Abstract
Objective: To test the working hypothesis that inflammation underlying precocious and severe coronary atherosclerotic disease in familial hypoalphalipoproteinaemia (FH) can be mediated by up regulation of the innate immune response.
Methods and results: 52 patients with FH were compared with 52 healthy controls with regard to immune system markers such as C reactive protein (CRP), soluble intercellular adhesion molecule-1 (sICAM-1), C3c, and C4. Patients differed from controls in their significantly lower concentrations of high density lipoprotein cholesterol (30.2 (4.0) v 50.5 (13.6) mg/dl, p < 0.0001) and apolipoprotein A I (113.2 (19.9) v 148.7 (25.1) mg/dl, p < 0.0001) and their higher triglyceride (139.3 (63.2) v 81.4 (41.7) mg/dl, p < 0.0001) and CRP plasma concentrations (median 0.33 mg/dl, range 0.02–4.66 mg/dl v median 0.07 mg/dl, range 0.02–0.85 mg/dl, p < 0.0001), but not in their total cholesterol and low density lipoprotein cholesterol concentrations. Concentrations of protein complement were higher in patients (C3: 150.8 (42.3) v 101.9 (17.4) mg/dl, p < 0.0001; C4: 35.5 (13.6) v 22.8 (6.4) mg/dl, p < 0.0001) and sICAM-1 concentrations were more than double those found in the controls (335.1 (107.5) v 159.5 (78.2) mg/dl, p < 0.0001).
Conclusions: Increased concentrations of sICAM-1, C3c, and C4 co-express with high concentrations of CRP in FH. The lack of signs and symptoms of inflammation in these patients may suggest that the immune response is up regulated as part of the pro-inflammatory mechanisms that are activated in this atherogenic condition.
Keywords: high density lipoprotein, HDL, hypoalphalipoproteinaemia, sICAM-1, soluble intercellular adhesion molecule-1, complement, atherosclerosis
We have recently shown that in patients with familial hypoalphalipoproteinaemia (FH), a condition in which premature and severe coronary atherosclerosis is usually established, there is a latent inflammatory state that is detected by increased C reactive protein (CRP) plasma concentrations.1 CRP is not simply a sensitive marker of inflammation but can also mediate complement activation.2 In the recent debate about the interrelation between molecules with a central role in the development of inflammation underlying atherosclerosis, an increased expression of CRP has been considered the first step along the pathway that leads to increased concentrations of intercellular adhesion molecule-1 (ICAM-1), with complement acting as a biochemical intermediate.3,4 These queries, crucial for understanding atherosclerotic processes, are still unanswered as far as human pathology is concerned. Complement has long been suspected to have a role in atherogenesis.5 Atherosclerosis has been experimentally produced by the synergic action of allergenic injury to arteries and by a lipid rich diet.6 A series of subsequent reports are in agreement with the immunological view of atherosclerosis.5,7–9
In 1990, Seifert et al10 showed that lipid fractions rich in cholesterol from atheromatous material of human aorta induce complement activation in a dose dependent manner, suggesting a direct association between lipid accumulation and immune system activation. Increased concentrations of complement components of either the early or the late (lytic complex constituents) complement cascade have been reported at the site of atherosclerotic lesions.11–13 Lastly, it has been reported repeatedly that ICAM-1 expression, known to be induced by cytokines such as tumour necrosis factor α and interleukin 1β14 as well as by lipoprotein components,15 can increase in endothelial cells because of complement deposition.16
The ICAM-1 molecules, as counterreceptors of lymphocyte function related antigen-1, are constitutive molecules of the immune system.17 Their role is crucial in the early phase of atherosclerosis when circulating monocytes and T lymphocytes invade the subendothelial space; ICAM-1 too, have been reported in human atherosclerotic plaques, co-expressed in macrophages and endothelial cells.18 Lastly, ICAM-1 molecules are definite prognostic markers of atherosclerosis.19
ICAM-1 and complement components then cooperate actively with inflammatory phenomena in the atherosclerotic process and may have the common potentiality to mirror events at the site of the arterial wall.
FH, because of the increased CRP and cholesterol content issued into the arterial wall (due to a defective efflux), is a particularly appropriate condition for verifying the theory that the inflammatory atherogenic reaction, consequent to abnormal lipid homeostasis, may be associated with up regulation of the immune response.
Therefore, in the clinical setting of FH we investigated whether there were also increased plasma concentrations of soluble ICAM-1 (sICAM-1) and complement fractions, such as C3 and C4, above the increased concentrations of CRP. These molecules not only are exquisite representatives of the immune system but are also easily and accurately measurable in peripheral blood; on the other hand, the search for potential sites of intervention through which atherosclerosis progression can be inhibited or slowed down cannot be disassociated from the determination of biohumoral effectors of the inflammatory response within the vascular tree.
METHODS
Patients
We recruited 52 patients with FH (44 men and eight women, mean (SD) age 52.7 (16.8) years) who had been referred to our lipid clinic.
The diagnosis of FH had been based on at least three consecutive analyses indicating high density lipoprotein (HDL) concentrations under the 10th centile, as well as on documentation of the vertical transmission of the trait through two (18 kindred) or three (five kindred) generations. All patients had a normal body mass index and were free from other forms of organ, systemic, chronic, or recurrent disease and additional risk factors for atherosclerosis; none had experienced acute myocardial infarction in the six months before the study. Patients were excluded from the study if they had CRP concentrations over 5.00 mg/dl, as well as other markers of inflammation, indicating the presence of intervening infections. None of the patients in either the control or patient group was taking a hormone or β blocker.
In seven patients, the low HDL concentrations were also associated with raised triglyceride plasma concentrations (200 < triglyceride < 320 mg/dl); 26 had ischaemia with coronary artery disease, documented by angiography.
The control group consisted of 52 healthy volunteers (44 men and eight women, mean (SD) age 50.1 (8.6) years) recruited from healthy laboratory personnel who underwent a complete medical examination to exclude organ, systemic, or recurrent diseases. Moreover, they had normal lipid profiles and a reduced probability of coronary artery disease (< 5%) according to the criteria of Diamond and Forrester,20 since they had no risk factors, no chest pain, and a negative ECG during exercise.
Laboratory analysis
Serum total cholesterol, triglyceride, and HDL cholesterol concentrations were assessed by standard procedures. low density lipoprotein cholesterol concentration was calculated according to Friedewald and colleagues.21 Apolipoprotein A I, Lp(a) lipoprotein, high sensitivity CRP (0.02–6.00 mg/dl), C3, and C4 were assayed in serum stored at −80°C, in one batch, by rate nephelometry (Behring BN 100, Dade Behring, Milan, Italy). Enzyme linked immunosorbent assays (ELISAs) were used for sICAM-1 (Bender Med System, Vienna, Austria). Routine chemical clinical analyses were performed by standard methods under strict quality control. The interassay coefficients of variation were < 5% for every type of measurement.
Statistical analysis
Data are expressed as mean (SD) or median and range for non-normal distributed variables. Quantitative variables not normally distributed were logarithmically transformed. Analysis of variance was used to evaluate differences in age, body mass index, lipid profiles, and CRP, C3, C4, and sICAM-1 concentrations between the two groups. Qualitative variables were analysed by the χ2 test.
Linear regression and Pearson’s correlation in univariate analysis assayed the relations between HDL, CRP, C4, C3, and sICAM-1 concentrations. The independent effect of each variable was then analysed by a set of multiple linear regressions and partial correlation coefficients.
RESULTS
Table 1 lists the physical characteristics and complete lipid profile of patients with FH and of the control group. The two groups differed significantly, as expected, in HDL cholesterol, triglyceride, and apolipoprotein A I concentrations and in the ratios of total cholesterol to HDL cholesterol and of HDL cholesterol to apolipoprotein A I. There was no difference in total cholesterol and low density lipoprotein concentrations.
Table 1.
Baseline characteristics and lipid profiles of the study groups
| FH (n = 52) | Controls (n = 52) | p Value | |
| Age (years) | 52.7 (16.8) | 50.1 (8.6) | NS |
| Body mass index (kg/m2) | 25.4 (4.4) | 24.8 (2.6) | NS |
| Total cholesterol (mg/dl) | 179.9 (36.9) | 190.6 (30.3) | NS |
| Triglycerides (mg/dl) | 139.3 (63.2) | 81.4 (41.7) | <0.0001 |
| HDL-C (mg/dl) | 30.2 (4.0) | 50.5 (13.6) | <0.0001 |
| LDL-C (mg/dl) | 122.6 (31.4) | 121.7 (31.6) | NS |
| TC/HDL-C | 6.00 (1.18) | 4.00 (1.11) | <0.0001 |
| HDL-C/apolipoprotein A I | 0.27 (0.04) | 0.34 (0.08) | <0.0001 |
| Apolipoprotein A I (mg/dl) | 113.2 (19.9) | 148.7 (25.1) | <0.0001 |
| Apolipoprotein B (mg/dl) | 113.7 (28.0) | 100.7 (23.5) | 0.0130 |
| Lipoprotein (a) (mg/dl) | 11.3 (9.6–133.5) | 11.9 (9.6–63.7) | NS |
| Hs-CRP (mg/dl) | 0.33 (0.02–4.66) | 0.07 (0.02–0.85) | <0.0001 |
Values are mean (SD) or median (range).
FH, familial hypoalphalipoproteinaemia; HDL-C, high density lipoprotein cholesterol; Hs-CRP, C reactive protein detected with high sensitivity measurement; LDL, low density lipoprotein cholesterol; NS, not significant; TC, total cholesterol.
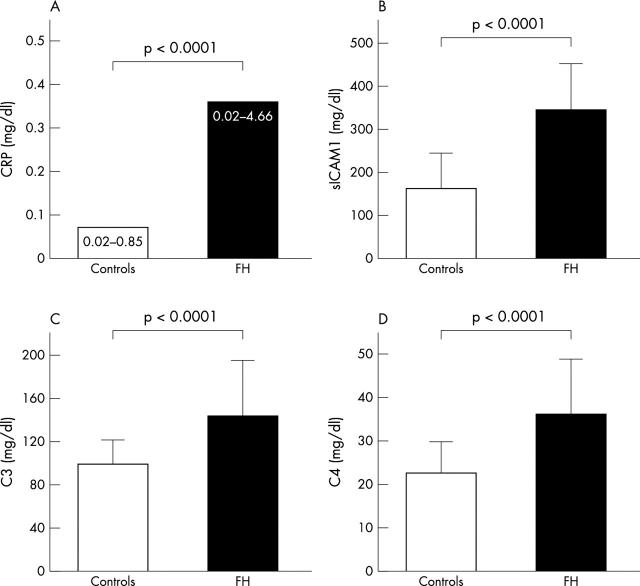
Figure 1 shows CRP, C3, C4, and sICAM-1 concentrations in the FH and control groups, which clearly illustrates that all these parameters are significantly higher in the patients with FH: CRP concentrations were more than four times higher (median 0.33 mg/dl, range 0.02–4.66 mg/dl v median 0.07 mg/dl, range 0.02–0.85 mg/dl, p < 0.0001), C3 concentrations increased by 49.8% (150.8 (42.3) mg/dl v 101.9 (17.4) mg/dl, p < 0.0001), C4 concentrations increased by 55.7% (35.5 (13.6) mg/dl v 22.8 (6.4) mg/dl, p < 0.0001), and sICAM-1 concentrations were more than doubled (110.1%, 335.1 (107.5) mg/dl v 159.5 (78.2) mg/dl, p < 0.0001).
Figure 1.
C reactive protein (CRP) (median, range) and C3, C4, and soluble intercellular adhesion molecule-1 (sICAM-1) (mean (SD)) concentrations in the control group and in patients with familial hypoalphalipoproteinaemia (FH).
To evaluate the circuit linking HDL, CRP, C4, C3, and sICAM-1, their correlation was analysed for all participants (fig 2). The HDL result was significant and was negatively correlated with all the other parameters (fig 2D, E, F, G). A significant and positive relation was observed between CRP and C4 (fig 2C), C3 and sICAM-1 (fig 2A), and C4 and sICAM-1 (fig 2B).
Figure 2.
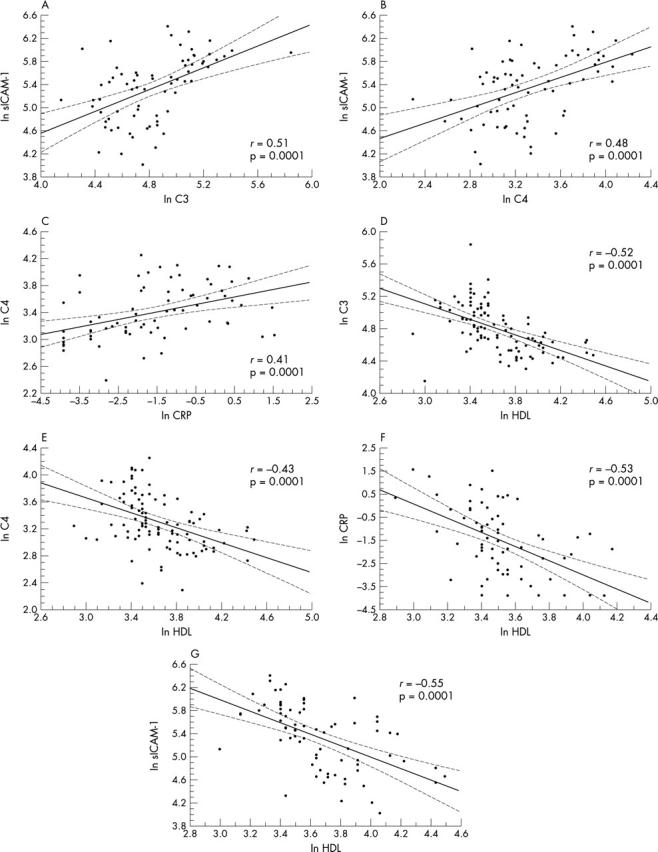
Linear regression analysis between high density lipoprotein (HDL), CRP, C4, C3, and sICAM-1 for all study participants.
Relations between HDL and CRP (r = 0.41, p = 0.002), between sICAM-1 and C4 (r = 0.37, p = 0.036), and between sICAM-1 and C3 (r = 0.41, p = 0.019) were also significant in the FH group despite the narrow range of value distributions.
C4 and C3 were highly correlated with each other in the entire population studied (r = 0.71, p < 0.0001), as well as in the FH group alone (r = 0.60, p < 0.0001), to the extent that they eliminated each other reciprocally in the multivariate analysis.
Multiple regression analysis showed a direct, negative relation of HDL with CRP (partial r = −0.53, p < 0.001), C3 (partial r = −0.34, p < 0.001), and sICAM-1 (partial r = −0.35, p < 0.01), of C3 and C4 with sICAM-1 (partial r = −0.30, p < 0.05 and r = 0.29, p < 0.05, respectively), of C3 with C4 (partial r = 0.63, p < 0.001), and of CRP with C4 (partial r = 0.28, p < 0.05).
DISCUSSION
We report that patients with FH have very high plasma concentrations of sICAM-1, C3, and C4 complement fractions, in addition to the high CRP concentrations that we reported previously.1
The presence of any possible secondary cause of inflammation (due to organ or systemic, chronic, or recurrent disease) was carefully excluded in the patients under study; therefore, the increase in these markers must be interpreted as being associated primarily with FH.
The overexpression of these markers in patients relative to the control group is very strong. Since these are constitutive components of the immune system, they probably indicate that the atherosclerotic–inflammatory reactions that develop in this condition may be characterised by up regulation of the immune response.
While the experimental setting used provides evidence of an association between FH and high plasma concentrations of these immune system markers, it only leads to speculation as regards the mechanism(s) producing such an increased plasma expression.
Data in the literature suggest that CRP, C3, C4, and sICAM-1 are linked by a biological pathway through which an increased expression of CRP enhances inflammation by activating complement and that this, among other effects, stimulates the expression of ICAM 1.3 Applying multivariate analysis, it emerges that HDL may directly influence sICAM-1, C3, and CRP. The data reported here suggest that low HDL concentrations might have acted on FH as a primary stimulus that triggered the chain of events in which high CRP concentrations activated complement and that, in turn, stimulated sICAM-1 expression; however, this may have been directly influenced by low HDL concentrations as well.
These data appear consistent with reports in the literature that several products of the activated complement system (C3b, iC3b, and C5a) can modulate endothelial cell function by influencing leucocyte activation and adherence by mediating the increased expression of ICAM-1 and by triggering local cytokine secretion; in short, by acting as pro-inflammatory mediators to atherogenesis.14
The suggestion that the complement system constitutes the bridge, or one of the bridges, between the inflammatory stimuli (in this case, the low HDL concentrations) and atherosclerotic lesions is based on results obtained mainly in vitro and, above all, through many different approaches aimed at evaluating the involvement of the complement system.
Data for human atherosclerosis are either sparse or have been obtained by incomparable methods as far as the activation of complement cascade is concerned.11–13
Our decision to measure C3 and C4 stems from two considerations: they are both at the beginning of the complement cascade; and they are involved both in the classical and in the alternative pathway of complement activation. In both instances, their increase may be interpreted as an increased secretion in response to chronic, continuous overconsumption; in effect, they are accurately measurable. Whether their increase in plasma can be interpreted as the result of an increased synthesis or release due to chronic constant consumption, albeit at a low level, is nevertheless a realistic possibility.
The possibility of accurately evaluating plasma markers of immune system involvement in atherogenesis would facilitate ex vivo study in humans aimed at prognostic evaluation and treatment intervention.
FH, a genetic trait characterised by extremely low plasma HDL concentrations that are associated with early and severe cardiovascular disease, is a good clinical model for investigating the expression of circulating forms of these molecules, with the aim of furthering understanding of the immune system’s involvement within the complexity of the inflammatory reactions in atherogenesis.
Acknowledgments
We are grateful to the Guidotti Laboratory Medical Department for their scientific collaboration.
Abbreviations
CRP, C reactive protein
ELISA, enzyme linked immunosorbent assay
FH, familial hypoalphalipoproteinaemia
HDL, high density lipoprotein
ICAM-1, intercellular adhesion molecule-1
sICAM-1, soluble intercellular adhesion molecule-1
REFERENCES
- 1.Sampietro T, Bigazzi F, Dal Pino B, et al. Increased plasma C-reactive protein in familial hypoalphalipoproteinemia: a proinflammatory condition? Circulation 2002;105:11–4. [DOI] [PubMed] [Google Scholar]
- 2.Wolbink GJ, Brouwer MC, Buysmann S, et al. CRP-mediated activation of complement in vivo: assessment by measuring circulating complement-C-reactive protein complexes. J Immunol 1996;157:473–9. [PubMed] [Google Scholar]
- 3.Yeh ET, Anderson HV, Pasceri V, et al. C-reactive protein: linking inflammation to cardiovascular complications. Circulation 2001;104:974–5. [DOI] [PubMed] [Google Scholar]
- 4.Lagrand WK, Niessen HW, Nijmeijer R, et al. Role for complement as an intermediate between C-reactive protein and intercellular adhesion molecule-1 expression? Circulation 2001;104:E46. [DOI] [PubMed] [Google Scholar]
- 5.Pang AA, Katz A, Minta JO. C3 deposition in cholesterol-induced atherosclerosis in rabbits: a possible etiologic role for complement in atherogenesis. J Immunol 1979;123:1117–22. [PubMed] [Google Scholar]
- 6.Smith EB. The relationship between plasma and tissue lipids in human atherosclerosis. Adv Lipid Res 1974;12:1–49. [DOI] [PubMed] [Google Scholar]
- 7.Seifert PS, Kazatchkine MD. The complement system in atherosclerosis. Atherosclerosis 1988;73:91–104. [DOI] [PubMed] [Google Scholar]
- 8.Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature 1993;362:801–9. [DOI] [PubMed] [Google Scholar]
- 9.Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med 1999;340:115–26. [DOI] [PubMed] [Google Scholar]
- 10.Seifert PS, Hugo F, Tranum-Jensen J, et al. Isolation and characterization of a complement-activating lipid extracted from human atherosclerotic lesions. J Exp Med 1990;172:547–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hollander W, Colombo MA, Kirkpatrick B, et al. Soluble proteins in the human atherosclerotic plaque: with spectral reference to immunoglobulins, C3-complement component, alpha 1-antitrypsin and alpha 2-macroglobulin. Atherosclerosis 1979;34:391–405. [DOI] [PubMed] [Google Scholar]
- 12.Vlaicu R, Rus HG, Niculescu F, et al. Immunoglobulins and complement components in human aortic atherosclerosis intima. Atherosclerosis 1985;55:35–50. [DOI] [PubMed] [Google Scholar]
- 13.Niculescu F, Hugo F, Rus HG, et al. Quantitative evaluation of the terminal C5b-9 complement complex by ELISA in human atherosclerotic plaques. Clin Exp Immunol 1987;69:477–83. [PMC free article] [PubMed] [Google Scholar]
- 14.Kilgore KS, Shen JP, Miller BF, et al. Enhancement by the complement membrane attack complex of tumor necrosis factor-α-induced endothelial cell expression of E selectin and ICAM-1. J Immunol 1995;155:1434–41. [PubMed] [Google Scholar]
- 15.Kume N, Cybulsky MI, Gimbrone MA Jr. Lysophosphatidylcholine, a component of atherogenic lipoproteins, induces mononuclear leukocyte adhesion molecules in cultured human and rabbit arterial endothelial cells. J Clin Invest 1992;90:1138–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Niessen HW, Lagrand WK, Visser CA, et al. Upregulation of ICAM-1 on cardiomyocytes in jeopardized human myocardium during infarction. Cardiovasc Res 1999;41:603–10. [DOI] [PubMed] [Google Scholar]
- 17.Springer TA. Adhesion receptors of the immune system. Nature 1990;346:425–34. [DOI] [PubMed] [Google Scholar]
- 18.Poston RN, Haskard DO, Coucher JR, et al. Expression of intercellular adhesion molecule-1 in atherosclerotic plaques. Am J Pathol 1992;140:665–73. [PMC free article] [PubMed] [Google Scholar]
- 19.Ridker PM, Hennekens CH, Roitman-Johnson B, et al. Plasma concentration of soluble intercellular adhesion molecule 1 and risks of future myocardial infarction in apparently healthy men. Lancet 1998;351:88–92. [DOI] [PubMed] [Google Scholar]
- 20.Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med 1979;300:1350–8. [DOI] [PubMed] [Google Scholar]
- 21.Friedewald WT, Levy RR, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499–502. [PubMed] [Google Scholar]