of Saccharomyces cerevisiae 5.8 S ribosomal ribonucleic acid. J. Biol. Chem. 1973;248:3860–3875. [PubMed] [Google Scholar]
- Saito K., Fujimura-Kamada K., Furuta N., Kato U., Umeda M., Tanaka K. Cdc50p, a protein required for polarized growth, associates with the Drs2p P-type ATPase implicated in phospholipid translocation in Saccharomyces cerevisiae. Mol. Biol. Cell. 2004;15:3418–3432. doi: 10.1091/mbc.E03-11-0829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciorra V. A., Audhya A., Parsons A. B., Segev N., Boone C., Emr S. D. Synthetic genetic array analysis of the PtdIns 4-kinase Pik1p identifies components in a Golgi-specific Ypt31/rab-GTPase signaling pathway. Mol. Biol. Cell. 2005;16:776–793. doi: 10.1091/mbc.E04-08-0700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seaman M. N., Marcusson E. G., Cereghino J. L., Emr S. D. Endosome to Golgi retrieval of the vacuolar protein sorting receptor, Vps10p, requires the function of the VPS29, VPS30, and VPS35 gene products. J. Cell Biol. 1997;137:79–92. doi: 10.1083/jcb.137.1.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sikorski R. S., Hieter P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics. 1989;122:19–27. doi: 10.1093/genetics/122.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siniossoglou S., Peak-Chew S. Y., Pelham H. R. Ric1p and Rgp1p form a complex that catalyses nucleotide exchange on Ypt6p. EMBO J. 2000;19:4885–4894. doi: 10.1093/emboj/19.18.4885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siniossoglou S., Pelham H. R. An effector of Ypt6p binds the SNARE Tlg1p and mediates selective fusion of vesicles with late Golgi membranes. EMBO J. 2001;20:5991–5998. doi: 10.1093/emboj/20.21.5991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spelbrink R. G., Nothwehr S. F. The yeast GRD20 gene is required for protein sorting in the trans-Golgi network/endosomal system and for polarization of the actin cytoskeleton. Mol. Biol. Cell. 1999;10:4263–4281. doi: 10.1091/mbc.10.12.4263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens T., Esmon B., Schekman R. Early stages in the yeast secretory pathway are required for transport of carboxypeptidase Y to the vacuole. Cell. 1982;30:439–448. doi: 10.1016/0092-8674(82)90241-0. [DOI] [PubMed] [Google Scholar]
- Toi H., Fujimura-Kamada K., Irie K., Takai Y., Todo S., Tanaka K. She4p/Dim1p interacts with the motor domain of unconventional myosins in the budding yeast, Saccharomyces cerevisiae. Mol. Biol. Cell. 2003;14:2237–2249. doi: 10.1091/mbc.E02-09-0616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsukada M., Will E., Gallwitz D. Structural and functional analysis of a novel coiled-coil protein involved in Ypt6 GTPase-regulated protein transport in yeast. Mol. Biol. Cell. 1999;10:63–75. doi: 10.1091/mbc.10.1.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdivia R. H., Baggott D., Chuang J. S., Schekman R. W. The yeast clathrin adaptor protein complex 1 is required for the efficient retention of a subset of late Golgi membrane proteins. Dev. Cell. 2002;2:283–294. doi: 10.1016/s1534-5807(02)00127-2. [DOI] [PubMed] [Google Scholar]
- van Dam E. M., Stoorvogel W. Dynamin-dependent transferrin receptor recycling by endosome-derived clathrin-coated vesicles. Mol. Biol. Cell. 2002;13:169–182. doi: 10.1091/mbc.01-07-0380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vida T. A., Emr S. D. A new vital stain for visualizing vacuolar membrane dynamics and endocytosis in yeast. J. Cell Biol. 1995;128:779–792. doi: 10.1083/jcb.128.5.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W., Ferro-Novick S. A Ypt32p exchange factor is a putative effector of Ypt1p. Mol. Biol. Cell. 2002;13:3336–3343. doi: 10.1091/mbc.01-12-0577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wendland B., Emr S. D., Riezman H. Protein traffic in the yeast endocytic and vacuolar protein sorting pathways. Curr. Opin. Cell Biol. 1998;10:513–522. doi: 10.1016/s0955-0674(98)80067-7. [DOI] [PubMed] [Google Scholar]
- Wiederkehr A., Avaro S., Prescianotto-Baschong C., Haguenauer-Tsapis R., Riezman H. The F-box protein Rcy1p is involved in endocytic membrane traffic and recycling out of an early endosome in Saccharomyces cerevisiae. J. Cell Biol. 2000;149:397–410. doi: 10.1083/jcb.149.2.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilcox C. A., Redding K., Wright R., Fuller R. S. Mutation of a tyrosine localization signal in the cytosolic tail of yeast Kex2 protease disrupts Golgi retention and results in default transport to the vacuole. Mol. Biol. Cell. 1992;3:1353–1371. doi: 10.1091/mbc.3.12.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yahara N., Ueda T., Sato K., Nakano A. Multiple roles of Arf1 GTPase in the yeast exocytic and endocytic pathways. Mol. Biol. Cell. 2001;12:221–238. doi: 10.1091/mbc.12.1.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K., Jigami Y. Mutation of TRS130, which encodes a component of the TRAPPII complex, activates transcription of OCH1 in Saccharomyces cerevisiae. Curr. Genet. 2002;42:85–93. doi: 10.1007/s00294-002-0336-5. [DOI] [PubMed] [Google Scholar]
- Zhang C. J., Bowzard J. B., Greene M., Anido A., Stearns K., Kahn R. A. Genetic interactions link ARF1, YPT31/32 and TRS130. Yeast. 2002;19:1075–1086. doi: 10.1002/yea.903. [DOI] [PubMed] [Google Scholar]
- Zhang F. L., Casey P. J. Protein prenylation: molecular mechanisms and functional consequences. Annu. Rev. Biochem. 1996;65:241–269. doi: 10.1146/annurev.bi.65.070196.001325. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.