Abstract
Objective: To examine the relation between plasma concentrations of interleukin-18 (IL-18), the interferon γ inducing factor, and clinical instability of coronary artery disease.
Design and setting: Observational study in a university hospital.
Patients: 11 patients with unstable angina and negative troponin I, 21 patients with acute non-Q wave myocardial infarction (MI), 21 patients with acute Q wave MI, 9 patients with stable angina, and 11 controls.
Main outcome measures: Plasma IL-18 concentrations and their relation to clinical instability and myocardial dysfunction.
Results: Plasma concentrations of IL-18 were significantly increased in the unstable angina and MI groups in comparison with the stable angina and control groups (p < 0.01). No difference in IL-18 concentrations were found between patients with unstable angina, patients with non-Q wave MI, and patients with Q wave MI. Plasma IL-18 concentrations significantly correlated with decreased left ventricular ejection fraction (p = 0.01).
Conclusions: Plasma IL-18 concentrations are increased in patients with acute coronary syndromes and correlate with the severity of myocardial dysfunction.
Keywords: myocardial infarction, acute coronary syndromes, inflammation, interleukin-18, IL-18
Inflammatory mechanisms have an important role in the pathogenesis of atherosclerotic disease and the occurrence of acute ischaemic syndromes.1 However, because of our limited knowledge about the critical inflammatory pathways involved in the pathogenesis and progression of ischaemic heart disease in humans, no specific anti-inflammatory treatment can be advocated at this time. Interleukin-18 (IL-18), identified as the interferon γ inducing factor, is produced by many cell types, particularly macrophages, and has direct proinflammatory activities in diverse cell types including inflammatory and vascular cells.2,3 We recently reported increased expression of IL-18 and its receptor in human atherosclerotic plaques,4 and Pomerantz and colleagues5 identified a deleterious role for IL-18 in an in vitro model of ischaemia–reperfusion injury. In addition, Seta and associates6 reported increased plasma concentrations of IL-18 in patients with acute myocardial infarction (MI), further supporting a role for IL-18 in this context. However, whether patients with other acute coronary syndromes have increased concentrations of IL-18 is unknown. In the present study, we examined the relation between plasma concentrations of IL-18 and the clinical instability of coronary artery disease. We also examined the relation between IL-18 concentrations and the extent of myocardial dysfunction.
METHODS
Patient characteristics
We studied 53 consecutive patients admitted to an intensive care unit for a chest pain syndrome at rest and ST segment modifications suggestive of myocardial ischaemia or necrosis. On the basis of serial serum troponin I measurements, the patients were classified as having had unstable angina (negative tests for troponin I, n = 11, 7 men and 4 women, mean (SEM) 69.9 (4.2) years old) or acute MI (positive troponin I tests, n = 42, 21 non-Q wave and 21 Q wave MI, 29 men and 13 women, 62.6 (1.9) years old). Patients with Q wave MI were admitted less than six hours after symptoms onset. Those with other ischaemic signs were admitted within 24 hours. To examine the association between IL-18 concentrations and plaque instability, we included a group of patients with documented stable coronary artery disease (n = 9, 6 men and 3 women, 65.8 (5.4) years old). Table 1 shows the patients’ characteristics and treatment during their hospital stay. In addition, five non-coronary patients (one with non-ischaemic chest pain, one with mitral stenosis, one with sinoauricular block, and two with acute pericarditis) and six healthy subjects formed the non-coronary control group (n = 11, 7 men and 4 women, 36.1 (4.0) years old). All patients and subjects gave informed consent.
Table 1.
Baseline clinical characteristics of the patients
| Non-coronary group | Stable angina group | Unstable angina group | Myocardial infarction group | |
| Age (years) | 36.1 (4.0) | 65.8 (5.4) | 69.9 (4.2) | 62.6 (1.9) |
| Number (male/female) | 11 (7/4) | 9 (6/3) | 11 (7/4) | 42 (29/13) |
| Medical history | ||||
| Dyslipidaemia | 0 (0%) | 5 (55.6%) | 3 (27.3%) | 20 (47.6%) |
| Hypertension | 1 (9%) | 6 (66.7%) | 6 (54.5%) | 18 (42.9%) |
| Diabetes mellitus | 0 (0%) | 3 (33.3%) | 1 (9.1%) | 9 (21.4%) |
| Smokers | 3 (27%) | 3 (33.3%) | 5 (45.5%) | 21 (50.0%) |
| Familial CAD | 0 (0%) | 0 (0%) | 2 (18.2%) | 7 (16.7%) |
| Number of diseased coronary arteries* | ||||
| 1 | ND | 3 (33.3%) | 5 (45.5%) | 11 (26.2%) |
| 2 | ND | 2 (22.2%) | 3 (27.3%) | 14 (33.3%) |
| 3 | ND | 3 (33.3%) | 1 (9.1%) | 9 (21.4%) |
| Thrombus | ND | 0 (0%) | 0 (0%) | 23 (54.8%) |
| Left ventricular ejection fraction (%)† | ND | 55.5 (5.0) | 55.1 (3.6) | 51.8 (1.8) |
| Treatment during hospital stay | ||||
| Primary coronary angioplasty | NA | 0 (0%) | 3 (27.3%) | 12 (28.6%) |
| Delayed coronary angioplasty | NA | 0 (0%) | 3 (37.3%) | 22 (52.4%) |
| Thrombolysis (accelerated rtPA) | NA | 0 (0%) | 0 (0%) | 5 (11.9%) |
| Unfractionated heparin | NA | 0 (0%) | 1 (9.1%) | 13 (31.0%) |
| Low molecular weight heparin | NA | 0 (0%) | 9 (81.8%) | 29 (69.0%) |
| Abciximab | NA | 0 (0%) | 0 (0%) | 14 (33.3%) |
| Aspirin | 1 (9%) | 7 (77.8%) | 11 (100%) | 41 (97.6%) |
| Ticlopidine or clopidogrel | 0 (0%) | 1 (11.1%) | 6 (54.5%) | 26 (61.9%) |
| Intravenous vasopressors | NA | 0 (0%) | 0 (0%) | 4 (9.5%) |
| Nitrates | 0 (0%) | 2 (22.2%) | 4 (36.4%) | 11 (26.2%) |
| Diuretics | 1 (9%) | 0 (0%) | 0 (0%) | 6 (14.3%) |
| β Blockers | 0 (0%) | 5 (55.6%) | 9 (81.8%) | 33 (78.6%) |
| ACE inhibitors | 1 (9%) | 3 (33.3%) | 3 (27.3%) | 19 (45.2%) |
| Calcium inhibitors | 0 (0%) | 5 (55.6%) | 3 (27.3%) | 2 (4.8%) |
| Statins | 0 (0%) | 3 (33.3%) | 3 (27.3%) | 29 (69.0%) |
| Insulin | 0 (0%) | 2 (22.2%) | 1 (9.1%) | 6 (14.3%) |
Age and left ventricular ejection fraction presented as mean (SEM).
*Twelve patients did not undergo coronary angiography during the hospital stay (three patients because of refusal, eight patients because of lack of approval by their physicians, and one patient because of non-fatal haemorrhagic stroke). Nine of these patients had documented coronary artery disease (CAD) on a previous coronary angiography and all patients with unstable angina had ischaemic ECG changes during hospitalisation.
†Left ventricular ejection fraction was measured using two dimensional echocardiography.
ACE, angiotensin converting enzyme; NA, not applicable; ND, not determined; rtPA, recombinant tissue plasminogen activator.
Determination of plasma IL-18 concentrations
Blood samples were collected by venepuncture in 5 ml citrated tubes during hospitalisation between 6 00 and 8 00 am. The interval between admission and blood sampling for IL-18 ranged from 0–9 days with a median at 2 days. Plasma samples were prepared for determination of IL-18 concentrations using a commercially available enzyme linked immunosorbent assay (ELISA) kit (MBL, Nagoya, Japan).
Statistics analysis
Results are expressed as medians and ranges. Groups were compared by the Kruskal-Wallis and Mann-Whitney tests. Simple regression analysis was used to analyse the relation between plasma values of IL-18 and left ventricular ejection fraction or C reactive protein concentrations.
RESULTS
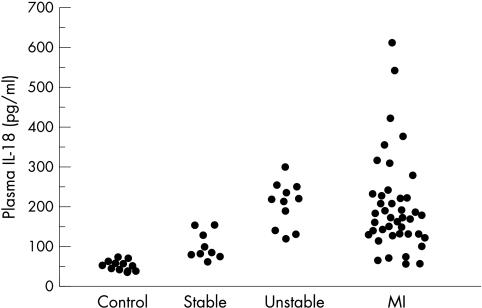
Figure 1 presents the data distribution. IL-18 concentrations were not associated with age, sex, risk factors, medications, or time from admission to sampling. The median IL-18 concentrations in control non-coronary subjects (46.8 pg/ml, range 34.2–68.2 pg/ml) were significantly different from those in patients with stable angina (85.7 pg/ml, range 56.0–157.7 pg/ml, p < 0.01), suggesting that IL-18 concentrations in patients with stable coronary disease may be associated with the presence of advanced coronary artery disease. In the group of patients with unstable angina, the median IL-18 concentrations (214.7 pg/ml, range 116.6–297.0 pg/ml) were significantly higher than those in the control group (p < 0.001) or the group with stable angina (p = 0.0012). In the group of patients with MI, the median IL-18 concentrations (164.6 pg/ml, range 53.6–602.5 pg/ml) were also significantly higher than those in the control group (p < 0.001) or the group with stable angina (p < 0.01). The IL-18 concentrations did not differ significantly between the group with unstable angina and the group with MI or between patients with Q wave and patients with non-Q wave MI.
Figure 1.
Distribution of plasma IL-18 concentrations in the patient groups.
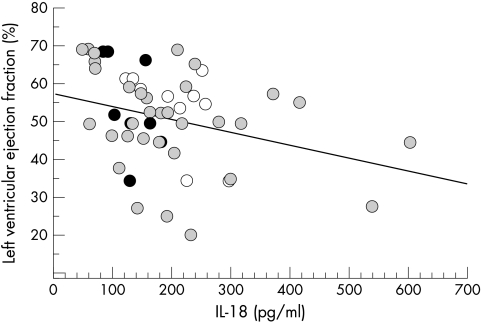
Plasma IL-18 concentrations correlated significantly with the severity of myocardial dysfunction as assessed by the determination of ventricular ejection fraction (r = −0.35, p = 0.011) (fig 2).C reactive protein concentrations were available for 20 patients with unstable angina or MI (median 24.5 μg/ml, range 5.0–105.0 μg/ml). We found no correlation between IL-18 and C reactive protein concentrations (p = 0.38).
Figure 2.
Relation between left ventricular ejection fraction and plasma IL-18 concentrations (r = −0.35, p = 0.011) in patients with stable angina (black circles), unstable angina (white circles), or myocardial infarction (grey circles).
DISCUSSION
Two potentially important findings were observed in the present study. Firstly, plasma concentrations of IL-18 are increased in patients with acute coronary syndromes with or without myocardial necrosis. Secondly, plasma concentrations of IL-18 correlate with the severity of myocardial dysfunction.
Although these findings are preliminary and need confirmation in a large multicentre study, we believe that they should be considered seriously for several reasons. IL-18 has the potential to promote both atherosclerotic plaque instability4 and systemic inflammatory responses through activation of monocytes/macrophages, lymphocytes, and endothelial cells. Indeed, we have recently showed that in vivo inhibition of IL-18 signalling greatly decreases plaque development and induces a switch to a stable plaque phenotype.7 In the human myocardium, IL-18 is upregulated following ischaemia and contributes to postischaemic myocardial dysfunction in vitro.5 IL-18 may aggravate the proinflammatory response within the myocardium through increased expression of endothelial cell adhesion molecules8 and production of proinflammatory mediators such as IL-1β, IL-8, tumour necrosis factor α, and inducible nitric oxide synthase.2,9 These proinflammatory mediators, particularly tumour necrosis factor α and nitric oxide, have already been implicated in myocardial contractile depression10 and loss of cardiomyocytes.11,12 In addition, IL-18 upregulates membrane Fas ligand expression13 and may therefore contribute to Fas mediated apoptosis of Fas expressing cardiomyocytes. Interestingly, this pathway may further induce IL-18 production14 and has already been involved in both cardiomyocyte apoptosis15 and cardiomyocyte arrhythmogenicity16 related to heart disease. Finally, IL-18 is emerging as a potent antiangiogenic cytokine17 and may therefore severely affect myocardial neoangiogenesis following ischaemia, with its expected deleterious consequences on myocardial functional recovery and remodelling.
In conclusion, this preliminary study suggests an important role for IL-18 in unstable coronary artery disease and ischaemic myocardial dysfunction. These results should pave the way for future studies aimed at defining the precise roles of IL-18 or its endogenous inhibitor, IL-18 binding protein, in the initiation or progression of ischaemic heart disease.
Acknowledgments
This study was supported by Action Concertée Incitative Jeunes Chercheurs, ACI 2000, Ministére de la Recherche, France.
Abbreviations
ELISA, enzyme linked immunosorbent assay
IL, interleukin
MI, myocardial infarction
REFERENCES
- 1.Lusis AJ. Atherosclerosis. Nature 2000;407:233–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Okamura H, Tsutsui H, Kashiwamura S, et al. Interleukin-18: a novel cytokine that augments both innate and acquired immunity. Adv Immunol 1998;70:281–312. [DOI] [PubMed] [Google Scholar]
- 3.Dinarello CA. Interleukin-18, a proinflammatory cytokine. Eur Cytokine Netw 2000;11:483–6. [PubMed] [Google Scholar]
- 4.Mallat Z, Corbaz A, Scoazec A, et al. Expression of interleukin-18 in human atherosclerotic plaques and relation to plaque instability. Circulation 2001;104:1598–603. [DOI] [PubMed] [Google Scholar]
- 5.Pomerantz BJ, Reznikov LL, Harken AH, et al. Inhibition of caspase 1 reduces human myocardial ischemic dysfunction via inhibition of IL-18 and IL-1beta. Proc Natl Acad Sci USA 2001;98:2871–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Seta Y, Kanda T, Tanaka T, et al. Interleukin 18 in acute myocardial infarction. Heart 2000;84:668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mallat Z, Corbaz A, Scoazec A, et al. Interleukin-18/interleukin-18 binding protein signaling modulates atherosclerotic lesion development and stability. Circ Res 2001;89:E41–5. [DOI] [PubMed] [Google Scholar]
- 8.Vidal-Vanaclocha F, Fantuzzi G, Mendoza L, et al. IL-18 regulates IL-1beta-dependent hepatic melanoma metastasis via vascular cell adhesion molecule-1. Proc Natl Acad Sci USA 2000;97:734–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Puren AJ, Fantuzzi G, Gu Y, et al. Interleukin-18 (IFNgamma-inducing factor) induces IL-8 and IL-1beta via TNFalpha production from non-CD14+ human blood mononuclear cells. J Clin Invest 1998;101:711–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kelly RA, Smith TW. Cytokines and cardiac contractile function. Circulation 1997;95:778–81. [DOI] [PubMed] [Google Scholar]
- 11.Krown KA, Page MT, Nguyen C, et al. Tumor necrosis factor alpha-induced apoptosis in cardiac myocytes: involvement of the sphingolipid signaling cascade in cardiac cell death. J Clin Invest 1996;98:2854–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Koglin J, Granville DJ, Glysing-Jensen T, et al. Attenuated acute cardiac rejection in NOS2 -/- recipients correlates with reduced apoptosis. Circulation 1999;99:836–42. [DOI] [PubMed] [Google Scholar]
- 13.Hashimoto W, Osaki T, Okamura H, et al. Differential antitumor effects of administration of recombinant IL-18 or recombinant IL-12 are mediated primarily by Fas-Fas ligand- and perforin-induced tumor apoptosis, respectively. J Immunol 1999;163:583–9. [PubMed] [Google Scholar]
- 14.Tsutsui H, Kayagaki N, Kuida K, et al. Caspase-1-independent, Fas/Fas ligand-mediated IL-18 secretion from macrophages causes acute liver injury in mice. Immunity 1999;11:359–67. [DOI] [PubMed] [Google Scholar]
- 15.Jeremias I, Kupatt C, Martin-Villalba A, et al. Involvement of CD95/Apo1/Fas in cell death after myocardial ischemia. Circulation 2000;102:915–20. [DOI] [PubMed] [Google Scholar]
- 16.Felzen B, Shilkrut M, Less H, et al. Fas (CD95/Apo-1)-mediated damage to ventricular myocytes induced by cytotoxic T lymphocytes from perforin-deficient mice: a major role for inositol 1,4,5-trisphosphate. Circ Res 1998;82:438–50. [DOI] [PubMed] [Google Scholar]
- 17.Cao R, Farnebo J, Kurimoto M, et al. Interleukin-18 acts as an angiogenesis and tumor suppressor. FASEB J 1999;13:2195–202. [DOI] [PubMed] [Google Scholar]