Abstract
Activation of the Hedgehog (Hh) pathway has been identified in several cancers, including medulloblastoma, but the mechanisms by which this pathway affects tumor survival and growth are incompletely understood. We investigated whether Hedgehog might promote survival of medulloblastoma cells via up-regulation of BclII. We found that mRNA levels of the Hedgehog pathway effector Gli1 were significantly associated with BclII expression in medulloblastoma and that Gli1 and BclII are both present in regions of decreased apoptosis in nodular medulloblastoma. Transient overexpression of Gli1 and Gli2 in medulloblastoma cultures induced a BclII transcriptional reporter and increased BclII protein levels, whereas stable overexpression of Gli1 was associated with increased BclII mRNA. The Hedgehog antagonist cyclopamine blocked expression of the Hh pathway targets PTCH1 and Gli1, lowered BclII levels, and increased apoptosis in DAOY and UW228 medulloblastoma cells. Apoptotic induction caused by cyclopamine could be rescued in part by enforced expression of Gli1 or BclII. Hh pathway blockade also sensitized medulloblastoma to the effects of the proapoptotic agent lovastatin. These data demonstrate that BclII is an important mediator of Hh activity in medulloblastoma and suggest new strategies for combined chemotherapeutic regimens.
Hedgehog (Hh) pathway blockade has emerged as a promising therapy for multiple types of cancer, including brain tumors and carcinomas of the prostate, digestive tract, and lung.1–5 Gli transcription factors are critical effectors of the canonical Hh signaling pathway and regulate expression of multiple targets, including N-myc, cyclin D, PTCH1, and Gli1.6,7 The Hh pathway is frequently activated in the nodular medulloblastoma subtype by genetic changes inactivating the inhibitory PTCH1 receptor, although nonnodular medulloblastoma also can harbor PTCH1 mutations.8–10 Expression analyses have detected elevated levels of the Hh pathway targets PTCH1, Gli1, and N-myc in nodular medulloblastoma.11,12 However, Hh pathway targets are expressed in medulloblastoma of the classic subtype as well.11,13,14
Several groups have shown that Hh pathway blockade inhibits the growth of murine and human medulloblastoma cells in culture and in xenografts.1,15–17 It has also recently been demonstrated that cyclopamine or synthetic Hh antagonists can cause regression of primary medulloblastoma arising in PTCH heterozygous transgenic mice.16,17 Although most studies have concentrated on the effects of Hh inhibition on cell cycle regulation and cellular differentiation, increased apoptosis after Hh blockade has been noted as well.1,16,17 In this report, we focus on the mechanism by which Hh signaling regulates apoptosis, implicating BclII in the process. Increased expression of BclII has been associated with Hh signaling in keratinocytes and renal epithelial cells.18,19 More recently, two groups have shown that Gli1 and Gli2 directly bind to sequences in the BclII promoter in epithelial cells and induce transcription, indicating that this regulation is direct.20,21 Several studies have suggested that BclII is an important regulator of apoptosis in the cerebellar granule cell precursors from which medulloblastomas are thought to arise.22,23 Given these data and the direct linkage between Hh activity and BclII expression in nonneoplastic tissues, we investigated whether Hh might regulate cell survival in medulloblastoma via this anti-apoptotic protein.
Materials and Methods
Clinical Specimens
Immunohistochemistry was performed on freshly cut, deparaffinized tumor sections after antigen retrieval using Antigen Unmasking Solution (Vector Laboratories, Burlingame, CA). Primary antibody was detected using the Vectastain Elite kit (Vector Laboratories). The following antibodies were used: BclII, clone Ab-1 (1:500; Calbiochem, La Jolla, CA); Gli1, clone C-18 (1:100; Santa Cruz Biotechnology, Santa Cruz, CA); Gli2, clone N-20 (1:100, Santa Cruz Biotechnology); and cleaved caspase 3, clone Asp175 (1:100; Cell Signaling Technologies, Beverly, MA). Snap-frozen primary medulloblastoma tissues were obtained from the Department of Pathology, The Johns Hopkins University School of Medicine, with institutional review board approval. RNA was extracted from medulloblastoma samples using Trizol reagent (Invitrogen, Carlsbad, CA) and further purified using RNeasy columns (Qiagen, Valencia, CA) according to the manufacturer’s instructions.
Cell Culture
The DAOY cell line was obtained from American Type Culture Collection (Rockville, MD) and maintained in minimal essential medium supplemented with 10% fetal bovine serum (FBS). UW228 cells were the kind gift of Dr. J.R. Silber (University of Washington, Seattle, WA) and were maintained in Dulbecco’s modified Eagle’s medium/F12 supplemented with 10% FBS. To isolate granule cell precursors, cerebella of 7-day-old C57/B6 pups were microdissected and minced in ice-cold Hanks’ buffer and washed once with 12 ml of ice-cold phosphate-buffered saline (PBS). To digest connective tissue, cerebella pieces were incubated in 15 ml of Hanks containing 0.25% trypsin (Mediatech, Herndon, VA) and 1.5 mg of DNase I (Roche, Nutley, NJ) for 7 minutes at 37°C with gentle agitation. Fifteen ml of Hanks with 10% FBS was then added to the suspension to inactivate the trypsin. After two additional washes in ice-cold Hanks, a single-cell suspension was generated by trituration with a fire-polished Pasteur pipette in 5 ml of ice-cold Hanks containing 1.5 mg of DNase I. The cell suspension was filtered through a 70-μm nylon mesh (BD Falcon, Bedford, MA), and the cells concentrated by centrifugation at 700 rpm for 5 minutes in a table-top centrifuge. Granule cell precursors were resuspended in neurobasal medium supplemented with B27 growth supplement (Invitrogen) to a final concentration of 1 × 106/ml. Granule cell precursors (5 × 105) were treated in poly-l-lysine-coated six-well plates (Corning, Acton, MA). SHH was produced as previously described.24
Cells were transfected using Fugene6 reagent (Promega, Madison, WI) with plasmids encoding either BclII under control of the CMV promoter25 or Gli-expressing pcDNA4 plasmids.21 Stable subclones were selected by 28 days of treatment with 1 mg/ml G418. Lovastatin (Sigma, St. Louis, MO) was solubilized in ethanol. The BclII reporter assay was performed 48 hours after transfection using a dual-luciferase reporter assay system (Promega) and normalized to Renilla luciferase activity. Cells were plated in 24-well plates at 3.5 × 104 cells per well. On the next day, they were transfected with 475 ng of BclII-luciferase reporter [95% (w/w)] or an empty vector as negative control and with 25 ng of Renilla luciferase [5% (w/w)] as a transfection control. For mRNA quantification or apoptotic assays, cells were plated in six-well plates (RNA) or 24-well plates (apoptosis) and incubated overnight, and then washed once with PBS and switched to low serum (0.5% FBS) media. The following day (normally 16 hours after serum reduction), this was replaced with medium supplemented with 0.5% FBS and either ethanol (designated either vehicle or 0 μmol/L in the figures) or cyclopamine (5 or 10 μmol/L) in ethanol. RNA was extracted at various time points after cyclopamine treatment using Qiagen RNeasy kits. Quantification of mid-apoptotic and dead cells was performed 2 and 4 days after cyclopamine treatment with the Guava-PCA flow cytometry system and ViaCount reagent according to the manufacturer’s instructions (Guava Technologies, Hayward, CA). In brief, cells were trypsinized and spun at 1200 rpm for 5 minutes, and then 200 μl of ViaCount reagent was added to the cell pellets. Analysis was performed 10 to 20 minutes after the addition of the reagent. Maximal apoptotic induction was observed after 2 days in DAOY cells, and after 4 days in UW228 cells, and these time points are shown in the results.
For BclII short interfering RNA (siRNA) experiments, 4 × 104 DAOY cells were plated in triplicate and then incubated overnight, then transfected according to the manufacturer’s directions using RNAiFect (Qiagen) with 1 μg/well of a pool of scrambled, nonspecific siRNA (Dharmacon, Lafayette, CO) as negative control or BclII siRNA (Dharmacon) with the following oligo sequences: BclII forward 5′-GGGAGAUAGUGAUGAAGUAUU-3′ and BclII reverse 5′-P-UACUUCAUCACUAUCUCCCUU-3′. Plates were incubated for 8 hours, and then washed once with PBS and switched to low serum (0.5% FBS) media. The following day, this was replaced with medium supplemented with 0.5% FBS and either ethanol or cyclopamine. Apoptosis analysis was performed 48 hours after treatment.
RNA and Protein Analyses
RNA levels were analyzed by real-time polymerase chain reaction (PCR) analysis performed in triplicate with SYBR Green reagents (Applied Biosystems, Foster City, CA) according to the manufacturer’s instructions on an I-Cycler IQ real-time detection system (Bio-Rad, Hercules, CA). To minimize contaminating genomic DNA, a 15-minute on-column DNase step (Qiagen RNase-free DNase kit) was included during RNA extraction. The standard curve method was used to determine expression levels, and all values were normalized to actin. Oligo sequences were as follows: human Gli1 forward: 5′-ACCCGGGGTCTCAAACTG-3′; human Gli1 reverse: 5′-GGCTGACAGTATAGGCAGAGC-3′; human Gli2 forward: 5′-CCACATGGAGCACTACCT-3′; human Gli2 reverse: 5′-CCTGGCTGCAGAGATCATGG-3′; human Ptch1B forward: 5′-GACGCCGCCTTCGCTCTG-3′; human Ptch1B reverse: 5′-GCCCACAACCAAGAACTTGCC-3′; human BclXL forward: 5′-GTCGGATCGCAGCTTGGA-3′; human BclXL reverse: 5′-GCTGCATTGTTCCCATAGAGTTC-3′; human BclII forward: 5′-TACCTGAACCGGCACCTG-3′; human BclII reverse: 5′-GCCGTACAGTTCCACAAAGG-3′; mouse BclII forward: 5′-GGTGGTGGAGGAACTCTTCA-3′; mouse BclII reverse: 5′-CAGATGCCGGTTCAGGTACT-3′; human actin-β forward: 5′-CCCAGCACAATGAAGATCAA-3′; human actin-β reverse: 5′-GATCCACACGGAGTACTTG-3′. Hprt forward: 5′-CTTTGCTGACCTGCTGGATT-3′; and Hprt reverse: 5′-GTTGAGAGATCATCTCCACC-3′. Immunoblot analysis of BclII (clone Ab-1, 1:500) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (clone 6C5, 1:500,000; Research Diagnostics, Concord, MA) was performed on lysates (50 μg) prepared using standard techniques either 2 days (DAOY) or 4 days (UW228) after cyclopamine administration and quantified using Quantity-One densitometry software (Bio-Rad).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 4 (GraphPad Software, San Diego, CA). Data graphed with error bars represent mean and SE of mean from three replicates. All experiments were repeated at least two times, with similar results. Unless otherwise specified, a two-sided Student’s t-test was used to determine the significance of any differences.
Results
Gli and BclII Are Preferentially Expressed in the Internodular Regions of Desmoplastic Medulloblastoma
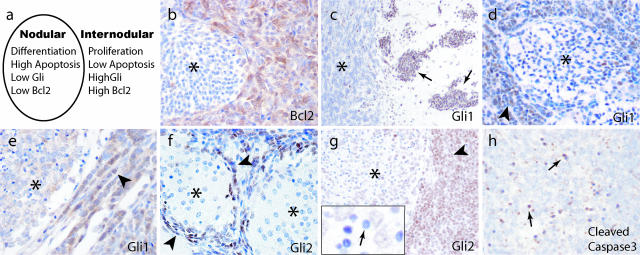
In nodular/desmoplastic medulloblastoma, the internodular regions are similar to the external germinal layer of the developing cerebellum in that they are comprised of primitive-appearing, highly proliferative cells. Nodules, in contrast, are characterized by cell cycle exit and increases in both apoptosis and neuronal differentiation, and resemble maturing granule cell precursors (Figure 1a).26,27 BclII levels are increased in the internodular regions (Figure 1b),28 and we investigated whether the Hh pathway was active here as well. We used expression of Gli1 and Gli2 as markers of Hh activity, and detected these pathway targets immunohistochemically in medulloblastoma cells, but not in adjacent nonneoplastic cerebellar tissue (Figure 1c and data not shown). In 5 of 10 nodular lesions, nuclear Gli1 staining was more intense in the internodular regions than within nodules (Figure 1, d and e). A similar expression pattern was observed for Gli2 in five of eight lesions, and in one very nodular tumor Gli2 expression was essentially restricted to internodular cells (Figure 1f). In other tumors, a fraction of cells within nodules were weakly positive for Gli2, although the small cells with condensed nuclei were always immunonegative for Gli1 and Gli2 (Figure 1g). We believe these small lymphocyte-like cells within nodules are undergoing programmed cell death because they are immunopositive for cleaved caspase 3 (Figure 1h) and have been previously shown to be terminal dUTP nick-end labeling-positive.29 The fact that both Gli and BclII proteins are expressed in internodular regions of decreased apoptosis suggests that Hh activity may regulate cell death in tumors via BclII.
Figure 1.
Expression of BclII and the Hh targets Gli1 and Gli2 is increased in the internodular regions of nodular/desmoplastic medulloblastoma. a: A cartoon modeling molecular and cellular factors in nodular medulloblastoma. Cells within medulloblastoma nodules have decreased levels of Gli and BclII, and either differentiate into neurocytes or undergo programmed cell death. b: BclII expression is low in nodules (asterisk) and high in internodular regions. c: Medulloblastoma cells invading the cerebellar molecular layer (arrows) are strongly immunopositive for Gli1, whereas nonneoplastic internal granule layer neurons (asterisk) are negative. d and e: Gli1 staining was frequently strong in internodular regions (arrowhead) but weak within nodules (asterisk). f and g: Expression of Gli2 was also higher between (arrows) compared with within (asterisks) nodules. Apoptotic cells with condensed nuclei lacked Gli2 protein (small arrow, inset). h: Cleaved caspase 3 was detected in lymphocyte-like apoptotic cells (small arrows). Original magnifications: ×200 (b, d, f, g); ×100 (c); ×400 (e, h).
Gli1 Expression Is Significantly Associated with BclII in Medulloblastoma
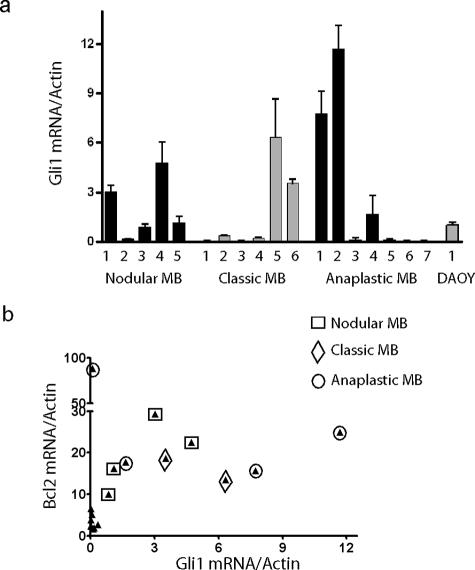
We used quantitative reverse transcriptase (RT)-PCR to more precisely measure expression of the Hh pathway target Gli1 in and a second, independent set of medulloblastoma samples. It has previously been suggested that Gli1 expression is elevated in desmoplastic medulloblastoma as compared with classic ones.12 Consistent with this, Gli1 expression was high in four of the five desmoplastic tumors we tested (Figure 2a). In our series, however, two of six classic medulloblastomas and three of seven anaplastic medulloblastomas also had relatively high Gli1 mRNA levels, and the differences between nodular/desmoplastic lesions and these groups were not statistically significant. Thus, activation of the Hh pathway does not seem to be restricted to the nodular/desmoplastic medulloblastoma subtype.
Figure 2.
Analysis of Gli and BclII mRNA expression. a: Gli1 expression was measured by quantitative RT-PCR in RNA extracted from five nodular, six classic, and seven anaplastic snap-frozen medulloblastomas, as well as the DAOY cell line. b: BclII mRNA levels in these 18 samples are compared with those of Gli1, with each tumor represented by a filled triangle. The subtype of tumors with above-median Gli1 or BclII expression is further designated using an open square, diamond, or circle.
To address the relationship between Hh activity and BclII expression, we compared Gli1 mRNA levels in these 18 primary medulloblastomas with BclII expression (Figure 2b). Interestingly, of the nine tumors with above-median Gli1 mRNA levels, eight also had above-median BclII levels, a statistically significant association (P = 0.003, two-tailed Fisher’s exact test). The one medulloblastoma with low Gli1 levels but high BclII expression was a diffusely anaplastic tumor that quickly metastasized. We have previously shown that anaplastic medulloblastomas have an increased number of chromosomal abnormalities,30 and other genetic alterations might account for the elevated BclII levels in this single case with low Hh activity. Indeed, although the correlation between Gli1 and BclII was not significant in the entire cohort, when the anaplastic tumor with extremely high BclII levels was excluded, they were significantly correlated in the remaining 17 cases (Pearson’s r = 0.69, P = 0.002). Overall, our data indicate that increased Hh activity is associated with higher BclII expression, potentially resulting in decreased apoptosis.
Gli1 and Gli2 Positively Regulate BclII Expression in Medulloblastoma
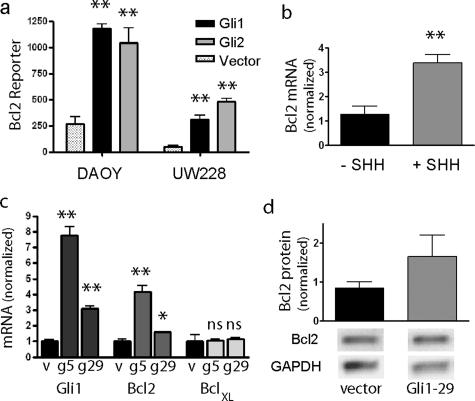
We next sought to determine whether Gli1 and Gli2 regulate BclII expression in medulloblastoma cultures. The DAOY cell line was initially derived from a nodular medulloblastoma and has recently been shown using luciferase-based reporter assays to have Gli transcriptional activity.31,32 We also easily detected Gli1 mRNA in UW228 cells, although at significantly lower levels than in DAOY. Using a reporter containing the BclII promoter region, we found that transient transfection of Gli1 or Gli2 into either cell line significantly induced reporter activity (Figure 3a). Gli1 and Gli2 had similar induction maxima, and we did not observe preferential induction of the reporter by Gli2 as was previously described in keratinocytes.33 We also found that induction of Hh signaling in cultured granule cell precursors by application of sonic hedgehog (SHH) ligand induced expression of BclII mRNA, suggesting that anti-apoptotic effects of Hh signaling in tumors may parallel mechanisms operating during normal cerebellar development (Figure 3b).
Figure 3.
Gli positively up-regulates BclII expression. a: Transient transfection of constructs encoding Gli1 or Gli2 into DAOY or UW228 cell lines significantly induced transcription of a BclII promoter-luciferase reporter as compared with empty vector. b: Stimulation of Hh signaling in cerebellar granule cell precursor cultures using SHH ligand results in a significant increase in BclII mRNA levels measured using quantitative RT-PCR. c: Both Gli1 and BclII mRNA levels are significantly increased in two DAOY subclones (g5, g29) stably expressing Gli1 compared with a vector-transfected clone (v). d: BclII protein expression, normalized to GAPDH, is elevated in DAOY cells stably transfected with Gli1. Mean levels from two experiments are shown above representative bands. *P < 0.05, **P < 0.005 for two-sided t-tests; ns, not significant.
We next generated subclones of DAOY stably transfected with Gli1 expressed under the control of a constitutive promoter. Two representative clones (Gli1-5, Gli1-29) had Gli1 mRNA levels increased approximately sevenfold and threefold greater than those in vector-transfected cells (Figure 3c). We were not able to isolate subclones with higher levels of Gli1 mRNA, suggesting that DAOY cells may not tolerate extremely elevated Gli1 expression, nor were we able to isolate clones expressing Gli2. In agreement with our transient transfection experiments, stable increases in Gli1 expression were associated with significantly higher BclII mRNA expression (Figure 3c). BclII luciferase reporter activity was increased approximately twofold in the Gli1-overexpressing cells (data not shown). The effect of Hh signaling on anti-apoptotic proteins seemed to be somewhat selective because increased Gli1 expression did not up-regulate the Bcl family member BclXL (Figure 3c). BclII protein levels were also increased in the DAOY clone with elevated Gli1 (Figure 3d).
Pharmacological Blockade of Hh Signaling Decreases BclII Levels and Induces Apoptosis in Medulloblastoma
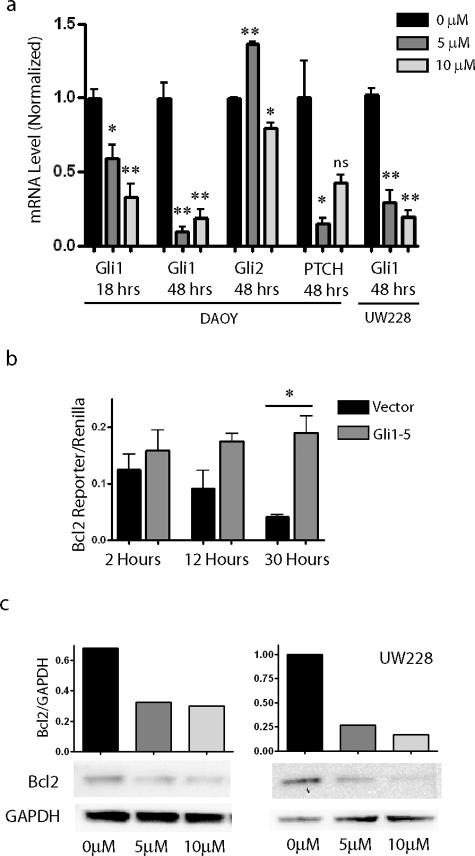
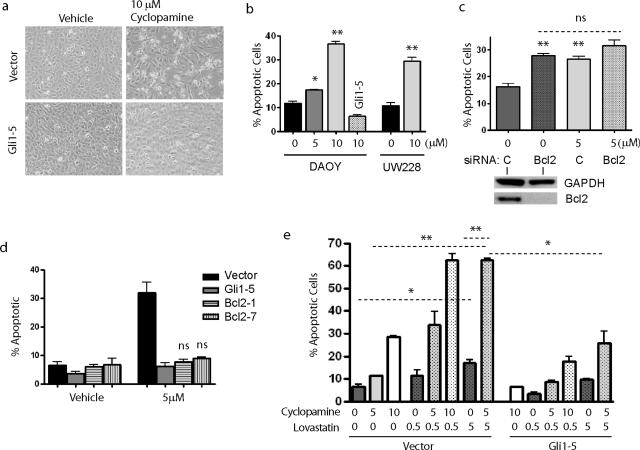
We next investigated whether Hh pathway blockade in medulloblastoma cells using the steroidal alkaloid cyclopamine would reduce BclII expression. A significant reduction in Gli1 mRNA level was seen after 18 hours of cyclopamine therapy, whereas 48 hours of treatment further reduced expression of Gli1 and a second pathway target, PTCH1 (Figure 4a). In separate experiments, Gli1 levels were also significantly lowered in UW228 cells by cyclopamine. Basal levels of Gli2 were not reduced by Hh pathway blockade in these cells. Indeed, a significant increase in Gli2 level was seen using the lower dose of drug. PTCH levels also increased significantly when DAOY cells were treated with 10 μmol/L cyclopamine compared with a 5 μmol/L. The cause(s) of these increases are not clear, but could relate to feedback loops specific to Gli2 and PTCH. Gli1 mRNA levels in the DAOY-Gli1 stable transfectants remained above those in untreated DAOY cells, even in the presence of 10 μmol/L cyclopamine (data not shown). BclII levels were also reduced in DAOY cultures after cyclopamine administration, consistent with our hypothesis that expression of this anti-apoptotic factor is dependent on a basal level of Gli1 activity. Normalized BclII reporter levels were lowered more than fourfold after 30 hours of treatment with 5 μmol/L cyclopamine, whereas stably transfected cells expressing Gli1 under the control of a constitutive promoter were refractory to such inhibition (Figure 4b). In UW228 cells, application of 5 μmol/L cyclopamine lowered BclII-reporter activity twofold throughout 3 days compared with vehicle-treated cells (data not shown). Western blot analysis confirmed that BclII protein levels in DAOY and UW228 cells after Hh pathway suppression were half or less of those in controls (Figure 4c).
Figure 4.
Analysis of Hh targets and BclII expression after cyclopamine treatment. a: Levels of Gli1 and PTCH1, but not Gli2, are significantly reduced by 5 μmol/L cyclopamine compared with vehicle-treated controls. b: BclII luciferase reporter assays after 5 μmol/L cyclopamine exposure reveal progressive inhibition of BclII expression throughout 30 hours. BclII expression was not inhibited in DAOY cells stably expressing Gli1 (Gli1-5). c: Hedgehog pathway blockade in DAOY and UW228 cells by cyclopamine for 48 hours also inhibited BCL2 protein expression normalized to GAPDH. *P < 0.05, ** P < 0.005 for two-sided t-tests; ns, not significant.
Because decreased Hh activity and BclII expression are observed in medulloblastoma nodules with increased cell death (Figure 1), we examined whether the reduced BclII expression in culture after cyclopamine treatment also occurred in concert with apoptotic induction. Floating or refractile cells were more numerous in cyclopamine-exposed cultures, suggesting the compound was causing cell death (Figure 5a). Indeed, a statistically significant increase in the percentage of apoptotic DAOY and UW228 cells was documented using ViaCount reagents after 48 hours of cyclopamine exposure (Figure 5b). Similar results were seen when apoptotic induction was measured using annexin assays after cyclopamine treatment (data not shown). Enforced Gli1 expression rescued DAOY cells from increased cell death. The Gli1-5 and Gli1-29 stable subclones both blocked apoptotic induction after addition of 5 μmol/L cyclopamine (data not shown), but only the Gli1-5 subclone expressing higher levels of Gli1 and BclII was able to rescue this effect at the 10 μmol/L dose (Figure 5b). These data suggest that the effect of cyclopamine on medulloblastoma apoptosis is mediated through the Hh pathway.
Figure 5.
Hh pathway blockade induces apoptosis in medulloblastoma. a: Vector-transfected DAOY cell cultures exposed to 10 μmol/L cyclopamine for 48 hours contained numerous refractile or floating cells. Cell death seemed more limited in cultures treated with vehicle, and in cyclopamine-treated DAOY cells stably expressing Gli1 (Gli1-5). b: Quantification of apoptotic cells after cyclopamine treatment confirmed that it significantly induces apoptosis. Enforced Gli1 expression blocked this increase in cell death in the stable DAOY Gli1-5 subclone. c: DAOY cells were transfected with siRNA targeting BclII or scrambled control (C) siRNA, and then treated with vehicle or 5 μmol/L cyclopamine. The addition of cyclopamine to cells already depleted of BclII did not cause significant additional apoptosis. Decreased levels of BclII protein are documented in a Western blot positioned below the first two bars. d: Two subclones stably transfected with a BclII-expressing plasmid were not sensitive to apoptotic induction by cyclopamine. e: Combined treatment with cyclopamine and lovastatin acts synergistically to induce apoptosis in medulloblastoma. *P < 0.05, **P < 0.005 for two-sided t-tests; ns, not significant.
To further define the role BclII plays in apoptosis induced by Hh pathway blockade, we transiently reduced BclII expression in DAOY cells using siRNA. Quantitative RT-PCR analysis confirmed a more than 80% decrease in BclII transcript levels in DAOY and UW228 cells transfected with targeted siRNA compared with control siRNA (data not shown). This reduction in BclII was associated with a significant increase in the percentage of apoptotic cells (Figure 5c). Cyclopamine also significantly increased apoptosis in cells transfected with control siRNA, although the level of induction was less than other experiments, perhaps because of the siRNA transfection protocol. If down-regulation of BclII via decreased Gli activity is a major proapoptotic mechanism of cyclopamine, one would predict that in the context of already-reduced BclII, Hh blockade would not cause a significant additional increase in cell death. We therefore exposed cells treated with BclII siRNA to 5 μmol/L cyclopamine and saw only a small, nonsignificant increase in apoptosis in both DAOY (Figure 5c) and UW228 (data not shown), suggesting that BclII was a major mediator of this effect. To further address the role of BclII in cyclopamine-mediated apoptosis, we introduced a construct encoding BclII under the control of a constitutive promoter into DAOY cultures then selected two stable subclones (BclII-1, BclII-7). Constitutive expression of BclII fully protected tumor cells against the cyclopamine-induced increase in programmed cell death (Figure 5d). These data strongly suggest that apoptotic induction after Hh blockade is dependent to a significant degree on BclII down-regulation.
Hh Blockade Sensitizes Medulloblastoma Cells to Combined Chemotherapeutic Regimens
Chemotherapeutic drugs are often given in combination, and the observation that BclII levels are reduced in cells treated with cyclopamine suggests that Hh inhibition may sensitize tumor cells to other proapoptotic stimuli. We tested this hypothesis by co-treatment of DAOY cultures with cyclopamine and the HMG-CoA reductase inhibitor lovastatin, which has been shown to induce apoptosis in medulloblastoma.34 Apoptosis was significantly induced from 7 to 17% of cells by 5 μmol/L lovastatin (Figure 5e). In addition, co-treatment with both 5 μmol/L cyclopamine and 5 μmol/L lovastatin induced apoptosis in 63% of cells compared with less than 20% programmed cell death in cultures treated with the same concentration of either agent alone (Figure 5e). This increase in sensitivity was partially rescued by constitutively expressing Gli1 because only 26% of Gli1-5 cultures were apoptotic when treated with 5 μmol/L levels of both drugs, a statistically significant decrease from 63%. Interestingly, enforced Gli1 expression significantly diminished the effect of lovastatin treatment alone (P = 0.02), suggesting Hh effectors act downstream of lovastatin in protecting against cell death. It is possible that lovastatin could influence the Hh pathway directly because sterol depletion has been shown to reduce Smoothened activity and diminish responsiveness to the Hh signal.35
Discussion
Medulloblastomas are the most common malignant brain tumors in children and are associated with significant morbidity and mortality.36 Current treatment regimens use radiation and chemotherapeutic agents that affect dividing cells in a relatively nonspecific manner and are associated with serious long-term side effects.37,38 This has prompted great interest in mechanism-based therapies that might target tumor cells more selectively. The Hh pathway plays a critical role in both normal cerebellar development,39 and in medulloblastoma formation.8,9,15,40 Several studies have demonstrated that Hh pathway inhibitors can slow or arrest the growth of both human and murine medulloblastomas.1,15–17 However, for these promising new agents to be used most effectively, we must first determine what groups of medulloblastoma patients might benefit from their use. A better understanding of the mechanism(s) by which Hh blockade regulates the survival of tumor cells will also be critical if we are to rationally develop such therapies. In this study, we examine Hh pathway activity in a range of medulloblastoma subtypes, and the impact of Hh activity on BclII expression and apoptosis.
It has previously been shown that Hh activity, as evidenced by elevated expression of the Hh pathway targets PTCH1 and Gli1, is predominantly found in nodular/desmoplastic medulloblastomas.12 This suggests that Hh blockade might only affect tumors of this subtype, which accounts for ∼29% of cases.41 Our results contrast somewhat with this earlier study. We examined 18 primary medulloblastomas of the nodular/desmoplastic, classic, and anaplastic subtypes and detected mRNA encoding Gli1 in all cases. We observed high-level Gli1 expression in a larger percentage of the nodular/desmoplastic cases than classic or anaplastic ones, but several individual tumors in each of these other subtypes had elevated Gli1 levels as well. Differences between our study and the prior report could be attributable to sampling issues or to differences in techniques, as the earlier data came from oligonucleotide expression arrays, although we measured Gli1 levels using quantitative RT-PCR. The relatively widespread activation of the Hh pathway in medulloblastoma suggested by our Gli1 expression data are consistent with the broad response to cyclopamine previously documented in newly resected human medulloblastoma.1 Taken together, these data suggest that therapies targeting Hh may be effective in more than one subtype of medulloblastoma.
Apoptosis induced by Hh pathway blockade seems to be caused at least in part by changes in the expression of the anti-apoptotic protein BclII. We found that increased expression of the Hh targets Gli1 and Gli2 positively regulated BclII transcription, whereas pharmacological suppression of Hh activity resulted in deceased BclII expression and increased apoptosis. Our data provide a mechanism explaining the induction of apoptosis previously reported in murine medulloblastoma after Hh blockade,16 and extend the results to include human cell lines. Unlike previous observations in keratinocytes, we find that Gli1 is as potent as Gli2 in inducing BclII expression, suggesting that the effects of the various Gli factors may be somewhat tissue-dependent. The regulation of BclII by Gli1 also contrasts with the Patched-induced cell death reported in developing nonneoplastic neuroepithelial cells, in which Patched acts as a dependence receptor.42,43 In those experiments, cell death induced by the absence of SHH ligand did not seem to function via the canonical SMO/GLI transduction cascade. Our work does not exclude the possibility that Patched acts as a dependence receptor in tumors, but it does indicate that Gli1 also plays a role in regulating cell survival via BclII.
Our studies of primary tumor samples also support the concept that Gli factors regulate BclII expression and apoptosis in medulloblastoma. Gli and BclII proteins are often most highly expressed in the internodular regions of desmoplastic medulloblastoma. Interestingly, apoptosis is suppressed in these zones between nodules, perhaps attributable to the increase in BclII expression driven by Gli. In addition, we found that expression of Gli1 mRNA was significantly associated with that of BclII in a wide range of primary tumors, and 89% of cases with above-median Gli1 levels also had above-median BclII. Although the magnitude of Gli1 and BclII expression was not significantly correlated in the entire group, the fact that tumors with high Gli1 generally also had high BclII suggests that a certain threshold of Gli1 activity might drive BclII expression. A final result of potential clinical significance was the synergistic induction of cell death we observed after Hh blockade and lovastatin exposure. This suggests that cyclopamine or other Hh inhibitors might sensitize medulloblastoma to the pro-apoptotic effects of radiation or other chemotherapeutic agents.
Acknowledgments
We thank Dr. Lisa Oliver, Nantes University, France, for the kind gift of BclII expression plasmid; and Dr. Fritz Aberger, University of Salzburg, Salzburg, Austria, for the kind gift of Gli expression and BclII reporter plasmids.
Footnotes
Address reprint requests to Eli E. Bar or Charles G. Eberhart, Johns Hopkins University School of Medicine, Department of Pathology, 720 Rutland Ave., Ross Building 558, Baltimore, MD 21205. E-mail: ebar1@jhmi.edu and ceberha@jhmi.edu.
Supported in part by the National Institute of Neurological Disorders and Stroke (grant K08NS43279) and Burroughs Wellcome (Career Award in the Biomedical Sciences to C.E.).
References
- Berman DM, Karhadkar SS, Hallahan AR, Pritchard JI, Eberhart CG, Watkins DN, Chen JK, Cooper MK, Taipale J, Olson JM, Beachy PA. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science. 2002;297:1559–1561. doi: 10.1126/science.1073733. [DOI] [PubMed] [Google Scholar]
- Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K, Parker AR, Shimada Y, Eshleman JR, Watkins DN, Beachy PA. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature. 2003;425:846–851. doi: 10.1038/nature01972. [DOI] [PubMed] [Google Scholar]
- Karhadkar SS, Bova GS, Abdallah N, Dhara S, Gardner D, Maitra A, Isaacs JT, Berman DM, Beachy PA. Hedgehog signalling in prostate regeneration, neoplasia and metastasis. Nature. 2004;431:707–712. doi: 10.1038/nature02962. [DOI] [PubMed] [Google Scholar]
- Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernandez-del Castillo C, Yajnik V, Antoniu B, McMahon M, Warshaw AL, Hebrok M. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature. 2003;425:851–856. doi: 10.1038/nature02009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA, Baylin SB. Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature. 2003;422:313–317. doi: 10.1038/nature01493. [DOI] [PubMed] [Google Scholar]
- Hooper JE, Scott MP. Communicating with Hedgehogs. Nat Rev Mol Cell Biol. 2005;6:306–317. doi: 10.1038/nrm1622. [DOI] [PubMed] [Google Scholar]
- Lum L, Beachy PA. The hedgehog response network: sensors, switches, and routers. Science. 2004;304:1755–1759. doi: 10.1126/science.1098020. [DOI] [PubMed] [Google Scholar]
- Pietsch T, Waha A, Koch A, Kraus J, Albrecht S, Tonn J, Sorensen N, Berthold F, Henk B, Schmandt N, Wolf HK, von Deimling A, Wainwright B, Chenevix-Trench G, Wiestler OD, Wicking C. Medulloblastomas of the desmoplastic variant carry mutations of the human homologue of Drosophila patched. Cancer Res. 1997;57:2085–2088. [PubMed] [Google Scholar]
- Raffel C, Jenkins RB, Frederick L, Hebrink D, Alderete B, Fults DW, James CD. Sporadic medulloblastomas contain PTCH mutations. Cancer Res. 1997;57:842–845. [PubMed] [Google Scholar]
- Vorechovsky I, Tingby O, Hartman M, Stromberg B, Nister M, Collins VP, Toftgard R. Somatic mutations in the human homologue of Drosophila patched in primitive neuroectodermal tumours. Oncogene. 1997;15:361–366. doi: 10.1038/sj.onc.1201340. [DOI] [PubMed] [Google Scholar]
- Hallahan AR, Pritchard JI, Hansen S, Benson M, Stoeck J, Hatton BA, Russell TL, Ellenbogen RG, Bernstein ID, Beachy PA, Olson JM. The SmoA1 mouse model reveals that notch signaling is critical for the growth and survival of sonic hedgehog-induced medulloblastomas. Cancer Res. 2004;64:7794–7800. doi: 10.1158/0008-5472.CAN-04-1813. [DOI] [PubMed] [Google Scholar]
- Pomeroy SL, Tamayo P, Gaasenbeek M, Sturla LM, Angelo M, McLaughlin ME, Kim JY, Goumnerova LC, Black PM, Lau C, Allen JC, Zagzag D, Olson JM, Curran T, Wetmore C, Biegel JA, Poggio T, Mukherjee S, Rifkin R, Califano A, Stolovitzky G, Louis DN, Mesirov JP, Lander ES, Golub TR. Prediction of central nervous system embryonal tumour outcome based on gene expression. Nature. 2002;415:436–442. doi: 10.1038/415436a. [DOI] [PubMed] [Google Scholar]
- Lee Y, Miller HL, Jensen P, Hernan R, Connelly M, Wetmore C, Zindy F, Roussel MF, Curran T, Gilbertson RJ, McKinnon PJ. A molecular fingerprint for medulloblastoma. Cancer Res. 2003;63:5428–5437. [PubMed] [Google Scholar]
- Reifenberger J, Wolter M, Weber RG, Megahed M, Ruzicka T, Lichter P, Reifenberger G. Missense mutations in SMOH in sporadic basal cell carcinomas of the skin and primitive neuroectodermal tumors of the central nervous system. Cancer Res. 1998;58:1798–1803. [PubMed] [Google Scholar]
- Dahmane N, Sanchez P, Gitton Y, Palma V, Sun T, Beyna M, Weiner H, Ruiz i Altaba A. The sonic hedgehog-Gli pathway regulates dorsal brain growth and tumorigenesis. Development. 2001;128:5201–5212. doi: 10.1242/dev.128.24.5201. [DOI] [PubMed] [Google Scholar]
- Romer JT, Kimura H, Magdaleno S, Sasai K, Fuller C, Baines H, Connelly M, Stewart CF, Gould S, Rubin LL, Curran T. Suppression of the Shh pathway using a small molecule inhibitor eliminates medulloblastoma in Ptc1(+/−)p53(−/−) mice. Cancer Cell. 2004;6:229–240. doi: 10.1016/j.ccr.2004.08.019. [DOI] [PubMed] [Google Scholar]
- Sanchez P, Ruiz IAA. In vivo inhibition of endogenous brain tumors through systemic interference of Hedgehog signaling in mice. Mech Dev. 2005;122:223–230. doi: 10.1016/j.mod.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Fan H, Oro AE, Scott MP, Khavari PA. Induction of basal cell carcinoma features in transgenic human skin expressing Sonic Hedgehog. Nat Med. 1997;3:788–792. doi: 10.1038/nm0797-788. [DOI] [PubMed] [Google Scholar]
- Louro ID, Bailey EC, Li X, South LS, McKie-Bell PR, Yoder BK, Huang CC, Johnson MR, Hill AE, Johnson RL, Ruppert JM. Comparative gene expression profile analysis of GLI and c-MYC in an epithelial model of malignant transformation. Cancer Res. 2002;62:5867–5873. [PubMed] [Google Scholar]
- Bigelow RL, Chari NS, Unden AB, Spurgers KB, Lee S, Roop DR, Toftgard R, McDonnell TJ. Transcriptional regulation of bcl-2 mediated by the sonic hedgehog signaling pathway through gli-1. J Biol Chem. 2004;279:1197–1205. doi: 10.1074/jbc.M310589200. [DOI] [PubMed] [Google Scholar]
- Regl G, Kasper M, Schnidar H, Eichberger T, Neill GW, Philpott MP, Esterbauer H, Hauser-Kronberger C, Frischauf AM, Aberger F. Activation of the BCL2 promoter in response to hedgehog/GLI signal transduction is predominantly mediated by GLI2. Cancer Res. 2004;64:7724–7731. doi: 10.1158/0008-5472.CAN-04-1085. [DOI] [PubMed] [Google Scholar]
- Lossi L, Zagzag D, Greco MA, Merighi A. Apoptosis of undifferentiated progenitors and granule cell precursors in the postnatal human cerebellar cortex correlates with expression of BCL-2, ICE, and CPP32 proteins. J Comp Neurol. 1998;399:359–372. [PubMed] [Google Scholar]
- Tanabe H, Eguchi Y, Kamada S, Martinou JC, Tsujimoto Y. Susceptibility of cerebellar granule neurons derived from Bcl-2-deficient and transgenic mice to cell death. Eur J Neurosci. 1997;9:848–856. doi: 10.1111/j.1460-9568.1997.tb01434.x. [DOI] [PubMed] [Google Scholar]
- Fuse N, Maiti T, Wang B, Porter JA, Hall TM, Leahy DJ, Beachy PA. Sonic hedgehog protein signals not as a hydrolytic enzyme but as an apparent ligand for patched. Proc Natl Acad Sci USA. 1999;96:10992–10999. doi: 10.1073/pnas.96.20.10992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cartron G, Dacheux L, Salles G, Solal-Celigny P, Bardos P, Colombat P, Watier H. Therapeutic activity of humanized anti-CD20 monoclonal antibody and polymorphism in IgG Fc receptor FcgammaRIIIa gene. Blood. 2002;99:754–758. doi: 10.1182/blood.v99.3.754. [DOI] [PubMed] [Google Scholar]
- Eberhart CG, Kaufman WE, Tihan T, Burger PC. Apoptosis, neuronal maturation, and neurotrophin expression within medulloblastoma nodules. J Neuropathol Exp Neurol. 2001;60:462–469. doi: 10.1093/jnen/60.5.462. [DOI] [PubMed] [Google Scholar]
- Schuller U, Schober F, Kretzschmar HA, Herms J. Bcl-2 expression inversely correlates with tumour cell differentiation in medulloblastoma. Neuropathol Appl Neurobiol. 2004;30:513–521. doi: 10.1111/j.1365-2990.2004.00553.x. [DOI] [PubMed] [Google Scholar]
- Schiffer D, Cavalla P, Migheli A, Giordana MT, Chiado-Piat L. Bcl-2 distribution in neuroepithelial tumors: an immunohistochemical study. J Neurooncol. 1996;27:101–109. doi: 10.1007/BF00177472. [DOI] [PubMed] [Google Scholar]
- Schiffer D, Cavalla P, Chio A, Giordana MT, Marino S, Mauro A, Migheli A. Tumor cell proliferation and apoptosis in medulloblastoma. Acta Neuropathol (Berl) 1994;87:362–370. doi: 10.1007/BF00313605. [DOI] [PubMed] [Google Scholar]
- Eberhart CG, Kratz JE, Schuster A, Goldthwaite P, Cohen KJ, Perlman EJ, Burger PC. Comparative genomic hybridization detects an increased number of chromosomal alterations in large cell/anaplastic medulloblastomas. Brain Pathol. 2002;12:36–44. doi: 10.1111/j.1750-3639.2002.tb00420.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobsen PF, Jenkyn DJ, Papadimitriou JM. Establishment of a human medulloblastoma cell line and its heterotransplantation into nude mice. J Neuropathol Exp Neurol. 1985;44:472–485. doi: 10.1097/00005072-198509000-00003. [DOI] [PubMed] [Google Scholar]
- Di Marcotullio L, Ferretti E, De Smaele E, Argenti B, Mincione C, Zazzeroni F, Gallo R, Masuelli L, Napolitano M, Maroder M, Modesti A, Giangaspero F, Screpanti I, Alesse E, Gulino A. REN(KCTD11) is a suppressor of hedgehog signaling and is deleted in human medulloblastoma. Proc Natl Acad Sci USA. 2004;101:10833–10838. doi: 10.1073/pnas.0400690101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Regl G, Kasper M, Schnidar H, Eichberger T, Neill GW, Ikram MS, Quinn AG, Philpott MP, Frischauf AM, Aberger F. The zinc-finger transcription factor GLI2 antagonizes contact inhibition and differentiation of human epidermal cells. Oncogene. 2004;23:1263–1274. doi: 10.1038/sj.onc.1207240. [DOI] [PubMed] [Google Scholar]
- Wang W, Macaulay RJ. Cell-cycle gene expression in lovastatin-induced medulloblastoma apoptosis. Can J Neurol Sci. 2003;30:349–357. doi: 10.1017/s0317167100003061. [DOI] [PubMed] [Google Scholar]
- Cooper MK, Wassif CA, Krakowiak PA, Taipale J, Gong R, Kelley RI, Porter FD, Beachy PA. A defective response to hedgehog signaling in disorders of cholesterol biosynthesis. Nat Genet. 2003;33:508–513. doi: 10.1038/ng1134. [DOI] [PubMed] [Google Scholar]
- Gilbertson RJ. Medulloblastoma: signalling a change in treatment. Lancet Oncol. 2004;5:209–218. doi: 10.1016/S1470-2045(04)01424-X. [DOI] [PubMed] [Google Scholar]
- Stavrou T, Bromley CM, Nicholson HS, Byrne J, Packer RJ, Goldstein AM, Reaman GH. Prognostic factors and secondary malignancies in childhood medulloblastoma. J Pediatr Hematol Oncol. 2001;23:431–436. doi: 10.1097/00043426-200110000-00008. [DOI] [PubMed] [Google Scholar]
- Packer RJ, Cogen P, Vezina G, Rorke LB. Medulloblastoma: clinical and biologic aspects. Neuro-Oncology. 1999;1:232–250. doi: 10.1215/15228517-1-3-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler-Reya RJ, Scott MP. Control of neuronal precursor proliferation in the cerebellum by sonic hedgehog. Neuron. 1999;22:103–114. doi: 10.1016/s0896-6273(00)80682-0. [DOI] [PubMed] [Google Scholar]
- Goodrich LV, Milenkovic L, Higgins KM, Scott MP. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science. 1997;277:1109–1113. doi: 10.1126/science.277.5329.1109. [DOI] [PubMed] [Google Scholar]
- Eberhart CG, Kepner JL, Goldthwaite PT, Kun LE, Duffner PK, Friedman HS, Strother DR, Burger PC. Histopathologic grading of medulloblastomas: a Pediatric Oncology Group study. Cancer. 2002;94:552–560. doi: 10.1002/cncr.10189. [DOI] [PubMed] [Google Scholar]
- Thibert C, Teillet MA, Lapointe F, Mazelin L, Le Douarin NM, Mehlen P. Inhibition of neuroepithelial patched-induced apoptosis by sonic hedgehog. Science. 2003;301:843–846. doi: 10.1126/science.1085405. [DOI] [PubMed] [Google Scholar]
- Mehlen P, Mille F, Thibert C. Morphogens and cell survival during development. J Neurobiol. 2005;64:357–366. doi: 10.1002/neu.20167. [DOI] [PubMed] [Google Scholar]