Abstract
CTLA-4-deficient mice develop a fatal lymphoproliferative disorder, characterized by polyclonal expansion of peripheral lymphocytes. To examine the effect of restricting the CD4+ TCR repertoire on the phenotype of CTLA-4-deficient mice and to assess the influence of CTLA-4 on peptide-specific CD4+ T cell responses in vitro, an MHC class II-restricted T cell receptor (AND TCR) transgene was introduced into the CTLA-4−/− animals. The expression of the AND TCR transgene by CD4+ T cells delays but does not prevent the lymphoproliferation in the CTLA-4−/− mice. The CD4+ T cells become preferentially activated and expand. Interestingly, young AND TCR+ CTLA-4−/− mice carrying a null mutation in the rag-1 gene remain healthy and the T cells maintain a naive phenotype until later in life. We demonstrate that CTLA-4 regulates the peptide-specific proliferative response generated by naive and previously activated AND TCR+ RAG−/− T cells in vitro. The absence of CTLA-4 also augments the responder frequency of cytokine-secreting AND TCR+ RAG−/− T cells. These results demonstrate that CTLA-4 is a key regulator of peptide-specific CD4+ T cell responses and support the model that CTLA-4 plays a differential role in maintaining T cell homeostasis of CD4+ vs. CD8+ T cells.
Keywords: CD152, inhibitory costimulation, homeostasis, tolerance
Optimal T cell activation requires two signals, one via the T cell receptor (TCR) and the second via costimulatory molecules. The primary positive costimulatory molecule for T cell activation is CD28 (1). CD28-mediated costimulation functions to facilitate and sustain T cell-proliferative responses (1–3). Conversely, recent studies indicate that CD28 homologue CTLA-4 inhibits T cell responses (4). CTLA-4-mediated costimulation inhibits T cell-proliferative and cytokine responses in vitro, and blockade of CTLA-4/ligand interaction enhances T cell responses in vivo (4). Although CD28 and CTLA-4 share common ligands, namely CD80 (B7.1) and CD86 (B7.2), it appears that CTLA-4 does not function simply to counteract CD28-mediated costimulatory signals; rather, accumulating evidence indicates that CTLA-4 may directly modulate proximal TCR signaling (5–11).
CTLA-4-deficient mice die at 3–4 weeks of age as a result of a lymphoproliferative disorder caused by polyclonal T cell expansion (12–14). Peripheral CD4+ T cells are primarily responsible for the lymphoproliferative disorder in CTLA-4−/− mice expressing an unmanipulated TCR repertoire (15). Mice depleted of CD8+ T cells from birth by antibody treatment develop a lymphoproliferative disorder similar in onset and severity to unmanipulated CTLA-4−/− mice, whereas lymphoproliferation does not occur in CTLA-4−/− mice depleted of CD4+ T cells from birth. Moreover, the CD8+ T cells in these animals maintain a naive phenotype. These results suggest that CTLA-4 has differential effects on the CD4+ vs. the CD8+ T cells in vivo.
The nature of TCR–ligand interaction that initiates the lymphoproliferation and the extent to which individual naive CD4+ T cells become dysregulated by the absence of CTLA-4 in vivo is unknown. Based on the phenotype of the CTLA-4−/− mice, we proposed (15, 16) that CTLA-4 may provide an inhibitory signal that prevents CD4+ T cell activation during the continuous interaction of TCR and MHC that is shown to be required for T cell survival (17–21). Further, we have proposed a two-signal model for T cell activation in which T cell responses are regulated by the dynamic integration of TCR, CD28, and CTLA-4 signals (14). To test these models and to determine antigenic peptide reactivity of naive T cells in the absence of CTLA-4, we have initiated studies using MHC class I or class II-restricted TCR transgenic CTLA-4−/− mice.
The role of CTLA-4 in regulating homeostasis and antigen reactivity of CD8+ T cells has been examined in three different MHC class I-restricted TCR transgenic mouse strains (21–23). CTLA-4−/− mice expressing MHC class I-restricted TCR transgenes remain healthy for several months but eventually develop a lymphoproliferative disorder, apparently initiated by activation of the normally small population of CD4+ T cells present in these animals (22, 23). The CD8+ TCR transgenic CTLA-4−/− T cells maintain a naive phenotype in these animals (23, 24). The magnitude and the kinetics of the peptide-specific proliferative and cytokine responses by naive CD8+ peptide-specific responses are equivalent between the CTLA-4−/− and littermate control T cells (23, 24), but there is a dramatic increase in the magnitude of secondary peptide-specific responses by 2C TCR+ CD8+ CTLA-4−/− compared with wild-type T cells (23).
In this report we examine the effects of the CTLA-4 null mutation on the development and function of T cells expressing an MHC class II-restricted TCR. The CTLA-4+/− mice were crossed to an MHC class II-restricted TCR (AND TCR) (Vβ3Vα11) transgenic mouse strain that is selected on MHC class II Ab molecules and specific for pigeon cytochrome c (PCC88–104) in the context of I-Ek (25), to generate CTLA-4-deficient AND TCR transgenic mice (AND TCR+ CTLA4−/−). Further, we introduced a RAG null mutation in these mice to generate AND TCR+RAG−/− CTLA-4−/− mice that express only the transgenic TCR. We demonstrate that although there is a delay in the onset, AND TCR+ CTLA-4−/− mice develop a fatal lymphoproliferative disorder mediated primarily by activated CD4+ T cells whereas the minor CD8+ population present in these mice maintain a naive phenotype. Interestingly, CD4+ T cells in young AND TCR+ RAG−/− CTLA-4−/− mice were not activated, and these mice remained viable. These naive CD4+ CTLA-4−/− AND TCR+ T cells consistently exhibited an increase in the magnitude of the in vitro primary and secondary peptide-specific proliferative response compared with the CTLA-4+/− littermate controls. There also was an increase in the frequency of responding T cells, as measured by cytokine production. These findings indicate that CTLA-4 functions to regulate the magnitude of antigen-specific T cell responses by CD4+ T cells. Moreover, the results further strengthen the notion of a differential role of CTLA-4 in naive CD4 and CD8 T cells. The implications of the results on the integrative two-signal model of T cell activation are discussed.
METHODS AND MATERIALS
Mice.
CTLA-4+/− mice (26) were backcrossed six times to C57BL/6 before being crossed to B6 AND TCR transgenic mice (25). AND TCR+ CTLA-4+/− mice were intercrossed to obtain TCR transgenic CTLA-4+/+, +/−, and CTLA-4−/− animals. Animals were genotyped for the ctla-4 gene mutation by PCR and for the TCR transgene by flow cytometry of peripheral blood lymphocytes. Mice were backcrossed further to B6 RAG-1−/− (ref. 27; The Jackson Laboratory). Animals were maintained in microisolators in accordance with the Animal Care and Use Regulations, University of California, Berkeley.
Medium and Reagents.
Medium was prepared as described (15). Recombinant human IL-2, 3-amino-9-ethylcarbazole (AEC) substrate, and streptavidin-horseradish peroxidase (HRPO) were obtained from Cetus, Sigma, and Jackson ImmunoResearch, respectively. Peptides PCC88–104 (28) and MCC93–103 (29) were synthesized by solid-phase synthesis (Cancer Research Laboratory, Univ. of California Berkeley).
mAbs used were anti (α)-CD4-TC, αCD44-PE, H57-biotin, αCD69-FITC, αCD25-FITC, αCD62L-PE, αCD45RB-FITC, αVα11-FITC/PE, αVβ3-biotin, R46A2, and XMG1.2-biotin (all from PharMingen), αCD8α-TC, streptavidin-TC, and streptavidin-PE (Caltag, South San Francisco, CA), and αCD4–613 and αCD8–613 (GIBCO).
Immunofluorescence Staining and Flow Cytometry.
Samples from single cell suspensions from the lymph nodes and spleen were prepared as described (15). The samples were run on an XL (Coulter), and 40,000 events gated on live cells were collected. The files were analyzed by using elite and winmd software. Data are displayed as four logarithmic histograms or dotplots.
Proliferation Assays and ELISA.
LN cells from AND TCR+ RAG−/−CTLA-4−/−and littermate controls were isolated and were routinely >96% Vβ3+Vα11+ AND TCR+ T cells. LN cells were stimulated (2 × 104 cell per well) with irradiated L cells transfected with I-Ek [antigen-presenting cell (APC); ref. 30]. The L cells were sorted to obtain a stable cell line expressing high homogenous levels of I-Ek and low levels of endogenous B7.1. The plates were incubated for 96 hr and had been pulsed with [3H]thymidine (Amersham Pharmacia; 1 μCi/well) 12–14 hr before harvesting. In some experiments, supernatants were removed before pulsing and assayed on cytokine-specific ELISAs, as described (31). LN cells and splenocytes were stimulated (3 × 106 cell per well) with irradiated APC plus PCC88–104 (5 μM) and expanded for 7–10 days with rhIL-2 (10 units/ml). The cells were collected, reisolated, and used in proliferation assays and/or enzyme-linked immunospot (ELISPOT) assays. All cells assayed were AND TCR+ CD44hi after stimulation in vitro.
ELISPOT.
ELISPOT analysis was performed as described (32). Briefly, LNT cells from AND TCR+ RAG−/−CTLA-4−/− and littermate control mice were plated at the indicated dilutions in 96-well plates (HA-Millipore) coated with anti-IL-4 antibody (5 μg/ml) and stimulated with irradiated APC (5 × 104 cell per well) plus rhIL-2 (50 units/ml) ± peptide for 22 or 36 hr. The plates were washed and incubated overnight at 4°C with biotinylated anti-IL-4 antibody (5 μg/ml). The plates were washed and incubated with streptavidin-HRPO (1:1,000), and washed again, and AEC substrate was added and developed for 15 min. The spots were scored by using a dissecting microscope. No cytokine-specific spots were detected in wells with T cells only or with APC in the absence of specific peptide. All of the AND TCR+ RAG−/− T cells, independent of the CTLA-4 genotype, become activated by day 5 after stimulation under the conditions used in ELISPOT assays, as determined by carboxyl-fluorescein diacetate succinimidyl ester labeling and CD44 up-regulation (data not shown).
RESULTS
Phenotype of the AND TCR+ CTLA-4−/− Mice.
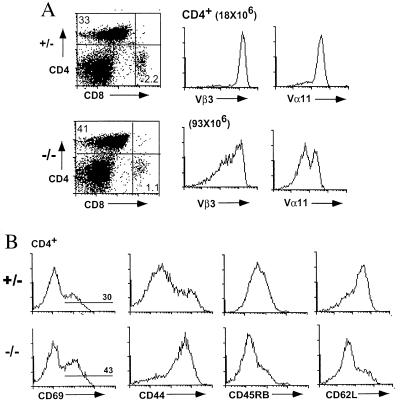
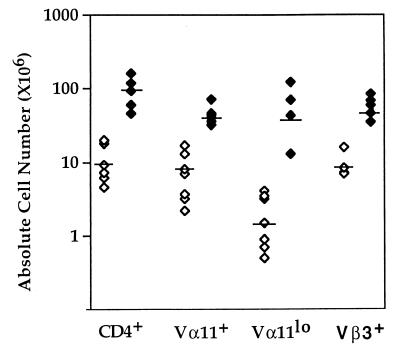
The CTLA-4 mutation was introduced into AND TCR+ mice and the mice were analyzed at 4–8 weeks of age. The expected skewing toward CD4+ T cells was observed in the AND TCR+ CTLA-4+/+ or CTLA-4+/− (AND TCR+ CTLA-4+) mice, and >90% of the lymph node T cells expressed high levels of the Vβ3 and Vα11 TCR transgene (Fig. 1A). In the absence of CTLA-4, there was a dramatic effect on the number and phenotype of the cells in AND TCR+ mice. Although the onset was delayed compared with CTLA-4−/− animals with an unmanipulated TCR repertoire, the lymphoproliferation was readily apparent at 1 month of age and the animals died at approximately 8–12 weeks of age, a delay of 5–8 weeks compared with CTLA-4−/− mice expressing an unmanipulated TCR repertoire. There was an increase of >10-fold in the absolute number of CD4+ lymph node T cells (Fig. 2), including the AND TCR+ T cells and the lymph node T cells expressing endogenous TCR chains (CD3+ H57+Vβ3lo and/or Vα11lo: >50-fold increase) (Figs. 1A and 2) in the CTLA-4−/− animals compared with the CTLA-4+ littermate controls.
Figure 1.
CD4+ T cells become activated in AND TCR+ mice in the absence of CTLA-4. Representative flow cytometry data of the lymph node cells from AND TCR+ CTLA-4−/− and littermate control animals. The expression of AND TCR transgene (A) and the phenotype of the CD4+ lymph node T cells (B) from an 8-week-old AND TCR+ CTLA-4−/− mouse and littermate control. The absolute number of the CD4+ T cells is indicated.
Figure 2.
Absolute cell number of CD4+ T cells expressing AND TCR+ and endogenous TCR chains increase in CTLA-4-deficient animals. Single cell suspensions were prepared from the spleen and lymph nodes and stained for CD4 and Vβ3, Vα11, and the absolute number of each subpopulation was determined. Each dot represents a single animal. AND TCR+ CTLA-4+/+, +/−, n = 6 (⋄); CTLA-4−/−, n = 5 (♦).
Most of the CD4+ T cells in the AND TCR+ CTLA-4+ mice had a naive phenotype and were CD44lo, MEL14hi, CD45RBhi (Fig. 1B). In contrast, virtually all of the CD4+ T cells in the AND TCR+ CTLA-4−/− mice, including the AND TCR+ and T cells expressing endogenous TCR chains, had an activated phenotype and were CD69+, CD44hi, CD62Llo, and CD45RBlo (Fig. 1B). The activated phenotype was observed by 4 weeks of age, the earliest time analyzed.
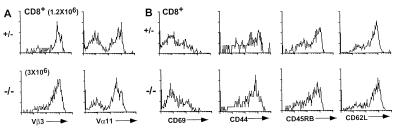
There was a small population (1–3%) of TCRαβ+ (H57+) CD8+ T cells in the lymph node of CTLA-4+ littermate control and CTLA-4−/− AND TCR+ mice (Fig. 1A). Virtually all of the CD8+ T cells expressed the Vβ3 TCR transgene, and routinely one-third to one-half of the CD8+ T cells expressed Vα11 of the AND TCR (Fig. 3A). The absolute number of CD8+ T cells eventually did increase in the AND TCR+ CTLA-4−/− animals (AND TCR+ CTLA-4+, 0.73 ± 0.6 × 106, n = 6; AND TCR+ CTLA-4−/−, 4.6 ± 3.2 × 106, n = 4), but they continued to decrease as a percentage of T cells in the lymph node and spleen. Further, the CD8+ T cells displayed a relatively naive phenotype that was indistinguishable between the CTLA-4-deficient and littermate control animals (Fig. 3B). This occurred despite the expression of endogenous TCR chains and the availability of CD4+-mediated T cell help.
Figure 3.
CD8+ T cells do not become activated in AND TCR+ mice in the absence of CTLA-4. Shown are representative flow cytometry data of the AND TCR transgene (A) and the phenotype (B) of CD8+ lymph node cells from AND TCR+ CTLA-4−/− and littermate control mice shown in Fig. 1. The absolute number of the CD8+ T cells is indicated.
AND TCR+ CTLA-4−/− RAG−/− LNT Cells Maintain a Naive Phenotype in Young Animals.
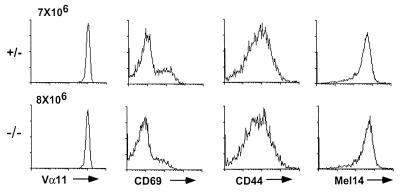
To examine the effect of restricting the TCR repertoire, the null mutation in RAG-1 (27) was introduced into the AND TCR+ CTLA-4+/− mice to obtain CTLA-4−/− and CTLA-4+ AND TCR+ RAG−/− mice. Unlike their RAG+ counterparts, T cells from AND TCR+ CTLA-4−/− RAG−/− mice express a single TCR complex. There was a noticeable (but not statistically significant) difference in the total number of AND TCR+ T cells in the lymph node of the CTLA-4−/− and littermate mice (AND TCR+ CTLA-4+ RAG−/−, 7.0 ± 3.8 × 106, n = 17; AND TCR+ CTLA-4−/− RAG−/−, 14 ± 7.5 × 106, n = 7; ages 4–9 weeks). Further, the T cells maintained a naive phenotype in the absence of CTLA-4 and had TCR levels equivalent to the littermate controls (Fig. 4). Preliminary data suggest that the CD4+ T cells eventually do become activated in aged AND TCR+ CTLA-4−/− RAG−/− mice (unpublished result). It remains to be determined whether the delay in the onset of T cell activation and absence of rampant lymphoproliferation in these animals is caused by the monoclonality of the TCR repertoire and/or unique lymphoid organ architecture in RAG−/− mice as a result of the absence of B cells.
Figure 4.
CD4+ AND TCR+ CTLA-4−/− RAG-1−/− T cells do not become activated in vivo. Shown are representative flow cytometry data of lymph node CD4+ AND TCR+ CTLA-4+/− and CTLA-4−/− T cells that are RAG-1−/− (7 weeks old). The absolute number of AND TCR+ RAG−/− T cells is indicated. The average numbers of lymph node T cells in these animals are: AND TCR+ CTLA-4+ RAG−/−, 7.0 ± 3.8 × 106, n = 17; AND TCR+ CTLA-4−/− RAG−/−, 14 ± 7.5 × 106, n = 7.
Primary and Secondary Antigen-Specific Proliferative Responses by CD4+ AND TCR+ RAG−/− T Cells Are Greater in the Absence of CTLA-4.
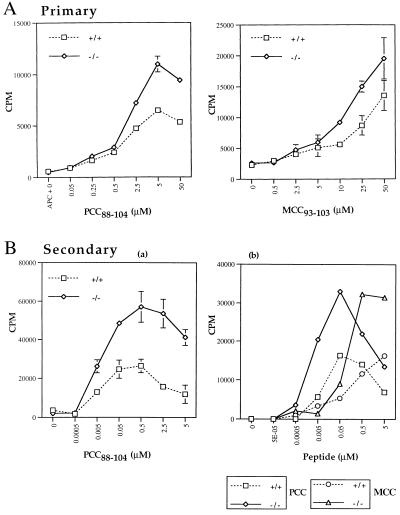
Because almost all of the AND TCR+ T cells in the RAG+ CTLA-4-deficient mice become activated in vivo, it is not possible to compare the peptide-specific responses of the naive T cells from the CTLA-4-deficient and littermate animals. However, naive AND TCR+ T cells could be obtained from young AND TCR+ RAG−/− CTLA-4−/− mice and were used to assess the effects of the absence of CTLA-4 on peptide-specific responses in vitro. Representative data of the primary proliferative response to peptides PCC88–104 and MCC93–103 presented by I-Ek+ B7.1lo L cells (APC) are shown in Fig. 5. The AND TCR+ RAG−/− CTLA-4+ T cells generate a response to PCC88–104 over a range of 0.5 to 50 μM (Fig. 5A), as reported previously (33). The response to PCC88–104 by AND TCR+ RAG−/− CTLA-4−/− T cells was similar to littermate control T cells at lower peptide concentrations; however, there was a small but consistent 1.5- to 2-fold increase in the magnitude of the proliferative response by the CTLA-4-deficient T cells at the highest peptide concentrations (Fig. 5A). Similar results were obtained with MCC93–103, although the minimal peptide concentration required to generate a measurable proliferative response was higher (2.5 μM; Fig. 5A).
Figure 5.
Primary and secondary proliferative responses by AND TCR+ RAG−/− T cells are greater in the absence of CTLA-4. (A) Naive AND TCR+ RAG−/− CTLA-4-deficient and littermate control lymph node T cells were isolated and stimulated (2 × 104 cell per well) with irradiated I-Ek-transfected L cells (2 × 104 cell per well) and pigeon or moth cytochrome c (PCC88–104 and MCC93–103, respectively). (B) Secondary response. AND TCR+ RAG−/− T cells from CTLA-4−/− and littermate control animals were expanded in vitro as described in Methods and Materials. The cells were collected, ficolled, and restimulated with PCC88–104 or MCC93–103 as described above. These are representative data of at least three experiments.
To examine the effect of the absence of CTLA-4 on the magnitude of the secondary peptide-specific response by naive CD4+ T cells, AND TCR+ RAG−/− CTLA-4−/− and littermate control LNT cells were stimulated with PCC (5 μM) and APC and expanded with IL-2 (15 units/ml) in vitro for 7–10 days. The cells then were harvested, ficolled to remove debris, and restimulated. Restimulation of the AND TCR+ RAG−/− CTLA-4+ littermate control cells with PCC88–104 or MCC93–103 generated a more robust response compared with the primary response, and there was routinely a 2- to 3-fold shift in the dose-response curve (maximal proliferation occurring at 0.05–0.5 μM compared with 5–50 μM in the primary response), demonstrating a typical secondary response (Fig. 5B). Most interestingly, in the absence of CTLA-4, the AND TCR+ RAG−/− T cells generated a greater peptide-specific secondary response compared with the littermate controls (Fig. 5B). There was not, however, a greater response by the CTLA-4−/− T cells at peptide concentrations to which the CTLA-4-littermate control mice did not respond, suggesting that below a certain level of TCR occupancy, the magnitude of the response is not influenced by CTLA-4.
Increase in the Responder-Precursor Frequency During the Peptide-Specific Response in the Absence of CTLA-4.
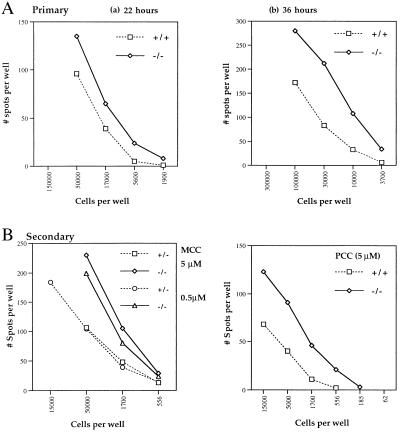
Another measure of T cell responsiveness is the ability to synthesize and secrete cytokines. To examine whether the absence of CTLA-4 influences the frequency of cytokine-secreting T cells, serial dilutions of AND TCR+ RAG-1−/− CTLA-4−/− and littermate control T cells were stimulated with APC ± peptide and the responder frequency was determined by ELISPOT. Using IL-2-, IFN-γ-, and IL-4-specific ELISAs, it was determined that the AND TCR+ RAG−/− T cells predominantly secrete IL-4 in response to primary (20 pg/ml; 50 μM peptide) and secondary (≥500 pg/ml; 5 μM peptide) stimulation with peptides PCC and MCC. ELISPOT assays for IL-2, IFN-γ, and IL-4 confirmed that IL-4 was the predominant cytokine secreted by both the littermate control and the CTLA-4-deficient T cells. Based on these results, ELISPOT assays to detect IL-4 secretion were performed. Representative data of the frequency of the IL-4 responders to primary stimulation of AND TCR+ RAG−/− CTLA-4+ T cells with MCC93–103 (50 μM) plus APC after 22 or 36 hr of incubation are shown in Fig. 6A. There was a consistently higher responder frequency of T cells secreting IL-4 in the absence of AND TCR+ RAG−/− CTLA-4−/− compared with the littermate control T cells (Fig. 6A). Stimulation with PCC88–104 yielded similar results (approximately 1:1,000 and 1:600 IL-4 secretors after 36-hr stimulation of naive AND TCR+ RAG-1−/− control and CTLA-4−/− T cells, respectively). The frequency of T cell responders was increased dramatically and required lower peptide concentrations upon restimulation of previously activated AND TCR+ RAG−/− control and CTLA-4−/− T cells with either PCC88–104 or MCC93–103 (Fig. 6B). Further, the frequency of T cell responders was 3- to 4-fold higher in the absence of CTLA-4 at most input cell doses (Fig. 6B). In some experiments the frequency of the responders was as high as 1 in 5 for littermate and 1 in 2 of CTLA-4−/− T cells plated (data not shown).
Figure 6.
Frequency of AND TCR+ T cells secreting IL-4 in response to primary and secondary PCC stimulation in vitro is increased in the absence of CTLA-4. (A) Naive AND TCR+ RAG−/− T cells were stimulated in vitro with I-Ek+ B7lo L cells plus MCC93–103 at the indicated cell number on plates coated with anti-IL-4 antibody, as described in Methods and Materials. After incubation for 22 (a) or 36 (b) hr, the plates were developed and scored for IL-4 secretion. (B) Previously activated AND TCR+ RAG−/− T cells were stimulated with MCC93–103 (5 or 0.5 μM) or PCC88–104 (0.5 μM) plus APC for 36 hr as described above. These are representative data of at least five experiments.
DISCUSSION
In this report we describe the influence of CTLA-4 on T cell homeostasis and antigen responsiveness in CTLA-4−/− mice expressing an MHC class II-restricted TCR transgene. The results indicate that (i) the TCR repertoire of the CD4+ T cells influences the manifestation of the CTLA-4 null phenotype in vivo; (ii) CTLA-4 regulates the magnitude of naive and previously activated CD4+ T cell responses; and (iii) CTLA-4 differentially affects CD4+ vs. CD8+ T cell subsets.
CTLA-4 Regulates the Magnitude of the Peptide-Specific Response by Monoclonal T Cells.
The absence of CTLA-4 results in an increase in the magnitude of primary and, to a greater degree, secondary responses of peptide-specific CD4+ T cells in vitro (Fig. 5). This observation is consistent with the hypothesis that CTLA-4 functions to attenuate T cell responses. In both the primary and secondary stimulations, the greater response by the CTLA-4-deficient T cells occurs at peptide concentrations that generate a detectable proliferative response by the littermate control T cells. This result suggests that under these experimental conditions, the level of TCR occupancy and not CTLA-4 is limiting T cell activation at the lowest peptide concentrations. It is possible that some threshold level of TCR stimulation is necessary for the up-regulation of CTLA-4 to functionally relevant levels that then could inhibit the TCR and/or the CD28-mediated signals. This result is observed with the two strong agonist peptides used in these experiments. It will be important to determine whether altering the strength of the TCR signal by stimulation with weak agonists or inhibition of T cell activation by antagonist peptides modulates the role of CTLA-4 in regulating T cell responses. Further, the regulation of the T cell response by CTLA-4 may vary when higher levels of ligand for CTLA-4/CD28 and/or the LFA-1 ligand intercellular adhesion molecule-1 are available.
CTLA-4 Regulates the Frequency of Responding Peptide-Specific T Cells.
An increase in the frequency of responding naive and previously activated CD4+ T cells in the absence of CTLA-4, as measured by cytokine secretion, was observed. This was detectable as early as 22 hr after stimulation (Fig. 5; earliest time tested). The increase in the frequency of responders in the absence of CTLA-4 may arise because of a higher frequency of activated T cells that produce cytokine in CTLA-4-deficient T cells and/or the CTLA-4−/− T cells exhibit more rapid kinetics for cytokine synthesis and secretion than normal T cells. Regardless of the mechanism, these results clearly demonstrate that the absence of CTLA-4 can influence T cell responses occurring within the first 24 hr of activation of naive CD4+ T cells and support the model that CTLA-4 plays a role in regulating the initiation of T cell responses (14).
It recently has been suggested that multiple cell divisions are required for the synthesis of IL-4 (34, 35). In this study IL-4 secretion was detected 22 hr after activation of the naive T cells from either the littermate control or CTLA-4−/− AND TCR+ RAG−/− T cells, before detectable T cell division (as determined by membrane labeling with carboxyl-fluorescein diacetate succinimidyl ester; data not shown). This result suggests that cell division is not an absolute requirement for the synthesis and secretion of IL-4. The disparity between these results and previous reports may be related to the genetic background of the animals, the stimulation conditions used in vitro, and/or different levels of sensitivity of the assays used to detect cytokine production.
CTLA-4 Deficiency Has a Differential Effect on CD4+ and CD8+ T Cells.
Previously we proposed that CTLA-4 deficiency has a differential effect on CD4+ compared with CD8+ T cells in young mice with an unmanipulated TCR repertoire (15). The results presented here and elsewhere (22, 23) clearly demonstrate that the absence of CTLA-4 has differential effects on CD4+ and CD8+ T cells in animals expressing MHC class II- and class I-restricted TCR repertoires. First, the CTLA-4−/− CD4+ T cells expressing an MHC class II-restricted TCR transgene become activated in the absence of nominal antigen stimulation, whereas CD8+ CTLA-4−/− T cells expressing MHC class I-restricted TCR transgenes maintain a naive phenotype and do not expand until the normally small CD4+ T cell population in the transgenic mice expands (23, 24). In MHC class I-restricted 2C TCR transgenic mice there is a small population (3–5%) of CD4+ T cells expressing the transgenic TCR and/or endogenous TCR chains. This “nonselected” population of CD4+ T cells becomes activated in the absence of CTLA-4, expanding dramatically as a percentage and in absolute number, and mediates the lymphoproliferative disorder in the MHC class I-restricted 2C TCR+ CTLA-4−/− animals (23). A similar expansion of CD4+ T cells was seen in lymphocytic choriomeningitis virus-specific or HY-specific transgenic CTLA-4−/− mice (22). In contrast, CD8+ T cells in the AND TCR+ CTLA-4−/− mice did not undergo dramatic activation and expansion. This occurred despite the fact that at least one-third of the CD8+ T cells express endogenous TCRα and/or TCRβ chains (Fig. 3A). These results are consistent with the notion that the absence of CTLA-4 differentially affects CD4+ and CD8+ T cells in vivo.
The lack of CD8+ T cell activation in the CTLA-4−/− mice in the absence of CD4+ T cells or in the MHC class II-restricted TCR transgenic mice does not mean, however, that CTLA-4 does not regulate CD8+ T cell responses. We have demonstrated previously that there was an increased response in vitro upon secondary stimulation of CD8+ 2C TCR+ T cells that have been activated previously in vitro or in vivo (23). Thus, the activational history of both CD4+ and CD8+ T cells influences the regulation of T cell responses by CTLA-4. At least two mechanisms can be invoked to explain this observation. First, previously activated T cells may express higher levels of CTLA-4. Recently, it was demonstrated that CD4+ T cells that have been activated previously in vivo express a basal level of CTLA-4 ex vivo and that CTLA-4 is up-regulated more quickly and to higher levels upon restimulation in vitro in these cells compared with naive CD4+ T cells (36). However, the functional significance of different levels of CTLA-4 expression is not yet clear. CTLA-4 crosslinking on T cell clones has been shown to have the same functional consequences despite vastly different levels of CTLA-4 expressed by different clones (37). Second, the functional consequences of CTLA-4 ligation may be more apparent in the secondary response, because previously activated CD4+ and CD8+ T cells have a decreased requirement for CD28-mediated costimulation (38). Alternatively, primary T cell activation in the absence/presence of CTLA-4 ligation may cause a differentiative or qualitative change, thereby altering T cell responses to subsequent stimulations. This might occur in both CD4+ and CD8+ T cells during primary stimulation but may be quantifiable in the CD8+ T cells only upon restimulation.
Phenotype of CTLA-4−/− Mice Is Altered When the TCR Repertoire Is Restricted.
Restriction of the TCR repertoire by the introduction of an MHC class II-restricted TCR transgene alters the onset of the lymphoproliferation (Fig. 2) observed in CTLA-4−/− mice. There are several explanations for these results. First, restriction of the TCR repertoire might decrease the frequency of overtly autoreactive T cells in the periphery of the CTLA-4-deficient animals by altering parameters of selection. However, thymocyte development appears normal before massive peripheral T cell lymphoproliferation in CTLA-4−/− mice (26), and no defects in thymocyte selection have been observed to date in MHC class I- or II-restricted TCR transgenic CTLA-4−/− mice (ref. 22; C.A.C., unpublished result). Second, because the TCR repertoire in the TCR transgenic mice presumably is capable of interacting with only a restricted set of self-MHC/self-peptide and/or environmental antigen ligands, it may simply statistically take longer for the T cells expressing a particular TCR to become activated and accumulate in the CTLA-4−/− mice. Finally, the AND TCR+ T cells may become activated via a second TCR containing endogenous α- or β-chains that arise because of the infidelity of allelic exclusion of the TCRα genes and, to a lesser extent, TCRβ genes (39–41) and subsequently down-regulate the TCR transgenes.
We have suggested previously that CTLA-4 may prevent the activation of CD4+ T cells during the continuous interaction with self-MHC known to be necessary for T cell survival (15–21). The observation that the young AND TCR+ CTLA-4−/− mice carrying a null mutation in the rag-1 gene fail to develop a lymphoproliferative disorder and the T cells maintain a naive phenotype seemingly appears to be inconsistent with this model. However, this result has two important caveats. The AND TCR+ RAG−/− CTLA-4−/− mice analyzed in these experiments were <10 weeks old. Preliminary data demonstrating that the majority of the T cells in aged (11 months) AND TCR+ RAG−/− CTLA-4−/− mice but not the CTLA-4+ littermate display an activated phenotype (CD69+, CD44hi, CD62Llo; C.A.C., unpublished result) support our model. This result also indicates that T cells expressing two TCRs, shown to be critical in some models of autoimmunity (42), are not required for the activation of CTLA-4-deficient T cells. Alternatively, the activation of the CTLA-4−/− T cells expressing a monoclonal repertoire may be delayed because of effects of the RAG−/− background not associated with T cell monoclonality. These mice also lack B cells and have an altered lymph node and splenic architecture. Normal lymphoid organ architecture may be important to facilitate, for example, the interactions between the T cells and the B and/or dendritic cells, leading to the activation of the CTLA-4−/− T cells. This also may account for the relatively moderate accumulation of the T cells in the aged AND TCR+ RAG−/− CTLA-4−/− mice, despite >70% of the T cells having an activated phenotype (unpublished data). Introduction of a null mutation in the TCRα locus did not prevent lymphoproliferation in AND TCR+ CTLA-4−/− mice (unpublished result), suggesting that the lymphoid microenvironment may be important. Adoptive transfer of AND TCR+ RAG−/− CTLA-4−/− and littermate control T cells into mice with normal lymphoid architecture and introduction of mutations in both the TCRα and TCRβ loci into the AND TCR+ CTLA-4−/− mice will distinguish between these possibilities and elucidate the basis for the lymphoproliferative disorder.
Implications for T Cell Homeostasis.
The size and TCR diversity of peripheral pools of naive and memory T cells remain amazingly stable throughout the lifetime of an animal (reviewed in ref. 43). This occurs despite the continuous ligation of TCR necessary for T cell survival and the dramatic expansion and contraction of antigen-specific T cells during the generation and resolution of immune responses. It has been demonstrated recently that the magnitude of the T cell expansion in the primary immune response has a critical impact on the generation of the effector and memory T cell populations (32, 44). Thus, seemingly small increases in the magnitude of a primary T cell response, such as observed with the AND TCR+ CTLA-4−/− T cells in vitro, may have a profound impact on the magnitude of secondary or memory responses in vivo. The nature of primary T cell activation also can greatly influence the resultant responses (45–47); thus, alterations in T cell priming may have dramatic consequences for peptide-specific T cell responses in vivo.
These results demonstrate that CTLA-4 is a key regulator of peptide-specific CD4+ T cell responses and suggest that CTLA-4 may play an important role in regulating memory T cell responses. Elucidating the influence of positive vs. inhibitory costimulatory signals on different T cell subsets, such as CD4+ vs. CD8+ T cells and naive vs. memory and/or effector T cells will be important for understanding the role of CTLA-4 in establishing the threshold for T cell activation, tolerance, and maintaining T cell homeostasis. Further, elucidation of the relationship between the levels of TCR occupancy, positive costimulation, and the integration of the signaling pathways will facilitate the manipulation of these responses for immunotherapy.
Acknowledgments
We thank J. Grein for technical assistance and Dr. Joonsoo Kang for critically reading the manuscript. This work was supported by the Human Frontier Science Program Organization (C.A.C.) and National Institutes of Health Grant CA40041 (J.P.A.). J.P.A. is an investigator of the Howard Hughes Medical Institute.
ABBREVIATIONS
- AND TCR
MHC class II-restricted T cell receptor
- APC
antigen-presenting cell
- ELISPOT
enzyme-linked immunospot assay
- CTLA-4
cytotoxic T lymphocyte antigen-4
References
- 1.Lenschow D J, Walunas T L, Bluestone J A. Annu Rev Immunol. 1996;14:233–258. doi: 10.1146/annurev.immunol.14.1.233. [DOI] [PubMed] [Google Scholar]
- 2.Wülfing C, Davis M M. Science. 1998;282:2266–2269. doi: 10.1126/science.282.5397.2266. [DOI] [PubMed] [Google Scholar]
- 3.Viola A, Schroeder S, Sakakibara Y, Lanzavecchia A. Science. 1999;283:680–682. doi: 10.1126/science.283.5402.680. [DOI] [PubMed] [Google Scholar]
- 4.Chambers C A, Allison J P. Curr Opin Cell Biol. 1999;11:203–210. doi: 10.1016/s0955-0674(99)80027-1. [DOI] [PubMed] [Google Scholar]
- 5.Marengére L E M, Waterhouse P, Duncan G S, Mittrucker H-W, Feng G-S, Mak T W. Science. 1996;272:1170–1173. doi: 10.1126/science.272.5265.1170. [DOI] [PubMed] [Google Scholar]
- 6.Lin H, Rathmell J C, Gray G S, Thompson C B, Leiden J M, Alegre M-L. J Exp Med. 1998;188:199–204. doi: 10.1084/jem.188.1.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fallarino F, Fields P E, Gajewski T F. J Exp Med. 1998;188:205–210. doi: 10.1084/jem.188.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Saito K, Sakurai J, Ohata J, Kohsaka T, Hashimoto H, Okumura K, Abe R, Azuma M. J Immunol. 1998;160:4225–4231. [PubMed] [Google Scholar]
- 9.Lee K-M, Chuang E, Griffin M, Khattri R, Hong D K, Zhang W, Straus D, Samelson L E, Thompson C B, Bluestone J A. Science. 1998;282:2263–2266. doi: 10.1126/science.282.5397.2263. [DOI] [PubMed] [Google Scholar]
- 10.Blair P J, Riley J L, Levine B L, Lee K P, Craighead N, Francomano T, Perfetto S J, Gray G S, Carreno B M, June C H. J Immunol. 1998;160:12–15. [PubMed] [Google Scholar]
- 11.Brunner M, Chambers C A, Chan F, Hanke J, Winoto A, Allison J P. J Immunol. 1999;162:5813–5820. [PubMed] [Google Scholar]
- 12.Waterhouse P, Penninger J M, Timms E, Wakeham A, Shahinian A, Lee K P, Thompson C B, Griesser H, Mak T W. Science. 1995;270:985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 13.Tivol E A, Borriello F, Schweitzer A N, Lynch W P, Bluestone J A, Sharpe A H. Immunity. 1995;3:541–546. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 14.Chambers C A, Krummel M F, Boitel B, Hurwitz A A, Sullivan T J, Fournier S, Cassell D, Brunner M, Allison J P. Immunol Rev. 1996;153:27–46. doi: 10.1111/j.1600-065x.1996.tb00919.x. [DOI] [PubMed] [Google Scholar]
- 15.Chambers C A, Sullivan T J, Allison J P. Immunity. 1997;7:885–895. doi: 10.1016/s1074-7613(00)80406-9. [DOI] [PubMed] [Google Scholar]
- 16.Chambers C A, Allison J P. Curr Opin Immunol. 1997;9:396–404. doi: 10.1016/s0952-7915(97)80087-8. [DOI] [PubMed] [Google Scholar]
- 17.Takeda S, Rodewald H-R, Arakawa H, Bluethmann H, Shimizu T. Immunity. 1996;5:217–228. doi: 10.1016/s1074-7613(00)80317-9. [DOI] [PubMed] [Google Scholar]
- 18.Tanchot C, Lemonnier F A, Pérarnau B, Freitas A A, Rocha B. Science. 1997;276:2057–2062. doi: 10.1126/science.276.5321.2057. [DOI] [PubMed] [Google Scholar]
- 19.Brocker T. J Exp Med. 1997;186:1223–1232. doi: 10.1084/jem.186.8.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kirberg J, Berns A, von Boehmer H. J Exp Med. 1997;186:1269–1275. doi: 10.1084/jem.186.8.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Markiewicz M A, Girao C, Opferman J T, Sun J, Hu Q, Agulnick A A, Bishop C E, Thompson C B, Ashton-Rickardt P G. Proc Natl Acad Sci USA. 1998;95:3065–3070. doi: 10.1073/pnas.95.6.3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Waterhouse P, Bachmann M E, Penninger J M, Ohashi P S, Mak T W. Eur J Immunol. 1997;27:1887–1892. doi: 10.1002/eji.1830270811. [DOI] [PubMed] [Google Scholar]
- 23.Chambers C A, Sullivan T J, Truong T, Allison J P. Eur J Immunol. 1998;28:3137–3143. doi: 10.1002/(SICI)1521-4141(199810)28:10<3137::AID-IMMU3137>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 24.Bachmann M F, Waterhouse P, Speiser D E, McKall-Faienza, Mak T W, Ohashi P S. J Immunol. 1998;160:95–100. [PubMed] [Google Scholar]
- 25.Kaye J, Hsu M-L, Sauron M-E, Jameson S C, Gascoigne N R J, Hedrick S M. Nature (London) 1989;341:746–749. doi: 10.1038/341746a0. [DOI] [PubMed] [Google Scholar]
- 26.Chambers C A, Cado D, Truong T, Allison J P. Proc Natl Acad Sci USA. 1997;94:9296–9301. doi: 10.1073/pnas.94.17.9296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mombaerts P, Iacmini J, Johnson R S, Herrup K, Tonegawa S, Papaioannou V E. Cell. 1992;68:869–877. doi: 10.1016/0092-8674(92)90030-g. [DOI] [PubMed] [Google Scholar]
- 28.Matsui K, Boniface J J, Steffner P, Reay P A, Davis M M. Proc Natl Acad Sci USA. 1994;91:12862–12866. doi: 10.1073/pnas.91.26.12862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reay P A, Kantor R M, Davis M M. J Immunol. 1994;152:3946–3957. [PubMed] [Google Scholar]
- 30.Shastri N, Hood L. Proc Natl Acad Sci USA. 1985;82:5885–5889. doi: 10.1073/pnas.82.17.5885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Krummel M F, Allison J P. J Exp Med. 1996;183:2533–2540. doi: 10.1084/jem.183.6.2533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Murali-Krishna K, Altman J D, Suresh M, Sourdive D J, Zajac A J, Miller J D, Slansky J, Ahmed R. Immunity. 1998;8:177–187. doi: 10.1016/s1074-7613(00)80470-7. [DOI] [PubMed] [Google Scholar]
- 33.Croft M, Duncan D D, Swain S S. J Exp Med. 1992;176:1431–1437. doi: 10.1084/jem.176.5.1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bird J J, Brown D R, Mullen A C, Moskowitz N H, Mahowald M A, Sider J R, Gajewski T F, Wang C-R, Reiner S L. Immunity. 1998;9:229–237. doi: 10.1016/s1074-7613(00)80605-6. [DOI] [PubMed] [Google Scholar]
- 35.Gett A V, Hodgkin P D. Proc Natl Acad Sci USA. 1998;95:9488–9493. doi: 10.1073/pnas.95.16.9488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Metz D P, Farber D L, Taylor T, Bottomly K. J Immunol. 1998;161:5855–5861. [PubMed] [Google Scholar]
- 37.Alegre M-L, Shiels H, Thompson C B, Gajewski T F. J Immunol. 1998;161:3347–3356. [PubMed] [Google Scholar]
- 38.Croft M. Curr Opin Immunol. 1994;6:431–437. doi: 10.1016/0952-7915(94)90123-6. [DOI] [PubMed] [Google Scholar]
- 39.Bluthmann H, Kisielow P, Uematsu Y, Malissen M, Krimpenfort P, Berns A, von Boehmer H, Steinmetz M. Nature (London) 1988;334:156–159. doi: 10.1038/334156a0. [DOI] [PubMed] [Google Scholar]
- 40.Berg L J, de St. Groth B F, Ivars F, Coodnow C, Gilfillan S, Garchon H-J, Davis M M. Mol Cell Biol. 1988;8:5459–5469. doi: 10.1128/mcb.8.12.5459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Balomenos D, Balderas R S, Mulvany K P, Kaye J, Kono D H, Theofilopoulos A N. J Immunol. 1995;155:3308–3312. [PubMed] [Google Scholar]
- 42.Sarukhan A, Garcia C, Lanoue A, von Boehmer H. Immunity. 1998;8:563–570. doi: 10.1016/s1074-7613(00)80561-0. [DOI] [PubMed] [Google Scholar]
- 43.Tanchot C, Rocha B. Immunol Today. 1998;19:575–579. doi: 10.1016/s0167-5699(98)01344-9. [DOI] [PubMed] [Google Scholar]
- 44.Opferman J T, Ober B T, Ashton-Richardt P G. Science. 1999;283:1745–1748. doi: 10.1126/science.283.5408.1745. [DOI] [PubMed] [Google Scholar]
- 45.Jenkins M K, Pardoll D M, Mizuguchi J, Quill H, Schwartz R H. Immunol Rev. 1987;95:113–1135. doi: 10.1111/j.1600-065x.1987.tb00502.x. [DOI] [PubMed] [Google Scholar]
- 46.Perez V L, Van Parijs L, Biuckians A, Zheng X X, Strom T B, Abbas A K. Immunity. 1997;6:411–417. doi: 10.1016/s1074-7613(00)80284-8. [DOI] [PubMed] [Google Scholar]
- 47.Pape K, Mercia R, Mondino A, Khoruts A, Jenkins M K. J Immunol. 1998;160:4719–4729. [PubMed] [Google Scholar]