Abstract
OBJECTIVE—To establish a quality assessment programme for the diagnosis of crystal arthropathies by synovial fluid (SF) microscopy. METHODS—Three or four cytocentrifuge slides prepared from suitable patient SF specimens were distributed to 25-47 predominantly Finnish clinical laboratories once a year. Sodium urate crystals were included in every survey. RESULTS—Returns for the years 1989-1996 were reviewed. Laboratories that participated in > four surveys made on an average one error a year (range 0.25-2). The error rate for specimens containing abundant crystals was acceptable but it increased considerably for specimens showing few crystals per microscope field. No laboratory characteristic predictive of successful performance was found. CONCLUSION—Errors in quality assessment results for crystal identification were much more frequent than in the fields of, for example, clinical chemistry or microbiology. Despite efforts to provide educational feedback, no improvement was seen during the study period. Because of the dearth of data from other parts of the world it is not known for certain whether this study has merely pinpointed a local problem or if the same trend applies elsewhere. Keywords: synovial fluid; joint fluid; quality control; crystals
Full Text
The Full Text of this article is available as a PDF (74.2 KB).
Figure 1 .
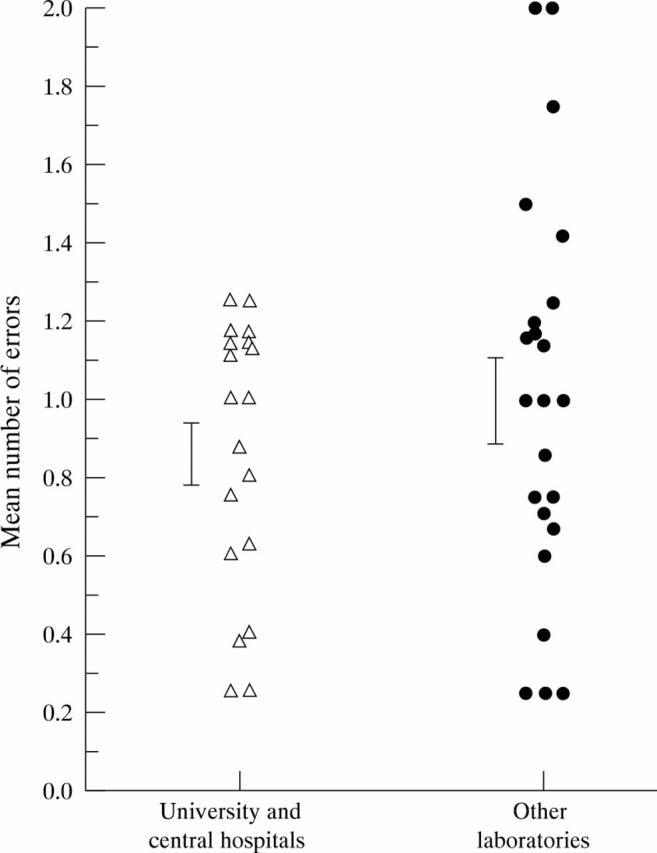
Mean annual number of errors for 19 university and central hospital laboratories and 23 other clinical laboratories that participated in >4 SF crystal identification surveys. Bars = standard error.


