Reperfusion treatment in acute myocardial infarction represents the main indication for thrombolytic therapy. The most common thrombolytic agents have been streptokinase (first generation thrombolytic agent) and alteplase (tissue type plasminogen activator, t-PA, second generation thrombolytic agent). In the meantime third generation thrombolytic agents have reached clinical practice. Many of them are derivatives of alteplase, the current gold standard for thombolytic therapy in acute coronary syndromes with ST segment elevation. The most prominent among them are reteplase, tenecteplase, and lanoteplase.
RETEPLASE
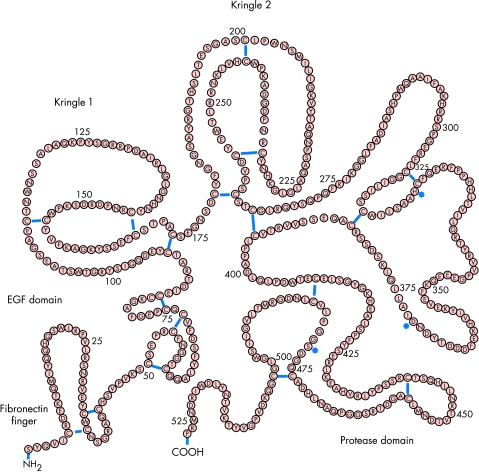
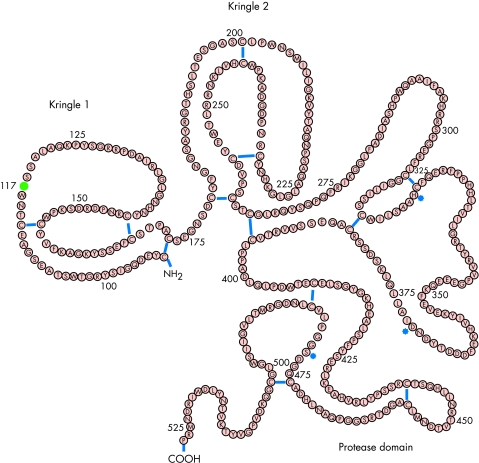
Reteplase (recombinant plasminogen activator, r-PA) is a single chain deletion mutant of alteplase that is expressed in Escherichia coli and, therefore, is expressed as an unglycosylated protein (table 1, figs 1 and 2). Reteplase includes 355 amino acids with a total molecular weight of 39 kDa. The molecule consists of cringle 2 and the protease domain of the alteplase molecule. Because of the deletion of the fibronectin finger region, the binding of reteplase to fibrin is significantly reduced in comparison with that of alteplase. Although kringle 2 (known to stimulate protease in the presence of fibrin) is part of the reteplase molecule, reteplase is stimulated in the presence of fibrin to a lower extent than alteplase, suggesting that the fibronectin finger is involved in the stimulation of the protease as well. Reteplase, in comparison with alteplase, is characterised by reduced fibrin selectivity. In the abscence of fibrin, reteplase and alteplase do not differ with respect to their activity as plasminogen activators, nor do they differ with respect to their inhibition by the plasminogen activator inhibitor type 1 (PAI-1).
Table 1.
Alteplase: structure of the molecule and function of its components
| Component | Function |
| Fibronectin finger | Binding to fibrin |
| Epidermal growth factor domain | Elimination by hepatocytes |
| Kringle 1 | Elimination by liver endothelial cells |
| Kringle 2 | Stimulation of protease by fibrin |
| Protease domain | Splitting of plasminogen |
| Inhibition by PAI-1 | |
| Carbohydrate side chain | Elimination from plasma |
Reteplase, tenecteplase, and lanoteplase are derivatives of alteplase, components of which have been deleted or changed.
PAI-1, plasminogen activator inhibitor type 1.
Figure 1.
Alteplase.
Figure 2.
Reteplase.
The elimination of reteplase from the circulating plasma predominantly occurs in the liver. Because of the deletion of the fibronectin finger region, the epidermal growth factor domain and kringle 1, as well as the carbohydrate side chains, the hepatic elimination of the molecule is reduced. Consequently, plasma half life is increased to 14–18 minutes (versus 3–4 minutes with alteplase). This allows reteplase to be administered as boli (versus as an initial bolus followed by an infusion, as with alteplase). Since early reocclusions of the infarct related coronary artery had been observed with the single bolus administration, it was replaced by a double bolus one. The best results have been obtained with a double bolus of 10 U each 30 minutes apart in the case of an acute myocardial infarction.
Reteplase has been approved for the thrombolytic treatment of acute myocardial infarction. In the RAPID I (recombinant plasminogen activator angiographic phase II international dose-finding study) trial reteplase was superior to alteplase (administered over three hours) with respect to patency of the infarct related coronary artery. With reteplase patency was reached earlier and more frequently than with alteplase. This was confirmed in the RAPID II (reteplase vs alteplase patency investigation during myocardial infarction) trial where alteplase was administered in the accelerated regimen (over 1.5 hours).1 However, in the GUSTO (global use of strategies to open occluded coronary arteries) III trial mortality after 30 days did not differ between patients treated with reteplase and those treated with alteplase.2 This discrepancy may be explained by more frequent patent infarct related vessels that reoccluded more often, leading to the same net benefit. In summary, reteplase in comparison with alteplase is equal in efficacy and superior in its application as a double bolus that also facilitates prehospital initiation of reperfusion therapy.
TENECTEPLASE
Tenecteplase is also called the TNK-mutant of alteplase. The molecule does not constitute a deletion mutant of alteplase (as reteplase does). Instead, it consists of the alteplase molecule with the exception of three point mutations (table 1, fig 3). At position 103 of the polypeptide the aminoacid threonine has been replaced by asparagine leading to a new glycosylation site. The carbohydrate chain that is linked to this site enlarges the molecule, thereby reducing its elimination and prolonging its plasma half life. At position 117 asparagine has been replaced by glutamine. By the exchange of this amino acid the carbohydrate side chain that facilitates hepatic elimination has been removed. Hence, plasma half life is further prolonged. Finally, at position 296–299 the amino acids lysine, histidine, arginine, and arginine have been replaced by four amino acids alanine. Consequently, the inhibition by PAI-1 is reduced 80 times in comparison with alteplase. The amino acids that were replaced at the three positions are called T, N, and K according to the one letter code for amino acids, which leads to the expression TNK-mutant. Since the molecule is expressed in CHO (Chinese hamster ovary) cells, it is expressed with carbohydrate side chains linked to the glycosylation sites of the polypeptide.
Figure 3.
Tenecteplase.
The relatively long plasma half life of tenecteplase (approximately 17 minutes) allows for single bolus application in the thrombolytic treatment of acute myocardial infarction. Compared with the alteplase molecule, no domain is missing in the tenecteplase molecule, so its fibrin selectivity is relatively high.
Tenecteplase has been tested extensively in clinical trials. In the ASSENT-1 (assessment of safety and efficacy of a new thrombolytic agent) trial in patients with acute myocardial infarction, single bolus tenecteplase proved to be as safe as the gold standard of thrombolytic therapy, the accelerated regimen of alteplase (initial bolus followed by an infusion over 90 minutes).3 With respect to intracranial bleeding complications the rate in patients treated within six hours of the onset of myocardial infarction was 0.56% with 30 mg tenecteplase and 0.58% with 40 mg tenecteplase. In the TIMI-10B (thrombolysis in myocardial infarction) trial single bolus administration of 40 mg tenecteplase achieved the same rate of patency at 90 minutes after the initiation of thrombolytic therapy as alteplase in the accelerated regimen did.4 In the ASSENT-2 trial tenecteplase and alteplase were equal with respect to total mortality after 30 days.5 As with reteplase, tenecteplase in comparison with alteplase is equal in efficacy and superior in its application as a single bolus that also facilitates prehospital initiation of reperfusion therapy.
LANOTEPLASE
The plasminogen activator lanoteplase (novel plasminogen activator, n-PA) is another deletions mutant of the alteplase molecule that also exhibits an additional, single point mutation (table 1, fig 4).6 In comparison with alteplase the fibronectin finger region and the epidermal growth factor domain have been deleted in the lanoteplase molecule. In addition, in kringle 1, at position 117 the aminoacid asparagine has been replaced by glutamine. Because of this point mutation the glycosylation site that is responsible for facilitated hepatic elimination is lost. Consequently, the plasma half life of lanoteplase is increased. All other carbohydrate side chains are incorporated because lanoteplase is expressed in CHO cells, just as is tenecteplase. CHO cells are eukaryotic cells, in contrast to prokaryotic cells such as E coli.
Figure 4.
Lanoteplase.
The plasma half life of lanoteplase is about 10 times that of alteplase and may reach 45 minutes. In the thrombolytic treatment of an acute myocardial infarction lanoteplase can be administered as a single bolus.
In the InTIME-1 (intravenous n-PA for treatment of infarcting myocardium early) trial, treatment with 120 kU lanoteplase per kg body weight resulted in a higher patency rate of the infarct related coronary artery at 90 minutes than treatment using alteplase in the accelerated regimen.7 However, with respect to overall mortality at 30 days lanoteplase and alteplase were equally effective (InTIME-2 trial).8 The latter trial also demonstrated an increased rate of haemorrhagic stroke with lanoteplase (1.12%) than with alteplase (0.64%, p = 0.004). This increase in the most severe complication of thrombolytic therapy has stopped lanoteplase from entering the market to date.
COMBINATION THERAPY
In the field of reperfusion therapy in acute myocardial infarction, the term “combination therapy” is most often used to describe the combined use of reduced dose plasminogen activators and full dose glycoprotein (Gp) IIb/IIIa inhibitors. The latter block the Gp IIb/IIIa receptors at the surface of activated platelets and, subsequently, platelet aggregation, the major mechanism in reocclusion.9 Since the activated receptor constitutes the final common pathway of platelet activation, the Gp IIb/IIIa inhibitors form the most potent antiplatelet therapy now available. Among them abciximab, eptifibatide, and tirofiban have proven their clinical efficacy.
In the TIMI 14 trial alteplase at half dose (15 mg as an initial bolus, 35 mg as an infusion over 60 minutes) combined with abciximab at full dose (0.25 mg/kg as an initial bolus, 10 μg/min as an infusion over 12 hours) yielded the highest patency rate 90 minutes after the initiation of treatment (TIMI 3 flow in 76% of treated patients) without increasing the risk of severe bleeding complications.10 Additional analysis of the trial showed that microvascular reperfusion (quantified by resolution of ST segment elevation) was re-established more often with the combination therapy than with alteplase treatment alone.11 In the SPEED (strategies for patency enhancement in the emergency department) trial a patent coronary artery could be achieved more often with the combination of reteplase in half dose (a double bolus of 5 U each, 30 minutes apart) and abciximab in full dose than with reteplase in full dose alone.12 However, in the GUSTO V trial the higher patency rate of this regimen could not be translated into reduced mortality after 30 days.13 In the ASSENT 3 trial half dose tenecteplase combined with abciximab was compared with full dose tenecteplase alone.14 With respect to the primary end point (a composite end point combining 30 day mortality, in-hospital reinfarction, or in-hospital refractory ischaemia), the combination therapy was superior to monotherapy with plasminogen activator but without Gp IIb/IIIa inhibitor.
CONCLUSION
In comparison with alteplase in its accelerated dose (over 90 minutes) the plasminogen activators reteplase and tenecteplase are equal in their efficacy (mortality after 30 days) and superior in their application (reteplase as a double bolus, tenecteplase as a single bolus). The combination of reduced dose plasminogen activators and full dose Gp IIb/IIIa inhibitors may yield an even better outcome after thrombolytic therapy in acute myocardial infarction.
REFERENCES
- 1.Bode C, Smalling RW, Berg G, et al. Randomized comparison of coronary thrombolysis achieved with double-bolus reteplase (recombinant plasminogen activator) and front-loaded, accelerated alteplase (recombinant tissue plasminogen activator) in patients with acute myocardial infarction. Circulation 1996;94:891–8. [DOI] [PubMed] [Google Scholar]
- 2.GUSTO III Investigators.A comparison of reteplase for acute myocardial infarction, N Engl J Med 1997;337:1118–23. ▸ The landmark clinical study that established reteplase as a thrombolytic agent equally as effective as accelerated alteplase, the gold standard, with respect to mortality after 30 days, the most decisive end point in clinical studies optimising the treatment of acute myocardial infarction.9340503 [Google Scholar]
- 3.van de Werf F, Cannon CP, Luyten A, et al. Safety assessment of single-bolus administration of TNK tissue-plasminogen activator in acute myocardial infarction: the ASSENT-1 trial. Am Heart J 1999;137:786–91. [DOI] [PubMed] [Google Scholar]
- 4.Cannon CP, Gibson CM, McCabe CH, et al. TNK-tissue plasminogen activator compared with front-loaded alteplase in acute myocardial infarction: results of the TIMI 10B trial. Circulation 1998;98:2805–14. [DOI] [PubMed] [Google Scholar]
- 5.ASSENT-2 Investigators. Single-bolus tenecteplase compared with front-loaded alteplase in acute myocardial infarction: the ASSENT-2 double-blind randomised trial. Lancet 1999;354:716–22. ▸ The landmark clinical study that established tenecteplase as a thrombolytic agent in the treatment of acute myocardial infarction, proving as equally effective as accelerated alteplase with respect to mortality after 30 days. [DOI] [PubMed] [Google Scholar]
- 6.Nordt TK, Moser M, Kohler B, et al. Pharmacokinetics and pharmacodynamics of lanoteplase (n-PA). Thromb Haemost 1999;82(suppl):121–3. [PubMed] [Google Scholar]
- 7.den Heijer P, Vermeer F, Ambrosioni E, et al. Evaluation of a weight-adjusted single-bolus plasminogen activator in patients with myocardial infarction: a double blind, randomized angiographic trial of lanoteplase versus alteplase. Circulation 1998;98:2117–25. [DOI] [PubMed] [Google Scholar]
- 8.InTIME-II Investigators. Intravenous NPA for the treatment of infarcting myocardium early: InTIME-II, a double-blind comparison of single-bolus lanoteplase vs accelerated alteplase for the treatment of patients with acute myocardial infarction. Eur Heart J 2000;21:2005–13. [DOI] [PubMed] [Google Scholar]
- 9.Nordt TK, Moser M, Kohler B, et al. Augmented platelet aggregation as predictor of reocclusion after thrombolysis in acute myocardial infarction. Thromb Haemost 1998;80:881–6. ▸ A clinical study in patients with acute myocardial infarction treated with thrombolytic therapy, supporting the dominant role of platelets in reocclusion in comparison with plasmatic coagulation and the endogenous fibrinolytic system. [PubMed] [Google Scholar]
- 10.Antman EM, Giugliano RP, Gibson CM, et al. Abciximab facilitates the rate and extent of thrombolysis. Results of the TIMI 14 trial. Circulation 1999;99:2710–32. [DOI] [PubMed] [Google Scholar]
- 11.de Lemos JA, Antman EM, Gibson CM, et al. Abciximab improves both epicardial flow and myocardial reperfusion in ST-elevation myocardial infarction. Observations from the TIMI 14 trial. Circulation 2000;101:239–43. ▸ A clinical study that has brought the importance of myocardial microvascular reperfusion therapy in the treatment of myocardial infarction to new attention. [DOI] [PubMed] [Google Scholar]
- 12.SPEED Group. Trial of abciximab with and without low-dose reteplase for acute myocardial infarction. Circulation 2000;101:2788–94. [DOI] [PubMed] [Google Scholar]
- 13.GUSTO V Investigators. Reperfusion therapy for acute myocardial infarction with fibrinolytic therapy or combination reduced fibrinolytic therapy and platelet glycoprotein IIb/IIIa inhibition: the GUSTO V randomised trial. Lancet 2001;357:1905–14. [DOI] [PubMed] [Google Scholar]
- 14.ASSENT-3 Investigators. Efficacy and safety of tenecteplase in combination with enoxaparin, abciximab, or unfractionated heparin: the ASSENT-3 randomised trial in acute myocardial infarction. Lancet 2001;358:605–13. [DOI] [PubMed] [Google Scholar]