Abstract
Background: Cardiac microangiopathy may be involved in the development of heart failure in diabetes mellitus.
Objective: To evaluate the effect of angiotensin II receptor blockade on cardiac function and fine structures in diabetes.
Methods: Male Otsuka Long-Evans Tokushima Fatty (OLETF) rats (n = 30), a model of spontaneously developing diabetes mellitus, and their diabetes resistant counterparts (n = 20) were used. At 30 weeks of age, when the OLETF rats show hyperglycaemic obesity with hyperinsulinaemia, the animals were divided into two groups and given candesartan, an angiotensin II receptor blocker, 0.2 mg/kg/day, or vehicle for six weeks. Capillary density was evaluated in the left ventricular myocardium by electron microscopy, matrix metalloproteinase (MMP) activity by zymography, and cytokines by reverse transcriptase polymerase chain reaction.
Results: Compared with the control rats, the OLETF rats at 36 weeks showed decreased peak negative dP/dt (mean (SD): 2350 (250) v 3492 (286) mm Hg/s) and increased cardiomyocyte diameter (24.3 (0.6) v 18.9 (0.6) μm) (both p < 0.05). Thickening of the capillary basement membranes and decreased capillary density were observed. Angiotensin receptor blockade improved almost all the haemodynamic variables, and the histological findings became similar to those of the controls. Angiotensin receptor blockade also activated MMP-2 and prevented an increase of inflammatory cytokines, especially interleukin (IL)-1β and IL-6, in the diabetic heart.
Conclusions: Angiotensin II receptor blockade preserved left ventricular diastolic function. It was also potent at improving cardiomyocyte diameter and the thickening of the capillary basement membrane, increasing MMP-2 activity, and decreasing inflammatory cytokines. With all these changes, candesartan could contribute to cardioprotection in diabetes mellitus.
Keywords: angiotensin II receptor blocker, diabetes mellitus, heart, remodelling
Analysis of the Framingham data shows that the risk of developing heart failure increases substantially among diabetic patients.1 This suggests that metabolic abnormalities associated with diabetes may contribute to cardiomyocyte dysfunction.2 Microangiopathy in the myocardium could be involved in the development of heart failure, but the precise pathophysiological mechanisms have not been elucidated.
The extracellular matrix appears to be important for diastolic function, and matrix metalloproteinases (MMP) play a role in regulating the extracellular matrix. To date, 18 vertebrate MMP and human homologues have been identified.3 Gelatinases (MMP 2 and 9) are distinguished from other MMP by a fibronectin type II repeat insert that mediates their particular ability to bind to and degrade denatured collagens. Recently, MMP activity was reported to play a critical role in mediating angiogenesis in ischaemic tissue.4
In the treatment of patients with heart failure, inhibition of neurohormonal systems—such as the renin–angiotensin system and the sympathetic nervous system—has been shown to improve the prognosis, suggesting an important role for neurohormonal factors in the pathogenesis of heart failure.5,6 The renin–angiotensin system is upregulated in diabetes mellitus, and locally produced angiotensin II may lead to oxidative damage.7 Thus angiotensin II receptor blockade may work as a cardioprotective mechanism by blocking the cardiac renin–angiotensin system in diabetes.
The Otsuka Long-Evans Tokushima Fatty (OLETF) rat, a model of the spontaneous development of type II diabetes, develops hyperglycaemic obesity with hyperinsulinaemia and insulin resistance similar to the human condition.8,9 In the present study, we investigated cardiac function and fine structure in OLTEF diabetic rats, focusing on the capillaries and on MMP activity, and we evaluated the effect of angiotensin receptor blockade on left ventricular remodelling in the diabetic heart.
METHODS
Subjects
Male OLETF rats (n = 30) and, as controls, their diabetes resistant counterparts, LETO (Long-Evans Tokushima Otsuka) rats (n = 20) were used.10 All animals were kept in a specific pathogen-free facility under controlled temperature (20–24°C) and humidity (40–70%), with a 12 hour lighting cycle, and were given free access to standard laboratory rat chow (MF, Oriental Yeast, Tokyo, Japan) and tap water. All procedures were in accordance with institutional guidelines for animal research.
Experimental protocol
At the age of 30 weeks, when diabetes mellitus had been established in the OLETF rats,10,11 each group of OLETF or LETO rats was divided into two subgroups, with or without angiotensin II receptor blockade (ARB). In the treated groups (OLETF+ARB and LETO+ARB), candesartan 0.2 mg/kg/day was given intraperitoneally by an osmotic minipump (Alzet model 2002, Alza Co, Palo Alto, California, USA) for six weeks; in the control groups, vehicle solution in the same volume was given instead of candesartan. The type of minipump used works for two weeks, so it was renewed three times during the experimental period.
In all animals, cardiac catheterisation was done under anaesthesia with sodium pentobarbitone (pentobarbital) (50 mg/kg, intraperitoneally). A 2 French microtip catheter (model PR-249, Millar Instruments, Houston, Texas, USA) was introduced through the right carotid artery into the ascending aorta and left ventricular cavity to measure aortic and left ventricular systolic and end diastolic pressures, dP/dt, and heart rate. Blood samples were taken for measurement of thiobarbituric acid reactive substances (TBARS). A part of the left ventricle was snap frozen in liquid nitrogen and stored at −80°C for the measurement of MMP activity and the cytokine gene expression assay. The rest of the left ventricular myocardium and a part of the ascending aorta were used for light and electron microscopic studies.
Measurement of TBARS
The analysis was done using a previously published method.12 In brief, 50 μl plasma was mixed with 3 μl of 0.1 M 2,6-di-tert-butyl p-hydroxy-toluene in ethanol, 50 μl of 0.2 M phosphoric acid, and 6 μl of 0.1 M thiobarbituric acid. The mixture was then incubated at 90°C for 45 minutes. The samples were cooled on ice, after which 125 μl butanol and 10 μl saturated NaCl solution were added and the mixture was vortexed for 60 seconds. After centrifugation (one minute, 12 000 ×g), 62.5 μl of the supernatant fraction was measured in a fluorometer (λ530 nm excitation, λ590 nm emission).
Light and electron microscopic study
For the light microscopic study, the specimens were fixed in 10% formaldehyde, embedded in paraffin, and cut into 4 μm thick sections. The tissue sections were stained with haematoxylin and eosin, periodic acid-Schiff, and Mallory-azan, and examined by light microscopy.
For the transmission electron microscopic study, the specimens were fixed in 4% paraformaldehyde containing 0.25% glutaraldehyde and 4.5% sucrose, and ultrathin sections obtained from the embedded blocks were examined in a Hitachi H-7000 electron microscope.13
For the scanning electron microscopic study, the specimens were fixed in 3% glutaraldehyde and macerated in sodium hydroxide solution to eliminate the cellular components, according to a cell maceration method reported elsewhere.14 In each print of scanning electron microscopy, the interstitial collagen percentage per visual field was evaluated quantitatively using the NIH Image 1.60 program (National Institutes of Health, Bethesda, Maryland, USA).11
Immunohistochemistry for TIMP-2 and HO-1
For the immunohistochemical light microscopic study, additional sections were obtained from the paraffin block. The sections were placed on silanised glass slides (No S3003, Dako Japan, Kyoto, Japan), and the slides were dried, dewaxed in xylene, and rehydrated in graded concentrations of ethanol. After incubation with normal blocking serum, the sections were incubated overnight at 4°C with monoclonal antibody against the tissue inhibitors of MMP (TIMP-2) (Fuji-Yakuhin Co, Toyama, Japan), or polyclonal antibody against haem oxygenase (HO-1) (sc-1797, Santa Cruz Biotechnology Inc, Santa Cruz, California, USA). After incubation with a secondary antibody, the sections were allowed to react with Vectastain Elite ABC reagent (Vector Laboratories, Burlingame, California, USA), and then with the peroxidase substrate solution (Vectastain 3′3′-diaminobenzidine substrate kit, Vector Laboratories).15
Following immunohistochemical analysis, the staining grade was evaluated from 0 to 3+ under light microscopy by three observers who were blinded to the other available data, and the average of the semiquantitative score was calculated.16
Measurement of capillary density by morphometry
Using transmission electron microscopy, more than 15 negative films at ×1000 magnification were taken from each block by systematic random sampling, and the number of capillaries was determined according to the theory of morphometry. Myocyte density (N/mm2) was also measured, and the ratio of myocyte to capillary density calculated.17
Zymography
Zymography using gelatin containing polyacrylamide gel for detecting MMP activity was undertaken. Cardiac tissue extract was prepared by a previously published method18 and the protein content of the concentrated extract was determined using a Bio-Rad protein assay kit according to Bradford.19 MMP activity was indicated by clear zones on a blue background. The gels were scanned into a computer database and analysed with the NIH image.20
Cytokine gene expression assay system: quantitative RT-PCR
Cytokine mRNA concentrations were measured by a real time quantitative reverse transcriptase polymerase chain reaction (RT-PCR) method, with the ABI Prism 7700 sequence detection system (Perkin-Elmer Applied Biosystems, Shelton, Connecticut, USA).21 cDNA was synthesised from 100 ng of total RNA. Relative quantification and standardisation were achieved using a log10 dilution series of RNA extracted from rat spleen (calibrator). Relative quantification of 18S ribosomal, interleukin (IL)-1β, IL-6, transforming growth factor β (TGF-β), and tumour necrosis factor α (TNFα) mRNA was calculated by the comparative cycle threshold method outlined in the user manual provided by Perkin-Elmer.22 Because the values for the calibrator wells are always subtracted from themselves, their relative quantification values (cytokine gene/18S ribosomal gene) should be 1.
Statistical analysis
All results are expressed as mean (SD), and statistical analysis was done using one way analysis of variance. When significant differences were identified, the Scheffé’s multiple range test was applied to determine the level of significance. The results were considered significant at a probability value of p < 0.05.
RESULTS
Heart weight, body weight, blood sugar, and TBARS
Although the heart weight and body weight were increased in the OLETF rats, there was no significant difference in the ratio of heart weight to body weight among the four groups (table 1). The blood sugar concentration (mean (SD)) in the OLETF rats was significantly higher than in the LETO rats, at 436 (34) v 168 (52) mg/dl, but there was no significant difference between the groups with and without angiotensin receptor blockade (408 (48) mg/dl). TBARS measured as a marker of oxidative stress was significantly increased in the OLETF groups. Angiotensin receptor blockade in the OLETF rats modestly suppressed the increase in TBARS, but this did not reach significance (table 1).
Table 1.
Haemodynamic data, diameter of cardiomyocytes, capillary density, and thiobarbituric reducing substances
| Variable | LETO | LETO+ARB | OLETF | OLETF+ARB |
| Body weight (g) | 489 (22) | 461 (22) | 605 (26)* | 529 (25)† |
| Heart weight (g) | 1.3 (0.1) | 1.2 (0.1) | 1.7 (0.1)* | 1.3 (0.1)† |
| HW/BW (mg/g) | 2.6 (0.1) | 2.6 (0.1) | 2.8 (0.2) | 2.4 (0.1) |
| Heart rate (beats/min) | 412 (37) | 402 (30) | 263 (71)* | 294 (53)* |
| LVPsys (mm Hg) | 96 (4) | 87 (7) | 110 (6)* | 87 (5)† |
| LVEDP (mm Hg) | 6 (2) | 7 (2) | 11 (2)* | 7 (2)† |
| Max dP/dt (mm Hg/s) | 3196 (161) | 2860 (296) | 2390 (1205) | 3240 (746) |
| Min dP/dt (mm Hg/s) | 3492 (286) | 3220 (148) | 2350 (250)* | 3420 (1317)† |
| Diameter (μm) | 18.9 (0.6) | 18.4 (0.4) | 24.3 (0.6)* | 20.1 (1.1)† |
| Density (N/mm2) | 2587 (336) | 2889 (137) | 1768 (227)* | 3055 (151)† |
| TBARS (nM/ml) | 2.9 (0.6) | 2.8 (0.8) | 8.2 (1.5)* | 6.8 (1.2)* |
Values are mean (SD).
*p < 0.05 v LETO; †p < 0.05 v OLETF.
ARB, angiotensin II receptor blockade; BW, body weight; Density, capillary density; dP/dt, rate of change of pressure; diameter, mean diameter of cardiomyocytes; HW, heart weight; LETO, Long-Evans Tokushima Otsuka rats; LVEDP, left ventricular end diastolic pressure; LVPsys, left ventricular systolic pressure; max, maximum. min, minimum; OLETF, Otsuka Long-Evans Tokushima Fatty rats; TBARS, thiobarbituric acid reactive substances.
Haemodynamic data
Angiotensin receptor blockade had no haemodynamic effect on the LETO rats. The OLETF rats had a significant decrease in heart rate and peak negative dP/dt, and an increase in left ventricular systolic and end diastolic pressures. In the OLETF+ARB group, however, these variables (except for heart rate) improved significantly and became similar to those in the LETO groups (table 1).
Histological findings
No morphological abnormality was detected in the LETO rats (fig 1), and no pathological changes were found in the ascending aorta in either LETO or OLETF rats. In the OLETF rats, a thickened capillary basement membrane and perivascular fibrosis were prominent (fig 2). The mean diameter of the cardiomyocytes was increased, with bizarrely shaped mitochondria, myofibrillar lysis, and vacuolisation. Some mitochondria showed disorganisation of the matrix space between the cristae and amorphous matrix densities. All pathological findings were less severe in size and degree in the OLETF+ARB subgroup, although some degenerative changes were still observed in these rats (table 1, figs 1 and 2).
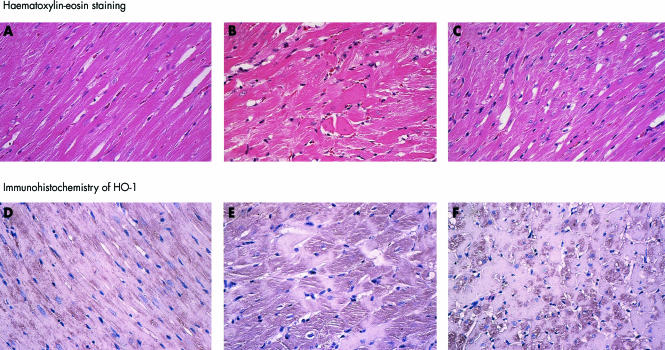
Figure 1.
Light micrographs. Upper panels (A, B, C): haematoxylin–eosin staining. Lower panels (D, E, F): immunohistochemistry for HO-1 (polyclonal antibody against haem oxygenase-1). The LETO rats showed normal morphology (A). In the LETO rats, immunoreactivity against HO-1 antibody was slight (D). In the OLETF rats, hypertrophied cardiomyocytes, disarray of myofibres, and scarcity of myofibrils were observed (B). HO-1 was increased in the cytoplasm of cardiocytes (E). All the above changes were smaller in the OLEFT+ARB subgroup (C and F). Magnification ×100. ARB, angiotensin II receptor blockade.
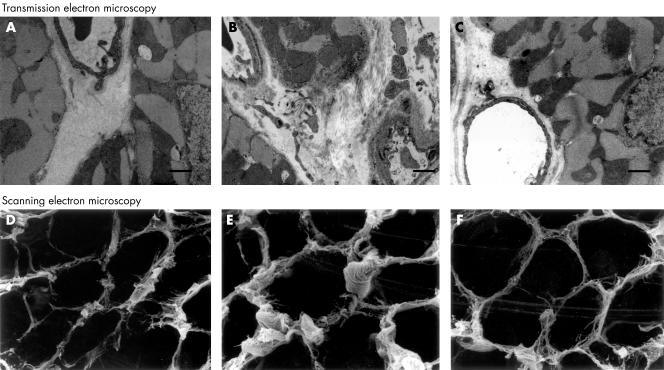
Figure 2.
Transmission and scanning electron micrographs. The LETO rats showed normal appearances (A and D). In the OLETF rats, thickened capillary basement membranes and interstitial fibrosis were prominent (B and E). All the changes were smaller in the OLETF+ARB subgroup (C and F). Scale bar = 1 μm. ARB, angiotensin II receptor blockade.
Under scanning electron microscopy, interstitial collagen fibres increased in the OLETF rats, especially around the small arteries. In the OLETF+ARB subgroup, the amount of collagen tended to be decreased but there was no significant difference in the percentage of collagen per visual field between the OLETF and the OLETF+ARB subgroups (fig 2).
Immunohistochemistry
Immunoreactivity against TIMP-2 was present heterogeneously in the cardiomyocytes in all the groups, and reactivity tended to increase only in the vicinity of the small arteries in the OLETF rats. The semiquantitative scores calculated from the grading score by three observers were 1.3 (0.2), 1.6 (0.3), and 1.3 (0.2) in LETO, OLETF, and OLETF+ARB groups, respectively.
HO-1 increased significantly in the cytoplasm of cardiomyocytes in the OLETF rats (score, 2.5 (0.2), v 1.0 (0.3) in LETO rats; p < 0.05), and the immunoreactivity tended to decrease in the OLETF+ARB subgroup (score, 2.1 (0.2)) (fig 1).
Capillary density
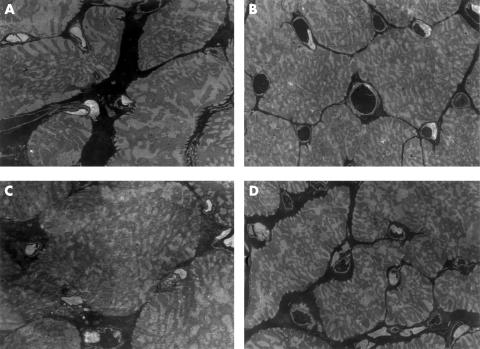
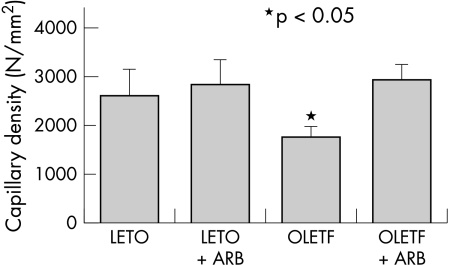
Capillary density was decreased only in the OLETF rats, and there was no significant difference among the LETO, LETO+ARB, and OLETF+ARB animals (figs 3 and 4). The myocyte to capillary ratio in each group ranged from 0.66 to 0.76, and there were no significant differences.
Figure 3.
Measurement of capillary density. More than 15 negative films at ×1000 magnification were taken by transmission electron microscopy according to the theory of morphometry (A, LETO; B, LETO+ARB; C, OLETF; D, OLETF+ARB). ARB, angiotensin II receptor blockade.
Figure 4.
Capillary density was significantly decreased in the OLETF rats. This was partially reversed by angiotensin II receptor blockade.
Zymography
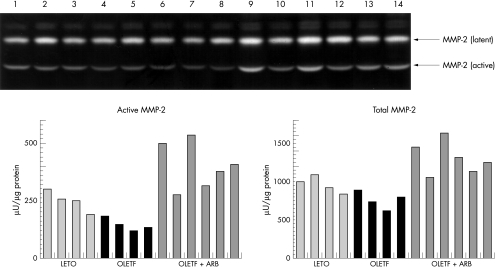
Mean MMP-2 activity tended to be reduced in the OLETF rats (144 (24) μU/μg), though not significantly compared with theLETO group (249 (47) μU/μg). However, MMP-2 activity was increased by angiotensin receptor blockade in the OLETF+ARB group (to 399 (101) μU/μg; p < 0.01 v LETO and OLETF). The latent type of MMP-2 and total MMP-2, defined as the sum of latent and active type, were similar in the quantification analysis (fig 5).
Figure 5.
Zymography of matrix metalloproteinases (MMP) in the LETO group (lanes 1 to 4), in the OLETF group (lanes 5 to 8), and in the OLETF+ARB group (lanes 9 to 14). The latent type (72 kDa) and active type (62 kDa) of MMP-2 tended to decrease in the OLETF group, an effect that was significantly reversed by angiotensin II receptor blockade. The bar graphs show active MMP-2 and total MMP-2, defined as the sum of latent and active type, which were quantified with the NIH image and expressed as μU/μg protein. ARB, angiotensin II receptor blockade.
Cytokine gene expression assay system: quantitative RT-PCR
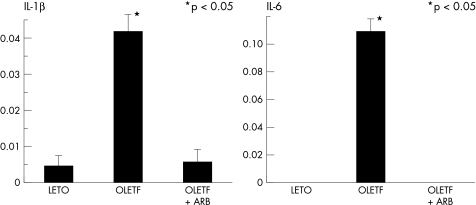
The expression of mRNA of inflammatory cytokines, particularly IL-1β and IL-6, was significantly enhanced only in the OLETF rats, an effect that was decreased by angiotensin receptor blockade (fig 6). Although TGF-β and TNF-α tended to increase in the OLETF group, there was no significant difference among the four groups.
Figure 6.
Cytokine gene expression. Expression of interleukin (IL)-1β and IL-6 mRNA was significantly enhanced in the OLETF group. This effect was decreased by angiotensin II receptor blockade.
DISCUSSION
OLETF rats
Earlier studies have shown that insulin resistance in OLETF rats is apparent at 12 weeks of age, before the impairment of pancreatic β cell function.10 Even in the pre-stage of type 2 diabetes, OLETF rats show alteration in left ventricular diastolic dysfunction,11 and in our study this was significantly impaired despite only mild deterioration of systolic function (table 1). We therefore considered that our model animal had an early stage of diabetic cardiomyopathy. Though we could not discount an effect of anaesthesia, the histological damage in the sinus node observed only two weeks after the induction of diabetes could have been related to the decreased heart rate in the OLETF rats.20
The large standard deviation of the peak positive dP/dt values implies the presence of animals with deteriorated systolic function in the OLETF group. We could not undertake subgroup analysis because of the small number of rats with systolic dysfunction. Further exploration of this effect, including the sarcoplasmic reticular Ca2+-ATPase and myosin isozyme analysis in the left ventricular myocardium, should be considered.23
In the OLETF+ARB subgroup, blood pressure was lowered and all the haemodynamic variables, including diastolic function, were similar to those in normal rats. It seems that angiotensin receptor blockade is effective in preventing diastolic dysfunction resulting from diabetes, but a blood pressure lowering effect might also be involved. However, Kamide and colleagues have reported that left ventricular hypertrophy in fructose fed rats may be less dependent on systemic elevation of blood pressure than on the renin–angiotensin system.24 Although the ratio of heart weight to body weight showed no significant change among the four groups, heart weight was reduced by angiotensin receptor blockade in our study. To exclude an effect of decreased blood pressure, the experiments should be repeated using smaller amounts of angiotensin receptor blockers.
Morphologically, in the absence of intervention a thickened capillary basement membrane was the most prominent feature, and there was moderate perivascular fibrosis and myocardial degeneration. Under electron microscopy, some mitochondria showed disorganisation of the matrix space between the cristae and amorphous matrix densities, similar to ischaemic changes. Increased collagen around the blood vessels might play a role in left ventricular diastolic dysfunction because of increased tangential wall stress and decreased oxygen supply. Though we did not observe atherosclerotic changes in the ascending aorta, a search for premature atherosclerosis of the peripheral vessels should be made.
MMP activity
Activation of MMP is controlled in three steps: gene expression, change of form from latent to active, and inhibition by tissue inhibitors of metalloproteinases (TIMP).25 TIMP are regulated by cytokines and growth factors, and decreased TIMP protein concentrations have been reported in the failing human heart.26 Although we observed increased reactivity only in the vicinity of the small arteries, there was no significant difference in the immunohistochemical staining of TIMP-2 between the OLETF and LETO rats, which might be related to disease stage. The exact role of TIMP in the failing heart is far from clear, insofar as it seems that under certain conditions TIMP may actually stabilise or localise MMP, which in turn could facilitate their activation.27 Another important point is that the extracellular matrix is a dynamic, interactive milieu which may send signals to influence myocardial cells. Other MMP and their inhibitors should be examined chronologically in this model.
Yagi and colleagues have reported that TGF-β mRNA was increased at 30 weeks,9 and Mizushige and associates reported an increase in TGF-β receptor at 15 weeks in the left ventricle of OLETF rats.11 We also found that TGF-β was slightly increased at 36 weeks, but there was no significant difference among the groups. On the other hand, the proinflammatory cytokines IL-1β and IL-6 were significantly increased in the OLETF rats, an effect that was suppressed by angiotensin II receptor blockade. It is not clear whether or not this effect depended on the timing of the sampling, but proinflammatory cytokines such as IL-1β and IL-6 also play an important role in the development of diabetic complications.28
In this study, the activity of MMP-2 tended to decrease in the OLETF rats, but was notably increased after angiotensin II receptor blockade. Singh and colleagues reported that MMP-2 activity was decreased in rat mesangial cells by glucose loading.29 We have previously shown that decreased MMP-2 activity in the left ventricular myocardium occurred in rats with streptozotocin induced diabetes.20 This could induce an increase in type IV collagen, causing thickening of the capillary basement membrane, followed by increased stiffness and diastolic dysfunction in the diabetic heart.
Capillary density
It has been reported that capillary density shows a progressive decrease of more than 20% over 26 weeks in streptozotocin induced diabetes.30 We also found a significant decrease in capillary density in the OLETF group, which might cause ischaemia in the cardiomyocytes (figs 3 and 4). Angiotensin receptor blockade in the OLETF rats increased capillary density, which would be expected to be beneficial to diabetic myocardial cells. However, most of the increased capillarisation could have been an indirect effect of a lesser degree of cardiac hypertrophy with an unchanged myocyte to capillary ratio among the groups.
Activation of MMP-2 and MMP-9 was reported recently to play a critical role in promoting ischaemia induced angiogenesis.4 Although we could not evaluate vascular endothelial growth factor in our study, the increased MMP-2 activity observed may helped to improve capillary density.
Oxidative stress
Oxidative stress may play a role in the effects of angiotensin receptor blockade in diabetes.31 There is increasing evidence that enhanced production or ineffective scavenging of reactive oxygen species can result in increased oxidative stress, and this may be crucial in the pathogenesis of cardiac abnormalities.32 The interaction of glycated proteins with their cell surface binding sites leads to oxidative stress and induction of the stress protein haem oxygenase (HO-1). HO-1 expression is increased by various oxidative insults.33 We observed that TBARS in plasma and HO-1 in the myocardium were significantly increased in OLETF rats. Locally produced angiotensin II might upregulate cardiac HO-1 through a pressor independent effect. Angiotensin receptor blockade was unable to prevent the increase of TBARS and HO-1 in this study, but higher dose of blocker might have an antioxidative effect.34
Although we could not show any relation between oxidative stress and MMP activity, aggressive treatment of oxidative stress might affect cytokines, causing the change in MMP activity. Inhibitors of MMP and cytokines should be evaluated to clarify the effect of angiotensin receptor blockade in the diabetic heart.
Conclusions
Cardesartan, an angiotensin II receptor blocker, lowered blood pressure and preserved left ventricular diastolic function in diabetic rat hearts. It was also potent at reducing the thickening of the capillary basement membrane, decreasing the diameter of the cardiomyocytes, increasing MMP-2 activity, and decreasing inflammatory cytokines. With all these changes, candesartan might contribute to cardioprotection in diabetes mellitus, independently of blood sugar concentration.
Acknowledgments
We express our deep gratitude to Dr Yumiko Kanzaki and Dr Nobuaki Okuda for their valuable assistance in the scanning electron microscopy, and to Dr Jules Constant for his advise and comments on the manuscript. We also thank Y Kitaguni and S Uchida for their expert technical assistance.
Abbreviations
ARB, angiotensin II receptor blockade
(HO)-1, haem oxygenase-1
LETO, Long-Evans Tokushima Otsuka rats
MMP, matrix metalloproteinase
OLETF, Otsuka Long-Evans Tokushima Fatty rats
RT-PCR, reverse transcriptase polymerase chain reaction
TBARS, thiobarbituric acid reactive substances
TIMP, tissue inhibitors of metalloproteinases
REFERENCES
- 1.Abbott RD, Donahue RP, Kannel WB, et al. The impact of diabetes on survival following myocardial infarction in men vs women. The Framingham study. JAMA 1988;260:3456–60. [PubMed] [Google Scholar]
- 2.Dhalla NS, Liu X, Panagia V, et al. Subcellular remodeling and heart dysfunction in chronic diabetes. Cardiovasc Res 1998;40:239–47. [DOI] [PubMed] [Google Scholar]
- 3.Sternlicht MD, Werb Z. Matrix metalloproteinases. In: Kreis T, Vale R, eds. Guidebook to the extracellular matrix, anchor, and adhesion proteins. Oxford: Oxford University Press, 1999:519–24.
- 4.Silvestre JS, Mallat Z, Tamarat R, et al. Regulation of matrix metalloproteinase activity in ischemic tissue by interleukin-10. Circ Res 2001;89:259–64. [DOI] [PubMed] [Google Scholar]
- 5.New RB, Sampson AC, King MK, et al. Effects of combined angiotensin II and endothelin receptor blockade with developing heart failure: effects on left ventricular performance. Circulation 2000;102:1447–53. [DOI] [PubMed] [Google Scholar]
- 6.Pfeffer MA. Enhancing cardiac protection after myocardial infarction: rationale for newer clinical trials of angiotensin receptor blockers. Am Heart J 2000;139:S23–8. [DOI] [PubMed] [Google Scholar]
- 7.Frustaci A, Kajstura J, Chimenti C, et al. Myocardial cell death in human diabetes. Circ Res 2000;87:1123–32. [DOI] [PubMed] [Google Scholar]
- 8.Kawano K, Hirashima T, Mori S, et al. Spontaneous long-term hyperglycemic rat with diabetic complications: Otsuka Long-Evans Tokushima Fatty (OLETF) strain. Diabetes 1992;41:1422–8. [DOI] [PubMed] [Google Scholar]
- 9.Yagi K, Kim S, Wanibuchi H, et al. Characteristics of diabetes, blood pressure, and cardiac and renal complications in Otsuka Long-Evans Tokushima Fatty rats. Hypertension 1997;29:728–35. [DOI] [PubMed] [Google Scholar]
- 10.Sato T, Asahi Y, Toide K, et al. Insulin resistance in skeletal muscle of the male Otsuka Long-Evans Tokushima Fatty Rat, a new model of NIDDM. Diabetologia 1995;38:1033–41. [DOI] [PubMed] [Google Scholar]
- 11.Mizushige K, Yao L, Noma T, et al. Alteration in left ventricular diastolic filling and accumulation of myocardial collagen at insulin-resistant prediabetic stage of a type II diabetic rat model. Circulation 2000;101:899–907. [DOI] [PubMed] [Google Scholar]
- 12.Jentzsch AM, Furst P, Biesalski HK. Improved analysis of malondialdehyde in human body fluids. Free Radical Biol Med 1996;20:251–6. [DOI] [PubMed] [Google Scholar]
- 13.Hayashi T, James TN, Buckingham DC. Ultrastructure and immunohistochemistry of the coronary chemoreceptor in human and canine hearts. Am Heart J 1995;129:946–59. [DOI] [PubMed] [Google Scholar]
- 14.Rossi MA, Abreu MA, Santoro LB. Connective tissue skeleton of the human heart: a demonstration by cell-maceration scanning electron microscope method. Circulation 1998;97:934–5. [DOI] [PubMed] [Google Scholar]
- 15.Hayashi T, Shimomura H, Toko H, et al. Collagen subtypes and matrix metalloproteinase in idiopathic restrictive cardiomyopathy. Int J Cardiol 1998;64:109–16. [DOI] [PubMed] [Google Scholar]
- 16.Hayashi T, Ijiri Y, Toko H, et al. Increased digitalis-like immunoreactive substances in patients with hypertrophic cardiomyopathy. Eur Heart J 2000;21:296–305. [DOI] [PubMed] [Google Scholar]
- 17.Rakusan K, Cicutti N, Maurin A, et al. The effect of treatment with low dose ACE inhibitor and/or diuretic on coronary microvasculature in stroke-prone spontaneously hypertensive rats. Microvasc Res 2000,59:243–54. [DOI] [PubMed] [Google Scholar]
- 18.Tyagi SC, Matsubara L, Weber KT. Direct extraction and estimation of collagenase(s) activity by zymography in microquantities of rat myocardium and uterus. Clin Biochem 1993;26:191–8. [DOI] [PubMed] [Google Scholar]
- 19.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–54. [DOI] [PubMed] [Google Scholar]
- 20.Hayashi T, Nozawa M, Sohmiya K, et al. Efficacy of pancreatic transplantation on cardiovascular alterations in diabetic rats: an ultrastructural and immunohistochemical study. Transplant Proc 1998;30:335–8. [DOI] [PubMed] [Google Scholar]
- 21.Bieche I, Olivi M, Champene MH, et al. Novel approach to quantitative polymerase chain reaction using real-time detection: application to the detection of gene amplification in breast cancer. Int J Cancer 1998;78:661–6. [DOI] [PubMed] [Google Scholar]
- 22.Holland PM, Abramson RD, Watson R, et al. Detection of specific polymerase chain reaction product by utilizing the 5′3′ exonuclease activity of Thermus aquaticus DNA polymerase. Proc Natl Acad Sci USA 1991;88:7276–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abe T, Ohga Y, Tabayashi N, et al. Left ventricular diastolic dysfunction in type 2 diabetes mellitus model rats. Am J Physiol Heart Circ Physiol 2002;282:H138–48. [DOI] [PubMed] [Google Scholar]
- 24.Kamide K, Rakugi H, Higaki J, et al. The renin–angiotensin and adrenergic nervous system in cardiac hypertrophy in fructose-fed rats. Am J Hypertens 2002;15:66–71. [DOI] [PubMed] [Google Scholar]
- 25.Libby P, Lee RT. Matrix matters. Circulation 2000;102:1874–6. [DOI] [PubMed] [Google Scholar]
- 26.Thomas CV, Coker ML, Zellner JL, et al. Increased matrix metalloproteinase activity and selective upregulation in LV myocardium from patients with end-stage dilated cardiomyopathy. Circulation 1998;97:1708–15. [DOI] [PubMed] [Google Scholar]
- 27.Mann DL, Taegtmeyer H. Dynamic regulation of the extracellular matrix after mechanical unloading of the failing human heart. Recovering the missing link in left ventricular remodeling. Circulation 2001;104:1089–91. [PubMed] [Google Scholar]
- 28.Esposito K, Nappo F, Marfella R, et al. Inflammatory cytokine concentrations are acutely increased by hyperglycaemia in humans. Circulation 2002;106:2067–72. [DOI] [PubMed] [Google Scholar]
- 29.Singh R, Song RH, Alavi N, et al. High glucose decreases matrix metalloproteinase-2 activity in rat mesangial cells via transforming growth factor-beta. Exp Nephrol 2001;9:249–57. [DOI] [PubMed] [Google Scholar]
- 30.Thompson EW. Quantitative analysis of myocardial structure in insulin-dependent diabetes mellitus: effects of immediate and delayed insulin replacement. Proc Soc Exp Biol Med 1994;205:294–305. [DOI] [PubMed] [Google Scholar]
- 31.Giugliano D, Ceriello A, Paolisso G. Oxidative stress and diabetic vascular complications. Diabetes Care 1996;19:257–67. [DOI] [PubMed] [Google Scholar]
- 32.Kaul N, Siveski-Iliskovic N, Hill M, et al. Free radicals in the heart.J Pharmacol Toxicol Methods 1993;30:55–63. [DOI] [PubMed] [Google Scholar]
- 33.Ishizaka N, Aizawa T, Mori I, et al. Heme oxygenase-1 is upregulated in the rat heart in response to chronic administration of angiotensin II. Am J Physiol Heart Circ Physiol 2000;279:H672–8. [DOI] [PubMed] [Google Scholar]
- 34.Yanagitani Y, Rakugi H, Okamura A, et al. Angiotensin II type 1 receptor-mediated peroxide production in human macrophages. Hypertension 1999;33:335–9. [DOI] [PubMed] [Google Scholar]