Abstract
Objectives: To investigate the effect of prolonged neutralisation of tumour necrosis factor α (TNFα) on the radiological course in rheumatoid arthritis (RA). To assess whether the radiological course can be predicted by clinical variables or biological markers of cartilage and synovium turnover and of endothelial activation.
Patients and methods: Forty seven patients with active RA enrolled at our centre in monotherapy trials with adalimumab (D2E7), a fully human anti-TNFα monoclonal antibody, were studied for two years. Radiographs of hands and feet obtained at baseline and after one and two years were scored in chronological order by a single, blinded observer using the modified Sharp method. Radiological course was classified as stable or progressive using the smallest detectable difference as cut off point. The relation between radiological course and serum markers of cartilage and synovium turnover (metalloproteinases (MMP-1 and MMP-3), cartilage oligomeric matrix protein (COMP), human cartilage glycoprotein-39 (HC gp-39)), endothelial activation (soluble E-selectin and intercellular adhesion molecule (ICAM-1)), and integrated measures of disease activity were assessed using univariate and multivariate analysis.
Results: Radiological evaluation was performed in 36 patients with paired sets of radiographs at baseline and two years. After two years a total of 15/36 (42%) presented no radiological progression. More patients with stable radiological course were still receiving anti-TNFα treatment after two years (13/15 (87%) v 11/21 (52%); p=0.03) and had lower baseline COMP and sICAM-1 levels (p=0.01 and 0.04, respectively) than those in the group with progressive disease. In a logistic regression model the combination of sustained TNF neutralisation and baseline COMP and sICAM-1 levels was predictive for radiological outcome (p=0.03). C reactive protein and disease activity score area under the curve were significantly correlated with changes in radiological scores after two years (r=0.40 and 0.37, p<0.05). Long term TNFα neutralisation decreased the levels of COMP, sICAM, MMPs, and HC gp-39, but not sE-selectin.
Conclusion: The results suggest that long term monotherapy with anti-TNFα has a positive effect on radiological outcome and modulates cartilage and synovium turnover as measured by biological markers. Baseline serum sICAM-1 levels and COMP levels may be helpful to identify patients with progressive or non-progressive radiological outcome.
Full Text
The Full Text of this article is available as a PDF (151.1 KB).
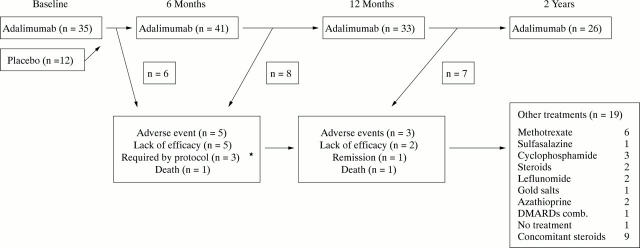
Figure 1 .
Scheme of patients receiving treatment, primary reasons for drop out, and treatments used thereafter (box) during the two year follow up. *Patients who did not respond after three administrations of adalimumab were withdrawn according to the study protocol.
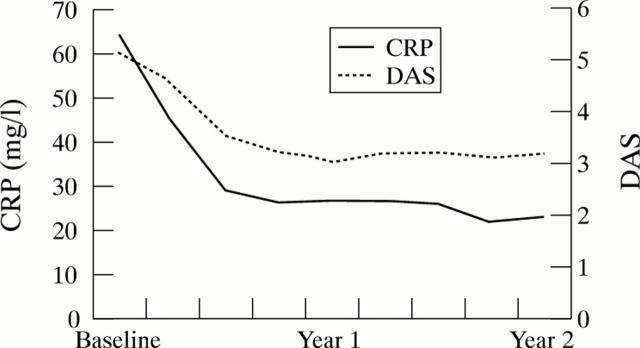
Figure 2 .
Course of the median DAS and CRP during the two year follow up. Intention to treat analysis.
Figure 3 .
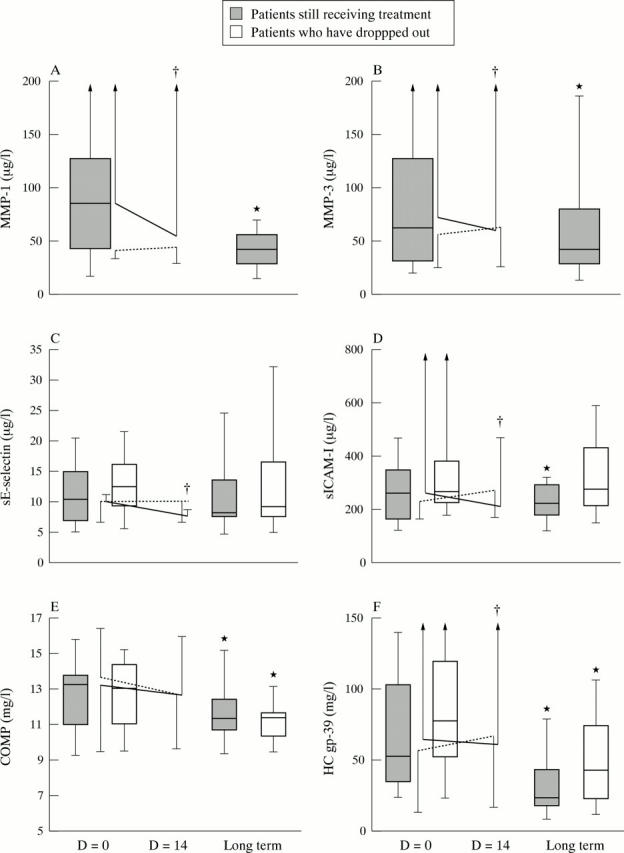
Short and long term changes in markers of synovium turnover (metalloproteinases MMP-1 (A), MMP-3 (B), endothelial activation (sE-selectin (C), sICAM-1 (D)) and cartilage and COMP (E), and HC gp-39 (F)). Short time changes: changes seen within two weeks (D=0 to D=14) after the first dose of anti-TNFα (continuous line) or placebo (dashed line) in the double blind phase. Long term changes: changes occurring between baseline and the two years follow up. The boxes illustrate the mean (horizontal bar), 25th and 75th centiles (box) and 5th and 95th centiles (bars). Significance is shown as p<0.05, tested within groups (*) and between groups (†).
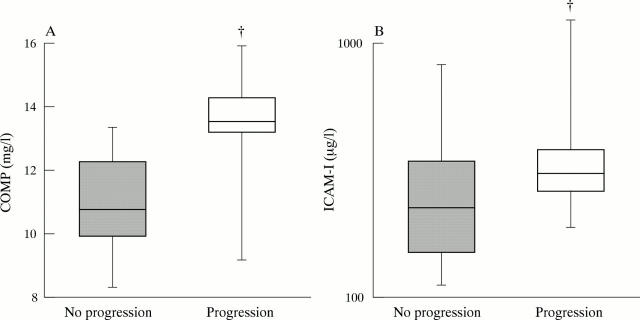
Figure 4 .
Concentrations of COMP (A) and sICAM-1 (B) in patients with and without radiological progression. The boxes illustrate the mean (horizontal bar), 25th and 75th centiles (box) and 5th and 95th centiles (bars). Significance is shown as p<0.05, tested between groups (†).
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aoki S., Imai K., Yachi A. Soluble intercellular adhesion molecule-1 (ICAM-1) antigen in patients with rheumatoid arthritis. Scand J Immunol. 1993 Nov;38(5):485–490. doi: 10.1111/j.1365-3083.1993.tb02592.x. [DOI] [PubMed] [Google Scholar]
- Arnett F. C., Edworthy S. M., Bloch D. A., McShane D. J., Fries J. F., Cooper N. S., Healey L. A., Kaplan S. R., Liang M. H., Luthra H. S. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988 Mar;31(3):315–324. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- Bathon J. M., Martin R. W., Fleischmann R. M., Tesser J. R., Schiff M. H., Keystone E. C., Genovese M. C., Wasko M. C., Moreland L. W., Weaver A. L. A comparison of etanercept and methotrexate in patients with early rheumatoid arthritis. N Engl J Med. 2000 Nov 30;343(22):1586–1593. doi: 10.1056/NEJM200011303432201. [DOI] [PubMed] [Google Scholar]
- Brennan F. M., Browne K. A., Green P. A., Jaspar J. M., Maini R. N., Feldmann M. Reduction of serum matrix metalloproteinase 1 and matrix metalloproteinase 3 in rheumatoid arthritis patients following anti-tumour necrosis factor-alpha (cA2) therapy. Br J Rheumatol. 1997 Jun;36(6):643–650. doi: 10.1093/rheumatology/36.6.643. [DOI] [PubMed] [Google Scholar]
- Dolhain R. J., van der Heiden A. N., ter Haar N. T., Breedveld F. C., Miltenburg A. M. Shift toward T lymphocytes with a T helper 1 cytokine-secretion profile in the joints of patients with rheumatoid arthritis. Arthritis Rheum. 1996 Dec;39(12):1961–1969. doi: 10.1002/art.1780391204. [DOI] [PubMed] [Google Scholar]
- Elliott M. J., Maini R. N., Feldmann M., Kalden J. R., Antoni C., Smolen J. S., Leeb B., Breedveld F. C., Macfarlane J. D., Bijl H. Randomised double-blind comparison of chimeric monoclonal antibody to tumour necrosis factor alpha (cA2) versus placebo in rheumatoid arthritis. Lancet. 1994 Oct 22;344(8930):1105–1110. doi: 10.1016/s0140-6736(94)90628-9. [DOI] [PubMed] [Google Scholar]
- Fex E., Eberhardt K., Saxne T. Tissue-derived macromolecules and markers of inflammation in serum in early rheumatoid arthritis: relationship to development of joint destruction in hands and feet. Br J Rheumatol. 1997 Nov;36(11):1161–1165. doi: 10.1093/rheumatology/36.11.1161. [DOI] [PubMed] [Google Scholar]
- Forslind K., Eberhardt K., Jonsson A., Saxne T. Increased serum concentrations of cartilage oligomeric matrix protein. A prognostic marker in early rheumatoid arthritis. Br J Rheumatol. 1992 Sep;31(9):593–598. doi: 10.1093/rheumatology/31.9.593. [DOI] [PubMed] [Google Scholar]
- Hulsmans H. M., Jacobs J. W., van der Heijde D. M., van Albada-Kuipers G. A., Schenk Y., Bijlsma J. W. The course of radiologic damage during the first six years of rheumatoid arthritis. Arthritis Rheum. 2000 Sep;43(9):1927–1940. doi: 10.1002/1529-0131(200009)43:9<1927::AID-ANR3>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- Kavanaugh A. F., Davis L. S., Jain R. I., Nichols L. A., Norris S. H., Lipsky P. E. A phase I/II open label study of the safety and efficacy of an anti-ICAM-1 (intercellular adhesion molecule-1; CD54) monoclonal antibody in early rheumatoid arthritis. J Rheumatol. 1996 Aug;23(8):1338–1344. [PubMed] [Google Scholar]
- Kempeni J. Preliminary results of early clinical trials with the fully human anti-TNFalpha monoclonal antibody D2E7. Ann Rheum Dis. 1999 Nov;58 (Suppl 1):I70–I72. doi: 10.1136/ard.58.2008.i70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyszer G., Lambiri I., Nagel R., Keysser C., Keysser M., Gromnica-Ihle E., Franz J., Burmester G. R., Jung K. Circulating levels of matrix metalloproteinases MMP-3 and MMP-1, tissue inhibitor of metalloproteinases 1 (TIMP-1), and MMP-1/TIMP-1 complex in rheumatic disease. Correlation with clinical activity of rheumatoid arthritis versus other surrogate markers. J Rheumatol. 1999 Feb;26(2):251–258. [PubMed] [Google Scholar]
- Lassere M., Boers M., van der Heijde D., Boonen A., Edmonds J., Saudan A., Verhoeven A. C. Smallest detectable difference in radiological progression. J Rheumatol. 1999 Mar;26(3):731–739. [PubMed] [Google Scholar]
- Leeuwenberg J. F., Smeets E. F., Neefjes J. J., Shaffer M. A., Cinek T., Jeunhomme T. M., Ahern T. J., Buurman W. A. E-selectin and intercellular adhesion molecule-1 are released by activated human endothelial cells in vitro. Immunology. 1992 Dec;77(4):543–549. [PMC free article] [PubMed] [Google Scholar]
- Lipsky P. E., van der Heijde D. M., St Clair E. W., Furst D. E., Breedveld F. C., Kalden J. R., Smolen J. S., Weisman M., Emery P., Feldmann M. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group. N Engl J Med. 2000 Nov 30;343(22):1594–1602. doi: 10.1056/NEJM200011303432202. [DOI] [PubMed] [Google Scholar]
- Littler A. J., Buckley C. D., Wordsworth P., Collins I., Martinson J., Simmons D. L. A distinct profile of six soluble adhesion molecules (ICAM-1, ICAM-3, VCAM-1, E-selectin, L-selectin and P-selectin) in rheumatoid arthritis. Br J Rheumatol. 1997 Feb;36(2):164–169. doi: 10.1093/rheumatology/36.2.164. [DOI] [PubMed] [Google Scholar]
- Lorenz H. M., Antoni C., Valerius T., Repp R., Grünke M., Schwerdtner N., Nüsslein H., Woody J., Kalden J. R., Manger B. In vivo blockade of TNF-alpha by intravenous infusion of a chimeric monoclonal TNF-alpha antibody in patients with rheumatoid arthritis. Short term cellular and molecular effects. J Immunol. 1996 Feb 15;156(4):1646–1653. [PubMed] [Google Scholar]
- Molenaar E. T., Boers M., van der Heijde D. M., Alarcón G., Bresnihan B., Cardiel M., Edmonds J., Felson D., Furst D. E., Kirwan J. Imaging in rheumatoid arthritis: results of group discussions. J Rheumatol. 1999 Mar;26(3):749–751. [PubMed] [Google Scholar]
- Moreland L. W., Schiff M. H., Baumgartner S. W., Tindall E. A., Fleischmann R. M., Bulpitt K. J., Weaver A. L., Keystone E. C., Furst D. E., Mease P. J. Etanercept therapy in rheumatoid arthritis. A randomized, controlled trial. Ann Intern Med. 1999 Mar 16;130(6):478–486. doi: 10.7326/0003-4819-130-6-199903160-00004. [DOI] [PubMed] [Google Scholar]
- Månsson B., Carey D., Alini M., Ionescu M., Rosenberg L. C., Poole A. R., Heinegård D., Saxne T. Cartilage and bone metabolism in rheumatoid arthritis. Differences between rapid and slow progression of disease identified by serum markers of cartilage metabolism. J Clin Invest. 1995 Mar;95(3):1071–1077. doi: 10.1172/JCI117753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paleolog E. M., Hunt M., Elliott M. J., Feldmann M., Maini R. N., Woody J. N. Deactivation of vascular endothelium by monoclonal anti-tumor necrosis factor alpha antibody in rheumatoid arthritis. Arthritis Rheum. 1996 Jul;39(7):1082–1091. doi: 10.1002/art.1780390703. [DOI] [PubMed] [Google Scholar]
- Paleolog E. M., Young S., Stark A. C., McCloskey R. V., Feldmann M., Maini R. N. Modulation of angiogenic vascular endothelial growth factor by tumor necrosis factor alpha and interleukin-1 in rheumatoid arthritis. Arthritis Rheum. 1998 Jul;41(7):1258–1265. doi: 10.1002/1529-0131(199807)41:7<1258::AID-ART17>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- Posthumus M. D., Limburg P. C., Westra J., Cats H. A., Stewart R. E., van Leeuwen M. A., van Rijswijk M. H. Serum levels of matrix metalloproteinase-3 in relation to the development of radiological damage in patients with early rheumatoid arthritis. Rheumatology (Oxford) 1999 Nov;38(11):1081–1087. doi: 10.1093/rheumatology/38.11.1081. [DOI] [PubMed] [Google Scholar]
- Saxne T., Heinegård D. Cartilage oligomeric matrix protein: a novel marker of cartilage turnover detectable in synovial fluid and blood. Br J Rheumatol. 1992 Sep;31(9):583–591. doi: 10.1093/rheumatology/31.9.583. [DOI] [PubMed] [Google Scholar]
- So A., Chamot A. M., Péclat V., Gerster J. C. Serum MMP-3 in rheumatoid arthritis: correlation with systemic inflammation but not with erosive status. Rheumatology (Oxford) 1999 May;38(5):407–410. doi: 10.1093/rheumatology/38.5.407. [DOI] [PubMed] [Google Scholar]
- Tak P. P., Taylor P. C., Breedveld F. C., Smeets T. J., Daha M. R., Kluin P. M., Meinders A. E., Maini R. N. Decrease in cellularity and expression of adhesion molecules by anti-tumor necrosis factor alpha monoclonal antibody treatment in patients with rheumatoid arthritis. Arthritis Rheum. 1996 Jul;39(7):1077–1081. doi: 10.1002/art.1780390702. [DOI] [PubMed] [Google Scholar]
- Taylor D. J., Cheung N. T., Dawes P. T. Increased serum proMMP-3 in inflammatory arthritides: a potential indicator of synovial inflammatory monokine activity. Ann Rheum Dis. 1994 Nov;53(11):768–772. doi: 10.1136/ard.53.11.768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vos K., Steenbakkers P., Miltenburg A. M., Bos E., van Den Heuvel M. W., van Hogezand R. A., de Vries R. R., Breedveld F. C., Boots A. M. Raised human cartilage glycoprotein-39 plasma levels in patients with rheumatoid arthritis and other inflammatory conditions. Ann Rheum Dis. 2000 Jul;59(7):544–548. doi: 10.1136/ard.59.7.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamanaka H., Matsuda Y., Tanaka M., Sendo W., Nakajima H., Taniguchi A., Kamatani N. Serum matrix metalloproteinase 3 as a predictor of the degree of joint destruction during the six months after measurement, in patients with early rheumatoid arthritis. Arthritis Rheum. 2000 Apr;43(4):852–858. doi: 10.1002/1529-0131(200004)43:4<852::AID-ANR16>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- Youssef P. P., Triantafillou S., Parker A., Coleman M., Roberts-Thomson P. J., Ahern M. J., Smith M. D. Effects of pulse methylprednisolone on cell adhesion molecules in the synovial membrane in rheumatoid arthritis. Reduced E-selectin and intercellular adhesion molecule 1 expression. Arthritis Rheum. 1996 Dec;39(12):1970–1979. doi: 10.1002/art.1780391205. [DOI] [PubMed] [Google Scholar]
- van Der Heijde D., Boonen A., Boers M., Kostense P., van Der Linden S. Reading radiographs in chronological order, in pairs or as single films has important implications for the discriminative power of rheumatoid arthritis clinical trials. Rheumatology (Oxford) 1999 Dec;38(12):1213–1220. doi: 10.1093/rheumatology/38.12.1213. [DOI] [PubMed] [Google Scholar]
- van Leeuwen M. A., van Rijswijk M. H., van der Heijde D. M., Te Meerman G. J., van Riel P. L., Houtman P. M., van De Putte L. B., Limburg P. C. The acute-phase response in relation to radiographic progression in early rheumatoid arthritis: a prospective study during the first three years of the disease. Br J Rheumatol. 1993 Jun;32 (Suppl 3):9–13. doi: 10.1093/rheumatology/32.suppl_3.9. [DOI] [PubMed] [Google Scholar]
- van der Heijde D. M., van 't Hof M. A., van Riel P. L., Theunisse L. A., Lubberts E. W., van Leeuwen M. A., van Rijswijk M. H., van de Putte L. B. Judging disease activity in clinical practice in rheumatoid arthritis: first step in the development of a disease activity score. Ann Rheum Dis. 1990 Nov;49(11):916–920. doi: 10.1136/ard.49.11.916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Heijde D. M., van Riel P. L., Nuver-Zwart I. H., Gribnau F. W., vad de Putte L. B. Effects of hydroxychloroquine and sulphasalazine on progression of joint damage in rheumatoid arthritis. Lancet. 1989 May 13;1(8646):1036–1038. doi: 10.1016/s0140-6736(89)92442-2. [DOI] [PubMed] [Google Scholar]
- van der Heijde D., Boers M., Lassere M. Methodological issues in radiographic scoring methods in rheumatoid arthritis. J Rheumatol. 1999 Mar;26(3):726–730. [PubMed] [Google Scholar]
- van der Heijde D. How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol. 1999 Mar;26(3):743–745. [PubMed] [Google Scholar]