Abstract
Objective: To evaluate prospectively the influence of an angiotensin I converting enzyme (ACE) gene polymorphism on long term clinical outcome of patients with established coronary artery disease treated by percutaneous coronary intervention.
Design and setting: Prospective observational study in a university hospital.
Patients: Consecutive series of 1010 patients with symptomatic coronary artery disease who underwent successful coronary stent placement from November 1996 to April 1998.
Main outcome measures: Long term clinical outcome was obtained and the rates of major adverse cardiac events (death, non-fatal acute myocardial infarction, unstable angina, and revascularisation) were compared according to the insertion/deletion (I/D) polymorphism of the ACE gene.
Results: Of the 1010 patients 29% had the DD genotype, 51% had the ID genotype, and 20% had the II genotype. All baseline clinical angiographic and procedural characteristics were identical in the three groups of patients. Event-free survival during the follow up period (median two years) was identical in patients with the II genotype compared to those with one or two D alleles. The predictors of long term survival were age, diabetes, ejection fraction, and extension of coronary artery disease. ACE genotype had no influence on the long term survival. Additional analyses assuming dominant and recessive effects of the D allele also failed to find any association; nor did the examination of low risk subgroups.
Conclusions: The ACE I/D polymorphism does not influence the long term prognosis of patients with coronary disease treated by percutaneous coronary intervention, and screening patients for this gene polymorphism is not useful for secondary prevention strategies.
Keywords: ACE genotype, polymorphism, stent, angioplasty, long term outcome, coronary artery disease
The angiotensin I converting enzyme (ACE) insertion/deletion (I/D) gene polymorphism has been implicated in the clinical manifestations of coronary atherosclerosis.1–4 Subjects bearing the ACE D allele have a higher concentration of circulating enzyme than those bearing the I allele.5 Early studies suggested that ACE D allele bearers may have a higher risk of myocardial infarction,1 sudden death,6 and in-stent restenosis,7,8 although these results have not been consistently confirmed in larger studies.9,10 Recent studies with ACE inhibitors have reinforced the hypothesis that the ACE enzyme may be a key enzyme in various aspects of coronary artery disease.11,12 No data are available concerning the long term clinical outcome after percutaneous coronary intervention (PCI) of patients bearing the D allele. The potential role of a genetic variant of the ACE gene as a pathogenetic factor in the occurrence of major adverse cardiac events (MACE) after PCI is of particular interest given the large number of patients treated around the world by PCI and because of the ready availability of target specific pharmacological agents in the form of ACE inhibitors. Clearly the clinical implications of this finding would be more far reaching if the ACE genotype were not only associated with the occurrence of MACE after PCI but was also an identifiable and modifiable risk factor for secondary prevention. The potential importance of the finding mandates that it be based on strong epidemiological and molecular genetic standards. A few years ago we decided to conduct a large prospective study to assess the potential influence of the ACE gene polymorphism on long term clinical outcome in patients with documented coronary artery disease undergoing PCI.
METHODS
Primary end point
This prospective observational study was designed to examine the relation between ACE I/D gene polymorphism and the long term risk of MACE (death, acute myocardial infarction, unstable angina, and coronary revascularisation) in patients with symptomatic coronary artery disease treated by PCI. The primary outcome was a composite of MACE. Secondary end points were individual and combined outcomes of death, acute coronary syndromes, or any revascularisation.
Patients
From November 1996 to April 1998, 1039 consecutive white patients with symptomatic coronary artery disease who had successful PCI with stent implantation were prospectively studied. All patients gave written informed consent for the study according to our institutional ethics committee recommendations. Among the study population eight patients (0.8%) were definitively lost to clinical follow up and genotype was not available or not identified in 21 patients (2%). The remaining 1010 patients constituted the study cohort.
Data collection
The hospital records of patients included in the study were systematically reviewed by a physician who recorded demographic and clinical data including age, sex, medical history, cardiovascular risk factors, complications during hospitalisation, ECG findings, angiographic characteristics, dilatation procedures, and medications.
Genetic analysis
Blood samples were obtained at the end of the procedure after stent implantation. Genomic DNA from each patient was prepared from peripheral leucocytes by the salt precipitation method.13 The D and I alleles were identified on the basis of polymerase chain reaction (PCR) amplification of the respective fragments from intron 16 of the ACE gene as previously described.5,14 Amplification of the D allele results in a 190 bp fragment and amplification of the I allele results in a 490 bp fragment. Each sample that had the DD genotype underwent PCR amplification with a primer pair that recognises an insertion specific sequence.15 If the DNA was mistyped, a PCR product of 335 bp is present. Otherwise, no PCR product appears.
Follow up
Follow up was obtained by mailed questionnaires and scripted telephone interviews conducted by a physician. Events were verified by contacting the patient’s primary physician and reviewing medical records and death certificates. The primary clinical end point considered was a composite of hard cardiac events defined as non-fatal Q wave myocardial infarction (new pathological Q wave), unstable angina (shown by myocardial ischaemia and need for rehospitalisation), death (regardless of cause), and need for coronary revascularisation (PCI or coronary artery bypass surgery). The clinical follow up and event adjudication were realised with no knowledge of the patient’s genetic status.
Statistical analysis
Baseline characteristics of the study population are presented as counts and percentages for categorical variables and as mean (SD) for continuous variables. Differences in percentages were evaluated by the χ2 test and means by analysis of variance. Validity conditions were checked for each comparison. No striking deviation from the Hardy-Weinberg equilibrium was observed in the distribution of ACE I/D gene polymorphism (χ2 = 1.19, p > 0.50). Univariate analysis with the Kaplan-Meier product limit method and the log rank or Breslow tests was used to compare the genotype groups.
For identification of prognostic factors related to survival time and the relation between ACE I/D polymorphism and outcome, Cox’s proportional hazards models were used after adjustment for variables proved to be significant with univariate statistical analysis (p < 0.20). The relative risks are given with a 95% confidence interval. Significance was covered by an α error of 0.05. All calculations were performed with SPSS Professional Statistics 10.0.7 (SPSS Inc, Chicago, Illinois, USA).
RESULTS
Baseline characteristics of the study population
There were no differences in baseline clinical and angiographic characteristics between the three groups of genotypes (table 1). The study population was mainly treated and dilated for acute coronary syndromes (acute myocardial infarction or unstable angina) with the same proportion of patients in each group (74%, 79%, and 80% for DD, ID, and II genotypes, respectively). Diabetic patients were similarly represented in the three groups of patients. At hospital discharge, 35–44% of patients were receiving ACE inhibitors with no significant difference between the three groups of patients. Table 1 shows the distribution of several other risk factors in the three groups of genotypes. The distribution reflects the expected prevalence of recognised risk factors in patients with established coronary artery disease. No comparison between the three groups of genotypes was significant.
Table 1.
Baseline characteristics of the study population according to angiotensin converting enzyme (ACE) genotype
| ACE I/D genotype | |||
| DD (n=297) | ID (n=517) | II (n=196) | |
| Men (%) | 79 | 81 | 81 |
| Mean (SD) age (years) | 65 (11) | 64 (11) | 63 (12) |
| Indication for PCI (%) | |||
| Unstable angina | 36 | 38 | 37 |
| Acute MI | 38 | 41 | 43 |
| Stable angina | 26 | 21 | 19 |
| Risk factors (%) | |||
| Hypertension | 46 | 40 | 38 |
| Diabetes mellitus | 14 | 13 | 11 |
| Hypercholesterolaemia | 50 | 48 | 46 |
| Smoking | 51 | 49 | 51 |
| Familial history of CAD | 23 | 29 | 32 |
| BMI (kg/m2) >25 | 65 | 64 | 64 |
| Prior CABG | 9 | 9 | 8 |
| Prior MI | 30 | 32 | 31 |
| Prior PTCA | 14 | 14 | 12 |
| LVEF <40% | 23 | 23 | 18 |
BMI, body mass index; CABG, coronary artery bypass graft; CAD, coronary artery disease; I/D, insertion/deletion; LVEF, left ventricular ejection fraction; MI, myocardial infarction; PCI, percutaneous coronary intervention; PTCA, percutaneous transluminal coronary angioplasty.
Frequencies of alleles and genotypes
The frequencies of the I and D alleles were 45% and 55%, respectively. The frequencies of the DD (29%), ID (51%), and II (20%) genotypes were virtually identical to those predicted by the Hardy-Weinberg equilibrium. None of the recognised risk factors shown in table 1 differed in distribution or in mean value from those of the ACE genotype.
Clinical follow up of patients
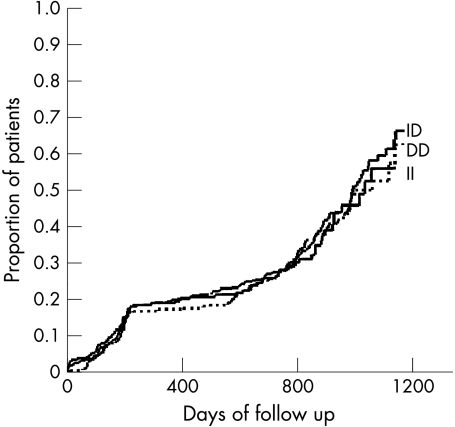
During follow up the primary clinical end point, a composite of MACE, was reached in 35%, 37%, and 34% of patients with the DD, ID, and II genotypes, respectively, with no significant difference. During a mean follow up period of 2 (1) years (maximum 4 years) a mortality of 5%, 8%, and 6% was observed in patients with the DD, ID, and II genotypes, respectively, with no significant difference between groups. Table 2 details other MACE, with no differences between genotypes. Kaplan-Meier analysis showed that patients in the three groups of genotypes had the same probability of death, revascularisation rates, or composite of MACE during the entire follow up period (fig 1). Ischaemia driven target and non-target vessel revascularisations were not influenced by the ACE genotype (results not shown). Additional analyses assuming a dominant or a recessive effect of the D allele did not change the results. Analyses were also performed with adjustment for PCI indication and ACE inhibitor treatment and in a low risk subgroup defined by a body mass index below 25 kg/m2 and with no dyslipidaemia. No association between ACE genotype and clinical outcome was observed (data not shown).
Table 2.
Incidence of the primary outcome and other outcomes according to ACE genotype
| DD (n=297) | ID (n=517) | II (n=196) | |
| Composite of MACE (%) | 35 | 37 | 34 |
| Death (%) | 5 | 8 | 6 |
| Revascularisation (%) | |||
| PTCA | 15 | 18 | 16 |
| CABG | 4 | 3 | 3 |
| Unstable angina (%) | 17 | 17 | 14 |
| AMI (%) | 1 | 1.5 | 1.5 |
MACE, major adverse cardiac events.
Figure 1.
Kaplan-Meier estimates of the composite outcome of major adverse cardiac events in relation to angiotensin converting enzyme insertion/deletion genotypes with no significant difference observed.
Multivariate analysis
Multivariate analyses of the relation between clinical data, angiographic findings, genetic markers, conventional risk factors, and the risk of MACE showed that four variables were significant: the presence of diabetes, prior percutaneous transluminal coronary angioplasty, low ejection fraction, and multivessel disease. ACE I/D genotype was not associated with deleterious prognosis either in dominant or in recessive assumptions. Table 3 shows the hazard ratios and the 95% confidence intervals for the independent prognostic variables.
Table 3.
Relative risk (RR) for composite of MACE: results of multivariate Cox regression model
| Composite of MACE | |||
| Risk factor | RR | 95% CI | p Value |
| Diabetes | 1.35 | 1.00 to 1.84 | 0.05 |
| LVEF <40% | 1.36 | 1.06 to 1.77 | 0.01 |
| Multivessel disease | 1.51 | 1.10 to 1.77 | 0.03 |
| Prior PTCA | 1.52 | 1.10 to 2.09 | 0.01 |
| DD v ID v II | |||
| DD v II | 1.02 | 0.73 to 1.42 | 0.90 |
| ID v II | 1.02 | 0.76 to 1.38 | 0.87 |
| DD + ID v II* (dominant) | 1.02 | 0.77 to 1.36 | 0.96 |
| DD v ID+II* (recessive) | 1.00 | 0.79 to 1.28 | 0.98 |
CI, confidence interval.
*Second and third Cox’s proportional hazards models.
DISCUSSION
As expected from the results of previous studies concerning patients with documented coronary artery disease, we found that several baseline variables including the presence of diabetes, the extent of coronary artery disease, and left ventricular ejection fraction were important and independent predictors of the risk of MACE after PCI.16,17 However, in this large prospective observational study we failed to observe any influence of the ACE I/D genotype on the long term risk of MACE after PCI.
Although there is evidence of a genetic predisposition for coronary artery disease, the impact of single genetic polymorphisms on the pathogenesis and natural history of ischaemic heart disease remains uncertain. The association of ACE I/D gene polymorphism with the risk of ischaemic heart disease in a case control study1 was provocative and generated great interest, although larger prospective studies in different study populations found no interaction.9,18 Because there is a strong correlation between ACE I/D genotypes and plasma ACE activity,5 and given the enormous number of studies that support the concept that blocking ACE by ACE inhibitors is beneficial for patients with congestive heart failure,19 acute myocardial infarction,20,21 and coronary artery disease,11 the concept that the DD genotype confers an increased risk of myocardial ischaemia is plausible. Indeed several biological actions of ACE may be involved in the pathogenesis of coronary artery disease: both the transformation of angiotensin I to angiotensin II, a potent vasoconstrictor, and the inactivation of bradykinin potentially result in decreased myocardial perfusion22; angiotensin II induced stimulation of plasminogen activator 1 may favour the formation of occlusive thrombi23; and angiotensin mediated promotion of smooth muscle cells growth may be involved in the progression of coronary artery disease.24 However, our findings in a large prospective study do not support the postulated role of the ACE genotype as a marker for long term risk of MACE in patients with established coronary artery disease who need PCI. Up to now, the influence of the ACE I/D gene polymorphism on the long term risk of MACE has not been studied. Some studies have reported an interaction of this polymorphism with in-stent restenosis7,8 but these results have not been confirmed by larger studies.10,25
In the present study no difference was observed in the various outcomes, including revascularisation rates, between the three genotypes. A recent report on a small series of patients suggested that ACE inhibitors given to patients with DD genotype may be associated with an unexpected deleterious effect on angiographic restenosis and with a trend for an increased risk of MACE.26 Our study, in a large patient cohort, does not support the hypothesis that an interaction between ACE inhibitors and different genotypes has any significant effect on long term clinical outcome.
Our study focused on the long term clinical outcome of a large consecutive series of patients with angiographically documented coronary artery disease. In this well selected high risk study population, with nearly 35% of patients reaching the primary study composite outcome and with a global mortality rate of 6.3% over the clinical follow up period, no influence of the ACE I/D gene polymorphism was found. Because it has been suggested that the II genotype may delay the onset of acute coronary syndromes,27 we compared the rate of MACE during the study period. However, as shown by the Kaplan-Meier estimates, the three groups of genotypes had the same probability of death, revascularisation rates, or composite of MACE during the entire follow up period.
Our study does not rule out the possibility that certain mutant alleles of the ACE gene may be associated with a predisposition to MACE after PCI; our results simply indicate that the ACE I/D polymorphism does not serve as a useful marker of a putative disease causing mutation on the ACE gene. Recently, a number of small case–control studies found associations between the prevalence of the ACE genotype and various aspects of coronary artery disease with contradictory results. It is important to recognise that these case control studies of linkage disequilibrium (association) are highly sensitive to the selection of a genetically appropriate control sample. In the prospective evaluation of a cohort of patients the sample size is also critical. It is interesting to note that our earliest preliminary results were flawed by a chance of major events in the DD patients in a small cohort of 368 patients.28 This initial interpretation was corrected once the sample was expanded.
Coronary artery disease is a multifactorial disease. Many environmental and, potentially, genetic factors contribute to the development of ischaemic heart disease. Our study of a large cohort of patients with clearly established ischaemic heart disease examined a study population coming from a single high volume centre characterised by regional environmental influences. Our study population may differ from those of multicentre studies with heterogeneous populations (for example, the World Health Organization sponsored MONICA (monitoring trends and determinants in cardiovascular disease) survey). The negative results of our study underline the concept that other important factors can overcome the overall impact of a particular single gene polymorphism, such as the ACE I/D polymorphism. However, screening for new susceptibility genetic risk factors and long term risk of MACE in ischaemic heart disease may allow us to identify in the future the specific individual genetic risk of our patients. Apart from biological explanations, statistical considerations are also important in assessing the power of a study to detect or to exclude a potential effect. From this perspective our study had 90% power (α = 0.05) to detect a 1.3-fold increase in risk of MACE associated with the DD genotype. Thus, our findings suggest that, at least in our regional population, the ACE I/D gene polymorphism is not a useful marker for assessing the long term risk of patients with ischaemic heart disease.
On the basis of the successful reduction of morbidity and mortality after myocardial infarction or in high risk patients by treatment with ACE inhibitors,11,20,29 we believe that the renin-angiotensin system probably has an important role in ischaemic heart disease.30 However, the mechanism has so far remained elusive. Our results do not exclude a pathogenic role for mutations of the ACE gene in ischaemic heart disease, but indicate for the first time that the ACE I/D polymorphism is not a useful marker for assessing the long term risk of MACE after PCI or for secondary prevention strategies.
Abbreviations
ACE, angiotensin I converting enzyme
MACE, major adverse cardiac events
MONICA, monitoring trends and determinants in cardiovascular disease
PCI, percutaneous coronary intervention
PCR, polymerase chain reaction
REFERENCES
- 1.Cambien F, Poirier O, Lecerf L, et al. Deletion polymorphism in the gene for angiotensin converting enzyme is a potent risk factor for myocardial infarction. Nature 1992;359:641–4. [DOI] [PubMed] [Google Scholar]
- 2.Samani NJ, Thompson JR, O’Toole L, et al. A meta-analysis of the association of the deletion allele of the angiotensin-converting enzyme gene with myocardial infarction. Circulation 1996;94:708–12. [DOI] [PubMed] [Google Scholar]
- 3.Oike Y, Hata A, Ogata Y, et al. Angiotensin converting enzyme as a genetic risk factor for coronary artery spasm. J Clin Invest 1995;96:2975–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Arbustini E, Grasso M, Fasani R, et al. Angiotensin converting enzyme gene deletion allele is independently and strongly associated with coronary atherosclerosis and myocardial infarction. Br Heart J 1995;74:584–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rigat B, Hubert C, Alhenc-Gelas F, et al. An insertion/deletion polymorphism in the angiotensin converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest 1990;86:1343–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Evans AE, Poirier O, Kee F, et al. Polymorphisms of the angiotensin-converting-enzyme gene in subjects who die from coronary heart disease. QJM 1994;87:211–4. [PubMed] [Google Scholar]
- 7.Ribichini F, Steffenino G, Dellavalle A, et al. Plasma activity and insertion/deletion polymorphism of angiotensin I converting enzyme: a major risk factor and a marker of risk for coronary stent restenosis. Circulation 1998;97:147–54. [DOI] [PubMed] [Google Scholar]
- 8.Amant C, Bauters C, Bodart JC, et al. D allele of the angotensin I-converting enzyme is a major risk factor for restenosis after coronary stenting. Circulation 1997;96:56–60. [DOI] [PubMed] [Google Scholar]
- 9.Linpainter K, Pfeffer MA, Kreutz R, et al. A prospective evaluation of an angiotensin-converting enzyme gene polymorphism and the risk of ischemic heart disease. N Engl J Med 1995;332:706–11. [DOI] [PubMed] [Google Scholar]
- 10.Zee RY, Fernandez-Ortiz A, Macaya C, et al. Ace D/I polymorphism and incidence of post-PTCA restenosis: a prospective, angiography-based evaluation. Hypertension 2001;37:851–5. [DOI] [PubMed] [Google Scholar]
- 11.The Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N Engl J Med 2000;342:145–53. [DOI] [PubMed] [Google Scholar]
- 12.O’Keefe JH, Wetzel M, Moe RR, et al. Should an angiotensin-converting enzyme inhibitor be standard therapy for patients with atherosclerotic disease. J Am Coll Cardiol 2001;37:1–8. [DOI] [PubMed] [Google Scholar]
- 13.Miller S, Dyker D, Poleshy G. Simple salting-out procedure for extracting DNA from human nucleated cells. Nucl Acids Res 1988;16:1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hamon M, Bauters C, Amant C, et al. Relation between the deletion polymorphism of the angiotensin converting enzyme gene and late luminal narrowing after coronary angioplasty. Circulation 1995;92:296–9. [DOI] [PubMed] [Google Scholar]
- 15.Shanmugan V, Sell KW, Saha BK. Mistyping ACE heterozygotes. PCR Methods Appl 1993;3:120–1. [DOI] [PubMed] [Google Scholar]
- 16.Van Domburg TR, Foley DP, de Feyter PJ, et al. Long-term clinical outcome after coronary balloon angioplasty. Identification of a population at low risk of recurrent events during 17 years of follow-up. Eur Heart J 2001;22:934–41. [DOI] [PubMed] [Google Scholar]
- 17.Mathew V, Farkouh M, Grill DE, et al. Clinical risk stratification correlates with the angiographic extent of coronary artery disease in unstable angina. J Am Coll Cardiol 2001;37:2053–8. [DOI] [PubMed] [Google Scholar]
- 18.Keavney B, Mc Kenzie C, Parish S, et al. Large-scale test of hypothesised associations between the angiotensin-converting-enzyme insertion/deletion polymorphism and myocardial infarction in about 5000 cases and 6000 controls. International studies of infarct survival (ISIS). Lancet 2000;355:434–42. [DOI] [PubMed] [Google Scholar]
- 19.Anon. Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. The SOLVD Investigators. N Engl J Med 1992;327:685–91. [DOI] [PubMed] [Google Scholar]
- 20.Pfeffer MA, Braunwald E, Moye LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE investigators. N Engl J Med 1992;327:669–77. [DOI] [PubMed] [Google Scholar]
- 21.Ambrosioni E, Borghi C, Magnani B. The effect of the angiotensin-converting-enzyme inhibitor zofenopril on mortality and morbidity after anterior myocardial infarction. The survival of myocardial infarction long-term evaluation (SMILE) study investigators. N Engl J Med 1995;332:80–5. [DOI] [PubMed] [Google Scholar]
- 22.Ehlers MRW, Riordan JF. Angiotensin converting enzyme: new concepts concerning its biological role. Biochemistry 1989;28:5311–8. [DOI] [PubMed] [Google Scholar]
- 23.Ridker PM, Gaboury CL, Conlin PR, et al. Stimulation of plasminogen activator inhibitor in vivo by infusion of angiotensin II: evidence of a potential interaction between the renin-angiotensin system and fibrinolytic function. Circulation 1993;87:1969–73. [DOI] [PubMed] [Google Scholar]
- 24.Daemen MJ, Lombardi DM, Bosman FT, et al. Angiotensin II induces smooth muscle cell growth in the normal and injured rat arterial wall. Circ Res 1991;68:450–6. [DOI] [PubMed] [Google Scholar]
- 25.Koch W, Kastrati A, Mehilli J, et al. Insertion/deletion polymorphism of the angiotensin I-converting enzyme gene is not associated with restenosis after coronary stent placement. Circulation 2000;102:197–202. [DOI] [PubMed] [Google Scholar]
- 26.Meurice T, Bauters C, Hermant X, et al. Effect of ACE inhibitors on angiographic restenosis after coronary stenting (PARIS): a randomised, double-blind, placebo-controlled trial. Lancet 2001;357:1321–4. [DOI] [PubMed] [Google Scholar]
- 27.Iwai N, Tamaki S, Ohmichi N, et al. The II genotype of the angiotensin converting enzyme gene delays the onset of acute coronary syndromes. Arterioscl Thromb Vasc Biol 1997;17:1730–3. [DOI] [PubMed] [Google Scholar]
- 28.Denizet A, Fradin S, Filippi-Codaccioni E, et al. The insertion/deletion ACE gene polymorphism and long term clinical outcome after coronary stenting [abstract]. Am J Cardiol 2000;86(suppl 8A):86i.10867099 [Google Scholar]
- 29.Kober L, Torp-Pedersen C, Carlsen JE, et al. A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril cardiac evaluation (TRACE) study group. N Engl J Med 1995;333: 1670–6. [DOI] [PubMed] [Google Scholar]
- 30.Dzau VJ. Theodore Cooper Lecture: tissue angiotensin and pathobiology of vascular disease: a unifying hypothesis. Hypertension 2001;37: 1047–52. [DOI] [PubMed] [Google Scholar]