Abstract
Background/Aims: The clinical definition of an atypical naevus (“dysplastic naevus” or “naevus with architectural disorder and cytological atypia of melanocytes”) stresses size larger than 5 mm in diameter as a major diagnostic criterion. Because malignant melanomas and their precursors may arise in smaller lesions, a histological study of melanocytic lesions smaller than 4 mm in diameter was conducted to evaluate their histological appearance.
Methods: Two hundred and sixty one naevi smaller than 4 mm in diameter were collected and characterised by histological examination into benign naevi without architectural disorder and naevi with architectural disorder and mild, moderate, and severe atypical melanocytes according to criteria used on larger lesions.
Results: Small melanocytic naevi covered the same complex histological spectrum from benign naevi to severely atypical naevi when compared with larger lesions. A high proportion of small naevi (72%) exhibited features diagnostic for naevi with architectural disorder and cytological atypia.
Conclusion: There is a discrepancy between histological and clinically defined atypical naevi. The same generally accepted criteria for the histological diagnosis of atypical naevi should be used for small melanocytic naevi in addition to large ones. Thus, small naevi exhibiting atypical features on histological examination should be categorised as atypical naevi, regardless of their small diameter.
Keywords: atypical naevi, dysplastic naevi, naevi with architectural disorder, grading of nuclear atypia
A cquired melanocytic naevi usually appear in the 1st years of life, enlarge, and increase in number until the 4th decade, after which they slowly disappear with age. More than 50% of adults have between 10 and 45 melanocytic lesions, and at least 10% have more than 50 naevi. The presence of atypical naevi is regarded as one of the major risk factors for the development of cutaneous malignant melanoma.1–5 Furthermore, melanocytic naevi serve not only as risk markers, but can also be precursor lesions for some malignant melanomas.6
The size of a melanocytic lesion has been used as one of the major criteria to identify atypical naevi and malignant melanomas. The decision to remove a pigmented lesion is usually based on a detailed morphological analysis of the clinical characteristics of the lesion. In 1992, the National Institutes of Health consensus conference on the diagnosis and treatment of early melanoma recommended the use of the ABCD checklist for the detection of suspicious lesions, referring to asymmetry, border irregularity, colour variegation, and diameter greater than 6 mm.7 Atypical (dysplastic) naevi were considered to represent an intermediate step in tumour progression from common acquired melanocytic naevus to malignant melanoma.8 Clark described them as lesions typically having a size of more than 5 mm in diameter.8 In this concept of tumour development, small naevi, less than 4 mm in width, tend to regress, whereas some large, clinically atypical naevi, more than 5 mm in width, may progress to melanoma. However, it has been demonstrated several times in the past that malignant melanomas with a diameter less than 6 mm do exist and may have the potential to metastasise.9–12 Usually, these lesions were clinically not suspicious for melanoma.10 The most common reason for excision was “newly discovered or changing lesion”.11 The 5 mm size criterion would have failed to detect these small diameter melanomas.
“The presence of atypical naevi is regarded as one of the major risk factors for the development of cutaneous malignant melanoma”
Surprisingly little is known about small pigmented lesions, especially with regard to their histopathological appearance. Most studies have been performed on clinically atypical naevi larger than 5 mm in diameter.13–15 Only one study focused on benign appearing lesions with a diameter less than 5 mm.16 Recently, it has been shown that the correlation between clinical atypia and histological dysplasia in acquired naevi is generally poor and partly related to size: the smaller the size, the greater the discrepancy.17 This prompted us to evaluate the histopathological characteristics of small diameter melanocytic naevi in our collection. In our study, the architectural and cytological features of a series of pigmented lesions ≤ 4 mm in diameter were microscopically analysed to determine whether they generally represent just common benign naevi or whether some may be naevi with architectural disorder and cytological atypia of variable degrees.
MATERIAL AND METHODS
Selection of cases
We screened the files from the division of dermatopathology at the New York Presbyterian Hospital, USA, in 1998, and the department of dermatology and allergology at the Technical University in Munich, Germany, between 1999 and 2001, for acquired melanocytic lesions measuring clinically ≤ 4mm in diameter. Initially excluded from the study were naevi in acral or genital locations. Also excluded after microscopic evaluation were blue naevi, Spitz and Reed naevi, naevi with congenital features, traumatised naevi, and naevi with incomplete biopsies precluding adequate evaluation of size. Two hundred and sixty one small diameter melanocytic lesions obtained from 206 patients remained for further analysis. Each specimen was routinely fixed in 3.7% buffered formalin, embedded in paraffin wax, sectioned at multiple levels, and stained with haematoxylin and eosin. The most “atypical” level was subjected to analysis. The diameter of ≤ 4 mm was reconfirmed microscopically in each case.
Definition of architectural and cytological features
The histopathological criteria of naevi with architectural disorder and the grading of cytological atypia were based on widely accepted, standard criteria and were adapted with minor changes.8,14,18–22
Definition of a naevus with architectural disorder
According to Clemente et al, the diagnosis of an atypical naevus requires two of two major criteria and two of four minor criteria to be satisfied.18 Major criteria are: (1) basilar proliferation of atypical melanocytes, exhibiting either a lentiginous or a nested pattern, and (2) (when there is a dermal component) the basilar proliferation by at least three rete ridges beyond the dermal component, thus exhibiting a so called shoulder. Minor criteria are: (1) the presence of concentric eosinophilic fibrosis or lamellar fibroplasia, (2) neovascularisation, (3) bridging of adjacent rete ridges, and (4) a dermal cellular inflammatory response.
Additional architectural features
Apart from these, we looked for the presence or absence of symmetry, regression, thickening of the stratum corneum, circumscription (bilaterally not sharp or sharp on one side), and pagetoid spread of melanocytic cells in upper levels of the epidermis. The rete ridge pattern was evaluated with regard to an increase in number and elongation of rete ridges, distortion, and regularity versus effacement (flattening of at least one rete ridge, resulting in an increased length of a suprapapillary plate containing numerous single melanocytes). The orientation of most of the nests was assessed (round/oval, horizontal, vertical, horizontal/vertical).
Cytological features
The shape of the melanocytic cells was judged according to whether they were predominantly round cells, spindle cells, or equal proportions of both. In the same way, the proportion of solitary melanocytes to melanocytes in nests (defined as a group of three or more cells) was roughly estimated. The location of melanocytes at the rete ridges was also recorded: whether they were at the tips and lateral aspects of the rete ridges or also involved the suprapapillary plates.
Grading of cytological atypia
Although grading of nuclear atypia is difficult and may only be poorly reproducible, we tried to adapt for small naevi the grading scheme used by the groups of Rhodes and Weinstock.21,22
Table 1 summarises the definitions of cellular morphology. The designation of slight, moderate, or severe atypia indicates the highest degree of atypia that applies to multiple melanocytes over at least two rete ridges at the epidermodermal junction, regardless of the manner in which they are grouped. No grading of dermal melanocytes was performed. The diagnosis of melanoma required a continuous proliferation of moderately to severely atypical melanocytes in a severely disordered pattern for a breadth of at least two suprapapillary plates connected by a flattened rete ridge.
Table 1.
Study definition of nuclear atypia of junctional melanocytes, adapted from groups of Weinstock et al and Rhodes and colleagues21,22
| Feature | Normal | Minimal | Moderate | Severe |
| Nuclear size* | <kn | >kn | 2×kn | >2×kn |
| Nuclear variability | Minimal | Some | Moderate | Severe |
| Chromatin | Homogenous/evenly dispersed, delicate | Hyperchromatic | Hyperchromatic/coarsely clumped/irregularity in nuclear contour (thickened or grooved nuclear membrane) | Hyperchromatic/coarsely clumped/irregularity in nuclear contour |
| Nucleolus | Not visible | Not prominent | Visible | Prominent and lavender in colour |
| Amount of cytoplasm | Scant | Scant | Abundant | Abundant |
*Size of melanocyte nucleus compared with the size of basal keratinocyte nucleus (kn).
Statistical analysis
All variables were analysed using χ2 for trend, with significance set at p < 0.05.
RESULTS
A total of 261 acquired melanocytic lesions obtained from 206 patients (125 female and 81 male patients; mean age, 41.6 years; range, 13–79) met the inclusion criteria. Forty three per cent of all lesions were junctional and 57% compound. The cases were first classified into three groups: benign melanocytic naevi without architectural disorder, benign naevi with architectural disorder, and melanoma according to the criteria of Clemente et al.18 Almost all lesions exhibited a lentiginous proliferation of melanocytes as individual cells, or nested or mixed patterns. Because a high percentage of the ordinary benign melanocytic naevi showed some bridging of rete ridges, in addition to slight inflammation, meeting two of the minor criteria for atypia, we found it more important to use distortion of rete ridges (instead of bridging) as a differentiating criterion of architectural disorder. With this change, 73 lesions (28%) were benign and 188 (72%) showed architectural disorder, and we designated these atypical naevi. Only 34% of the benign naevi had a dermal component. In contrast, 66% of the atypical naevi were compound, all of them showing a shoulder phenomenon. According to their cytological changes, the atypical naevi were graded mild in 103 cases (55%), moderate in 73 (39%), and severe in 12 cases (6%) (fig 1). There was no recognisable variability in age and sex distribution within the different groups (data not shown). A slight female preponderance for severely atypical melanocytic naevi was not significant and could be explained by the low number of cases within this group. Table 2 shows the anatomical site distribution. Table 3 shows the frequencies of the most relevant criteria. Other criteria such as regression and hyperkeratosis observed in around 10% and 26% of all cases, respectively, did not show pronounced differences between the groups.
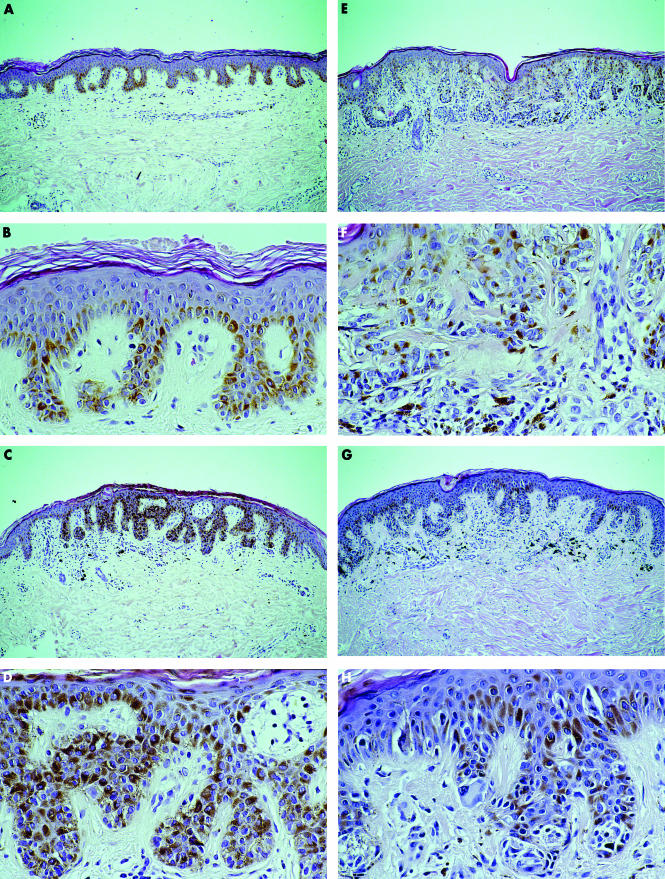
Figure 1.
Different categories of small melanocytic naevi: (A, B) ordinary benign naevi without architectural disorder; naevi with architectural disorder and (C, D) mild, (E, F) moderate, or (G, H) severe cytological atypia of melanocytes (haematoxylin and eosin stain; original magnification, ×10 for A, C, E, and G; ×40 for B, D, F, and H).
Table 2.
Anatomical site distribution of small melanocytic naevi
| All naevi | Benign | Mild | Moderate | Severe | |
| Total number | 261 (100) | 73 (100) | 103 (100) | 73 (100) | 12 (100) |
| Abdomen | 36 (14) | 7 (10) | 19 (18) | 8 (11) | 2 (17) |
| Chest, axilla | 30 (11) | 6 (8) | 11 (11) | 10 (14) | 3 (25) |
| Back, flank, buttock, shoulder | 105 (40) | 32 (44) | 41 (40) | 31 (42) | 1 (8) |
| Upper extremity | 24 (9) | 8 (11) | 8 (8) | 6 (8) | 2 (17) |
| Lower extremity | 51 (20) | 15 (20) | 18 (17) | 14 (19) | 4 (33) |
| Head or neck | 9 (3) | 3 (4) | 4 (4) | 2 (3) | 0 |
| Not specified | 6 (2) | 2 (3) | 2 (2) | 2 (3) | 0 |
Numbers in parenthesis are the percentage of cases in each column.
Table 3.
Frequency and percentage (in parenthesis) of histopathological features in small melanocytic naevi
| Atypical naevi | |||||
| Feature | Benign naevi | Mild | Moderate | Severe | p Value |
| Total number | 73 (100) | 103 (100) | 73 (100) | 12 (100) | |
| Fibrosis | |||||
| No | 65 (89) | 59 (57) | 43 (59) | 9 (75) | |
| Concentric eosinophilic | 7 (10) | 41 (40) | 26 (36) | 2 (17) | 0.0055 |
| Lamellar | 1 (1) | 2 (2) | 0 | 0 | – |
| Both | 0 | 1 (1) | 4 (5) | 1 (8) | 0.0019 |
| Neovascularisation | |||||
| No | 69 (95) | 91 (88) | 61 (84) | 6 (50) | |
| Yes | 4 (5) | 12 (12) | 12 (16) | 6 (50) | 0.003 |
| Inflammation | |||||
| No | 24 (33) | 3 (3) | 2 (3) | 1 (8) | |
| Only melanophages | 22 (30) | 27 (26) | 7 (9) | 1 (8) | 0.0107 |
| Scant | 24 (33) | 65 (63) | 43 (59) | 6 (50) | 0.0003 |
| Patchy | 3 (4) | 8 (8) | 21 (29) | 4 (34) | 0.0009 |
| Distortion of rete ridges | |||||
| No | 67 (92) | 4 (4) | 1 (1) | 1 (8) | |
| Yes | 6 (8) | 99 (96) | 72 (99) | 11 (92) | 0.0001 |
| Effacement | |||||
| No | 73 (100) | 99 (96) | 66 (90) | 6 (50) | |
| Yes | 0 | 4 (4) | 7 (10) | 6 (50) | 0.0001 |
| Pagetoid spread | |||||
| No | 73 (100) | 101 (98) | 69 (95) | 4 (33) | |
| Yes | 0 | 2 (2) | 4 (5) | 8 (67) | 0.0001 |
| Nuclear size | |||||
| <kn | 53 (72) | 53 (51) | 7 (10) | 0 | |
| ≤ kn <2 kn | 18 (25) | 42 (41) | 29 (40) | 1 (8) | 0.0001 |
| ≤2×kn | 2 (3) | 8 (8) | 37 (50) | 11 (92) | 0.0001 |
| Nuclear contour | |||||
| Regular oval | 61 (84) | 58 (56) | 25 (34) | 3 (25) | |
| Spindle shape | 12 (16) | 45 (44) | 48 (66) | 9 (75) | 0.0001 |
| Thickened membrane | 6 (8) | 11 (11) | 34 (47) | 8 (67) | 0.0001 |
| Indented | 0 | 5 (5) | 22 (30) | 8 (67) | 0.0001 |
| Chromatin | |||||
| Hyperchromatic | 14 (20) | 43 (42) | 49 (67) | 9 (75) | 0.0001 |
| Clumped | 1 (1) | 3 (3) | 19 (26) | 8 (67) | 0.0001 |
| Variability | |||||
| Minimal | 73 (100) | 92 (89) | 24 (33) | 0 | |
| Moderate | 0 | 11 (11) | 44 (60) | 6 (50) | 0.0001 |
| Severe | 0 | 0 | 5 (7) | 6 (50) | 0.147 |
| Nucleolus | |||||
| Not prominent | 70 (96) | 76 (74) | 20 (27) | 1 (8) | |
| Visible | 3 (4) | 27 (26) | 39 (54) | 8 (67) | 0.0001 |
| Prominent | 0 | 0 | 14 (19) | 3 (25) | 0.0068 |
| Cytoplasm | |||||
| Scant | 71 (97) | 85 (83) | 31 (42) | 0 | |
| Abundant | 2 (3) | 18 (17) | 42 (58) | 12 (100) | 0.0001 |
kn, size of melanocyte nucleus compared with the size of basal keratinocyte nucleus.
Some criteria were found to have a slightly increasing or decreasing frequency from benign to severe atypical naevi, but were not particularly useful for grading. For instance, most of the benign (90%), mild (77%), and moderate (62%) atypical lesions were symmetrical in contrast to the severe atypical lesions, which were asymmetrical in 54% of the cases. Ill defined borders were seen in 92% of benign, 89% of mild, 73% of moderate, and only 67% of severely atypical naevi. The orientation of nests was mostly round/oval, and the percentage of cases with horizontally oriented nests increased from 9% in benign, to 19% in mild, 38% in moderate, and 43% in severe atypical naevi. The cell shape of melanocytes was predominately round. A mixed pattern of round and spindle-type cells was seen in 18% of benign naevi, 27% of mildly atypical naevi, 48% of moderately atypical naevi, and 58% of severely atypical naevi. The naevus cells were preferentially located at the tips of the rete ridges in 88% of the benign and 82% of the mild cases. In addition, 48% of moderate and 75% of severe cases exhibited melanocytes in the suprapapillary plate regions. Some criteria, such as effacement of rete ridges or pagetoid spread, were mainly present in the centre of lesions in the severe group (50% and 67%, respectively) and almost non-existent in the others (benign, 0%; mild, 4% and 2%, respectively; moderate, 10% and 5%, respectively).
DISCUSSION
Our study focused on the histological appearance of small melanocytic lesions. Using widely accepted criteria for diagnosing and grading naevi with architectural disorder and cytological atypia (also known as “dysplastic naevi”), we found that small melanocytic naevi have the same complex spectrum as larger lesions, ranging from benign naevi to severely atypical naevi to malignant melanoma. An overall percentage of around 72% of the excised small naevi presenting with some atypical histological features is consistent with data established on naevi of any dimension.15,17
For practical purposes, we found useful the following stepwise manner for classifying and grading. First, the distinction between ordinary benign and atypical naevi was made on architectural features. Most of the atypical naevi showed distortion of rete ridges and a cellular lymphoid immune response, less often fibrosis, and only rarely neovascularisation. Grading of atypia was mainly based on cytological criteria. A naevus cell nucleus more than twice the size of a keratinocyte nucleus was a reliable marker to distinguish between moderately and mildly atypical naevi. The visibility of nucleoli was also an easily recognisable feature seen in naevus nuclei with at least moderate atypia. Because the measurement of the chromatin pattern, nuclear shape, and contours was too difficult and time consuming, the overall visual assessment of nuclear variability turned out to be a fair method for differentiating between mild and moderate/severe atypia, although no significance difference could be reached between moderately and severely atypical naevi. Whereas in most mildly atypical naevi melanocytes were found in nests at the tips and lateral aspects of the rete ridges, moderately atypical naevi had increasingly more melanocytes as single units in between rete ridges. In severely atypical naevi and in melanoma in situ this increase of single melanocytes at the suprapapillary plate region merged into effacement of rete ridges and an irregularly spaced rete pattern. Pagetoid spread of single melanocytes into upper parts of the epidermis was seen focally in the centre of lesions in the severe group, thus allowing differentiation from moderate atypical naevi.
It was noteworthy that no single histological feature studied here was absolutely diagnostic by itself for a clear cut distinction between the different groups. It was always the combination of features, rather than a single parameter that allowed classification of small melanocytic lesions.
Grading of melanocytic lesions itself can be problematical. Several analyses of grading atypical melanocytic naevi larger than 5 mm in diameter have reported only a low interobserver concordance and poor reproducibility, even when simple scoring systems were used.23–26 However, the clinical value of grading atypical naevi is not the subject of this study, grading only showed that melanocytic lesions do not automatically fit into the category of common benign naevi if they are small in size.
“In our opinion, if histological criteria have been formulated to define atypical naevi, then they should be used regardless of the size of the lesion”
The histopathological spectrum of small melanocytic lesions encompasses lesions that represent clearly benign naevi at one extreme and malignant melanoma at the other. In between is a large and heterogeneous group of small naevi exhibiting intermediate characteristics that fulfill the architectural and cytological abnormalities of atypical naevi. Several authors recommend that the diagnosis of an atypical naevus should be given only to lesions larger than 5 mm in size.19,20 This restrictive approach in diagnosing atypical naevi is advised probably because of a better correlation between large atypical naevi and a personal or family history of melanoma, and because of the higher predictive value as a marker for the development of melanoma. Several studies in the past have indeed verified that clinical atypical naevi larger than 5 mm in diameter are markers of an increased risk of developing melanoma.1,4,5 Numerous atypical naevi might be more predictive, but the number of small diameter naevi has also been found to be associated with an increased risk for melanoma. Recent studies indicated that the presence of more than 120 naevi less than 5 mm in diameter was associated with a relative risk for the development of melanoma of 19.6 compared with patients having fewer than 10 naevi.3 Tucker et al predicted a twofold higher risk for patients with more than 25 small naevi.27 That study also found a significant 2.6-fold risk associated with naevi smaller than 5 mm that had the clinical appearance of atypical naevi, but did not meet the size criterion. All these studies exclusively judged the relative risk by counting the number of naevi on the whole body and subdividing them into common benign or atypical naevi according to their clinical appearance. A histological examination for defining atypical naevi has not been included and no study has directly determined the risk associated with histological atypia as a specific marker for melanoma development in comparison with clinical atypia. Annessi et al recently studied the correlation between clinical atypia and histological dysplasia in acquired melanocytic naevi and found an overall disagreement of 39.8%, in part reciprocally related to size.17 In particular, 69.6% of clinically non-atypical naevi were found to be atypical on histiological examination and 33.3% of naevi judged histologically to be not atypical were diagnosed clinically as atypical. Others have also demonstrated histological features of atypia in clinically common naevi.16 Moreover, Piepkorn et al estimated the prevalence of atypical architectural features in naevi to be as high as 53% in the general population, regardless the size of the lesions and without cytological atypia as a required criterion.28 This poor correlation between clinical phenotype and histological appearance tends to question the concept of atypical naevus as a real entity and points towards the need for more exact definitions of both clinical and histological atypia.
Take home messages.
Small melanocytic naevi covered the same complex histological spectrum from benign naevi to severely atypical naevi as did larger lesions
A high proportion of small naevi (72%) had features diagnostic for naevi with architectural disorder and cytological atypia
Thus, the generally accepted criteria for the histological diagnosis of atypical naevi should be used for both small and large melanocytic naevi, and small naevi with atypical histological features should be categorised as atypical naevi, regardless of their small size
The present study confirms a high proportion of histological atypical features in small naevi. Even though our study presents consecutive cases collected over several years, a percentage of 72% atypical naevi may reflect a certain bias because patients who have had small naevi removed may more often have a positive personal or family history of melanoma. If this is the case, small lesions may indeed reflect an increased risk to develop melanoma. Alternatively, it has been argued that atypical features seen in small naevi do not indicate persistent atypia, but may be related to an early evolving state of a lesion that is actively proliferating.28 Because most of the small naevi in our study were excised because of changes over time or because they conformed to the “ugly duckling sign” (naevi that do not fit into the common profile of most naevi in a given patient),29 we expected to see some signs of proliferation. The clinical relevance of these lesions as precursors or markers for the risk of malignant melanoma cannot be estimated without appropriate studies, including their histological appearance. In our opinion, if histological criteria have been formulated to define atypical naevi, then they should be used regardless of the size of the lesion. Therefore, we assign small naevi exhibiting atypical features on histological examination to the category of naevi with architectural disorder and mild, moderate, or severe cytological atypia of melanocytes.
REFERENCES
- 1.Bataille V, Bishop JA, Sasieni P, et al. Risk of cutaneous melanoma in relation to the numbers, types and sites of naevi: a case–control study. Br J Dermatol 1996;73:1605–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Garbe C, Büttner P, Wieβ J, et al. Risk factors for developing cutaneous melanoma and criteria for identifying persons at risk: multicenter case–control study of the central malignant melanoma registry of the German Dermatological Society. J Invest Dermatol 1996:102:695–9. [DOI] [PubMed] [Google Scholar]
- 3.Grob JJ, Gouvernet J, Aymar D, et al. Count of benign melanocytic nevi as a major indicator of risk for nonfamilial nodular and superficial spreading melanoma. Cancer 1990;66:387–95. [DOI] [PubMed] [Google Scholar]
- 4.Schneider JS, Moore DH, Sagebiel RW. Risk factors for melanoma incidence in prospective follow-up. The importance of atypical (dysplastic) nevi. Arch Dermatol 1994;103:202–5. [PubMed] [Google Scholar]
- 5.Snels DG, Hille ET, Gruis NA, et al. Risk of cutaneous malignant melanoma in patients with nonfamilial atypical nevi from a pigmented lesions clinic. J Am Acad Dermatol 1999;40:686–93. [DOI] [PubMed] [Google Scholar]
- 6.Skender-Kalnenas TM, English DR, Heenan PJ. Benign melanocytic lesions: risk markers or precursors of cutaneous melanoma? J Am Acad Dermatol 1995;33:1000–7. [DOI] [PubMed] [Google Scholar]
- 7.NIH Consensus Conference. Diagnosis and treatment of early melanoma. JAMA 1992;268:1314–19. [DOI] [PubMed] [Google Scholar]
- 8.Clark WH, Jr, Tucker MA. Problems with lesions related to the development of malignant melanoma: common nevi, dysplastic nevi, malignant melanoma in situ, and radial growth phase malignant melanoma. Hum Pathol 1998;29:8–14. [DOI] [PubMed] [Google Scholar]
- 9.Bergman R, Katz I, Lichtig C, et al. Malignant melanomas with histologic diameters less than 6 mm. J Am Acad Dermatol 1992;26:462–6. [DOI] [PubMed] [Google Scholar]
- 10.Gonzalez A, West AJ, Pitha JV, et al. Small-diameter invasive melanomas: clinical and pathologic characteristics. J Cutan Pathol 1996;23:126–32. [DOI] [PubMed] [Google Scholar]
- 11.Kamino H, Kiryu H, Ratech H. Small malignant melanomas: clinicopathologic correlation and DNA ploidy analysis. J Am Acad Dermatol 1990;22:1032–8. [DOI] [PubMed] [Google Scholar]
- 12.Koch SE, Henneberry JM. Clinically subtle primary cutaneous melanoma. J Am Acad Dermatol 1999;40:252–4. [DOI] [PubMed] [Google Scholar]
- 13.Barnhill RL, Roush GC. Correlation of clinical and histopathologic features in clinical atypical melanocytic nevi. Cancer 1990;67:3157–64. [DOI] [PubMed] [Google Scholar]
- 14.Barnhill RL, Roush GC, Duray PH. Correlation of histologic architectural and cytoplasmic features with nuclear atypia in atypical (dysplastic) nevomelanocytic nevi. Hum Pathol 1990;21:51–8. [DOI] [PubMed] [Google Scholar]
- 15.Urso C. Atypical histologic features in melanocytic nevi. Am J Dermatopathol 2000;22:391–6. [DOI] [PubMed] [Google Scholar]
- 16.Klein LJ, Barr RJ. Histologic atypia in clinically benign nevi. J Am Acad Dermatol 1990;22:275–82. [DOI] [PubMed] [Google Scholar]
- 17.Annessi G, Cattaruzza MS, Abeni D, et al. Correlation between clinical atypia and histologic dysplasia in acquired melanocytic nevi. J Am Acad Dermatol 2001;45:77–85. [DOI] [PubMed] [Google Scholar]
- 18.Clemente C, Cochran AJ, Elder DE, et al. Histopathologic diagnosis of dysplastic nevi: concordance among pathologists convened by the World Health Organisation melanoma programme. Hum Pathol 1991;22:313–19. [DOI] [PubMed] [Google Scholar]
- 19.Mooi WJ. The dysplastic naevus. J Clin Pathol 1997;50:711–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Murphy GF, Mihm MC. Recognition and evaluation of cytological dysplasia in acquired melanocytic nevi. Hum Pathol 1999;30:506–12. [DOI] [PubMed] [Google Scholar]
- 21.Rhodes AR, Mihm MC, Weinstock MA. Dysplastic melanocytic nevi: a reproducible histologic definition emphasizing cellular morphology. Mod Pathol 1989;2:306–19. [PubMed] [Google Scholar]
- 22.Weinstock MA, Barnhill RL, Rhodes AR, et al. Reliability of the histopathologic diagnosis of melanocytic dysplasia. Arch Dermatol 1997;133:953–8. [PubMed] [Google Scholar]
- 23.Duncan LM, Berwick M, Bruijn JA, et al. Histopathologic recognition and grading of dysplastic melanocytic nevi: an interobserver agreement study. J Invest Dermatol 1993;100:318S–21S. [DOI] [PubMed] [Google Scholar]
- 24.Duray PH, DerSimonian R, Barnhill R, et al. An analysis of interobserver recognition of the histopathologic features of dysplastic nevi from a mixed group of nevomelanocytic lesions. J Am Acad Dermatol 1992;27:741–9. [DOI] [PubMed] [Google Scholar]
- 25.Hastrup N, Clemmensen OJ, Spaun E, et al. Dysplastic naevus: histological criteria and their inter-observer reproducibility. Histopathology 1994;24:503–9. [DOI] [PubMed] [Google Scholar]
- 26.Pozo L, Naase M, Cerio R, et al. Critical analysis of histologic criteria for grading atypical (dysplastic) melanocytic nevi. Am J Clin Pathol 2001;115:194–204. [DOI] [PubMed] [Google Scholar]
- 27.Tucker MA, Halpern A, Holly EA, et al. Clinically recognized dysplastic nevi. A central risk factor for cutaneous melanoma. JAMA 1997;277:1439–44. [PubMed] [Google Scholar]
- 28.Piepkorn M, Meyer JM, Goldgar D, et al. The dysplastic melanocytic nevus: a prevalent lesion that correlates poorly with clinical phenotype. J Am Acad Dermatol 1989;20:407–15. [DOI] [PubMed] [Google Scholar]
- 29.Grob JJ. The “ugly duckling” sign: identification of the common characteristics of nevi in an individual as a basis for melanoma screening. Arch Dermatol 1998;134:103–4. [DOI] [PubMed] [Google Scholar]