Abstract
Lesion of the “vestibular cortex” in the human posterior insula leads to a tilted perception of visual vertical but not to tilted body posture and loss of lateral balance. However, some stroke patients show the reverse pattern. Although their processing of visual and vestibular inputs for orientation perception of the visual world is undisturbed, they push away actively from the ipsilesional side (the side of lesion location), leading to a contraversive tilt of the body (tilt toward the side opposite to the lesion) and falling to that side. Recently, the origin of contraversive pushing was identified as an altered perception of the body's orientation in relation to gravity. These patients experience their body as oriented “upright” when actually tilted enormously to the ipsilesional side (18° on average). The findings argued for a separate pathway in humans for sensing body orientation in relation to gravity apart from the one projecting to the vestibular cortex. The present study aimed at identifying this brain area. The infarcted brain regions of 23 consecutively admitted patients with severe contraversive pushing were projected onto a template MRI scan, which had been normalized to Talairach space. The overlapping area of these infarctions centered on the posterolateral thalamus. Our finding necessitates reinterpretation of this area as being only a “relay structure” of the vestibular pathway on its way from the brainstem to the vestibular cortex. The ventral posterior and lateral posterior nuclei of the posterolateral thalamus (and probably its cortical projections) rather seem to be fundamentally involved in the neural representation of a second graviceptive system in humans decisive for our control of upright body posture.
Keywords: pusher syndrome, thalamus, gravity, vestibular, spatial neglect
Whereas most patients with hemiparesis have good trunk balance soon after the stroke, some patients may lose lateral balance and fall toward the paralyzed side even when sitting (1–4). The convergent observation in these studies was that such patients begin to list toward the hemiplegic side in an upright (sitting) position when the assistance given to prevent falling was withdrawn. This behavior has been termed the “listing phenomenon” (3).
It was Davies (5) who discovered that there are hemiplegic patients who exhibit the even more peculiar behavior of using the nonaffected arm or leg to push away actively from the nonparalyzed side. Without assistance, this contraversive pushing (toward the side opposite to the lesion) leads to loss of postural balance and falling toward the paralyzed side. When sitting or standing, these patients actively lean toward the hemiparetic side and resist any attempt to correct their tilted body posture. They use the nonparetic arm to resist actively attempts of passive correction toward the earth-vertical upright orientation and report the impression of lateral instability and the fear of falling toward the nonparalyzed side. In contrast, these patients show no fear when their active pushing leads to an unstable, tilted body position toward the contralesional side (the side contralateral to the lesion). Davies (5) termed this behavior the “pusher syndrome.” A systematic investigation of her observation in a large sample of acute stroke patients with hemiparesis (6) confirmed the existence of contraversive pushing. The authors (6) found the disorder in 10.4% of a large sample of 327 acute stroke patients with hemiparesis admitted in a 1-year period from a well-defined catchment area.
Recently, we identified the origin of contraversive pushing (7). Our study uncovered that the deficit is caused by an altered perception of the body's orientation in relation to gravity. With occluded eyes, subjects were rotated in the frontal (roll) plane sitting on a motor-driven chair. After a random offset, subjects were required to indicate when they reached upright body orientation. On average, pusher patients experienced their body as oriented upright when actually tilted 18° to the ipsilesional side (the side of lesion location). Surprisingly, these patients showed undisturbed processing of visual and vestibular inputs determining visual vertical. Thus, in contrast to their disturbed perception of upright body posture, orientation perception of the visual world was unaffected. This dissociation argued for a separate pathway in humans for sensing the orientation of gravity apart from the well-known for orientation perception of the visual world. The cortical representation of the latter system, the so-called vestibular cortex, has recently been identified in humans (8). The region responsible for vestibular function in the roll plane is found in the posterior insula, probably homologous to the parieto-insular vestibular cortex (PIVC) in the monkey (9, 10). Patients with lesions in this area show visual-vestibular dysfunction in terms of a perceptual tilt of the visual vertical but have no tilted body posture and subsequent loss of lateral balance (8). Interestingly, pusher patients show the opposite behavior. They have a severe tilt of body posture, but no visual-vestibular dysfunction. We (7) thus assumed that contraversive pushing must result from lesion of a brain area anatomically distinct from that described by Brandt et al. (8), and that this area is basically involved in control of upright body posture. The present study aimed at identifying the neural representation of this second graviceptive system in humans.
Patients and Methods
Thirty-one acute stroke patients with severe contraversive pushing, consecutively admitted over a 2-yr period were investigated. Twenty-three of these patients had a circumscribed unilateral lesion (see below). Contraversive pushing was assessed by using the standardized “Scale for Contraversive Pushing (SCP)” (11). (A translation of this scale is given in ref. 7.) Based on Davies' criteria (5), the SCP assesses (i) symmetry of spontaneous posture while sitting and standing, (ii) the use of the arm and/or the leg to extend the area of physical contact to the ground while sitting and standing, and (iii) resistance to passive correction of posture while sitting and standing. Patients were scored as having contraversive pushing if they reached at least a total score of 1 (maximum = 2, sitting plus standing) with respect to their spontaneous posture, at least a score of 1 (maximum = 2, sitting plus standing) concerning the use of the arm and/or the leg to extend the area of physical contact, and if they showed resistance to passive correction of posture while sitting and standing. (In six patients, pushing behavior while standing could not be quantified with SCP because of a complete inability to reach a standing position at the time of the present investigation.)
Patients were classified as having spatial neglect when they showed the typical clinical behavior such as (i) a spontaneous deviation of the head and eyes toward the ipsilesional side, (ii) orienting toward the ipsilesional side when addressed from the front or the left, and (iii) ignoring of contralesionally located people or objects. In addition to the evaluation of these characteristic manifestations of spatial neglect in the spontaneous behavior in all patients, the right-brain-damaged patients were further assessed with two cancellation tasks. The star cancellation test (12) presents 56 small stars pseudorandomly positioned among large stars, letters, and short words on a 21 × 29.7 cm large sheet of paper. The task is to cross out all small stars. In the letter cancellation test (13), various letters are randomly dispersed on a sheet of paper of the same size. There are 60 target letter A's on this page (30 on either half-side). The subjects were asked to mark all of the letter A's. In both tests, an omission score of at least 25% neglected targets on the contralesional side (the side opposite to the lesion) was taken as the criterion for spatial neglect.
Brain lesions were identified by computed tomography (CT) or magnetic resonance imaging (MRI). Patients with diffuse or bilateral brain lesions, patients with tumors, as well as patients in whom CT or MRI scans revealed no manifest lesion were excluded. The lesions were mapped by using MRICRO software (www.psychology.nottingham.ac.uk/staff/crl/mricro.html). The lesions were drawn manually on slices of a template MRI scan from the Montreal Neurological Institute (www.bic.mni.mcgill.ca/cgi/icbm_view), which is based on 27 T1-weighted MRI scans of the same individual that had been normalized to Talairach space. This scan is distributed with SPM99 (www.fil.ion.bpmf.ac.uk/spm/spm99.html) and has become a popular template for normalization in functional brain imaging. For superimposing of the individual brain lesions, the same MRICRO software was used.
The neuroanatomical findings in patients with contraversive pushing were contrasted with a sample of 23 acute stroke patients admitted in the same period who did not exhibit contraversive pushing but showed the same characteristics as the pusher patients with regard to age, etiology of lesion, presence of hemiparesis, spatial neglect, and aphasia (Table 1).
Table 1.
Demographic and clinical data of the patients with right brain damage (RBD) and with left brain damage (LBD)
| RBD-pusher | RBD-controls | LBD-pusher | LBD-controls | |||
|---|---|---|---|---|---|---|
| Number | 15 | 15 | 8 | 8 | ||
| Sex | 7 f, 8 m | 9 f, 6 m | 2 f, 6 m | 2 f, 6 m | ||
| Age | Median (range) | 71 (39–81) | 65 (62–79) | 68 (38–89) | 63.5 (38–81) | |
| Etiology | 8 Infarct | 11 Infarct | 4 Infarct | 7 Infarct | ||
| 7 Hemorrhage | 4 Hemorrhage | 4 Hemorrhage | 1 Hemorrhage | |||
| SCP-posture | Sitting | Median (range) | 1 (.75–1) | 0* | 1 (.75–1) | 0* |
| Standing | Median (range) | 1* | 0 (0–.25) | 1* | 0* | |
| SCP-extension | Sitting | Median (range) | 1 (.5–1) | 0* | 1 (.75–1) | 0* |
| Standing | Median (range) | 1 (.5–1) | 0* | 1* | 0* | |
| SCP-resistance | Sitting | Median (range) | 1* | 0* | 1* | 0* |
| Standing | Median (range) | 1* | 0* | 1* | 0* | |
| Spontaneous nystagmus | % present | 0 | 0 | 13 | 0 | |
| Skew deviation | % present | 0 | 0 | 0 | 0 | |
| Vertical gaze paresis | % present | 7 | 0 | 0 | 0 | |
| Hemiparesis of | % present | 100 | 100 | 100 | 100 | |
| Contralesional side† | Arm | Median (range) | 0 (0–3) | 2 (0–4.5) | 0 (0–3.5) | .75 (0–4.5) |
| Leg | Median (range) | 2.5 (0–4.5) | 3.5 (0–4.5) | 3 (0–4) | 1.5 (0–4) | |
| Babinski | % positive | 93 | 60 | 88 | 75 | |
| Somatosensory deficit | % t.n.p. | 0 | 13 | 38 | 75 | |
| Of contralesional side | Arm | % Present | 80 | 73 | 62 | 25 |
| (touch) | Leg | % Present | 80 | 67 | 62 | 25 |
| Aphasia | % Present | 7 | 0 | 100 | 100 | |
| Spatial neglect | % Present | 80 | 80 | 0 | 13 | |
f, female; m, male; SCP, scale for contraversive pushing (11); t.n.p., testing not possible.
No variation in data.
Paresis was scored with the usual clinical ordinal scale, where 0 stands for no trace of movement and 5 for normal movement.
Results
Demographics and Clinical Findings.
Because the SCP (11) does not need verbal instructions, we had no drop-outs of pusher patients because of communicational problems (i.e., no drop-out of patients with aphasia after left hemispheric infarcts). In 65% of our sample of 23 pusher patients, we found a unilateral right-sided lesion; 35% had a unilateral left-sided lesion. All left- and right-brain-damaged pusher patients suffered from severe paresis of the upper and lower contralesional extremities (Table 1). Somatosensory deficits on the contralesional side were present in 80% of the right-brain-damaged pusher patients and in 62% of those with left brain damage. Eighty percent of the right-brain-damaged pusher patients exhibited additional spatial neglect. Neglect was severe, with a mean omission score in the letter cancellation test of 93% (SD 10.7) and of 87% (SD 25.5) in the star cancellation test. All left-brain-damaged pusher patients also suffered from aphasia. None of this latter group showed any behavioral symptoms characteristic for additional spatial neglect (see above for criteria). Contralesional visual field defects (loss of at least one quadrant) were present in only 7% of the right-brain-damaged pusher patients. Due to aphasia, visual fields could not be tested in four of our eight left-brain-damaged patients. Of the remaining four patients, only one had a visual field defect.
Lesion Location.
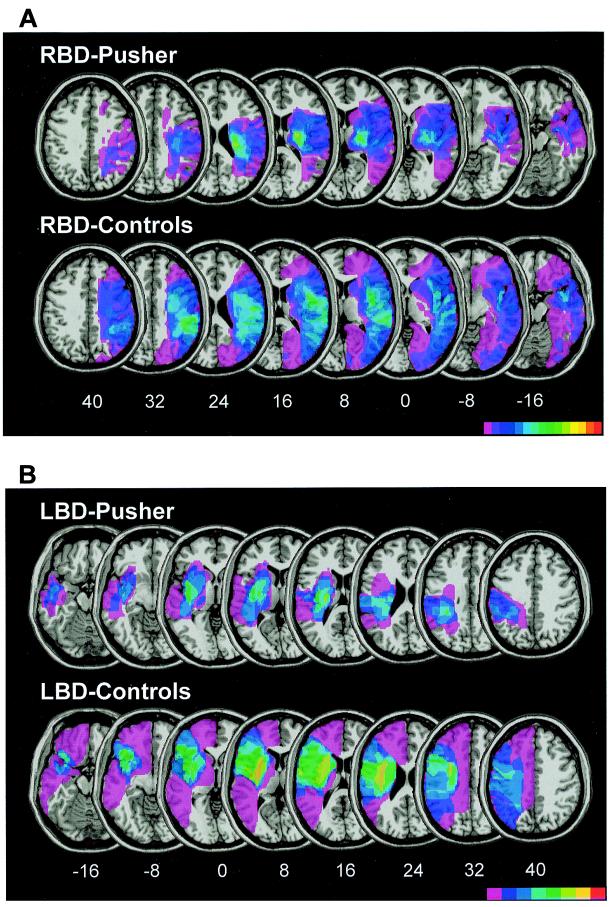
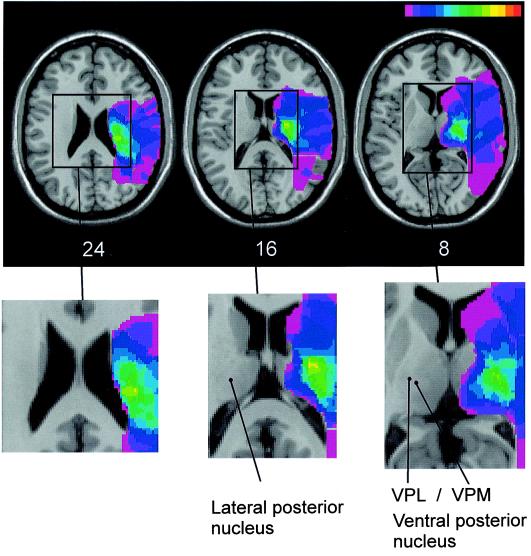
For lesion analysis, the patients with contraversive pushing were contrasted with a sample of patients without pushing behavior but with comparable demographic and clinical data (see Table 1). Fig. 1A illustrates the superimposed lesion plots of the 15 right-brain-damaged patients with contraversive pushing and their 15 controls. The center of overlap was defined as those voxels in the template MRI that were lesioned concurrently in 8 or more of the 15 pusher patients (green-yellow area in Figs. 1 and 2). Fig. 2 gives an exploded view of this center; Table 2 documents its boundaries in Talairach space (14). In clear contrast to controls, the center of lesion overlap in the pusher patients was located in the ventral posterior and lateral posterior nuclei of the posterolateral thalamus. Laterally and dorsally it extended into the posterior crus of the internal capsule, dorsally also slightly into the corpus of the caudate nucleus.
Figure 1.
(A) Overlay plots of the infarcted areas of 15 patients with contraversive pushing after unilateral right hemispheric lesions and their 15 controls without pushing. Using MRICRO software (www.psychology.nottingham.ac.uk/staff/crl/mricro.html), the lesions were mapped on slices of a template MRI scan from the Montreal Neurological Institute (www.bic.mni. mcgill.ca/cgi/icbm_view), which was normalized to Talairach space (14). The z-coordinates of each transverse section are given. The number of overlapping lesions is illustrated by different colors coding increasing frequencies from violet (n = 1) to red (n = 15). RBD, right-brain-damaged. (B) Overlay plots of the infarcted areas of 8 patients with contraversive pushing after unilateral left hemispheric lesions and their 8 controls without pushing. LBD, left-brain-damaged.
Figure 2.
Exploded view of the center of lesion overlap in the 15 right brain-damaged patients with contraversive pushing. The same conventions are used as for Fig. 1. VPL, ventral posterolateral nucleus; VPM, ventral posteromedial nucleus.
Table 2.
Talairach coordinates (mm) of overlap area in the right-brain-damaged pusher patients
| Borders | x | y | Anatomical structure | |
|---|---|---|---|---|
| z = 0 | ||||
| Anterior | 20 | −13 | Internal capsule, posterior crus | |
| Posterior | 26 | −23 | Internal capsule, posterior crus | |
| z = 8 | ||||
| Anterior | 17 | −15 | Thalamus, VPL* | |
| Medial | 15 | −19 | Thalamus, VPM† | |
| Lateral | 24 | −19 | Internal capsule, posterior crus | |
| Posterior | 26 | −25 | Internal capsule, posterior crus | |
| z = 16 | ||||
| Anterior | 17 | −11 | Thalamus, LP‡ | |
| Medial | 17 | −19 | Thalamus, LP‡ | |
| Lateral | 26 | −15 | External capsule | |
| Posterior | 24 | −29 | Internal capsule, posterior crus | |
| z = 24 | ||||
| Anterior | 20 | −15 | Periventricular white matter | |
| Medial | 20 | −27 | Fasciculus occipito-frontalis | |
| Lateral | 27 | −25 | Periventricular white matter | |
| Posterior | 27 | −36 | Periventricular white matter |
For each transverse section, the borders of the overlap area are given. VPL, ventral posterolateral nucleus; VPM, ventral posteromedial nucleus; LP, lateral posterior nucleus. In the human nomenclature of Hassler (27).
Nucleus ventrocaudalis externus.
Nucleus ventrocaudalis internus.
Nucleus dorso-intermedius externus.
Fig. 1B illustrates the superimposed lesion plots of the eight left brain-damaged patients with contraversive pushing and their eight controls. Because the number of pusher patients with left-sided lesions was low (half of the sample size of those with right-sided lesions), their overlay plot must be regarded with caution. Nevertheless, a tendency of lesion concentration in the same area as in the right brain-damaged patients with contraversive pushing was evident.
Discussion
The study of contraversive pushing by Pedersen et al. (6) in a large sample of acute stroke patients with hemiparesis revealed no evidence for a regular combination of contraversive pushing with other neuropsychological deficits such as spatial neglect, anosognosia, aphasia, or apraxia. Moreover, they found the disturbance equally frequent with left and with right brain damage. In contrast, we found an asymmetry between right- and left-sided lesion location in the present sample of 23 consecutively admitted patients with severe contraversive pushing. Sixty-five percent of our sample suffered from a right hemispheric lesion. Eighty percent of these right brain-damaged pusher patients exhibited spatial neglect, but neglect was not present in any of the pusher patients with left-sided lesions. However, all subjects with left-sided lesions suffered from aphasia.
Because of the differences in the study design, a direct comparison of our demographic and clinical findings with those obtained by Pedersen et al. (6) is difficult. Nevertheless, it is noteworthy that we found a strong association between contraversive pushing and the additional presence of spatial neglect (in right-brain-damaged patients) and aphasia (in left-brain-damaged patients). However, like the findings of Pedersen et al. (6), our data strongly argue against the assumption that spatial neglect might cause contraversive pushing. Twenty percent of the pusher patients with right-sided brain lesions exhibited no neglect, nor did all pusher patients with left-sided lesions.
We (7) discovered that the disturbance underlying contraversive pushing is an altered perception of the body's orientation in relation to gravity. Our results argued for a separate pathway in humans for sensing the orientation of gravity apart from the one for orientation perception of the visual world. How could one imagine that two such graviceptive systems are implemented in the brain? One possible assumption is that both systems rely on the same peripheral (visual, vestibular, eye- and neck-proprioceptive) input sources but that this same afferent input is projected to two anatomically separate neural networks that process the input in different ways. Whereas the first system processes the orientation of the visual world and the head to the vertical, the second system processes the posture of the trunk. An alternative assumption would be that both graviceptive systems rely on (at least in part) different input sources. In fact, the latter has been suggested by Mittelstaedt (15, 16). He proposed that the orientation of the visual world and the head to the vertical is exclusively perceived through our (visual, vestibular, and proprioceptive) sense organs in the head and the neck, whereas the posture of the trunk is mainly perceived through sense organs in the trunk. Such a truncal graviceptive system is known to exist in pigeons (17–19). He assumed the afferent input from the kidneys and the information through the inertia of a mass in the body as possible candidates for such truncal graviceptors in humans. Interestingly, the assumption of such a separate graviceptive system is in accordance with the observations of a recent study recording from the vestibular nuclei of cats (20). The animals had undergone a combined bilateral labyrinthectomy and vestibular neurectomy. While recording, neck movements were eliminated, and, in two cases, the C1−C3 dorsal roots were cut bilaterally in addition. Despite this complete removal of vestibular and neck proprioceptive input, the authors (20) still found a modulation by postural tilt in one third of the neurons examined in the “vestibular” nuclei.
The above possibilities of implementation of the two graviceptive systems in the brain must be further investigated in the future. Nevertheless, the present findings unequivocally demonstrate the anatomical correlate of contraversive pushing. The overlap area of infarctions in 23 consecutive patients with severe contraversive pushing very clearly centered on the ventral posterior and lateral posterior nuclei of the posterolateral thalamus. We propose that this structure is fundamentally involved in our control of upright body posture and is part of the neural representation of the human second pathway for sensing the orientation of gravity.
The center of overlap found in the superimposed lesion plots extended from the posterolateral thalamus into the posterior crus of the internal capsule, which explains the severe hemiparesis present in all of our pusher patients. When Pedersen et al. (6) compared lesion location in their hemiparetic patients with and without contraversive pushing, they found a difference between both groups only for the posterior crus of the internal capsule. For lesion analysis, they divided the entire brain into only eight different sections and calculated the frequency of their involvement in the individual computed tomography lesions. Because such a procedure does not allow for high resolution of lesion location, their failure to identify the neural substrate of contraversive pushing can easily be explained.
Does “contraversive pushing” describe the same behavioral disorder that Masdeu and Gorelick (21) previously had termed “thalamic astasia”? In 15 patients with unilateral, predominantly posterolateral thalamic lesions, they (21) found an inability to stand unsupported. Eight of the patients “could not even sit up by themselves and had marked truncal instability, falling backward or to the affected side from a sitting position when left without support. Typically, when asked to sit up, rather than using the axial muscles, these patients would grasp the side rail of the bed with the unaffected hand or with both hands to pull themselves up” (ref. 21, p. 597). This detailed description of the typical characteristics of thalamic astasia allows us to conclude that Masdeu and Gorelick observed a behavior different from contraversive pushing. When patients with contraversive pushing are asked to sit up, they never grasp something “with the unaffected hand or with both hands to pull themselves up.” Pusher patients do exactly the opposite. When at rest and also when asked to sit up, pusher patients extend the unaffected arm and use it to push away actively from the nonparetic side. Moreover, they use the nonparetic arm to resist actively against attempts of passive correction toward the earth-vertical upright orientation. A further difference between patients with contraversive pushing and those patients described by Masdeu and Gorelick (21) is the presence of hemiparesis. Whereas all of our 23 consecutively admitted patients with contraversive pushing also suffered from severe paresis of the contralateral arm and leg, those patients of Masdeu and Gorelick had only very mild or no motor weakness.
Unfortunately, the origin of astasia in the patients of Masdeu and Gorelick (21) is not known. It would have been interesting to know the perception of the subjective visual vertical (SVV) and subjective postural vertical in these patients. Dieterich and Brandt (22) recently investigated the SVV in 35 patients with acute thalamic infarctions (14 paramedian, 17 posterolateral, and 4 anterior polar). In 64% of the patients with paramedian and in 69% of those with posterolateral infarcts, the authors found a tilt of the SVV. Whereas the patients with paramedian infarcts showed a contraversive SVV tilt of 11° (together with a complete contraversive ocular tilt reaction, i.e., lateral head tilt, skew deviation, and ocular torsion), the SVV tilt of the patients with posterolateral lesions was not direction specific. In seven of the cases with posterolateral lesions, the tilt was ipsiversive (2.4°), and in four patients contraversive (4°). On the basis of their results, Dieterich and Brandt (22) speculated that the instability of upright posture in the patients with thalamic astasia described by Masdeu and Gorelick (21) might have been due to vestibular dysfunction as expressed by either a tilt of the SVV alone or by an ocular tilt reaction, i.e., the triad of head tilt, skew deviation, and ocular tortion, together with the associated tilt of the SVV.
Unfortunately, we cannot contribute to further clarification of this assumption. Like those patients of Masdeu and Gorelik (21) and 17 of those investigated by Dieterich and Brandt (22), our patients with contraversive pushing also showed an overlap of lesion location in the posterolateral thalamus. However, our present and recent (7) results demonstrate that patients with contraversive pushing (i) are clinically not identical with those described by Masdeu and Gorelik (21) (they exhibit the opposite motor behavior), and (ii) are obviously different from those 69% of the 17 patients who showed a tilt of the SVV after posterolateral thalamic lesion (22) [pusher patients have a tilted subjective postural vertical but undisturbed SVV (7)]. Masdeu and Gorelik (21), as well as Dieterich and Brandt (22), did not test the subjective postural vertical in their patients and also did not investigate for possible pushing behavior in a standardized manner. Thus, it must remain the issue of future studies to clarify whether (slightly differing) lesion locations in the posterolateral thalamus may indeed result in three different clinical syndromes because of disturbance of three different functional systems represented in the posterolateral thalamus, or (more likely) whether some of these inconsistencies are simply due to the different variables measured in the patients of these studies.
The active pushing away with the nonparetic extremities distinguishes pusher patients from those patients with lateropulsion in Wallenberg's syndrome (23, 24). Dieterich and Brandt (24) investigated 36 such patients with acute unilateral medullary brainstem infarctions. Contrary to patients with contraversive pushing (7), they found an ipsiversive tilt of the SVV in 94% of the patients (ranging from 2.7° to 53.3°) and a corresponding lateropulsion (defined as a tendency to fall sideways) with an ipsiversive deviation of the center of gravity (determined by means of posturography). As in those patients with thalamic astasia (21) and in those with infarctions of the paramedian and posterolateral thalamus (22), Dieterich and Brandt (24) did not find active pushing away and resistance against passive correction in the patients with Wallenberg's syndrome. Moreover, the tendency of the latter patients to fall sideways is to the opposite side (ipsiversively), compared with patients with pushing (contraversively).
In conclusion, the present data suggest that the posterolateral thalamus must be regarded as a structure basically involved in our control of upright body posture. From neurophysiological work in monkeys, we know that some nuclei in this portion of the thalamus are sensitive to vestibular stimulation (25, 26). These nuclei [nuclei ventrointermedii, nucleus zentrolateralis intermedius, nucleus ventrocaudalis externus, and nucleus dorso-intermedius externus and internus (human nomenclature of Hassler, ref. 27)] therefore had been regarded as vestibular relay structures to the cortex (e.g., ref. 8). In line with this idea was the observation that 69% of patients with acute infarctions of this area showed a tilt of the subjective visual vertical (22) as did the patients with lesions of the central vestibular system in the brainstem (24) and cortex (8). Further, we know that electrical stimulation in the nuclei ventrointermedii and the nucleus zentrolateralis intermedius elicits rotation or spinning of the eyes, head, or body in humans (27, 28).
The present data teach us that the posterolateral thalamus obviously does not serve only as a simple relay structure of the vestibular pathway on its way to the cortex. It would also be too narrow to regard it as the relay structure of various sensory pathways (from the body and the head) to the primary sensory cortex (29). The ventral posterior and lateral posterior nuclei of the posterolateral thalamus rather seem to be fundamentally involved in our control of upright body posture. Patients suffering from severe contraversive pushing showed a clear overlap of their infarctions in this portion of the thalamus. It is obvious that this structure is anatomically distinct from the vestibular cortex identified by Brandt and coworkers (8) in the posterior insula. Also, the clinical findings in patients with such lesions are different. Whereas lesion of the vestibular cortex in humans leads to a tilt of the subjective visual vertical but not to contraversive pushing and falling to that side (8), a lesion of the second system induces the opposite pattern. Those patients with contraversive pushing show a normal perception of visual vertical but a severe tilt of perceived body verticality in the frontal plane, with pushing and subsequent falling to that side (7). Thus, both graviceptive systems obviously not only are anatomically distinct but also seem to process afferent sensory information from peripheral input sources differently.
Future studies have to investigate the possible role of diaschesis (30) induced by the thalamic lesion of pusher patients. Thalamocortical axons arising in the ventral posterolateral and ventral posteromedial nuclei (cf. Table 2) project to the primary somatosensory cortex in the postcentral gyrus (areas 3a, 3b, 1, and 2), to the secondary somatosensory cortex in the parietal operculum, and to the insula (29, 31). The lateral posterior nucleus (LP; cf. Table 2) projects to the posterior parts of areas 5 and 7 of the superior and inferior parietal lobules (31). The thalamic lesion found in the patients with contraversive pushing might lead to additional functional or metabolic abnormalities in some of these structurally intact regions of the cortex. In addition to structural imaging in lesioned patients, functional imaging and other metabolic measures might help to assess whether there are such additional critical substrates in the cortex.
Acknowledgments
We are grateful to Doris Brötz, Annette Götz, and the team of physiotherapists for their valuable support with the investigation of the patients. We also thank Ute Amann for her help in the tomography archives, Chris Rorden for his advice when using mricro software, and one of the referees for the thoughtful comments on an earlier version of the manuscript. This work was supported by Grant KA 1258/2-1 from the Deutsche Forschungsgemeinschaft awarded to H.-O.K.
Abbreviations
- SCP
Scale for Contraversive Pushing
- SVV
subjective visual vertical
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.240279997.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.240279997
References
- 1.Beevor C E. Br. Med. J. 1909. April 10th, 881–885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Perry J. Clin Orthop. 1969;63:23–31. [PubMed] [Google Scholar]
- 3.Brunnstrom S. Movement Therapy in Hemiplegia: A Neurophysiological Approach. New York: Harper & Row; 1970. [Google Scholar]
- 4.Bohannon R W, Cook A C, Larkin P A, Dubuc W E, Smith M B, Horton M G, Hypes C. Neurol Rep. 1986;10:43–44. [Google Scholar]
- 5.Davies P M. Steps to Follow: A Guide to the Treatment of Adult Hemiplegia. New York: Springer; 1985. [Google Scholar]
- 6.Pedersen P M, Wandel A, Jorgensen H S, Nakayama H, Raaschou H O, Olsen T S. Arch Phys Med Rehabil. 1996;77:25–28. doi: 10.1016/s0003-9993(96)90215-4. [DOI] [PubMed] [Google Scholar]
- 7.Karnath, H.-O., Ferber, S. & Dichgans, J. (2000) Neurology55, in press. [DOI] [PubMed]
- 8.Brandt T, Dieterich M, Danek A. Ann Neurol. 1994;35:403–412. doi: 10.1002/ana.410350406. [DOI] [PubMed] [Google Scholar]
- 9.Grüsser O-J, Pause M, Schreiter U. J Physiol (London) 1990;430:537–557. doi: 10.1113/jphysiol.1990.sp018306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grüsser O-J, Pause M, Schreiter U. J Physiol (London) 1990;430:559–583. doi: 10.1113/jphysiol.1990.sp018307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karnath, H.-O., Brötz, D. & Götz, A. (2001) Nervenarzt, 72, in press. [DOI] [PubMed]
- 12.Halligan P W, Marshall J C, Wade D T. Lancet. 1989;14:908–911. doi: 10.1016/s0140-6736(89)91561-4. [DOI] [PubMed] [Google Scholar]
- 13.Weintraub S, Mesulam M-M. In: Principles of Behavioral Neurology. Mesulam M-M, editor. Philadelphia: Davis; 1985. pp. 71–124. [Google Scholar]
- 14.Talairach J, Tournoux P. Co-Planar Stereotaxic Atlas of the Human Brain: 3-Dimensional Proportional System—An Approach to Cerebral Imaging. New York: Thieme; 1988. [Google Scholar]
- 15.Mittelstaedt H. Ann N Y Acad Sci. 1992;656:124–139. doi: 10.1111/j.1749-6632.1992.tb25204.x. [DOI] [PubMed] [Google Scholar]
- 16.Mittelstaedt H. Neurosci Biobehav Rev. 1998;22:473–478. doi: 10.1016/s0149-7634(97)00032-8. [DOI] [PubMed] [Google Scholar]
- 17.Mittelstaedt H. Symp Soc Exp Biol. 1964;18:365–385. [PubMed] [Google Scholar]
- 18.Biederman-Thorson M, Thorson J. J Comp Physiol. 1973;83:103–122. [Google Scholar]
- 19.Delius J D, Vollrath F. J Comp Physiol. 1973;83:123–143. [Google Scholar]
- 20.Yates B J, Jian B J, Cotter L A, Cass S P. Exp Brain Res. 2000;130:151–158. doi: 10.1007/s002219900238. [DOI] [PubMed] [Google Scholar]
- 21.Masdeu J C, Gorelick P B. Ann Neurol. 1988;23:596–603. doi: 10.1002/ana.410230612. [DOI] [PubMed] [Google Scholar]
- 22.Dieterich M, Brandt T. Neurology. 1993;43:1732–1740. doi: 10.1212/wnl.43.9.1732. [DOI] [PubMed] [Google Scholar]
- 23.Bjerver K, Silfverskiöld B P. Acta Neurol Scand. 1968;44:91–100. doi: 10.1111/j.1600-0404.1968.tb07446.x. [DOI] [PubMed] [Google Scholar]
- 24.Dieterich M, Brandt T. Ann Neurol. 1992;31:399–408. doi: 10.1002/ana.410310409. [DOI] [PubMed] [Google Scholar]
- 25.Deeke L, Schwarz D W F, Fredrickson J M. Exp Brain Res. 1974;20:88–100. doi: 10.1007/BF00239019. [DOI] [PubMed] [Google Scholar]
- 26.Büttner U, Henn V. Brain Res. 1976;103:127–132. doi: 10.1016/0006-8993(76)90692-2. [DOI] [PubMed] [Google Scholar]
- 27.Schaltenbrand G, Wahren W. Atlas for Stereotaxy of the Human Brain: Architectonic Organisation of the Thalamic Nuclei by Rolf Hassler. Stuttgart: Thieme; 1977. [Google Scholar]
- 28.Tasker R R, Organ L W, Hawrylyshyn P A. The Thalamus and Midbrain of Man: A Physiological Atlas Using Electrical Stimulation. Springfield: Thomas; 1982. [Google Scholar]
- 29.Engelborghs S, Marien P, Martin J-J, De Deyn P P. Acta Neurol Belg. 1998;98:252–265. [PubMed] [Google Scholar]
- 30.Feeney D M, Baron J-C. Stroke. 1986;17:817–830. doi: 10.1161/01.str.17.5.817. [DOI] [PubMed] [Google Scholar]
- 31.Jones E G. The Thalamus. New York: Plenum; 1985. [Google Scholar]