Abstract
Purpose.
The purpose of this study is to determine the ability of single-value metrics of retinal image quality of the eye to predict visual performance as measured by high (HC) and low (LC) -contrast acuity at photopic (P) and mesopic (M) light levels in eyes with 20/17 and better visual acuity.
Methods.
Forty-nine normal subjects in good health ranging in age from 21.8 to 62.6 with 20/17 or better monocular high-contrast logarithm of the minimum angle of resolution (logMAR) acuity served as subjects. Wavefront error through the 10th Zernike radial order over a 7-mm pupil was measured on each test eye using a custom-built Shack/Hartmann wavefront sensor. For each eye, 31 different single-value retinal image quality metrics were calculated. Visual acuity was measured using HC (95%) and LC (11%) logMAR at photopic (270 cd/m2) and mesopic (0.75 cd/m2) light levels. To determine the ability of each metric of retinal image quality to predict each type of logMAR acuity (P HC, P LC, M HC, and M LC), each acuity measure was regressed against each optical quality metric.
Results.
The ability of the metrics of retinal image quality to predict logMAR acuity improved as luminance and/or contrast is lowered. The best retinal image quality metric (logPFSc) accounted for 2.6%, 15.1%, 27.6%, and 40.0% of the variance in P HC, P LC, M HC, and M LC logMAR acuity, respectively.
Conclusions.
In eyes with 20/17 and better P HC acuity, P HC logMAR acuity is insensitive to variations in retinal image quality compared with M LC logMAR acuity. Retinal image quality becomes increasingly predictive of logMAR acuity as contrast and/or luminance is decreased. Everyday life requires individuals to function over a large range of contrast and luminance levels. Clinically, the impact of retinal image quality as a function of luminance and contrast is readily measurable in a time-efficient manner with M LC logMAR acuity charts.
Keywords: aberration, wavefront error, optical quality metrics, visual performance, visual acuity
Recently, several investigators 1,2 (first noted by Thibos LN et al. in a presentation at the 2001 Fall vision meeting) reported that there was essentially no correlation between photopic high-contrast acuity (P HC) and measures of retinal image quality in eyes with supernormal P HC acuity (defined as 20/15 or better acuity). Such a finding is not surprising for a variety of reasons elaborated here. However, the fact that retinal image quality in these eyes does not impact visual performance for high-contrast targets should not be interpreted to mean that retinal image quality is uncorrelated with visual performance for other test conditions that are relevant to daily life. These tasks tend to be lower contrast and lower luminance than the task of photopic HC acuity and have been previously been shown to correlate better with retinal image quality in a large population.3
Although 20/15 is frequently described as supernormal, in fact, this is the average acuity of normal best-corrected healthy individuals under the age of 60.4 Moreover, 20/15 acuity is close to the neural sampling limit estimated from anatomic measurements of cone spacing.5 A precise estimate of this neural limit to acuity is difficult to achieve because of individual variation in retinal structure and the complexity of predicting the sampling limit for letter stimuli.6,7 When the subject population is limited to individuals with average or better than average P HC visual acuity, neural factors create a physiological limit that confines optical effects to a narrow acuity range of typically between 20/15 and 20/10. Recently, using an adaptive mirror to minimize the wavefront error of a normal eye with a dilated pupil, acuities have been measured to slightly better than 20/8 (Rossi E. The limits of high contrast photopic letter acuity with adaptive optics. Presented at the International Congress on Wavefront Sensing and Ideal Refractive Corrections, 2006.) Thus, it is not surprising that weak correlations between optical quality and P HC acuity have been reported in the literature. However, these ceiling effects are reduced when letter contrast is reduced or when the luminance is reduced to mesopic levels. These changes in stimulus conditions reduce acuity8-10 but of course have no effect on anatomic sampling density, and therefore, the range of acuity values that might be accounted for by optical factors should increase and correlations improve. Decreasing the luminance to mesopic levels also allows the pupil to dilate, which, in the typical normal eye, decreases retinal image quality11,12 and acuity,13 thereby increasing the predictive power of optical quality analysis.
In daily life, individuals have to operate in a visual world that varies widely in luminance and contrast. Evaluating whether optical aberrations play a significant role in visual performance for P HC acuity targets fails to reflect the importance of lower contrast and luminance levels when operating in the real world. Clinically, one could begin to assess the impact of these factors by measurement of visual performance for low-contrast photopic targets and for high- and low-contrast targets at mesopic light levels. In this way, we may discover that optical factors are more predictive of acuity under challenging viewing conditions. Accordingly, our purpose was to determine the predictive power of metrics of retinal image quality under a variety of viewing conditions that are representative of the range of conditions in daily life.
METHODS
Before data collection, all subjects signed a University Houston Institutional Review Board-approved informed consent. The subjects of this study are the subset of 148 subjects from the Texas Investigation of Normal and Cataract Optics (TINCO study) that had dilated high-contrast logMAR acuity of 20/17 or better. These 49 normal subjects ranged in age from 21.8 to 62.6 (average age, 43.4; standard deviation [SD], 11.4) with monocular high contrast logMAR acuity of -0.06 (20/17) or better (average, -0.12 [20/15]; SD, 0.04; range, -0.22 [20/12] to -0.08 [20/17]) in their self-reported preferred eye served as subjects. All subjects were in good ocular and systemic health for their age.
The preferred eye of each subject was dilated with one drop of 1% tropicamide and 10% Neo-Synephrine. Wavefront error through the 10th Zernike radial order over a 7-mm pupil was measured using a custom-built Shack/Hartmann (S/H) wavefront sensor. The fundamental design principles are published.14-16 In overview, the S/H ocular wavefront sensor images the entrance pupil of the subject through a 1:1 relay telescope onto a lenslet array. The lenslet array is a set of side-by-side small lenses each having a diameter of 400 μm and a focal length of 24 mm. Each lenslet samples a portion of the wavefront originating from a small retinal guide star (approximately 0.75 mm) created by a diode laser (λ= 830 nm, 18 μW incident on the cornea) and images the sampled wavefront onto a CCD camera. Subject alignment is maintained using a pupil camera, which allows the operator to align the subject's entrance pupil conjugate with the entrance aperture of the lenslet array. With the subject in a chin and forehead rest, such an arrangement allows the instrument to be moved to the subject, by motorized drives, as opposed to moving the subject to the instrument.
The displacement of each spot image of the retinal guide star from a known calibration position is used to determine the local slope of the wavefront. These local slopes are in turn used to calculate a representation of the wavefront error over the pupil in the form of a normalized Zernike expansion to the 10th radial order (66 modes) for the specification of ocular wavefront error following the recommended Optical Society of America standards,17 which evolved into the ANSI Z80.28 standard.
For each eye, 30 different single-value retinal image quality metrics were calculated as described previously18 using the measured higher-order aberrations (third to 10th radial orders) over a 7-mm pupil. The reader is referred to Thibos et al.18 for calculation details of all 30 metrics.
Immediately before acuity testing, each subject was trial frame-refracted in the experimental setting (i.e., at 12-feet acuity testing distance) using the end point criteria of maximum plus to best visual acuity. The mean and standard deviation of sphere component of the cycloplegic refraction at 12 feet of the 49 eyes was -0.73 D, ± 2.21 D (range, -6.25 D to +3.00 D). The mean and standard deviation of cylinder component of the cycloplegic refraction at 12 feet of the 49 eyes was -0.74 D, ± 0.84 D (range, -5.00 D to 0.00 D). Table 1 displays all spherocylindrical corrections for all 49 subjects. All acuity testing was performed in one setting taking approximately 10 min while wearing this trial frame refraction.
TABLE 1.
Each subject’s spherocylindrical correction in diopters
| Count | Subject | Sphere | Cylinder | Axis |
|---|---|---|---|---|
| 1 | T3_016 | 0.50 | -0.25 | 150 |
| 2 | T7_024 | 1.75 | -0.75 | 090 |
| 3 | T3_030 | 0.00 | 0.00 | |
| 4 | T3_041 | 0.00 | -0.25 | 178 |
| 5 | T3_015 | 0.50 | -0.25 | 090 |
| 6 | T3_019 | 0.25 | -0.37 | 095 |
| 7 | T3_117 | -4.00 | -0.50 | 180 |
| 8 | T4_106 | 0.75 | -0.75 | 015 |
| 9 | T4_137 | 0.00 | 0.00 | |
| 10 | T5_143 | 2.25 | -0.50 | 180 |
| 11 | T7_121 | 0.25 | -0.75 | 062 |
| 12 | T7_114 | -3.25 | -1.00 | 165 |
| 13 | T6_053 | -0.75 | -1.25 | 098 |
| 14 | T3_034 | 0.75 | 0.00 | |
| 15 | T6_042 | -4.00 | -2.00 | 123 |
| 16 | T5_090 | 3.00 | -0.50 | 180 |
| 17 | T4_107 | -3.00 | 0.00 | |
| 18 | T3_014 | 0.50 | -0.25 | 180 |
| 19 | T3_152 | -0.50 | -0.25 | 016 |
| 20 | T4_072 | -2.00 | -1.25 | 084 |
| 21 | T5_038 | 2.50 | -0.50 | 023 |
| 22 | T6_111 | 1.00 | -0.37 | 125 |
| 23 | T4_187 | 0.50 | -0.25 | 060 |
| 24 | T5_031 | 1.50 | 0.00 | |
| 25 | T4_089 | 1.75 | -0.25 | 100 |
| 26 | T6_149 | -0.75 | -1.50 | 092 |
| 27 | T6_025 | 0.00 | -0.25 | 095 |
| 28 | T4_116 | -1.25 | -1.50 | 004 |
| 29 | T5_028 | -4.50 | -0.75 | 020 |
| 30 | T6_054 | 0.50 | -2.25 | 090 |
| 31 | T5_094 | 1.00 | -0.50 | 010 |
| 32 | T5_062 | -4.50 | -5.00 | 075 |
| 33 | T6_080 | -1.75 | -0.75 | 082 |
| 34 | T5_173 | -1.25 | -1.50 | 008 |
| 35 | T5_135 | -1.25 | -1.00 | 044 |
| 36 | T6_039 | 1.75 | -0.25 | 075 |
| 37 | T5_175 | 0.00 | -0.50 | 030 |
| 38 | T5_098 | -1.00 | -0.25 | 038 |
| 39 | T5_055 | 0.75 | -0.25 | 008 |
| 40 | T5_099 | -1.75 | -0.50 | 173 |
| 41 | T4_177 | -6.25 | -0.50 | 145 |
| 42 | T5_091 | -1.75 | -0.50 | 045 |
| 43 | T6_113 | -0.50 | -1.75 | 093 |
| 44 | T6_059 | -3.50 | -1.50 | 005 |
| 45 | T5_176 | 0.37 | -0.50 | 110 |
| 46 | T5_064 | -2.00 | -1.50 | 090 |
| 47 | T6_069 | 1.75 | -0.25 | 180 |
| 48 | T5_168 | -5.25 | 0.00 | |
| 49 | T6_045 | -5.00 | -0.75 | 030 |
| Average | -0.73 | -0.74 | ||
| Standard deviation | 2.21 | 0.84 | ||
| Minimum | -6.25 | -5.00 | ||
| Maximum | +3.00 | 0 |
Visual acuity (dark letters on a white background) was measured on each subject through their dilated pupil using Bailey/Lovie logMAR acuity charts having high (95%) and low (11%) -contrast letters (HC and LC, respectively) at both photopic (270 cd/m2) and mesopic (0.75 cd/m2) light levels (P and M, respectively) at 12 feet. The charts were placed in the original light boxes designed for the Early Treatment Diabetic Retinopathy Study (ETDRS). These light boxes front illuminate the eye charts with two Phillips 40-watt F40T12 fluorescent tubes. Illumination was reduced by closing doors having a series of pinholes over the fluorescent tubes. Thus, the color temperature was unchanged from photopic to mesopic levels. As noted previously, neither Bailey and Lovie19 nor Ferris et al.20 detail a definitive end point criteria for logMAR acuity testing.21 To maximize precision, the acuity testing protocol used a forced-choice paradigm, strict assessment of errors, and a definitive end point criterion. The number of letters correct was counted until the subject missed a total of five letters starting from the smallest line that could be read without error. Final logMAR acuity was determined by giving credit to each letter read correctly and adjusting for the 12-foot test distance.
To determine the ability of each metric of retinal image quality to predict each type of logMAR acuity (P HC, P LC, M HC, and M LC), each acuity measure was regressed against each optical quality metric and coefficients of determination (R2) calculated. In k and n are regression parameters. To determine the best image quality metrics for predicting all four types of measured logMAR acuity, we averaged the coefficient of determination for each metric's ability to predict each type of acuity. The retinal image quality metric with the highest average coefficient of determination was taken as the best metric for measuring all types of acuity. The second best metric had the next highest average ranking and so on.
RESULTS
Table 2 describes the essence of each of the 10 best retinal image quality metrics for predicting logMAR acuity. Logarithmic transformations were applied to many of the metrics to improve their distribution for linear regression against logMAR.
TABLE 2.
A brief description of each of the top 10 retinal image quality metrics for predicting logMAR acuity
| logPFSc | Log of the pupil fraction; pupil fraction is defined as the concentric area for which root mean squared error of the slopes of the wavefront error is <1 arcmin divided by total available pupil area |
| log Area OTF | Log of the area of visibility for the radially averaged optical transfer function (normalized to diffraction-limited case); the radially averaged optical transfer function (OTF) is determined by integrating the full two-dimensional OTF over orientation; because the phase transfer function component of the OTF is taken into account when computing rOTF, this metric is intended to capture spatial phase errors in the image |
| SFcOTF | Spatial frequency cutoff of radially averaged OTF |
| log PFSt | Log of the pupil fraction when pupil fraction is defined as the area of a subaperture that satisfies the criterion horizontal slope and vertical slope are both <1 min arc divided by total available pupil area |
| log VSOTF | Log of the visual Strehl ratio computed in frequency domain using the OTF method in which the OTF is weighted by the contrast sensitivity function |
| log SRMTF | Log of the Strehl ratio calculated using the MTF method |
| log STD | Log of the standard deviation of intensity values in the point spread function (PSF) normalized to standard deviation of the intensity values in the PSF for the diffraction-limited case |
| log VSMTF | Log of the visual Strehl ratio computed in frequency domain using the modulation transfer function (MTF) method in which the MTF is weighted by the contrast sensitivity function |
| log NS | Log neural sharpness is defined as the PSF weighted by spatial sensitivity function of the neural system and normalized by the diffraction-limited PSF |
| RMSw | Root mean squared error of the wavefront error; this is equivalent to the standard deviation of the wavefront error |
Figure 1 shows typical regressions of logMAR acuity against singlevalue metrics of retinal image quality. In Figure 1A, the test conditions were photopic, high-contrast (P HC) letters and in Figure 1B, the test conditions were mesopic, low-contrast letters (M LC). Notice that P HC acuity varied over a very narrow range (seven letters,) whereas M LC acuity for the same eyes varied over a range that is nearly three times larger (20 letters). Furthermore, notice that although both regressions indicate that acuity declines as image quality declines, the ability of the retinal image quality metric to account for variance in acuity is over eight times poorer for P HC acuity than it is for M LC acuity (R2 = 0.045 and 0.374, respectively) for the particular metric shown in Figure 1 (log [area under OTF]).
FIGURE 1.
Variation of photopic, high-contrast logMAR acuity (A) and mesopic, low-contrast logMAR acuity (B) with retinal image quality as measured with the metric log(area optical transfer function). For photopic high-contrast (P HC) conditions, the best linear regression reveals a weak correlation (R2= 0.045) indicating that better retinal image quality (high values of the metric) is weakly correlated with better acuity (low values of logMAR). For mesopic low-contrast (M LC) conditions, the best regression reveals a relatively strong correlation (R2= 0.374). Note that M LC acuity varies over a larger range than does P HC acuity. One line of logMAR acuity is shaded on both graphs for reference.
Figure 2 plots the values of the coefficient of determination (R2) for the top 10 retinal image quality metrics for each of the four types of logMAR acuity (P HC, P LC, M HC, and M LC). Notice that as luminance and or contrast is decreased, the coefficient of determination increases. For P HC, P LC, M HC, and M LC acuity, the average R2 value for all 10 metrics is 0.038, 0.130, 0.240, and 0.334, respectively. Overall, the most predictive metric of retinal image quality (logPFSc) accounts for 40% of the variance in M LC logMAR acuity and only 2.6% of the variance in P HC acuity.
FIGURE 2.
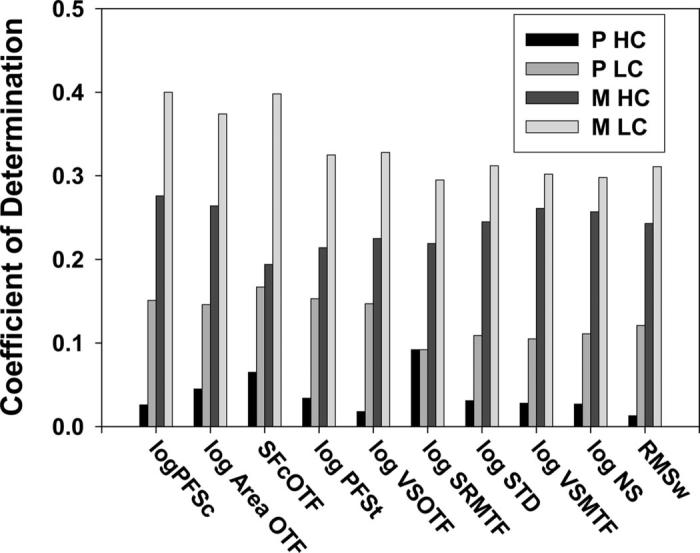
The coefficient of determination (R2) with which the top 10 retinal image quality metrics predict four types of logMAR visual acuity: photopic high contrast, photopic low contrast, mesopic high contrast, and mesopic low contrast logMAR acuity. The reader's attention is drawn to fact that as luminance and/or contrast are decreased, the amount of variance in acuity accounted for by these metrics of retinal image quality increases.
DISCUSSION
The results of our study confirm previous findings1 (Thibos LN, et al. 2001 Fall Vision Meeting; Artal P. Wavefront Congress 2005; Artal P, et al. ARVO 2005) that monochromatic image quality is a relatively poor predictor of logMAR acuity for photopic, high-contrast letters in observers with normal (i.e., better than 20/17) acuity. Importantly and unlike the previous work, extending the visual performance into ranges of contrast and luminance more common to everyday life revealed that the predictive power of image quality improves (i.e., R2 increases) when the contrast and/or mean luminance of the target is reduced (task becomes more demanding, information available to the subject less redundant).
To account for this effect of stimulus conditions on the coefficients of determination, R2, we note two qualitative features of the data in Figure 1 that were typical of similar regression curves for all the top 10 retinal image-quality metrics. First, the regression slope is steeper for the M LC condition than for the P HC condition. Second, the variability of the individual data points from the linear regression is greater for the M LC condition than for the P HC condition. These are important observations because in the linear regression model logMAR = m*Q+b+noise, where Q is an image quality metric (e.g., log[area under OTF]), the coefficient of determination depends principally on the ratio of slope parameter m to the standard deviation of the additive noise. For example, if slope increases, R2 increases; if noise increases, R2 decreases; but if slope increases and noise increases while their ratio remains constant, then R2 is unchanged. Thus, the increased regression slope in Figure 1B compared with Figure 1A would be expected to increase R2, as we observed experimentally. However, the increased variability in the data in Figure 1B would be expected to decrease R2, which is opposite to what we observed. Thus, the increased variability observed under mesopic conditions with low-contrast letters cannot account for the improved predictability of logMAR from metrics of image quality. Instead, the greater predictability is the result of the greater slope, which in turn is the result of the greater range of logMAR values encountered in our study population because the range of the image quality values in Figures 1A and 1B were exactly the same.
Our results indicate that the correlation between image quality and visual acuity is higher for letters of low contrast on a dim background because of the greater individual variation in logMAR performance under these conditions in the normal population with 20/17 or better acuity. The implication for everyday life is that under dim illumination conditions where the pupil is larger, and/or for low-contrast targets, visual acuity is affected by the optical quality of the retinal image.
There are several limitations to the current study. None of the limitations negate the principal findings. They are nonetheless important to consider as future studies are designed and conducted. First, testing was performed with a pupil drug dilated. The dilated pupil diameter of the 49 subjects was on average 7.94 mm with a standard deviation of ± 0.70 mm. Wavefront error was calculated over a fixed 7.0-mm pupil for all subjects. Consequently, acuity was measured through pupils that were somewhat variable in diameter and always slightly larger than the pupil diameter over which the wavefront error was measured (7 mm). It is anticipated that if pupil diameter had been fixed with a 7-mm artificial pupil, the correlations would improve between acuity and metrics of retinal image quality. A corollary that would not be solved by use of a large artificial pupil is that a drug dilated pupil is falsely large (compared with physiological pupils), particularly for photopic light levels. Larger pupils have poorer retinal image quality than do smaller pupils. It therefore is likely that the correlations reported for the photopic levels are actually better (metrics of retinal image quality vary more widely the larger the pupil) under the experimental paradigm reported here than they will be when the pupil is allowed to constrict as it would naturally under photopic testing. As a consequence, it is anticipated that the correlations between retinal image quality and photopic HC acuity are even worse under physiological pupils than reported here, further emphasizing the need for the use of more sensitive tests of visual performance than high-contrast acuity.
Second, the correlations between retinal image quality and visual performance reported here are likely to further improve when residual lower-order aberrations are added to the calculations of retinal image quality for several reasons. Consider the fact that because a dilated refraction is typically performed in 0.25-D steps, spherical and cylindrical errors are not reduced to zero. Residual spherocylindrical errors on the order of 0.125 to 0.25 D are particularly significant when the pupil is large and diminish in significance as pupil size decreases. Also consider the fact that patients use sphere and cylinder to improve retinal image quality by balancing higher-order aberrations with appropriate amounts of sphere and/or cylinder. 22 During a refraction, the subject is simply answering the question: “Which is better, choice one or choice two?” By answering this question, a patient is balancing higher-order aberrations with a spherocylindrical correction. Consequently, ignoring residual lower-order aberrations during the calculation of retinal image quality as done in this article does not entirely reflect the actual retinal image quality present when the patient is wearing their trial frame correction. We expect that refining our technique by measuring the residual optical aberrations while wearing a trial frame correction will further improve the correlations reported here.
Third, our monochromatic analysis of image quality is only an approximation to the actual image quality of a polychromatic retinal image. As the field continues to mature, need to include the polychromatic nature of light in our modeling of retinal image quality will grow.
Finally, it is important to state that it was not the purpose of this study to examine the correlation between physiological pupil sizes and visual performance in a normal population with good acuity. Consequently, further testing is required to verify these predictions under normal physiological conditions in which the pupil size varies physiologically between subjects and with ambient illumination. Nonetheless, it is fully expected that correlations between acuity and metrics of retinal image quality will improve as contrast is lowered (moving measured acuity away from sampling limits imposed by the retinal mosaic) and luminance is lowered (allowing the pupil to dilate naturally).
As the ophthalmic community continues to improve methods of wavefront-guided corrections, it will become increasingly important to use tests of visual performance that are sensitive to the induced changes in retinal image quality. Based on the results of our study, we recommend M LC acuity as a sensitive clinically viable measure of visual performance. The use of sensitive visual acuity testing is particularly attractive in the clinical environment because acuity testing is 1) well understood by clinicians and patients, 2) easy to administer by a trained technician, 3) time-efficient, and 4) inexpensive.
CONCLUSIONS
P HC logMAR acuity is relatively insensitive to variations in retinal image quality present in eyes with 20/17 and better visual acuity. Retinal image quality becomes increasingly predictive of logMAR acuity as contrast and/or luminance is decreased. Everyday life requires individuals to function over a large range of contrast levels and luminance. Individuals with good P HC acuity (20/17 or better) with good retinal image quality had better M LC acuity than those with poorer retinal image quality. When measuring visual performance using logMAR visual acuity, the positive or negative impact of small changes in retinal image quality in eyes are best reflected in a corresponding change in mesopic low contrast acuity. We do not live in a high-contrast, high-luminance world, resulting in the fact that the quality of the retinal image in eyes with good visual acuity does impact visual performance in everyday life.
ACKNOWLEDGMENTS
This work was supported by: NIH R01 EY05280 to RAA, NIH R01 EY05109 to LNT, EY07024 to JDM, NIH/NEI P30 EY007551 to College of Optometry, University of Houston, and the Visual Optics Institute at College of Optometry, University of Houston.
Drs. Applegate and Thibos have proprietary interests in single value metrics of optical performance.
REFERENCES
- 1.Levy Y, Segal O, Avni I, Zadok D. Ocular higher-order aberrations in eyes with supernormal vision. Am J Ophthalmol. 2005;139:225–8. doi: 10.1016/j.ajo.2004.08.035. [DOI] [PubMed] [Google Scholar]
- 2.Artal P, Villegas EA, Alcon E, Benito A. Better than normal visual acuity does not require perfect ocular optics. Invest Ophthalmol Vis Sci. 2005;46 E-abstract 3615. [Google Scholar]
- 3.Pesudovs K, Marsack JD, Donnelly WJ, 3rd, Thibos LN, Applegate RA. Measuring visual acuity—mesopic or photopic conditions, and high or low contrast letters? J Refract Surg. 2004;20:S508–14. [PubMed] [Google Scholar]
- 4.Elliott DB, Yang KC, Whitaker D. Visual acuity changes throughout adulthood in normal, healthy eyes: seeing beyond 6/6. Optom Vis Sci. 1995;72:186–91. doi: 10.1097/00006324-199503000-00006. [DOI] [PubMed] [Google Scholar]
- 5.Williams DR, Porter J, Yoon G, Guirao A, Hofer H, Chen L, Cox I, MacRae S. How far can we extend the limits of human vision? In: Krueger RR, Applegate RA, MacRae SM, editors. Wavefront Customized Visual Correction: The Quest for Super Vision II. Slack; Thorofare, NJ: 2004. pp. 19–38. [Google Scholar]
- 6.Anderson RS, Thibos LN. Sampling limits and critical bandwidth for letter discrimination in peripheral vision. J Opt Soc Am (A) 1999;16:2334–42. doi: 10.1364/josaa.16.002334. [DOI] [PubMed] [Google Scholar]
- 7.Anderson RS, Thibos LN. Relationship between acuity for gratings and for tumbling-E letters in peripheral vision. J Opt Soc Am (A) 1999;16:2321–33. doi: 10.1364/josaa.16.002321. [DOI] [PubMed] [Google Scholar]
- 8.Haegerstrom-Portnoy G. The Glenn A. Fry Award Lecture 2003: Vision in elders—summary of findings of the SKI study. Optom Vis Sci. 2005;82:87–93. doi: 10.1097/01.opx.0000153162.05903.4c. [DOI] [PubMed] [Google Scholar]
- 9.Regan D. High and low contrast acuity. Optom Vis Sci. 1990;67:650–6. doi: 10.1097/00006324-199008000-00018. [DOI] [PubMed] [Google Scholar]
- 10.Brown B, Lovie-Kitchin JE. High and low contrast acuity and clinical contrast sensitivity tested in a normal population. Optom Vis Sci. 1989;66:467–73. doi: 10.1097/00006324-198907000-00010. [DOI] [PubMed] [Google Scholar]
- 11.Campbell FW, Green DG. Optical and retinal factors affecting visual resolution. J Physiol (Lond) 1965;181:576–93. doi: 10.1113/jphysiol.1965.sp007784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Charman WN. Wavefront aberration of the eye: a review. Optom Vis Sci. 1991;68:574–83. doi: 10.1097/00006324-199108000-00002. [DOI] [PubMed] [Google Scholar]
- 13.Haegerstrom-Portnoy G, Brabyn J, Schneck ME, Jampolsky A. The SKILL Card. An acuity test of reduced luminance and contrast. Smith-Kettlewell Institute Low Luminance. Invest Ophthalmol Vis Sci. 1997;38:207–18. [PubMed] [Google Scholar]
- 14.Liang J, Williams DR. Aberrations and retinal image quality of the normal human eye. J Opt Soc Am (A) 1997;14:2873–83. doi: 10.1364/josaa.14.002873. [DOI] [PubMed] [Google Scholar]
- 15.Liang J, Grimm B, Goelz S, Bille JF. Objective measurement of wave aberrations of the human eye with the use of a Hartmann-Shack wave-front sensor. J Opt Soc Am (A) 1994;11:1949–57. doi: 10.1364/josaa.11.001949. [DOI] [PubMed] [Google Scholar]
- 16.Salmon TO, Thibos LN, Bradley A. Comparison of the eye's wave-front aberration measured psychophysically and with the Shack-Hartmann wave-front sensor. J Opt Soc Am (A) 1998;15:2457–65. doi: 10.1364/josaa.15.002457. [DOI] [PubMed] [Google Scholar]
- 17.Thibos LN, Applegate RA, Schwiegerling JT, Webb R. Report from the VSIA taskforce on standards for reporting optical aberrations of the eye. J Refract Surg. 2000;16:S654–5. doi: 10.3928/1081-597X-20000901-34. [DOI] [PubMed] [Google Scholar]
- 18.Thibos LN, Hong X, Bradley A, Applegate RA. Accuracy and precision of objective refraction from wavefront aberrations. J Vis. 2004;4:329–51. doi: 10.1167/4.4.9. [DOI] [PubMed] [Google Scholar]
- 19.Bailey IL, Lovie JE. New design principles for visual acuity letter charts. Am J Optom Physiol Opt. 1976;53:740–5. doi: 10.1097/00006324-197611000-00006. [DOI] [PubMed] [Google Scholar]
- 20.Ferris FL, 3rd, Kassoff A, Bresnick GH, Bailey I. New visual acuity charts for clinical research. Am J Ophthalmol. 1982;94:91–6. [PubMed] [Google Scholar]
- 21.Applegate RA, Howland HC, Sharp RP, Cottingham AJ, Yee RW. Corneal aberrations and visual performance after radial keratotomy. J Refract Surg. 1998;14:397–407. doi: 10.3928/1081-597X-19980701-05. [DOI] [PubMed] [Google Scholar]
- 22.Applegate RA, Marsack JD, Ramos R, Sarver EJ. Interaction between aberrations to improve or reduce visual performance. J Cataract Refract Surg. 2003;29:1487–95. doi: 10.1016/s0886-3350(03)00334-1. [DOI] [PubMed] [Google Scholar]



