Abstract
Objective: To assess the effects of intravenous vitamin C administration on the vasomotor responses to intracoronary l-arginine infusion in epicardial coronary arteries.
Methods: 28 patients with coronary artery disease and stable angina were enrolled in the study. Eight patients received intracoronary infusions of 150 μmol/min l-arginine before and after intravenous infusion of vitamin C, 10 patients received intracoronary infusions of 150 μmol/min l-arginine before and after intravenous infusion of normal saline, and 10 patients received intracoronary normal saline before and after intravenous infusion of vitamin C. The diameter of proximal and distal coronary artery segments was measured by quantitative angiography.
Results: Infusion of l-arginine caused significant dilatation of both proximal (4.87 (0.96)%, p < 0.01 v normal saline) and distal (6.33 (1.38)%, p < 0.01 v normal saline) coronary segments. Co-infusion of vitamin C and l-arginine dilated proximal coronary segments by 8.68 (1.40)% (p < 0.01 v normal saline, p < 0.01 vl-arginine) and distal segments by 13.07 (2.15)% (p < 0.01 v normal saline, p < 0.01 vl-arginine). Intravenous infusion of vitamin C caused a borderline increase in proximal and distal coronary segment diameters (1.93 (0.76)% and 2.09 (1.28)%, respectively, not significant).
Conclusions: l-Arginine dependent coronary segment vasodilatation was augmented by the antioxidant vitamin C in patients with coronary artery disease. Thus, vitamin C may have beneficial effects on nitric oxide bioavailability induced by l-arginine.
Keywords: nitric oxide, endothelium, antioxidants, vasodilatation, coronary disease
Nitric oxide is synthesised from the amino acid l-arginine by a family of enzymes through the l-arginine–nitric oxide pathway.1l-Arginine is the substrate for the production of nitric oxide synthase.
It has been shown that l-arginine administration improves endothelium dependent vasodilatation in patients with risk factors for atherosclerosis, such as hypercholesterolaemia,2 smoking,3 aging,4 and hypertension5 and in patients with coronary artery disease6–8 (including dilatation of coronary stenoses),9 microvascular angina pectoris,10 and peripheral arterial disease.11 In animal models, l-arginine has improved endothelial dysfunction after coronary angioplasty12 and protected against ischaemia–reperfusion injury.13
Increased oxidative stress has been implicated as a potential mechanism for abnormal endothelial vasomotor function14 and inactivation of nitric oxide.15 Vitamin C is the main water soluble antioxidant in human plasma16 and it has been shown to reverse nitric oxide dependent endothelial dysfunction in patients with risk factors for atherosclerosis, including hypercholesterolaemia,17 smoking,18 hypertension,19 diabetes,20 and hyperhomocysteinaemia,21 as well as in patients with atherosclerosis and coronary artery disease.22 It effectively scavenges superoxide and other reactive oxygen species, thus playing an important part in the regulation of intracellular redox state through its interaction with glutathione.23 Whether administration of combined administration of vitamin C and l-arginine can lead to additional improvement of endothelial function in patients with atherosclerosis has not been investigated. We therefore examined the effects of intravenous vitamin C infusion on the response of the epicardial segments to l-arginine in patients with coronary artery disease and stable angina.
METHODS
Patients
The study population consisted of 28 patients (19 men, nine women) with chronic stable angina, coronary artery disease, and a positive treadmill exercise test (⩾ 0.1 mV ST segment depression) at between 5–7 metabolic equivalents with the modified Bruce protocol. Patients were excluded from the study if they had diabetes mellitus, history of coronary spasm, recent myocardial infarction (< 6 months), left ventricular hypertrophy (on echocardiography), three vessel coronary artery disease, left ventricular dysfunction (left ventricular ejection fraction < 50%), or valvar heart disease. Table 1 presents patients’ baseline characteristics. The protocol was approved by the research ethics committee and each patient gave written and informed consent. The investigation conforms with the principles outlined in the Declaration of Helsinki.
Table 1.
Clinical characteristics of the patients
| Characteristic | Group A: l-arg + vit C (n = 8) | Group B: l-arg (n = 10) | Group C: vit C (n = 10) |
| Men/women | 6/2 | 7/3 | 6/4 |
| Age (years) | 59 (3.3) | 57 (2.6) | 56 (2.2) |
| Diabetes | 0 | 0 | 0 |
| Dyslipidaemia | 6 | 3 | 7 |
| Smoking | 5 | 4 | 7 |
| Hypertension | 4 | 8 | 8 |
| History of MI | 2 | 3 | 3 |
| Extent of CAD | |||
| 1 vessel | 6 | 7 | 8 |
| 2 vessel | 2 | 3 | 2 |
| Cholesterol (mmol/l) | 6.10 (0.36) | 5.08 (0.64) | 6.50 (0.43) |
| Triglycerides (mmol/l) | 1.55 (0.14) | 1.58 (0.16) | 1.77 (0.13) |
Values expressed as mean (SEM) or number.
CAD, coronary artery disease; l-arg, l-arginine; MI, myocardial infarction; vit C, vitamin C.
There were no significant differences between the three groups.
Study protocol
Antianginal medication was stopped 48 hours before the study. The patients were allowed to use sublingual glyceryl trinitrate as necessary, but no study was performed within three hours of its administration. After diagnostic coronary angiography, an optimal radiographic projection was selected and kept constant for subsequent angiograms. The artery studied was chosen to comply with the research ethics committee’s requirements that coronary stenoses > 70% be avoided.
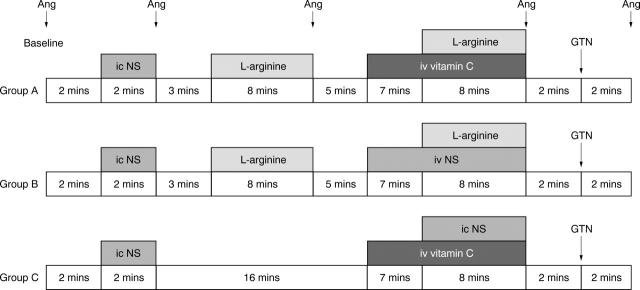
Patients were divided into one main group and two control groups, with 8–10 patients constituting each group (table 1). Two ECG leads were monitored continuously throughout the study. All infusions were administered into the coronary artery through a 7 French Judkin angioplasty guiding catheter. In the main group (group A: six men, two women, mean (SEM) age 59 (3.3) years) patients received an intracoronary infusion of 0.9% saline (2 ml/min) for two minutes, followed by an intracoronary infusion of 150 μmol/min of l-arginine for eight minutes, followed by a seven minute intravenous infusion of vitamin C (25 mg/min) and an eight minute combined infusion of intravenous vitamin C and intracoronary 150 μmol/min of l-arginine. Group B patients (seven men, three women, mean (SEM) age 57 (2.6) years) underwent the same protocol with the replacement of vitamin C with normal saline. Group C patients (six men, four women, mean (SEM) age 56 (2.2) years) received an intracoronary infusion of 0.9% saline (2 ml/min) for two minutes followed by a seven minute intravenous infusion of vitamin C (25 mg/min) and an eight minute combined infusion of intravenous vitamin C and intracoronary saline. Lastly, an intracoronary bolus dose of glyceryl trinitrate (250 μg) was administered to patients in all three groups. The distribution of risk factors for atherosclerosis (such as smoking, hypertension, lipid concentrations, or diabetes mellitus) did not differ between the groups (table 1). Antianginal medication (β blockers, aspirin, nitrates, statins, and angiotensin converting enzyme inhibitors) was similar in the three groups of patients. In a preliminary study of five patients we measured plasma concentration of vitamin C. After intravenous vitamin C administration serum vitamin C concentrations rose significantly from 41.8 (6.8) μmol/l to 120.2 (10.6) μmol/l (p < 0.05). This vitamin C concentration has been shown to protect human plasma from free radical mediated lipid peroxidation16 and to improve endothelium dependent vasodilatation in patients with hypercholesterolaemia,17 in chronic smokers,18 and in patients with diabetes mellitus.20Vitamin C concentrations were measured by high performance liquid chromatography after deproteinisation with 2% sulphur salicylic acid. Femoral arterial pressure and heart rate were recorded before and after l-arginine or l-arginine and vitamin C infusions and two minutes after glyceryl trinitrate administration. Angiography was performed (fig 1) with a hand injection of 6–8 ml non-ionic contrast medium. Before each angiogram, the catheter was emptied to avoid bolus administration of the infusate.
Figure 1.
Study protocol for the three groups of patients (A, B, and C) showing timing of angiography (Ang). GTN, glyceryl trinitrate; ic, intracoronary infusion; iv, intravenous infusion; NS, normal saline.
Quantitative coronary angiography
The arterial segments in each frame were analysed blindly in random order by quantitative computerised analysis with an automated edge contour detection analysis system (CAAS version 2V2; Pie Data Medical, Maastricht, the Netherlands).24 End diastolic frames from each arteriogram were selected for analysis. The angiographic catheter was used as a scaling device and this, together with pincushion distortion correction, allowed the diameters to be recorded as absolute values (expressed in millimetres). Special care was taken to avoid overlapping of coronary segments. The diameter of angiographically normal proximal and distal segments was recorded as follows: the proximal left anterior descending coronary artery diameter was measured just beyond the origin of the artery and the distal diameter was measured just distal to the second diagonal branch; the proximal left circumflex coronary artery diameter was measured just beyond the origin of the artery and the distal diameter just beyond the origin of the second obtuse marginal branch; and the proximal right coronary artery diameter was measured just beyond the origin of the artery and the distal diameter just beyond the posterior descending branch. Relatively straight coronary segments were preselected. Two proximal segments (one in the left circumflex coronary artery, another in the left anterior descending coronary artery) and two distal segments were usually selected for analysis from each left coronary arteriogram.
Two independent observers analysed the coronary arteriograms quantitatively and blindly re-analysed the films at a remote time for reproducibility of the method. Intra- and interobserver variabilities were non-significant (analysis of variance F = 0.3, p = 0.75).
Statistical analysis
For descriptive purposes data are expressed as mean (SEM). Analysis of variance for repeated measures was used to compare serial changes in heart rate and blood pressure. To test for differences in baseline values and percentage changes of proximal and distal segments within groups, the Friedman test and Wilcoxon sign rank test for repeated measures were applied; for differences in baseline values and percentage changes of proximal and distal segments between groups, the Kruskal-Wallis test and Mann-Whitney test were applied. A probability value of p < 0.05 (two tailed) was considered to indicate significance.
RESULTS
Clinical characteristics
During the l-arginine infusion the mean (SEM) heart rate (65 (3.4) v 69 (2.7) beats/min before and after, respectively, not significant) and systolic blood pressure (136 (9) v 133 (8) mm Hg before and after, respectively, not significant) remained unchanged. Similarly, after vitamin C infusion mean (SEM) heart rate (66 (3.0) v 67 (3.0) beats/min before and after, respectively, not significant) and systolic blood pressure (136 (4) v 135 (5) mm Hg before and after, respectively, not significant) did not differ.
Changes in epicardial coronary arteries
After saline infusion the luminal diameter of the proximal and distal segments of the epicardial coronary arteries did not change significantly in all groups (table 2).
Table 2.
Effects of intracoronary infusion of l-arginine, vitamin C, combined l-arginine and vitamin C, and nitrates on proximal and distal coronary segments
| Infusion | Group A: l-arg + vitamin C | Group B: l-arg + NS | Group C: vitamin C + NS | |||
| Diameter (mm) | Change from baseline (%) | Diameter (mm) | Change from baseline (%) | Diameter (mm) | Change from baseline (%) | |
| Proximal segment | ||||||
| Baseline | 3.02 (0.12) | 3.06 (0.12) | 2.92 (0.12) | |||
| NS | 3.04 (0.12) | 0.64 (0.30) | 3.08 (0.11) | 0.78 (0.41) | 2.92 (0.12) | −0.17 (0.84) |
| l-arginine | 3.16 (0.13) | 4.87 (0.96)** | 3.21 (0.13) | 5.20 (1.56)** | ||
| l-arg + NS | 3.23 (0.13) | 5.36 (1.33)** | ||||
| Vitamin C + NS | 2.98 (0.11) | 1.93 (0.76)* | ||||
| l-arg + vitamin C | 3.27 (0.12) | 8.68 (1.40)** † | ||||
| Nitrates | 3.59 (0.12) | 19.61 (1.94)** † | 3.49 (0.14) | 14.20 (2.15)** † | 3.39 (0.12) | 16.12 (1.22)** † |
| Distal segment | ||||||
| Baseline | 1.66 (0.09) | 1.53 (0.08) | 1.77 (0.06) | |||
| NS | 1.66 (0.09) | −0.08 (0.48) | 1.55 (0.08) | 1.21 (0.65) | 1.77 (0.06) | 0.37 (0.90) |
| l-arg | 1.76 (0.10) | 6.33 (1.38)** | 1.59 (0.07) | 4.23 (1.37)* | ||
| l-arg + NS | 1.62 (0.08) | 5.96 (1.52)** | ||||
| Vitamin C + NS | 1.80 (0.06) | 2.09 (1.28) | ||||
| l-arg + vitamin C | 1.87 (0.10) | 13.07 (2.15)** † | ||||
| Nitrates | 2.05 (0.10) | 24.64 (1.81)** † | 1.91 (0.10) | 24.78 (3.19)** † | 2.15 (0.06) | 22.38 (1.56)** † |
Values are expressed as mean (SEM).
*p<0.05, **p<0.01 versus baseline; †p<0.01 versus l-arg.
NS, normal saline.
Group A
The percentage increase in mean luminal diameter of the angiographically normal proximal (p < 0.01 v normal saline) and distal (p < 0.01 v normal saline) segments was significant after l-arginine administration (table 2, fig 2). With the co-infusion of vitamin C and l-arginine the diameter further increased significantly in proximal segments (p < 0.01 vl-arginine) and distal segments (p < 0.01 vl-arginine). The dilatation was significantly greater (p < 0.05) in distal than in proximal segments (fig 2). Nitrate infusion caused significant vasodilatation (p < 0.01) in both proximal and distal segments (table 2, fig 2).
Figure 2.
Graphs showing mean (SEM) luminal diameter percentage changes from baseline of proximal and distal segments after infusion of normal saline, l-arginine, combined factors (group A: l-arginine and vitamin C; group B: l-arginine and NS; group C: vitamin C and NS), and nitrates. Dilatation of epicardial arteries was significantly greater with l-arginine and vitamin C than with l-arginine alone (†p < 0.01). Distal segments dilated more than proximal segments (*p < 0.01).
Group B
The percentage increase in mean luminal diameter of the proximal (p < 0.01 v normal saline) and distal (p < 0.05 v normal saline) was significant after l-arginine administration (table 2, fig 2). The combined infusion of l-arginine during saline infusion did not further increase the percentage change of the luminal diameter in proximal (p = 0.758 vl-arginine) and distal (p = 0.365 vl-arginine) segments. Distal segments dilated less (p < 0.05) than in group A (fig 2). Nitrate infusion caused significant vasodilatation (p < 0.01) in both proximal and distal segments (table 2, fig 2).
Group C
The combination of intravenous infusion of vitamin C and intracoronary saline caused only a borderline increase in the diameter of proximal and distal coronary segments (not significant versus normal saline) (table 2, fig 2), which is significantly less than in the increases in groups A and B (fig 2). Nitrate infusion caused significant vasodilatation (p < 0.01) in both proximal and distal segments (table 2, fig 2). The response to nitrate infusion was similar in the three groups irrespective of vitamin C infusion both in the proximal and in the distal segments.
DISCUSSION
Our results showed that l-arginine significantly dilated epicardial coronary arterial segments in patients with atherosclerosis. The co-administration of l-arginine and antioxidant vitamin C significantly augmented this effect, whereas infusion of vitamin C alone caused only borderline dilatation. These findings indicate that vitamin C may increase nitric oxide availability induced by l-arginine administration.
Endothelial dysfunction and the l-arginine-nitric oxide pathway
l-Arginine is the substrate for nitric oxide production and has been shown to reduce vascular tone.25 The mechanism by which it exerts its vasodilator effects is controversial,26 but stimulation of the l-arginine-nitric oxide synthase (NOS)-nitric oxide pathway appears to be particularly important.25,26 Endothelial dysfunction may be partially attributed to l-arginine deficiency or the presence of l-arginine endogenous inhibitors such as asymmetric dimethylarginine.27
Effects of vitamin C on l-arginine-nitric oxide pathway
The present study showed that the antioxidant vitamin C increased nitric oxide availability induced by l-arginine administration in epicardial coronary arteries. Increased oxidative stress is known to be a potential mechanism for abnormal endothelial vasomotor function14 and inactivation of nitric oxide.15 Nitric oxide is inactivated by two basic mechanism. Firstly, nitric oxide reacts with reactive oxygen species. Superoxide radicals inactivate nitric oxide, leading to the formation of peroxynitrite—a powerful pro-oxidant that in high concentrations is toxic to cellular proteins and lipids.28 The amount of peroxynitrite production depends on the ratio of superoxide to nitric oxide.28 The second mechanism involves oxidised low density lipoprotein (LDL), which can also react directly with and inactivate nitric oxide.29 Oxidised LDL may interfere with signal transduction and agonist receptor dependent stimulation of NOS activity and with activation of guanylyl cyclase.30 Furthermore, oxidised LDL may induce a decreased uptake and local depletion of l-arginine, uncouple NOS, and potentially decrease production of nitric oxide.31 In the absence of sufficient concentrations of arginine or tetrahydrobiopterin, NOS itself can be a source of superoxide overproduction.32 In addition a competitive inhibition of nitric oxide synthesis through accumulation of factors such as asymmetric dimethylarginine may not be excluded.33
Vitamin C effectively inhibits the reaction of nitric oxide with superoxide only at very high concentrations, which in vivo are potentially achievable either in extracellular fluids by vitamin C infusion or in the intracellular milieu.34 Vitamin C increases the availability of tetrahydrobiopterin or the affinity of endothelial NOS (eNOS) for tetrahydrobiopterin and maintains high intracellular concentrations of glutathione primarily by a sparing effect, which may enhance the synthesis or increase the stabilisation of nitric oxide through formation of S-nitrosothiols.23,35 Vitamin C also exerts LDL specific antioxidant action by preventing its oxidation or by regenerating LDL associated α tocopherol, thereby inhibiting its pro-oxidant action.31
Thus, a balance between variations in eNOS protein concentrations, regulation of eNOS, and interaction of nitric oxide with superoxide controls the bioactivity of endothelium derived nitric oxide. The concentrations of l-arginine substrate and tetrahydrobiopterin, antioxidant enzymes such as glutathione, and other antioxidants such as vitamin C and α tocopherol may participate in this regulation.36 The main finding of the present study—that co-administration of vitamin C increased l-arginine induced epicardial artery dilatation in patients with coronary artery disease—may be explained by increased nitric oxide production in endothelial cells through both increased eNOS substrate l-arginine and depressed oxidative uncoupling of eNOS. Furthermore, vitamin C protects nitric oxide from oxidative modification to peroxynitrite, leading to a further increase in nitric oxide bioavailability. We also observed greater reactivity in distal than in proximal segments. This may be due to differences between proximal and distal segments in smooth muscle density, endothelial function, and vasomotor response to vasoactive stimuli.
Conclusions
Our results showed that l-arginine dependent coronary segment vasodilatation by the antioxidant vitamin C is augmented in patients with atherosclerosis, implying a synergistic action of this combination on nitric oxide bioactivity.
Abbreviations
eNOS, endothelial nitric oxide synthase
LDL, low density lipoprotein
NOS, nitric oxide synthase
REFERENCES
- 1.Palmer RMJ, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature 1988;333:664–6. [DOI] [PubMed] [Google Scholar]
- 2.Casino PR, Kilcoyne CM, Quyyimi AA, et al. Investigation of decreased availability of nitric oxide precursor as the mechanism responsible for impaired endothelium dependent vasodilation in hypercholesterolemic patients. J Am Coll Cardiol 1994;23:844–50. [DOI] [PubMed] [Google Scholar]
- 3.Campisi R, Czernin J, Schoder H, et al. L-Arginine normalizes coronary vasomotion in long term smokers. Circulation 1999;99:491–7. [DOI] [PubMed] [Google Scholar]
- 4.Chauhan A, More RS, Mullins PA, et al. Aging-associated endothelium dysfunction in humans is reversed by L-arginine. J Am Coll Cardiol 1996;28:1796–804. [DOI] [PubMed] [Google Scholar]
- 5.Higashi Y, Oshima T, Sasaki S, et al. Angiotensin-converting enzyme inhibition, but not calcium antagonism, improves a response of the renal vasculature to L-arginine in patients with essential hypertension. Hypertension 1998;32:16–24. [DOI] [PubMed] [Google Scholar]
- 6.Tousoulis D, Tentolouris C, Crake T, et al. Effects of L- and D-arginine on the basal tone of human diseased coronary arteries and their responses to substance P. Heart 1999;81:505–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tentolouris C, Tousoulis D, Toutouzas P, et al. Effects of acute L-arginine administration in coronary atherosclerosis response. Circulation 1999;99:1646–9. [PubMed] [Google Scholar]
- 8.Tentolouris C, Tousoulis D, Davies G, et al. Serum cholesterol level, cigarette smoking, and vasomotor responses to L-arginine in narrowed epicardial coronary arteries. Am J Cardiol 2000;85:500–3. [DOI] [PubMed] [Google Scholar]
- 9.Tousoulis D, Davies G, Tentolouris C, et al. Coronary stenosis dilation induced by L-arginine. Lancet 1997;349:1812–3. [DOI] [PubMed] [Google Scholar]
- 10.Egashira K, Hirooka Y, Kuga T, et al. Effects of L-arginine supplementation on endothelium dependent coronary vasodilation in patients with angina pectoris and normal coronary angiograms. Circulation 1996;94:130–4. [DOI] [PubMed] [Google Scholar]
- 11.Boger RH, Bode-Boger SM, Thiele W, et al. Restoring vascular nitric oxide formation by L-arginine improves the symptoms of intermittent claudication in patients with peripheral arterial occlusive disease. J Am Coll Cardiol 1998;32:1336–44. [DOI] [PubMed] [Google Scholar]
- 12.Hamon M, Vallet B, Bauters C, et al. Long-term administration of L-arginine reduces intimal thickening and enhances endothelium-dependent acetylcholine-induced relaxation after arterial injury. Circulation 1994;90:1357–62. [DOI] [PubMed] [Google Scholar]
- 13.Szabo G, Bahrle S, Batkai S, et al. L-arginine: effect on reperfusion injury after heart transplantation. World J Surg 1998;32:9–15. [DOI] [PubMed] [Google Scholar]
- 14.Ohara Y, Peterson TE, Harrison DG. Hypercholesterolemia increases endothelial superoxide anion production. J Clin Invest 1993;91:2546–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gryglewski RJ, Palmer RM, Moncada S. Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature 1986;320:454–6. [DOI] [PubMed] [Google Scholar]
- 16.Frei B, England L, Ames BN. Ascorbate is an outstanding anti-oxidant in human blood plasma. Proc Natl Acad Sci USA 1989;86:6377–81.2762330 [Google Scholar]
- 17.Ting HH, Timimi FK, Haley EA, et al. Vitamin C improves endothelium-dependent vasodilation in forearm resistance vessels of humans with hypercholesterolemia. Circulation 1997;95:2617–22. [DOI] [PubMed] [Google Scholar]
- 18.Heitzer T, Just H, Munzel T. Antioxidant vitamin C improves endothelial dysfunction in chronic smokers. Circulation 1996;94:6–9. [DOI] [PubMed] [Google Scholar]
- 19.Solzbach U, Hornig B, Jeserich M, et al. Vitamin C improves endothelial dysfunction of epicardial coronary arteries in hypertensive patients. Circulation 1997;96:1513–9. [DOI] [PubMed] [Google Scholar]
- 20.Ting HH, Timimi FK, Boles KS, et al. Vitamin C improves endothelium-dependent vasodilation in patients with non-insulin-dependent diabetes mellitus. J Clin Invest 1996;97:22–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chambers JC, McGregor A, Jean-Marie J, et al. Demonstration of rapid onset vascular endothelial dysfunction after hyperhomocysteinemia: an effect reversible with vitamin C therapy. Circulation 1999;99:1156–60. [DOI] [PubMed] [Google Scholar]
- 22.Levine GN, Frei B, Koulouris SN, et al. Ascorbic acid reverses endothelial vasomotor dysfunction in patients with coronary artery disease. Circulation 1996;93:1107–13. [DOI] [PubMed] [Google Scholar]
- 23.Meister A. Glutathione-ascorbic acid antioxidant system in animals. J Biol Chem 1994;269:9397–400. [PubMed] [Google Scholar]
- 24.Reiber JH, Serruys PW, Kooijman CJ, et al. Assessment of short-, medium, and long-term variations in arterial dimensions from computer-assisted quantitation of coronary cineangiograms. Circulation 1985;71:280–8. [DOI] [PubMed] [Google Scholar]
- 25.Creager MA, Gallagher SM, Girerd XJ, et al. L-arginine improves endothelium-dependent vasodilation in hypercholesterolemic humans. J Clin Invest 1992;90:1248–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Quyyumi A. Does acute improvement of endothelial dysfunction in coronary artery disease improve myocardial ischemia? A double-blind comparison of parenteral D- and L-arginine. J Am Coll Cardiol 1998;32:904–11. [DOI] [PubMed] [Google Scholar]
- 27.Miyazaki H, Matsuoka H, Cooke JP, et al. Endogenous nitric oxide synthase inhibitor: a novel marker of atherosclerosis. Circulation 1999;99:1141–6. [DOI] [PubMed] [Google Scholar]
- 28.Darley-Usmar VM, Hog N, O’Leavy VJ, et al. The simultaneous generation of superoxide and nitric oxide can initiate lipid peroxidation in human low density lipoprotein. Free Radic Res Commun 1992;17:9–20. [DOI] [PubMed] [Google Scholar]
- 29.Myers PR, Wright TF, Tanner MA, et al. The effects of native LDL and oxidized LDL on EDRF bioactivity and nitric oxide production in vascular endothelium. J Lab Clin Med 1994;124:672–83. [PubMed] [Google Scholar]
- 30.Keaney JF Jr, Guo Y, Cunningham D, et al. Vascular incorporation of alpha-tocopherol prevents endothelial dysfunction due to oxidized LDL by inhibiting protein kinase C stimulation. J Clin Invest 1996;98:386–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vergnani L, Hatrik S, Ricci F, et al. Effect of native and oxidized low-density lipoprotein on endothelial nitric oxide and superoxide production: key role of L-arginine availability. Circulation 2000;101:1261–6. [DOI] [PubMed] [Google Scholar]
- 32.O’Donnell VB, Chumley PH, Hogg N, et al. Nitric oxide inhibition of lipid peroxidation: kinetics of reaction with lipid peroxyl radicals and comparison with alpha-tocopherol. Biochemistry 1997;36:15216–23. [DOI] [PubMed] [Google Scholar]
- 33.Boger RH. The emerging role of asymmetric dimethylarginine as a novel cardiovascular risk factor. Cardiovasc Res 2003;59:824–33. [DOI] [PubMed] [Google Scholar]
- 34.Jackson TS, Xu A, Vita JA, et al. Ascorbate prevents the interaction of superoxide and nitric oxide only at very high physiological concentrations. Circ Res 1998;83:916–22. [DOI] [PubMed] [Google Scholar]
- 35.Heller R, Munscher-Paulig F, Grabner R, et al. L-ascorbic acid potentiates nitric oxide synthesis in endothelial cells. J Biol Chem 1999;274:8254–60. [DOI] [PubMed] [Google Scholar]
- 36.Takahashi S, Mendelsohn ME. Calmodulin-dependent and -independent activation of endothelial nitric-oxide synthase by heat shock protein 90. J Biol Chem 2003;278:9339–44. [DOI] [PubMed] [Google Scholar]