Abstract
CD4+ T cells can eliminate tumor cells in vivo in the absence of CD8+ T cells. We have CD4+ T cells specific for a MHC class II-restricted, tumor-specific peptide derived from a mutant ribosomal protein expressed by the UV light-induced tumor 6132A-PRO. By using neutralizing mAb specific for murine IFN-γ and adoptive transfer of CD4+ T cells into severe combined immunodeficient mice, we show that anti-IFN-γ treatment abolishes the CD4+ T cell-mediated rejection of the tumor cells in vivo. The tumor cells were MHC class II negative, and IFN-γ did not induce MHC class II expression in vitro. Therefore, the tumor-specific antigenic peptide must be presented by host cells and not the tumor cells. Tumor cells transduced to secrete IFN-γ had a markedly reduced growth rate in severe combined immunodeficient mice, but IFN-γ did not inhibit the growth of the tumor cells in vitro. Furthermore, tumor cells stably expressing a dominant-negative truncated form of the murine IFN-γ receptor α chain, and therefore insensitive to IFN-γ, nevertheless were rejected by the adoptively transferred CD4+ T cells. Thus, host cells, and not tumor cells, seem to be the target of IFN-γ. Together, these results show that CD4+ T cells can eliminate IFN-γ-insensitive, MHC class II-negative cancer cells by an indirect mechanism that depends on IFN-γ.
A single malignant cell can have multiple tumor-specific antigens (1–4); some are recognized by CD4+ T cells whereas others are recognized by CD8+ T cells (for review see ref. 5). CD4+ T cells can be critical for CD8+ T cells to (i) establish long-term, radio-resistant, specific memory immunity against tumors (6), (ii) sustain cytotoxic T cell responses during chronic viral infection (7), and (iii) maintain proliferation and viability of adoptively transferred CD8+ T cells in patients (8). In addition to this regulatory “helper” role, adoptively transferred CD4+ T cells can eliminate cancer cells in vivo in the absence of CD8+ T cells (9–12).
CD4+ T cells recognize a tumor-specific antigenic peptide on MHC class II molecules either when the antigen is acquired by a host antigen-presenting cell (APC) via the endocytic pathway, or when the peptide is endogenously produced in a tumor cell expressing MHC class II (11). CD4+ T cells can directly lyse such tumor cells. Because most human and animal tumors do not express MHC class II (13), this mechanism of direct destruction of tumor cells by CD4+ T cells would not apply generally. Interestingly, previous studies have shown that in the absence of CD8+ T cells, adoptive transfer of CD4+ T cells can lead to the elimination in vivo of tumor cells that do not express detectable levels of MHC class II antigens (9, 11, 14). Presumably, under these conditions tumor antigens are released from dead tumor cells and presented by “professional” host APC, which express high levels of MHC class II (11, 15–17). Recently, several MHC class II-restricted tumor-specific antigens have been identified as highly immunogenic peptides derived from mutant regions of proteins altered by tumor-specific somatic mutations (11, 18).
IFN-γ released by CD4+ T cells on antigenic stimulation has been proposed to be important in vivo for the anti-tumor effects of CD4+ T cells (10). IFN-γ induces the expression of several proteins in normal and neoplastic cells and thereby can alter the endogenous antigen processing machinery, up-regulate expression of MHC class I and II, activate macrophages, and inhibit cell proliferation (for review see ref. 19). By expressing a dominant-negative form of the IFN-γ receptor α chain in cancer cells (20), it has been shown that loss of sensitivity of tumor cells to IFN-γ can decrease immunogenicity and prevent tumor cell recognition and elimination (21). Furthermore, loss of sensitivity of the tumor cells to IFN-γ also prevented the anti-tumor effects of IL-12 (22).
Because of the frequent absence of MHC class II molecules on tumors and the effectiveness of CD4+ T cells in eliminating MHC class II-negative tumor cells in vivo, we have used three tools to investigate the possible involvement of IFN-γ in the CD4+ T cell-mediated rejection of MHC class II-negative tumors: (i) murine IFN-γ-neutralizing mAbs (23), (ii) a dominant negative form of the IFN-γ receptor α chain (20), and (iii) an efficient vector allowing stable, constitutive expression of IFN-γ in cells (24). We show here that IFN-γ is essential for the elimination of tumor cells by CD4+ T cells in vivo, but that this effect appears to be indirect because tumor cells remain MHC class II negative after stimulation with IFN-γ and still are rejected when made insensitive to IFN-γ.
MATERIALS AND METHODS
Mice and Cell Lines.
Female C3H/HeN MMTV− immunocompetent, severe combined immunodeficient (SCID), and nude mice were purchased from the National Cancer Institute’s Frederick Cancer Research Facility (Frederick, MD). The tumor cell lines used were the UV-induced fibrosarcoma 6132A-PRO (4, 25), also referred to as PRO, and the UV-induced fibrosarcoma 1591-PRO4L (26). The tumor cells were cultured in DMEM with 10% FCS and tested regularly for mycoplasma contamination by staining with Hoechst 33258 followed by fluorescence microscopy. To measure cell proliferation, 20 μl of MTS/PMS solution [(3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium]/(phenazine methosulfate)] (Promega) was added to cells growing in 96-well plates for 3 hr and analyzed at 490 nm versus 650 nm in an ELISA reader.
Generation of Lymph Node Cells (LNC).
The following two peptides were purchased from Chiron: NH2-LRRDFNHINVELSHLGKKKKRLRVD-CONH2 (mutant L9, mL9) and NH2-LRRDFNHINVELSLLGKKKKRLRVD-CONH2 (wild-type L9, wtL9). For immunization, 2.5 ng of either wild type or mutant L9 peptide in DMEM was mixed with complete Freund’s adjuvant and injected into footpads of immunocompetent C3H mice. Popliteal LNC were restimulated in vitro with either 10 μM peptide or 6132A-PRO cell lysate in T cell-DMEM as described (11). LNC proliferated in response to 6132A-PRO lysate or mL9 peptide but not to lysate of 1591-PRO4L cells or wtL9 peptide. LNC from mice immunized with 10 μM wtL9 peptide did not proliferate in response to either tumor cell lysate (data not shown). For measurement of IFN-γ secretion, 105 CD8-depleted LNC were incubated for 48 hr in 96-well plates coated with antibody 2C11 (anti-CD3 mAb, kindly provided by Jeffrey A. Bluestone, University of Chicago), and supernatants were analyzed in an ELISA as described (11).
Antibodies and Fluorometric Analyses.
For depletion of CD8+ cells in vitro, 107 cells/ml were incubated in DMEM with complement in the presence of supernatant from hybridoma 3.155 (anti-CD8) for 1 hr at 37°C. Antibody treatment in vivo was performed by injection i.p. of 0.3–0.7 ml of ascites fluid from nude mice bearing the hybridomas GK1.5 (anti-CD4), YTS169.4.2 (anti-CD8), XMG1.2 (anti-IFN-γ) (23), or 11B11(anti-IL-4) every 3–7 days. In vivo depletion treatments were started 3 days before tumor inoculation and continued for 3 weeks. Depletion was confirmed by flow cytometry (FACScan, Becton Dickinson Labware) before injection into animals (depletion in vitro) or at the end of the experiment (depletion in vivo) by using anti-CD4 or anti-CD8 antibodies coupled to FITC or phycoerythrin (PharMingen). MHC class I and class II expression were detected by flow cytometry using supernatant from the hybridomas 100.30R23 (anti-I-Kk/Dk) followed by goat anti-mouse antibody coupled to FITC, and the mAb 14–4-4S (anti-I-Ek) coupled to FITC, respectively. Magnetic-activated cells sorting (MACS) of CD4+ T cells was done according to the manufacturer’s instructions (Miltenyi Biotec, Auburn, CA) by using positive selection columns. After sorting, cells were incubated at 37°C overnight before being injected into animals or analyzed by flow cytometry.
Transfections and Retroviral Infections.
The plasmid encoding the transdominant negative IFN-γ receptor α chain (20) was transfected into 6132A-PRO cells by electroporation (Genepulser, Bio-Rad). G418-resistant clones were selected by fluorescence-activated cell sorting for increased IFN-γ receptor by using the antibody GR-20 (27). Positive clones then were analyzed for resistance to IFN-γ by examining MHC class I up-regulation induced by recombinant IFN-γ (250 units/ml) in vitro in a 48-hr assay. For retroviral infections, a 24-hr supernatant of MFG-IFN-γ retrovirus-producing cells (kindly provided by Glenn Dranoff, Dana–Farber Cancer Institute, Boston) was applied to 6132A-PRO cells for 4 hr in the presence of polybrene as described (24). Clones then were analyzed for secretion of IFN-γ by ELISA and for MHC class I surface expression by flow cytometry using the 100.30.R23 antibody.
Tumor Growth in Vivo.
6132A-PRO tumor cells (105) or 1591-PRO4L were injected s.c. into the flanks of C3H SCID mice. LNC (3–12 × 106) were injected i.p. the same day, and tumor growth was measured every 3–4 days by using a caliper. Tumor volume in cm3 was calculated as (abc/2) where a, b, and c are three orthogonal diameters, which approximates the volume of an ellipsoid: πabc/6.
Statistical Analyses.
One of the limitations of using highly purified T cell populations for adoptive transfer experiments in vivo is the relatively large number of cells required, resulting in repeated experiments with small group sizes. Therefore, Fisher’s Exact text was used to test the association in contingency with small sample sizes (28, 29). Statistics were performed by using Stata Statistical Software, 1999, Release 6.0 (Stata, College Station, TX) on an Apple Power Macintosh 7600/132.
RESULTS
CD8+ T Cells Are Not Required for Rejection of 6132A-PRO Tumor Cells.
In previous adoptive transfer experiments, we found that LNC induced by mL9 peptide caused rejection of 6132A-PRO tumor cells; however, mL9-specific CD4+ T cell clones inhibited the growth of tumor cells without eliminating them completely (11). To determine whether the stronger anti-tumor effects of LNC depended on the presence of CD8+ T cells in the LNC, we depleted the LNC of CD8+ T cells either before or after adoptive transfer into SCID mice. As summarized in Table 1, the nine SCID mice that were untreated all had progressively growing tumors. Ten of 11 mice that received either LNC depleted of CD8+ T cells or treatment with anti-CD8 antibodies in vivo after LNC transfer rejected the 6132A-PRO tumor. These results demonstrate that rejection of 6132A-PRO in SCID mice by adoptively transferred mL9-specific LNC does not depend on CD8+ T cells. No regrowth of tumors from the site of initial tumor challenge was seen during a 1-yr observation period in the mice that had rejected the tumors.
Table 1.
Tumor rejection by LNC from mL9-immunized mice does not require CD8+ T cells
SCID mice were innoculated s.c. with 105 6132A-PRO tumor cells and i.p. with 5-12 × 106 LNC from mL9-immunized mice as indicated.
LNC were depleted of T cell subsets by incubation with anti-CD8 antibodies and complement in vitro before adoptive transfer or by treatment of mice with anti-CD8 or anti-CD4 antibodies after adoptive transfer.
The differences in tumor incidence between the anti-CD8 antibody-depleted LNC-treated and untreated mice (P < 0.001) and between the untreated and undepleted LNC-treated group (P = 0.018) are statistically significant by Fisher’s Exact test.
CD4+ T Cells Are Required for Rejection of 6132A-PRO Tumor Cells.
In the next series of experiments, we determined whether or not adoptive transfer of CD4+ T cells present in LNC from mL9-immunized mice was sufficient for 6132A-PRO tumor rejection. We used MACS columns to obtain CD4+ and CD4− LNC fractions. With the exception of the first experiment, the purity of the CD4+ fraction was always above 95% as measured by flow cytometry (Fig. 1 and Table 2). In three different experiments, five of six SCID mice receiving CD4+ LNC rejected the tumor challenge, and the sixth mouse had a minimal tumor, the growth of which was arrested for 144 days (Table 2). In contrast, all six mice that received the CD4− fractions or no LNC had progressively growing tumors. Interestingly, even in the first experiment where the CD4− fraction still contained 45% CD4+ cells, tumors grew out slowly, indicating that even in the presence of a significant number of CD4+ cells, no other cell types present from the transferred LNC exerted significant anti-tumor effects or assisted in CD4+ T cell-mediated rejection of 6132A-PRO. Because CD4+ TCR-CD3− cells comprise less than 1% of LNC from mL9-immunized mice, these results strongly suggest that adoptive transfer of CD4+ T cells is necessary and sufficient for 6132A-PRO rejection in SCID mice.
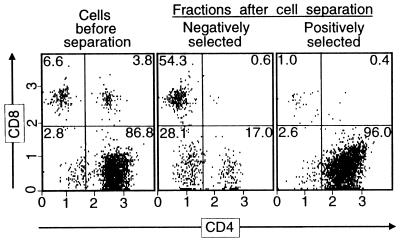
Figure 1.
Results of MACS of LNC from mL9-immunized mice. LNC from mL9 peptide-immunized mice were restimulated in vitro with 6132A-PRO tumor cell lysate and then incubated with microbead-coupled anti-CD4 antibodies for separation into CD4-positive and -negative fractions by using MACS positive selection columns. After overnight incubation, cell aliquots were stained with anti-CD4 and anti-CD8 antibodies and analyzed by flow cytometry. The numbers in the quadrants indicate the percentage of total cells that the different cell population in each quadrant comprises. Shown is the analysis of experiment 3 shown in Table 2.
Table 2.
CD4+ T cells from mL9-immunized mice mediate rejection of 6132A-PRO tumor cells in SCID mice
| Exp.† | Tumor incidence* after 28 days
|
||
|---|---|---|---|
| No LNC transferred | CD4-negative LNC transferred | CD4-positive LNC‡ transferred | |
| 1 | 1/1 | 1/1 | 0/2 |
| 2 | 1/1 | 1/1 | 1§/2 |
| 3 | 1/1 | 1/1 | 0/2 |
| Total | 3/3 | 3/3 | 1/6¶ |
Mice that showed no visible or palpable tumor at 2 months after tumor cell inoculation were considered tumor-free. All six mice that received no LNC or CD4-negative LNC developed progressive tumors and had to be killed at 28 days.
MACS-sorted LNC (3-4 × 106) from mL9-immunized mice were adoptively transferred in SCID mice inoculated with 105 6132A-PRO tumor cells.
Three independent experiments were performed, and the purity of the respective CD4+ fractions as analyzed by flow cytometry was 91% CD4+.
One mouse had a palpable nodule less than 3 mm in diameter that persisted for half a year and then slowly began to grow.
The Fisher Exact test for association of tumor incidence and type of cells transferred is statistically significant (P value = 0.015). Compared to the control mice or the CD4-negative LNC transferred group, both of which had to be killed at 28 days because of large tumor burdens (for typical growth curves consult Fig. 3), there was either complete rejection in the CD4-positive LNC receiving group (five mice) or complete arrest of growth for half a year (one mouse). For association of tumor rejection or arrest the P value was 0.002.
IFN-γ Increases MHC Class I Expression but Fails to Induce MHC Class II Expression in 6132A-PRO Tumor Cells.
IFN-γ is a potent up-regulator of MHC class I and II expression, and the mL9 antigen is presented by the MHC class II molecule I-Ek (11). Therefore, we determined expression levels for MHC class I and II on 6132A-PRO cells in the presence and absence of recombinant IFN-γ. As shown in Fig. 2, treatment of 6132A-PRO with IFN-γ (250 units/ml) for 48 hr strongly up-regulated MHC class I expression. However, there was no detectable expression of I-Ek even after IFN-γ treatment. As a positive control, we used 6132A-PRO cells transfected to express I-Ek under the control of the constitutive simian virus 40 promoter (11).
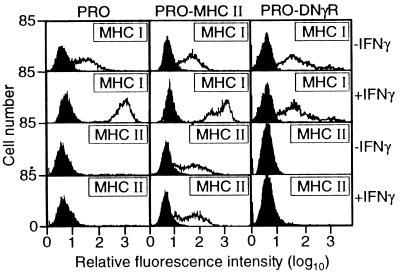
Figure 2.
6132A-PRO tumor cells expressing a dominant negative IFN-γ receptor (PRO-DNγR) are unresponsive to IFN-γ. 6132A-PRO cells were transfected with an expression vector for the transdominant-negative mutant IFN-γR α chain (20). PRO-DNγR, parental 6132A-PRO cells, and 6132A-PRO cells transfected to express I-Ek (PRO-MHC II, ref. 11) were cultured for 48 hr in the presence or absence of IFN-γ (250 units/ml) and then analyzed for expression of MHC class I and II by flow cytometry (open histograms). Controls (filled histograms) were incubated with second-step anti-Ig alone (control for MHC class I) or without antibody (control for MHC class II).
Rejection of 6132A-PRO Tumor Cells by CD4+ T Cells Requires IFN-γ.
Most mL9-specific CD4+ T cell clones secrete IFN-γ (11), and the same is true of LNC from mice immunized with mL9 peptide. To address the question of whether IFN-γ plays a role in the rejection of 6132A-PRO tumor cells, SCID mice challenged with 6132A-PRO tumor cells and reconstituted with CD8+ T cell-depleted LNC from mL9-immunized mice then were treated with antibodies against IFN-γ or IL-4. As shown in Table 3, anti-IFN-γ antibodies abolished the anti-tumor effect of LNC whereas anti-IL-4 antibodies had no effect on tumor rejection. Thus, IFN-γ is required for CD4+ LNC-mediated rejection of 6132A-PRO tumors in SCID mice.
Table 3.
IFN-γ is required for tumor rejection by LNC from mL9-immunized mice
| Group | LNC* | Antibody treatment† | Tumor incidence after 28 days‡ |
|---|---|---|---|
| 1 | − | — | 6/6 |
| 2 | + | — | 0/5 |
| 3 | + | Anti-IFNγ | 5/6 |
| 4 | + | Anti-IL-4 | 0/6 |
SCID mice were inoculated s.c. with 6132A-PRO tumor cells and received LNC from mL9-immunized mice.
Some animals were treated with anti-IFN-γ or anti-IL-4 antibodies as indicated in Materials and Methods, and tumor incidence was determined after 28 days.
The differences in tumor incidence between groups 1 and 2 (P = 0.002), between groups 2 and 3 (P = 0.015), between groups 3 and 4 (P = 0.015), and between groups 1 and 6 (P = 0.002) were significant by Fisher’s Exact test whereas treatment of mice that had received LNC with the control antibody (anti-IL-4), group 4, had no significant effect when compared with the tumor incidence in mice that received LNC but no further treatment.
6132A-PRO Tumor Cells Made Resistant to IFN-γ Are Still Rejected by CD4+ T Cells.
Sensitivity of tumor cells to IFN-γ can be important for tumor cell recognition and elimination (21, 22). However, in our adoptive transfer model, it was unclear whether IFN-γ acted directly or indirectly on 6132A-PRO tumor cells. Therefore, we generated an IFN-γ-insensitive tumor cell line, PRO-DNγR, by stably transfecting 6132A-PRO tumor cells with an expression vector for a mutant IFN-γ receptor α chain, IC-12 (20). This mutant receptor, which acts in a transdominant negative fashion, lacks all but 4 aa of its cytosolic domain and is unable to transmit intracellular signals on binding of IFN-γ (20). Fig. 2 shows that IFN-γ is unable to up-regulate MHC class I expression on PRO-DNγR cells, confirming their insensitivity to IFN-γ. PRO-DNγR also does not express any detectable MHC class II in the absence or presence of IFN-γ. Furthermore, there was no significant difference in proliferation in vitro between PRO-DNγR cells and the parental 6132A-PRO cells (Fig. 3, Left). Finally, the presence of various amounts of IFN-γ in the medium had little effect on the growth of the tumor cells in culture (Fig. 3, Middle), thereby demonstrating that IFN-γ did not affect the growth of tumor cells in vitro, even when they were IFN-γ sensitive based on up-regulation of MHC class I.
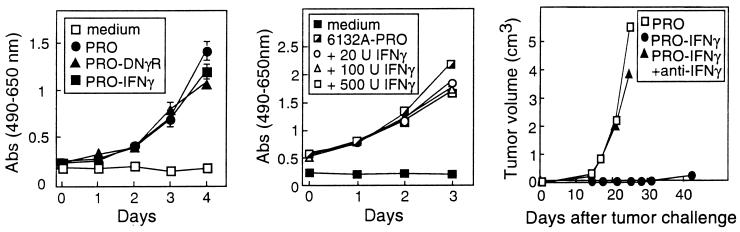
Figure 3.
(Left) IFN-γ-secreting and IFN-γ-insensitive 6132A-PRO tumor cells grow at similar rates in vitro as do unmodified 6132A-PRO parental cells. The in vitro growth of PRO-DNγR cells, PRO-IFN-γ cells, and 6132A-PRO cells (PRO) was analyzed in a [(3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium] (MTS) assay by measuring absorbance at 490 nm minus background absorbance at 650 nm. All experiments were done in triplicate, and SEM >0.08 are indicated. (Middle) IFN-γ is neither cytotoxic nor cytostatic for 6132A-PRO cells in vitro. The proliferation of 6132A-PRO tumor cells in vitro in the presence of varying amounts of IFN-γ was measured in an MTS assay by reading the absorbance at 490 nm minus the background absorbance at 650 nm. All experiments were performed in triplicate and SEM >0.08 are indicated. (Right) Growth of IFN-γ-secreting 6132A-PRO tumor cells in vivo. SCID mice were inoculated s.c with 105 6132A-PRO (PRO) or 3 × 105 IFN-γ-secreting PRO-IFN-γ, and tumor growth was measured every 3–4 days. Two of the five animals challenged with PRO-IFN-γ were treated with anti-IFN-γ antibodies. The SEM were always <0.03 and therefore are not shown. The tumor growth differs across the three groups (P value = 0.036) by Fisher’s Exact test considering rapid growth in the three PRO-challenged mice or the two mice challenged with PRO-IFN-γ cells but treated with anti-IFN-γ versus the three mice challenged with PRO-IFN-γ cells.
We then used the PRO-DNγR cells to determine whether direct effects of IFN-γ on tumor cells were required in vivo for CD4+ T cell-mediated tumor rejection. As shown in Table 4, the injected PRO-DNγR cells grew out as tumors in five of five mice, whereas all mice receiving LNC from mL9-immunized mice rejected the tumor challenge. The rejection of PRO-DNγR cells depended on IFN-γ, because anti-IFN-γ antibodies still abolished the anti-tumor effect of transferred LNC (Table 4). Reisolation of these tumors showed that PRO-DNγR cells were still insensitive to IFN-γ as measured by MHC class I up-regulation (data not shown). In summary, IFN-γ, while required for the rejection of 6132A-PRO tumors cells by LNC from mL9-immunized mice, does not act directly on the tumor cells.
Table 4.
LNC from mL9-immunized mice reject tumor cells made insensitive to IFN-γ (PRO-DNγR), but still require IFN-γ for this effect
SCID mice were inoculated s.c. with PRO-DNγR tumor cells and received LNC from mL9-immunized mice.
An additional animal treated with LNC and also with anti-IL-4 antibody rejected the tumor cell challenge similar to the LNC-treated mice treated with anti-IL-4 in Table 3.
The tumor incidence of PRO-DNγR for LNC-treated versus untreated mice was stitistically significant (P value = 0.008) by Fisher’s Exact test. The difference between tumor incidence for LNC-treated and LNC-treated plus anti-IFN-γ antibody is also statistically significant (P value = .048) by Fisher’s Exact test.
In the Absence of CD4+ T Cells, IFN-γ Inhibits, but Does Not Eliminate, the Growth of 6132A-PRO Tumor Cells in Vivo.
Finally, we investigated whether IFN-γ is sufficient for the rejection of 6132A-PRO tumor cells in mice. We therefore infected 6132A-PRO with a retrovirus containing the cDNA for IFN-γ (24) and obtained several 6132A-PRO clones that secreted large amounts of IFN-γ as determined by an IFN-γ-specific ELISA. The virally transduced cells showed MHC class I up-regulation by flow cytometry (data not shown). Clone 7, which secreted the largest amount of IFN-γ (PRO-IFN-γ), was chosen for further analysis. There was no significant difference in proliferation in vitro between PRO-IFN-γ cells and parental 6132A-PRO (PRO) cells (Fig. 3, Left). However, when we injected the PRO-IFN-γ cells s.c. at various doses into SCID mice, the growth of these tumors was greatly inhibited in vivo even in the absence of transferred LNC (Fig. 3, Right). This growth inhibition of PRO-IFN-γ tumor cells in vivo also was observed with higher tumor challenge doses and was independent of the particular tumor cell clone; two other PRO-IFN-γ cell lines showed equivalent growth inhibition in vivo (data not shown). Once again, growth inhibition clearly depended on IFN-γ because PRO-IFN-γ tumors grew as fast as the parental 6132A-PRO tumors in animals treated with anti-IFN-γ antibodies (Fig. 3, Right). Despite growth retardation, all SCID mice injected with PRO-IFN-γ tumor cells did develop slowly growing tumors after 3–4 weeks (Fig. 3, Right), whereas mice injected with CD4+ T cells remained tumor-free over a yearlong observation period. Reisolation of tumor cells from the animals confirmed that they continued to express and secrete IFN-γ (data not shown). Therefore, IFN-γ is essential, but not sufficient, for the rejection of 6132A-PRO tumor cells in SCID mice.
DISCUSSION
This study shows that IFN-γ is essential in vivo for the elimination of MHC class II-negative tumor cells by adoptively transferred CD4+ T cells. The participation of CD8+ T cells was not necessary because transfer of positively selected CD4+ T cells or CD8-depleted LNC from mL9-immunized mice still led to tumor rejection in SCID mice. The evidence suggests that neither the CD4+ T cells nor IFN-γ exerted inhibitory effects directly on the tumor cells. Tumor cells expressing the dominant-negative IFN-γ receptor still were rejected although such cells were unresponsive to IFN-γ. If IFN-γ acted directly on 6132A-PRO tumor cells to increase their immunogenicity or antigenicity, as proposed for the immune response to the murine tumor MethA (21), we would not expect that local release of IFN-γ by the transduced cells would inhibit their growth so markedly in mice lacking mature T and B cells. Furthermore, it is unlikely that the CD4+ T cells directly recognized and lysed the tumor cells, as the latter were MHC class II negative and resistant to lysis by the CD4+ T cells even after MHC class I expression on the tumor cells had been up-regulated by IFN-γ treatment. Even if MHC class II molecules were expressed at levels below the detectability of flow cytometry, these levels may not be functionally significant: cloned, mutant peptide-specific T cells produced large amounts of IFN-γ in vitro on specific stimulation with antigen and antigen-presenting cell, yet they lysed only those 6132A-PRO tumor cells that had been transfected to express the peptide-expressing I-Ek molecule (11).
Although the anti-tumor effects of CD4+ T cells and IFN-γ were indirect, the effects were localized to the site of the antigen-positive tumor cells because mL9-negative tumor cells at a separate site in the same host grew progressively, whereas the mL9-positive tumor cells were rejected (11). A mechanism that explains this CD4+ T cell-mediated, indirect growth inhibition is that CD4+ T cells encounter mL9 in the context of MHC class II on host antigen-presenting cell in the local tumor environment; this presentation results in IFN-γ release by the CD4+ T cells, which in turn stimulates host cells to eliminate the tumor. IFN-γ is known to induce monocytes, macrophages, fibroblasts, and certain tumor cells to produce Mig and IP-10, two CXC chemokines (30, 31). Both substances exert powerful antiangiogenic activity by damaging tumor vasculature, resulting in growth inhibition and tumor necrosis (32–34). Antiangiogenesis through IFN-γ-induced IP-10 and Mig is also the proposed mechanism by which IL-12 inhibits tumor cell growth (22, 35). However, the IL-12-mediated anti-tumor effects require tumor cells to be responsive to IFN-γ (22), whereas the CD4+ T cell-mediated effects observed here do not require tumor cells to be sensitive to IFN-γ. Antiangiogenic substances can arrest the growth of established tumors (36), and stromal antiangiogenic effects of IFN-γ would be consistent with the fact that adoptive transfer of CD4+ T cells can arrest the growth of even late established tumors (P.A.M. and H.S., unpublished data). Furthermore, IFN-γ activates macrophages to release TNF and nitric oxide and thus become tumoricidal (36–38), and macrophages are thought to participate in the anti-tumor activity of antigen-specific CD4+ T cells (10, 39, 40). Thus, we consider it likely that monocytes and macrophages in the tumor stroma are the key effector cells through which IFN-γ exerts its anti-tumor effect. Only nonspecific procedures such as treatment of mice with silica are available to inactivate macrophages in vivo, but such treatment does indeed reduce the tumoricial activity of adoptively transferred T cells (D.M. and H.S., unpublished data). Although our results suggest that IFN-γ is required for complete elimination of the tumor cells by CD4+ T cells, they also suggest that IFN-γ alone may not be sufficient. Thus, other cytokines released by antigen-stimulated CD4+ T cells, such as tumor necrosis factor α, also may participate and synergize with IFN-γ to release IP-10 and Mig and/or activate other tumoricidal cells (41, 42).
There are four reasons for the general importance of our findings for future treatment of human cancers. (i) The CD4+ T cell-recognized antigen mL9 seems to be prototypical of other CD4+ T cell-recognized, tumor-specific antigens in humans because other mutant proteins have been found to be the targets for CD4+ T cells in common human malignancies (18). Such mutant proteins represent altered self-antigens that can elicit high-affinity T cell clones because the latter have not been deleted during development. (ii) CD4+ T cell-recognized antigens are retained with tumor progression and thus represent stable targets for therapy (6). (iii) Normal host cells, which are genetically stable in comparison to tumor cells, process and present the tumor-specific antigen. Thus, this presentation pathway, which stimulates the CD4+ T cell-mediated, IFN-γ-dependent, indirect tumor inhibition, is unlikely to fail or to be subverted. (iv) Direct antigen presentation by tumor cells and tumor cell responsiveness to IFN-γ are not required for tumor cell elimination. Thus, this mechanism of elimination could be applicable to the majority of human malignancies that are MHC class II negative and may be or may become resistant to the direct effects of IFN-γ.
Acknowledgments
We thank Dr. Donald A. Rowley for critical review of the manuscript. We thank Dr. Glenn Dranoff for providing us with retrovirus encoding secreted IFN-γ and Dr. Patrick Fields for determining IFN-γ secretion levels by tumor cell lines. D.M. was supported by a postdoctoral fellowship from the Committee on Cancer Biology of the University of Chicago and from the Dr. Mildred Scheel Stiftung fur Krebsforschung. M.P. is supported by Graduate Training in Growth and Development Grant T32-HD07009. This work was supported by National Institutes of Health Grants RO1-CA-37516, RO1-CA-22677, PO1-CA74182, Training Grant GM-07281, and Core Grant CA-14599, and a gift of the Passis family.
ABBREVIATIONS
- LNC
lymph node cells
- mL9
mutant ribosomal protein L9
- SCID
severe combined immunodeficient
- MACS
magnetic-activated cell sorting
References
- 1.Wortzel R D, Philipps C, Schreiber H. Nature (London) 1983;304:165–167. doi: 10.1038/304165a0. [DOI] [PubMed] [Google Scholar]
- 2.Knuth A, Wölfel T, Klehmann E, Boon T, Meyer zum Büschenfelde K H. Proc Natl Acad Sci USA. 1989;86:2804–2808. doi: 10.1073/pnas.86.8.2804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Van den Eynde B, Hainaut P, Herin M, Knuth A, Lemoine C, Weynants P, van der Bruggen P, Fauchet R, Boon T. Int J Cancer. 1989;44:634–640. doi: 10.1002/ijc.2910440413. [DOI] [PubMed] [Google Scholar]
- 4.Ward P L, Koeppen H, Hurteau T, Schreiber H. J Exp Med. 1989;170:217–232. doi: 10.1084/jem.170.1.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schreiber H. In: Fundamental Immunology. 4th Ed. Paul W E, editor. Philadelphia: Lippincott; 1999. pp. 1247–1280. [Google Scholar]
- 6.Van Waes C, Urban J L, Rothstein J L, Ward P L, Schreiber H. J Exp Med. 1986;164:1547–1565. doi: 10.1084/jem.164.5.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Matloubian M, Concepcion R J, Ahmed R. J Virol. 1994;68:8056–8063. doi: 10.1128/jvi.68.12.8056-8063.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Walter E A, Greenberg P D, Gilbert M J, Finch R J, Watanabe K S, Thomas E D, Riddell S R. N Engl J Med. 1995;333:1038–1044. doi: 10.1056/NEJM199510193331603. [DOI] [PubMed] [Google Scholar]
- 9.Fujiwara H, Fukuzawa M, Yoshioka T, Nakajima H, Hamaoka T. J Immunol. 1984;133:1671–1676. [PubMed] [Google Scholar]
- 10.Greenberg P D, Kern D E, Cheever M A. J Exp Med. 1985;161:1122–1134. doi: 10.1084/jem.161.5.1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Monach P A, Meredith S C, Siegel C T, Schreiber H. Immunity. 1995;2:45–59. doi: 10.1016/1074-7613(95)90078-0. [DOI] [PubMed] [Google Scholar]
- 12.Frey A B. J Immunol. 1995;154:4613–4622. [PubMed] [Google Scholar]
- 13.Cabrera T, Ruiz-Cabello F, Garrido F. Scand J Immunol. 1995;41:398–406. doi: 10.1111/j.1365-3083.1995.tb03584.x. [DOI] [PubMed] [Google Scholar]
- 14.Greenberg P D, Cheever M A, Fefer A. J Exp Med. 1981;154:952–6314. doi: 10.1084/jem.154.3.952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Topalian S L, Rivoltini L, Mancini M, Ng J, Hartzman R J, Rosenberg S A. Int J Cancer. 1994;58:69–79. doi: 10.1002/ijc.2910580113. [DOI] [PubMed] [Google Scholar]
- 16.Schultz K R, Klarnet J P, Gieni R S, HayGlass K T, Greenberg P D. Science. 1990;249:921–923. doi: 10.1126/science.2118273. [DOI] [PubMed] [Google Scholar]
- 17.Kosugi A, Yoshioka T, Suda T, Sano H, Takahama Y, Fujiwara H, Hamaoka T. J Leukocyte Biol. 1987;42:632–641. doi: 10.1002/jlb.42.6.632. [DOI] [PubMed] [Google Scholar]
- 18.Pieper R, Christian R E, Gonzales M I, Nishimura M I, Gupta G, Settlage R E, Shabaovitz J, Rosenberg S A, Hunt D F, Topalian S L. J Exp Med. 1999;189:757–765. doi: 10.1084/jem.189.5.757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Billiau A. Adv Immunol. 1996;62:61–130. doi: 10.1016/s0065-2776(08)60428-9. [DOI] [PubMed] [Google Scholar]
- 20.Dighe A S, Farrar M A, Schreiber R D. J Biol Chem. 1993;268:10645–10653. [PubMed] [Google Scholar]
- 21.Dighe A S, Richards E, Old L J, Schreiber R D. Immunity. 1994;1:447–456. doi: 10.1016/1074-7613(94)90087-6. [DOI] [PubMed] [Google Scholar]
- 22.Coughlin C M, Salhany K E, Gee M S, LaTemple D C, Kotenko S, Ma X, Gri G, Wysocka M, Kim J E, Liu L, et al. Immunity. 1998;9:25–34. doi: 10.1016/s1074-7613(00)80585-3. [DOI] [PubMed] [Google Scholar]
- 23.Cherwinski H M, Schumacher J H, Brown K D, Mosmann T R. J Exp Med. 1987;166:1229–1244. doi: 10.1084/jem.166.5.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K, Jackson V, Hamada H, Pardoll D, Mulligan R C. Proc Natl Acad Sci USA. 1993;90:3539–3543. doi: 10.1073/pnas.90.8.3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ward P L, Koeppen H K, Hurteau T, Rowley D A, Schreiber H. Cancer Res. 1990;50:3851–3858. [PubMed] [Google Scholar]
- 26.Urban J L, Burton R C, Holland J M, Kripke M L, Schreiber H. J Exp Med. 1982;155:557–573. doi: 10.1084/jem.155.2.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.LeClaire R D, Basu M, Pinson D M, Redick M L, Hunt J S, Zavodny P J, Pace J L, Russell S W. J Leukocyte Biol. 1992;51:507–516. doi: 10.1002/jlb.51.5.507. [DOI] [PubMed] [Google Scholar]
- 28.Fisher R A. J R Stat Soc A. 1936;98:39–54. [Google Scholar]
- 29.Agresti A. Categorical Data Analysis. New York: Wiley; 1990. [Google Scholar]
- 30.Luster A D, Unkeless J C, Ravetch J V. Nature (London) 1985;315:672–676. doi: 10.1038/315672a0. [DOI] [PubMed] [Google Scholar]
- 31.Farber J M. Proc Natl Acad Sci USA. 1990;87:5238–5242. doi: 10.1073/pnas.87.14.5238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sgadari C, Angiolillo A L, Cherney B W, Pike S E, Farber J M, Koniaris L G, Vanguri P, Burd P R, Sheikh N, Gupta G, et al. Proc Natl Acad Sci USA. 1996;93:13791–13796. doi: 10.1073/pnas.93.24.13791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sgadari C, Farber J M, Angiolillo A L, Liao F, Teruya-Feldstein J, Burd P R, Yao L, Gupta G, Kanegane C, Tosato G. Blood. 1997;89:2635–2643. [PubMed] [Google Scholar]
- 34.Horton M R, McKee C M, Bao C, Liao F, Farber J M, Hodge-DuFour J, Pure E, Oliver B L, Wright T M, Noble P W. J Biol Chem. 1998;273:35088–35094. doi: 10.1074/jbc.273.52.35088. [DOI] [PubMed] [Google Scholar]
- 35.Kanegane C, Sgadari C, Kanegane H, Teruya-Feldstein J, Yao L, Gupta G, Farber J M, Liao F, Liu L, Tosato G. J Leukocyte Biol. 1998;64:384–392. doi: 10.1002/jlb.64.3.384. [DOI] [PubMed] [Google Scholar]
- 36.Folkman J. Proc Natl Acad Sci USA. 1998;95:9064–9066. doi: 10.1073/pnas.95.16.9064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Boehm U, Klamp T, Groot M, Howard J C. Annu Rev Immunol. 1997;15:749–795. doi: 10.1146/annurev.immunol.15.1.749. [DOI] [PubMed] [Google Scholar]
- 38.Xie Q W, Whisnant R, Nathan C. J Exp Med. 1993;177:1779–1784. doi: 10.1084/jem.177.6.1779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Frey A B. J Immunol. 1996;156:3841–3849. [PubMed] [Google Scholar]
- 40.Hung K, Hayashi R, Lafond-Walker A, Lowenstein C, Pardoll D, Levitsky H. J Exp Med. 1998;188:2357–2368. doi: 10.1084/jem.188.12.2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ohmori Y, Hamilton T. J Biol Chem. 1993;268:6677–6688. [PubMed] [Google Scholar]
- 42.Ohmori Y, Schreiber R, Hamilton T. J Biol Chem. 1997;272:14899–14907. doi: 10.1074/jbc.272.23.14899. [DOI] [PubMed] [Google Scholar]