Abstract
Aims: Tumour cell growth results from a disturbance in the balance between the rate of proliferation and cell death. In this study, proteins involved in the regulation of cell cycle arrest and apoptosis were studied as possible factors responsible for uncontrolled cell growth in colorectal cancer.
Methods: The expression of proteins involved in these processes was investigated in 48 metastases from patients with colorectal cancer and compared with eight normal colon mucosa samples and 14 primary tumours. Both primary tumours and metastases were obtained from eight patients. The expression of thymidylate synthase (TS), p53, retinoblastoma protein (Rb), Fas receptor, Fas ligand, bcl-2, mcl-1, bax, and bcl-x was measured using immunohistochemistry. Proliferation was determined by Ki67 staining, whereas apoptosis was assessed by M30 immunostaining, which recognises cleaved cytokeratin 18.
Results: In the limited number of cases in which paired comparisons were possible, the expression of TS and Ki67 was significantly higher in metastases than in the matched primary tumour samples (p = 0.014 and 0.016, respectively), whereas Rb expression was lower in metastases than in primary tumours (p = 0.024). Fas receptor expression was high in normal mucosa but absent in primary tumours and metastases, whereas the opposite was seen for p53. The expression of bax, mcl-1, and bcl-x in normal mucosa was more apical than that seen in malignant cells, where a more diffuse expression pattern was seen (p < 0.04). Apoptosis was more abundant in primary tumours than in metastases.
Conclusions: These results demonstrate that proliferation and apoptosis are disturbed during colorectal cancer progression, and this is accompanied by loss of Rb and Fas expression, the accumulation of p53 and TS, and changes in the expression patterns of bax, mcl-1, and bcl-xl.
Keywords: normal mucosa, colorectal tumours, cell cycle, apoptosis
Colorectal cancer is the third leading cause of cancer deaths in Western countries. Between 20% and 50% of patients with colorectal cancer will die within five years of diagnosis,1 usually as the result of extensive metastatic disease. At the time of diagnosis, 20% of patients have metastases in the liver, the predominant metastatic site for colorectal cancer.2 Systemic 5-fluorouracil (5-FU) based chemotherapy has become the most widely used treatment, with response rates of approximately 20%,3 although more recently irinotecan and oxaliplatin have been incorporated into first line chemotherapy regimens.4–6, The active metabolite of 5-FU, FdUMP, inhibits thymidylate synthase (TS), the essential enzyme for the conversion of deoxyuridine-5`-monophosphate (dUMP) to deoxythymidine-5`-monophosphate (dTMP), and therefore for DNA synthesis.7,8
“Between 20% and 50% of patients with colorectal cancer will die within five years of diagnosis, usually as the result of extensive metastatic disease”
Activation of specific oncogenes or the loss of tumour suppressor genes is associated with tumour aggressiveness and poor clinical outcome.9 Tumour kinetic studies suggest that tumour growth does not only result from increased rates of cell proliferation but also from decreased rates of apoptosis.10,11 Mutations in the tumour suppressor gene p53 are seen frequently in colorectal cancer and are thought to be a late event in the multistep tumorigenic process of colorectal cancer.12 In addition, the protooncogene bcl-2, an inhibitor of apoptosis, appears to be involved in colorectal tumorigenesis, thereby facilitating tumour cell progression.13 Other factors that regulate the processes of cell cycle progression (the retinoblastoma protein; Rb) and apoptosis (bax bcl-xl, mcl-1, Fas receptor, and Fas ligand) might therefore also contribute to tumour progression.
The tumour suppressor gene Rb can form a complex with E2F, thereby inhibiting its transcriptional activity. Phosphorylation of Rb by specific cyclin dependent kinases initiates the release of E2F, which subsequently initiates cell cycle progression.14 p53 can activate the expression of bax and the Fas receptor15,16 upon activation. Bax stimulates apoptosis, whereas bcl-2, bcl-xl, and mcl-1 protect cells against apoptosis.17,18 The death receptor, Fas, can be activated by the Fas ligand, resulting in the activation of caspase 8 and caspase 3,19,20 which induce apoptotic cell death.21 Deregulation of these factors may lead to deregulation of either cell cycle arrest or apoptosis, both of which result in uncontrolled cell growth.
In our study, we determined proliferation and apoptosis in normal colon mucosa, primary tumours, and liver metastases of patients with colorectal cancer and related this to the expression of cell cycle and apoptosis associated proteins.
PATIENTS AND METHODS
Patients
Tissue samples from primary colorectal carcinomas, liver metastases, and specimens of normal colon mucosa were taken from 63 patients with advanced colorectal cancer. Biopsies were obtained from both primary tumours and metastases in eight patients. From six other patients only primary tumour material was available, and from 41 patients only liver metastases could be retrieved. Normal colon mucosa (taken at a distance of 10 cm from the tumour) was obtained from an additional eight patients. Thirty six patients with metastases received a bolus injection of 5-FU before the biopsy was taken. As a result of this pretreatment, the expression of TS and Ki67 was downregulated in some metastases,22 but this did not influence our results. Informed consent was obtained from all patients for acquisition of tumour tissue for the analysis of protein expression.
Immunohistochemical staining
Biopsy specimens of normal colon mucosa, primary colon tumours, and liver metastases were immediately frozen in liquid nitrogen and subsequently stored at −80°C. Frozen tissue sections (4 μm) were mounted on poly-L-lysine (0.1%) coated slides and immunohistochemistry was performed as described previously.22 Briefly, slides were fixed for 10 minutes with 100% acetone (for TS, p53, Ki67, Fas receptor, Fas ligand, and bcl-2) or 4% paraformaldehyde (for Rb, bcl-xl, bax, and mcl-1). After blocking of endogenous peroxidase and biotin, slides were incubated for one hour for TS (R30; Dr Aherne, Sutton, UK), p53 (DO7; Dako, Glostrup, Denmark), Ki67 (polyclonal antibody; Dako), Fas receptor (DX2; Oncogene, Cambridge, USA), Fas ligand (clone 33; Transduction, Lexington, USA), M30 (M30 Cytodeath; Boehringer Mannheim, Mannheim, Germany), and bcl-2 (clone 124; Dako) and overnight for Rb (Rb-1; Novocastra, Newcastle, UK), bcl-xl (polyclonal antibody; Dako), mcl-1 (polyclonal antibody; Dako), and bax (polyclonal antibody; Dako). Primary antibodies were detected by the addition of biotinylated antimouse or antirabbit secondary antibodies for 30 minutes. The slides were developed by incubation for one hour with sABC-HRP (streptavidin-biotin conjugated horseradish peroxidase) and staining was visualised by diaminobenzidine (DAB; 0.5 mg/ml) with 0.025% H2O2 for three minutes. For Fas ligand an additional amplification step using the CARD (catalysed reporter deposition) enhancement kit (Dako) was needed.23 All slides were counterstained with haematoxylin. Negative controls were performed for each sample by omitting the primary antibody.
Immunohistochemical scoring
All slides were evaluated by an experienced pathologist (EB) and two investigators (DD and HB). Staining for p5313,24 and bcl-2 13,25 was evaluated as the proportion of positive normal colon mucosa or tumour epithelial cells in two categories (≤ 25%, > 25%). There were four categories for Rb24 and Ki67 staining (< 25%, 25–50%, 50–75%, and > 75%). The Rb antibody detects both hyperphosphorylated and hypophosphorylated Rb protein. For TS26,27 and Fas ligand28,29 the intensity of the staining was taken into account because these proteins were generally expressed in almost all cells. Intensity was divided into three categories: low, intermediate, or high. Bax,30 mcl-1, and bcl-xl30 were evaluated by their staining pattern: diffuse or perinuclear staining. The Fas receptor was evaluated as positive or negative. In all cases the interobserver variability was < 10%.
Positive controls included normal colon mucosa (for the Fas receptor, Rb, Ki67, mcl-1, bax, and bcl-xl), tonsils (for the Fas ligand, bcl-2, and bax), tumour cells with high protein expression (for all antibodies tested), and the internal controls in the samples such as normal liver (for Fas receptor and Rb) and lymphocytes (for bcl-2).
Statistical methods
Statistical evaluation was performed using the non-parametric Wilcoxon and Mann-Whitney U ranking tests. All values were based on two tailed statistical analysis. The first test was used to evaluate significant differences in protein expression between eight paired primary tumours and liver metastases from the same patients. The Mann-Whitney U test was used to test significant differences in protein expression between all primary tumours and liver metastases in 55 patients with colorectal cancer. A p value of < 0.05 was considered significant. All statistical procedures were carried out with SPSS 9.0 (SPSS Inc, Chicago, Illinois, USA).
RESULTS
Normal colon mucosa versus malignant colon tissue
Differences in the rates of proliferation and apoptosis and expression of cell cycle and apoptosis markers between normal and tumour cells were studied by comparing normal colon mucosa with primary and metastatic tumour biopsies. In normal mucosa, the distribution of various proteins showed a distinct pattern of spatial polarity within the crypt. Therefore, normal colon mucosa was divided into two regions: the lower region of colonic crypts containing the stem cells and the proliferative compartment and the middle and upper part of colonic crypts with absorptive epithelium and goblet cells. Considerable differences were found between normal colon mucosa and malignant colon tissue (tables 1 and 2). In contrast to normal mucosa, the proliferation marker Ki67 was expressed in more than 85% of all tumour biopsies in a random distribution pattern (p < 0.01; fig 1A–C). Apoptosis as determined by M30 staining, identifying cleaved cytokeratin 18, one of the target proteins of activated caspase 3. M30 staining was detected in the upper crypt cells in normal mucosa and randomly distributed in cells from primary tumours and metastases (fig 1D–F).
Table 1.
Staining pattern of cell cycle markers in normal colon mucosa, primary tumours, and liver metastases of patients with colorectal cancer
| Normal mucosa (n=8) | ||||
| Antigen | Basal crypt compartment | Upper 2/3 of the crypts | Primary tumours (n=14) | Metastases (n=48) |
| Ki67 | ||||
| <25% | 0 | 8 | 2 | 3 |
| 26%–50% | 0 | 0 | 7 | 12 |
| 51%–75% | 0 | 0 | 4 | 12 |
| >75% | 8 | 0 | 1 | 20 |
| Rb | ||||
| <25% | 0 | 0 | 0 | 3 |
| 26%–50% | 0 | 0 | 0 | 3 |
| 51%–75% | 0 | 0 | 0 | 8 |
| >75% | 8 | 8 | 14 | 34 |
| p53 | ||||
| ≤25% | 8 | 8 | 2 | 19 |
| >25% | 0 | 0 | 12 | 29 |
| TS | ||||
| Low | 8 | 8 | 2 | 10 |
| Intermediate | 0 | 0 | 6 | 15 |
| High | 0 | 0 | 4 | 21 |
The number of patients for the intensity of the staining or percentage of positive tumour cells is presented. In some metastases or primary tumour samples it was not possible to evaluate protein expression. Significant differences were found between normal mucosa and tumour cells (from primary tumours and metastases) for p53 (p < 0.005), Ki67 (p < 0.01), and TS (p < 0.001). For Ki67 (p = 0.011) and Rb (p = 0.024) significant differences between primary tumours and metastases were found.
Rb, retinoblastoma protein; TS, thymidylate synthase.
Table 2.
Staining pattern of apoptosis markers in normal colon mucosa, primary tumours, and liver metastases of patients with colorectal cancer
| Normal mucosa (n=8) | ||||
| Antigen | Basal crypt compartment | Upper 2/3 of the crypts | Primary tumours (n=14) | Metastases (n=48) |
| Bcl-2 | ||||
| ≤25% | 0 | 8 | 14 | 42 |
| >25% | 8 | 0 | 0 | 5 |
| Fas receptor | ||||
| Positive | 8 | 8 | 13 | 29 |
| Negative | 0 | 0 | 1 | 15 |
| Fas ligand | ||||
| Low | 8 | 0 | 5 | 15 |
| Intermediate | 0 | 8 | 7 | 19 |
| High | 0 | 0 | 1 | 11 |
| Bax | ||||
| Diffuse | 0 | 0 | 8 | 32 |
| Apical | 8 | 8 | 5 | 14 |
| mcl-1 | ||||
| Diffuse | 0 | 0 | 9 | 29 |
| Apical | 8 | 8 | 4 | 17 |
| Bcl-xl | ||||
| Diffuse | 0 | 0 | 7 | 29 |
| Apical | 8 | 8 | 6 | 18 |
The number of patients for the intensity of the staining or percentage of positive tumour cells is presented. In some metastases or primary tumour samples it was not possible to evaluate protein expression. Significant differences were found between normal mucosa and tumour cells (from primary tumours and metastases) for Fas receptor (p < 0.002), bax (p < 0.02), bcl-xl (p < 0.04), mcl-1 (p < 0.005), bcl-2 (when expression is > 25%; p < 0.001), and Fas ligand (when expression is low; p < 0.001). No significant differences were found between primary tumours and metastases.
Figure 1.
Immunostaining of normal and malignant colon tissue for Ki67, M30, and bcl-xl. Frozen tissue sections were immunostained with polyclonal antibodies against Ki67 and mcl-1. Antibodies were detected after visualisation with diaminobenzidine, which produced a brown colour. Nuclei were counterstained with haematoxylin. (A) Ki67 (original magnification, ×200) is expressed in the lower region of a crypt in normal colon mucosa, whereas expression is lower in (B) primary tumours than in (C) matched metastases. (D) Apoptosis as determined by M30 staining was detected in the upper crypt cells in normal mucosa (original magnification, ×100) and randomly distributed in cells from (E) primary tumours and (F) metastases (original magnification, ×400). (G) Normal colon mucosa cells express bcl-xl (original magnification, ×400) in the cytoplasm with an additional apical accumulation. (H) Apical staining was visible in only 14 of 46 metastases, whereas in the others (I) diffuse bcl-xl expression was seen.
TS expression was much lower in normal epithelial cells than in tumour cells (p < 0.001), as published previously.31 The tumour suppressor protein p53 was not expressed in normal mucosa, whereas 66% of tumour samples were p53 positive (p < 0.005). In contrast, the expression of the death receptor Fas was high in all mucosal cells but only a few tumour samples were Fas positive (p < 0.002). Although Fas ligand expression of lower crypt cells was low, the upper two thirds of normal epithelial crypt cells, in addition to tumour cells, expressed intermediate amounts of Fas ligand. In addition, striking differences were found in the pattern of mcl-1, bcl-xl, and bax staining. The typical apical expression found in normal mucosa was much less pronounced in malignant cells, in which diffuse expression was more common (p < 0.04). Tumour samples that tended to be more differentiated also showed apical expression (fig 1G–I). No differences in Rb and bcl-2 expression were seen between non-malignant and malignant biopsy specimens.
Primary tumours versus liver metastases
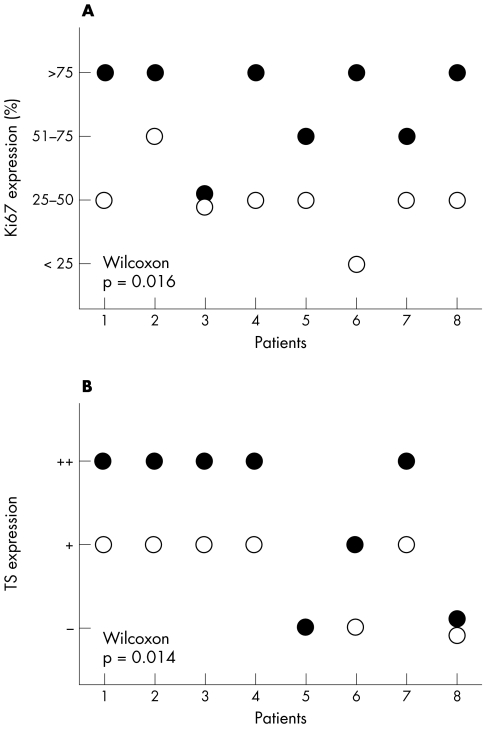
Eight patients biopsy specimens were obtained from both the primary tumour and liver metastasis. Significant differences were found between tumour cells from the primary tumour and those from metastases for Ki67 and TS expression only (fig 2) (p = 0.016 and 0.014, respectively). For all other proteins no differences were detected. Ki67 protein expression was significantly higher in metastases than in the corresponding primary tumour. In five metastases, more than 75% of the tumour cells were positive for Ki67, whereas in most primary tumours less than 50% of tumour cells were positive (figs 1B,C and 2A). TS protein expression was higher in six metastases compared with the corresponding primary tumour. Five metastases had high expression of TS, whereas five primary tumours had intermediate expression of TS (fig 2B).
Figure 2.
Ki67 and thymidylate synthase (TS) expression in primary tumours and matched liver metastases. In eight patients with both a primary tumour (open circle) and a liver metastasis (closed circle) a significant difference in Ki67 (A; p = 0.016; A) and TS (B; p = 0.014) protein expression was found with the Wilcoxon signed rank test. Ki67 was evaluated by scoring the proportion of positive tumour cells and TS by scoring the intensity of the staining (low (−), intermediate (+), and high (++)). In one case, TS expression could not be evaluated in the primary tumour.
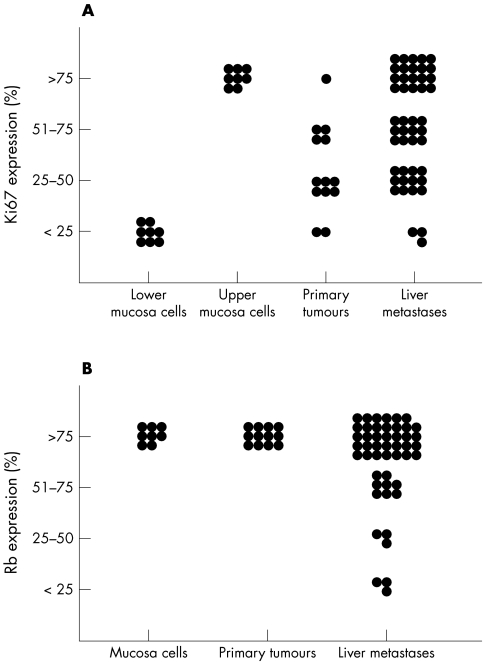
When all biopsy specimens were evaluated, a similar pattern was found for Ki67 expression. Only one of the 14 primary tumours had more than 75% positive tumour cells, whereas 20 of the 47 liver metastases specimens were positive. The overall expression of Ki67 was significantly lower in primary tumours than in metastases (fig 3A; p = 0.011). In addition, the expression of Rb was higher in primary tumours than in metastases. In all primary tumours, more than 75% of the tumour cells expressed Rb, whereas Rb expression was lower than 75% in one third of the liver metastases (fig 3B; p = 0.024). In contrast to proliferation, the rate of apoptosis was increased in primary tumours compared with metastases.
Figure 3.
Ki67 and retinoblastoma (Rb) expression in normal mucosa, primary tumours, and liver metastases. Expression of Ki67 in both the upper and lower crypt cells of the normal mucosa was significantly different to its expression in cells from primary tumours and metastases (p < 0.01; Mann-Whitney U rank test). No significant differences were found for Rb between normal mucosa and tumour cells. In 48 liver metastases and 14 primary tumours, a significant difference in Ki67 (A; p = 0.011) and Rb (B; p = 0.024) protein expression was found. Ki67 and Rb were evaluated by scoring the proportion of positive tumour cells.
However, no significant difference was found for the expression of TS or for the other proteins (table 2). Although only two of the 14 primary tumours were p53 negative compared with 19 of the 48 metastases, no significant difference was found between primary tumours and metastases for p53 staining. These results are in line with the p53 results obtained by Belluco and colleagues32 and De Jong et al.33 The expression of the Fas receptor was significantly higher in liver metastases than in primary tumours, but this was the result of the administration of 5-FU to 36 of the 44 patients, as we have described previously.22 No differences were found in untreated samples.
DISCUSSION
Our study demonstrates that proliferation and apoptosis are disturbed during colorectal cancer progression, and that this is accompanied by the loss of Rb and Fas expression, the accumulation of p53 and TS, and changes in the expression patterns of bax, mcl-1, and bcl-xl.
The role of proteins involved in apoptosis and the cell cycle has been studied previously in the adenoma–carcinoma sequence of colorectal tumorigenesis.12,13,31,34,35 In line with these studies, we found that the expression of p53, Ki67, and TS was higher in tumour samples than in normal colonic mucosa. Krajewska et al showed that in colon carcinomas bcl-x expression was increased and mcl-1 expression was decreased compared with normal mucosa or non-malignant adenomas.35 No differences were found for the expression of bax. In our study, differences were found in the staining pattern of these proteins; bax, mcl-1, and bcl-x staining was limited to apical areas in normal mucosa cells, whereas in 50–75% of tumour samples diffuse expression was observed. In addition, in metastases with a more differentiated phenotype apical staining was found. This suggests that in undifferentiated tumour cells with diffuse staining, the inhibition of apoptosis at the level of the mitochondria was disturbed, possibly resulting in enhanced tumour growth.
“The progression of primary tumours to metastases resulted in an increase in the rate of proliferation, as measured by Ki67, and a decrease in the rate of apoptosis using M30 staining”
A role in tumour progression seems very likely for another apoptosis related protein, the Fas receptor, because its expression was high in normal mucosa and negative in tumour cells.36 The absence of Fas would enable tumour cells to evade the immune destruction mediated by cytotoxic T cells (CTLs). In this way, CTLs expressing both Fas receptor and Fas ligand are unable to kill tumour cells via Fas mediated apoptosis. The high expression of the Fas ligand in tumour cells might be another immune escape mechanism by inducing apoptosis in CTLs.37,38
The progression of primary tumours to metastases resulted in an increase in the rate of proliferation, as measured by Ki67, and a decrease in the rate of apoptosis using M30 staining. These results are in line with data found in breast cancer progression from ductal carcinoma in situ to poorly differentiated invasive carcinoma.39 Although the expression of both cell cycle and apoptosis related proteins changes when normal colon mucosa cells progress into malignant cells, no differences were found in the expression of apoptosis related proteins between primary tumours and metastases. In contrast, increased expression of TS, a key enzyme during DNA synthesis, and of the proliferation marker Ki67, was seen in metastases compared with primary tumours, pointing to a deregulation of cell cycle mechanisms. Comparable data have previously been described for Ki6733 and for another cell cycle related protein, the proliferating nuclear antigen staining.40 The low Rb expression in metastases in comparison with primary tumours could be an important factor because this might result in increased concentrations of free E2F1 protein,14 which stimulates the transcription of cell cycle progression genes, including TS.41
In conclusion, these results provide a better insight into the growth and cell death kinetics of tumours, and suggest that proliferation and apoptosis are disturbed during colorectal cancer progression, which was accompanied by the loss of Rb and Fas expression, the accumulation of p53 and TS, and changes in the expression patterns of bax, mcl-1, and bcl-xl.
Take home messages .
Both proliferation and apoptosis are disturbed during colorectal cancer progression
Loss of Rb and Fas expression, the accumulation of p53 and TS, and changes in the expression patterns of bax, mcl-1, and bcl-xl also accompany colorectal cancer progression
Acknowledgments
This study was supported by a grant from the Dutch Cancer Society (VU 96–1240). We thank Dr PD Bezemer for his statistical advice and the department of surgery for help in obtaining samples (Professor S Meyer)
Abbreviations
CTL, cytotoxic lymphocyte
5FU, 5-fluorouracil
Rb, retinoblastoma protein
TS, thymidylate synthase
REFERENCES
- 1.Ballantyne GH, Quin J. Surgical treatment of liver metastases in patients with colorectal cancer. Cancer 1993;71:4252–66. [DOI] [PubMed] [Google Scholar]
- 2.Weiss L, Grundmann E, Torhorst J, et al. Hematogenous metastatic patterns in colonic carcinoma: an analysis of 1541 necropsies. J Pathol 1986;150:195–203. [DOI] [PubMed] [Google Scholar]
- 3.Advanced colorectal cancer meta-analysis project. Modulation of fluorouracil by leucovorin in patients with advanced colorectal cancer: evidence in terms of response rate. J Clin Oncol 1992;10:896–903. [DOI] [PubMed] [Google Scholar]
- 4.Peters GJ, Köhne CH. Fluoropyrimidines as antifolate drugs. In: AL Jackman, ed. Anticancer drug development guide: antifolate drugs in cancer therapy. Totowa: Humana Press, 1999:101–45.
- 5.Sobrero A, Kerr D, Glimelius B, et al. New directions in the treatment of colorectal cancer: a look to the future. Eur J Cancer 2000;36:559–66. [DOI] [PubMed] [Google Scholar]
- 6.Schmoll HJ, Buchele T, Grothey A, et al. Where do we stand with 5-fluorouracil? Semin Oncol 1999;26:589–605. [PubMed] [Google Scholar]
- 7.Spears CP, Gustavsson BG, Berne M, et al. Mechanisms of innate resistance to thymidylate synthase inhibition after 5-fluorouracil. Cancer Res 1988;48:5894–900. [PubMed] [Google Scholar]
- 8.Pinedo HM, Peters GJ. Fluorouracil: biochemistry and pharmacology. J Clin Oncol 1988;10:1653–64. [DOI] [PubMed] [Google Scholar]
- 9.Weinberg RA. The integration of molecular genetics into cancer management. Cancer 1992;71:1653–8. [DOI] [PubMed] [Google Scholar]
- 10.Deschner EE. Kinetics of normal, preneoplastic, and neoplastic colonic epithelium. San Diego: Academic Press, 1990.
- 11.Bedi A, Pasricha PJ, Akhtar AJ, et al. Inhibition of apoptosis during development of colorectal cancer. Cancer Res 1995;55:1811–16. [PubMed] [Google Scholar]
- 12.Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell 1990;61:759–67. [DOI] [PubMed] [Google Scholar]
- 13.Sinicrope FA, Ruan SB, Cleary KR, et al. Bcl-2 and p53 oncoprotein expression during colorectal tumorigenesis. Cancer Res 1995;55:237–41. [PubMed] [Google Scholar]
- 14.Weinberg RA. The retinoblastoma protein and cell cycle control. Cell 1995;81:323–30. [DOI] [PubMed] [Google Scholar]
- 15.Miyashita T, Krajewski M, Krajewska M, et al. Tumour suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene 1994;9:1799–805. [PubMed] [Google Scholar]
- 16.Müller M, Wilder S, Bannasch D, et al. P53 activates the CD95 (APO-1/Fas) gene in response to DNA damage by anticancer drugs. J Exp Med 1998;188:2033–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Korsmeyer SJ. Regulators of cell death. Trends Genet 1995;11:101–5. [DOI] [PubMed] [Google Scholar]
- 18.Allen RT, Cluck MW, Agrawal DK. Mechanisms controlling cellular suicide: role of bcl-2 and caspases. Cell Mol Life Sci 1998;54:427–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Salvesen GS, Dixit VM. Caspases: intracellular signaling by proteolysis. Cell 1997;91:443–6. [DOI] [PubMed] [Google Scholar]
- 20.Nagata S. Apoptosis by death factor. Cell 1997;88:355–65. [DOI] [PubMed] [Google Scholar]
- 21.Nagata S, Goldstein P. The Fas death factor. Science 1995;267:1449–56. [DOI] [PubMed] [Google Scholar]
- 22.Backus HHJ, Dukers DF, van Groeningen CJ, et al. 5-Fluorouracil induced Fas upregulation associated with apoptosis in liver metastases of colorectal cancer patients. Ann Oncol 2001;12:209–16. [DOI] [PubMed] [Google Scholar]
- 23.Bobrow MN, Harris TD, Shaughnessy KJ, et al. Catalyzed reporter deposition, a novel method of signal amplification. I. Application for immunoassays. J Immunol Methods 1989;125:279–85. [DOI] [PubMed] [Google Scholar]
- 24.Poller DN, Baxter KJ, Shepherd NA. p53 and Rb1 protein expression: are they prognostically useful in colorectal cancer? Br J Cancer 1997;75:87–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sinicrope FA, Hart J, Hsu HA, et al. Apoptotic and mitotic indices predict survival rates in lymph node-negative colon carcinomas. Clin Cancer Res 1999;5:1793–804. [PubMed] [Google Scholar]
- 26.Davies MM, Johnston PG, Kaur S et al. Colorectal liver metastasis thymidylate synthase staining correlates with response to hepatic arterial floxuridine. Clin Cancer Res 1999;5:325–8. [PubMed] [Google Scholar]
- 27.Lenz HJ, Danenberg KD, Leichman CG, et al. p53 and thymidylate synthase expression in untreated stage II colon cancer: associations with recurrence, survival, and site. Clin Cancer Res 1998;4:1227–34. [PubMed] [Google Scholar]
- 28.Yoong KF, Afford SC, Randhawa S, et al. Fas/Fas ligand interaction in human colorectal hepatic metastases. Am J Pathol 1999;154:693–703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mann B, Gratchev A, Böhm C, et al. FasL is more frequently expressed in liver metastases of colorectal cancer than in matched primary carcinomas. Br J Cancer 1999;79:1262–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ogura E, Senzaki H, Yamamoto D, et al. Prognostic significance of bcl-2, bcl-xL/S, bax and bak expressions in colorectal carcinomas. Oncol Rep 1999;6:365–9. [DOI] [PubMed] [Google Scholar]
- 31.Van Triest B, Loftus BM, Pinedo HM, et al. Thymidylate synthase expression in patients with colorectal carcinoma using a polyclonal thymidylate synthase antibody in comparison to the TS 106 monoclonal antibody. J Histochem Cytochem 2000;48:755–60. [DOI] [PubMed] [Google Scholar]
- 32.Belluco C, Guillem JG, Kemeny N, et al. p53 nuclear protein overexpression in colorectal cancer: a dominant predictor of survival in patients with advanced hepatic metastases. J Clin Oncol 1996;14:2696–701. [DOI] [PubMed] [Google Scholar]
- 33.De Jong KP, Stellema R, Karrenbeld A, et al. Clinical relevance of transforming growth factor α, epidermal growth factor receptor, p53, and Ki67 in colorectal liver metastases and corresponding primary tumors. Hepatology 1998;28:971–9. [DOI] [PubMed] [Google Scholar]
- 34.Hague A, Moorghen M, Hicks D, et al. Bcl-2 expression in human colorectal adenomas and carcinomas. Oncogene 1994;9:3367–70. [PubMed] [Google Scholar]
- 35.Krajewska M, Moss SF, Krajewska S, et al. Elevated expression of bcl-x and reduced bak in primary colorectal adenocarcinomas. Cancer Res 1996;56:2422–7. [PubMed] [Google Scholar]
- 36.Möller P, Koretz K, Leithäuser F, et al. Expression of apo-1 (CD-95), a member of the NGF/TNF receptor superfamily, in normal and neoplastic colon epithelium. Int J Cancer 1994;57:371–7. [DOI] [PubMed] [Google Scholar]
- 37.Shiraki K, Tsuji N, Shioda T, et al. Expression of Fas ligand in liver metastases of human colonic adenocarcinomas. Proc Natl Acad Sci U S A 1997;94:6420–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.O'Connell J, Bennett MW, O'Sullivan GC, et al. Fas ligand expression in primary colon adenocarcinomas: evidence that the Fas counterattack is a prevalent mechanism of immune evasion in human colon cancer. J Pathol 1998;186:240–6. [DOI] [PubMed] [Google Scholar]
- 39.Mommers ECM, van Diest PJ, Leonhart AM, et al. Balance of cell proliferation and apoptosis in breast carcinogenesis. Breast Cancer Res Treat 1999;58:163–9. [DOI] [PubMed] [Google Scholar]
- 40.Yang JL, Ow KT, Russell PJ, et al. Higher expression of oncoproteins c-myc, c-erb B-2/neu, PCNA and p53 in metastasizing colorectal cancer than in nonmetastasizing tumours. Ann Surg Oncol 1996;3:574–9. [DOI] [PubMed] [Google Scholar]
- 41.Degregori J, Kowalik T, Nevins JR. Cellular targets for activation by the E2F1 transcription factor include DNA synthesis- and G1/S-regulatory genes. Mol Cell Biol 1995;15:4215–24. [DOI] [PMC free article] [PubMed] [Google Scholar]