Abstract
Aims: To define epidemiology, clinical disease, and outcome of gemella bacteraemia by 16S rRNA gene sequencing. To examine the usefulness of the Vitek, API, and ATB systems in identifying two gemella species.
Methods: All α haemolytic streptococci other than Streptococcus pneumoniae isolated from blood cultures during a six year period were identified by conventional biochemical methods, the Vitek system, and the API system. 16S rRNA gene sequencing was performed on all isolates identified by both kits as gemella with ≥ 95% confidence or by either kit as any bacterial species with < 95% confidence. The ATB expression system was used to identify the two isolates that were defined as gemella species by 16S rRNA gene sequencing.
Results: Of the 302 α haemolytic streptococci other than S pneumoniae isolated, one was identified as Gemella morbillorum, and another as Gemella haemolysans by 16S rRNA gene sequencing. The patient with monomicrobial G morbillorum bacteraemia was a 66 year old man with community acquired infective endocarditis with septic thromboemboli. The patient with G haemolysans bacteraemia was a 41 year old woman with hospital acquired polymicrobial bacteraemia during the neutropenic period of an autologous bone marrow transplant for non-Hodgkin’s lymphoma, the first case of its kind in the English literature. The API and ATB expression systems only identified the second strain as G haemolysans at 94% and 99% confidence, respectively, whereas the Vitek system identified none of the two strains correctly at > 70% confidence.
Conclusions: Gemella bacteraemia is uncommon. 16S rRNA gene sequencing is the method of choice for identification of gemella and gemella-like isolates.
Keywords: Gemella, bacteraemia, 16S rRNA sequencing
Since the discovery of the polymerase chain reaction (PCR) and DNA sequencing, comparisons of the gene sequences of bacterial species have shown that the 16S rRNA gene is highly conserved within a species and among species of the same genus, and hence can be used as the new standard for speciation of bacteria.1 Using this new standard, phylogenetic trees, based on base differences between species, are constructed, and bacteria are classified and reclassified into new genera. Recently, we have reported the use of this technique for the identification of bacterial strains with ambiguous biochemical profiles,2–10 species that are rarely encountered clinically,11–15 and a bacterium that is non-cultivable.16 In addition, we have also reported the discovery of a novel clinical syndrome17,18 and three novel species,19–21 and the characterisation of β haemolytic Lancefield group G streptococcal bacteraemia and Granulicatella adiacens and Abiotrophia defectiva bacteraemia.22,23
“We have recently shown that a large number of bacterial strains identified as gemella species by commercially available phenotypic systems are actually Granulicatella adiacens”
Members of the genus gemella consist of catalase negative, facultative anaerobic, Gram positive cocci that phenotypically resemble viridans streptococci. Traditionally, the identification of, and hence the study in the epidemiology of and spectrum of clinical diseases caused by gemella species depend on the phenotypic characterisation of suspected isolates. However, we have recently shown that a large number of bacterial strains identified as gemella species by commercially available phenotypic systems are actually Granulicatella adiacens.23 No data are currently available that have used 16S rRNA gene sequencing as the gold standard. Therefore, in our study, we used 16S rRNA gene sequencing, assisted by traditional phenotypic tests, to describe the epidemiology, clinical diseases, and outcome of gemella bacteraemia. We also examined the usefulness of the Vitek system (bioMerieux Vitek, Hazelwood, Missouri, USA), the API system (bioMerieux Vitek), and the ATB expression system (bioMerieux Vitek), which are commonly used for microbial identification of α haemolytic streptococci in clinical microbiology laboratories, for the identification of the two gemella isolates.
METHODS AND MATERIALS
Patients and microbiological methods
The patients with gemella bacteraemia in our study were hospitalised at the Queen Mary Hospital in Hong Kong during a six year period (July 1995 to June 2001). All clinical data were collected as described in our previous publication.24 The BACTEC 9240 blood culture system (Becton Dickinson, Sparks, Maryland, USA) was used. To identify potential cases of gemella bacteraemia, in addition to identifying all blood culture isolates by standard conventional biochemical methods,25 the Vitek system (GPI; bioMerieux Vitek) and the API system (20 STREP; bioMerieux Vitek) were used for species identification of all α haemolytic streptococci other than Streptococcus pneumoniae isolated from blood cultures during the six year period. 16S rRNA gene sequencing was performed on all isolates that were identified by both kits as gemella with ≥ 95% confidence, or by either kit as any bacterial species with < 95% confidence. To examine the usefulness of the ATB expression system (ID32 STREP; bioMerieux Vitek) for the identification of gemella species, this system was also used for identification of the two isolates that were finally defined as gemella species by 16S rRNA gene sequencing. Antimicrobial susceptibility was undertaken by the E test for penicillin and the Kirby Bauer disk diffusion method for the other antibiotics using Muller Hinton agar supplemented with 5% horse blood, and the results were interpreted according to the NCCLS criteria for α haemolytic streptococci. Multiple positive blood cultures with the same isolate obtained from the same patient were counted only once.
Extraction of bacterial DNA for 16S rRNA gene sequencing
Bacterial DNA extraction was modified from our previously published protocol.26 Briefly, 80 μl of NaOH (0.05M) was added to 20 μl of bacterial cells suspended in distilled water and the mixture was incubated at 60°C for 45 minutes, followed by the addition of 6 μl of Tris/HCl (pH 7.0), to achieve a final pH of 8.0. The resultant mixture was diluted ×100 and 5 μl of the diluted extract was used for PCR.
PCR, gel electrophoresis, and 16S rRNA gene sequencing
PCR amplification and DNA sequencing of the 16S rRNA genes were performed according to our previous publications.9,20,22,23 Briefly, DNase I treated distilled water and the PCR master mix (which contains dNTPs, PCR buffer, and Taq polymerase) were used in all PCR reactions by adding 1 U of DNase I (Pharmacia, Uppsala, Sweden) to 40 μl of distilled water or PCR master mix, incubating the mixture at 25°C for 15 minutes, and subsequently at 95°C for 10 minutes to inactivate the DNase I. The bacterial DNA extracts and control were amplified with 0.5μM primers (LPW200, 5‘-GAGTTGCGAACGGGTGAG-3‘; and LPW205, 5‘-CTTGTTACGACTTCACCC-3‘; Gibco BRL, Rockville, Maryland, USA). The PCR mixture (50 μl) contained bacterial DNA, PCR buffer (10mM Tris/HCl, pH 8.3, 50mM KCl, 2mM MgCl2, and 0.01% gelatin), 200 μM of each dNTP and 1.0 U Taq polymerase (Boehringer Mannheim, Mannheim, Germany). The mixtures were amplified by 40 cycles of 94°C for one minute, 55°C for one minute, and 72°C for two minutes, with a final extension at 72°C for 10 minutes in an automated thermal cycler (Perkin-Elmer Cetus, Gouda, The Netherlands). DNase I treated distilled water was used as the negative control. A 10 μl sample of each amplified product was electrophoresed in a 1.0% (wt/vol) agarose gel, with a molecular size marker (Lambda DNA AvaII digest; Boehringer Mannheim) in parallel. Electrophoresis in Tris/borate/EDTA buffer was performed at 100 V for 1.5 hours. The gel was stained with ethidium bromide (0.5 μg/ml) for 15 minutes, rinsed, and photographed under ultraviolet light illumination.
The PCR products were gel purified using the QIAquick PCR purification kit (QIAgen, Hilden, Germany). Both strands of the PCR products were sequenced twice with an ABI 377 automated sequencer according to manufacturer’s instructions (Perkin-Elmer, Foster City, California, USA), using the primers LPW200, LPW205, LPW99 (5‘-TTATTGGGCGTAAAGCGA-3‘), and LPW273 (5‘-TTGCGGGACTTAACCCAAC-3‘). The sequences of the PCR products were compared with known 16S rRNA gene sequences in the GenBank by multiple sequence alignment using the CLUSTAL W program,27 and phylogenetic tree construction was performed using the PileUp method with GrowTree (Genetics Computer Group Inc, San Diego, USA).
RESULTS
16S rRNA gene sequencing
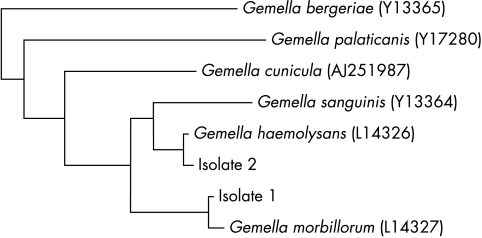
Among a total of 302 α haemolytic streptococci other than S pneumoniae isolated from blood cultures of patients admitted to Queen Mary Hospital during the six year period (July 1995 to June 2001), none was identified by both the Vitek system (GPI) and the API system (20 STREP) as any gemella species with ≥ 95% confidence. A total of 74 were identified by either kit as any species with < 95% confidence. PCR of the 16S rRNA genes of these isolates showed bands at approximately 1410 bp. Sequencing of the 16S rRNA genes revealed that one isolate (table 1; patient 1) had > 99% nucleotide identity with the 16S rRNA genes of G morbillorum (GenBank accession number L14327), and another (table 1; patient 2) had > 99% nucleotide identity with the 16S rRNA genes of G haemolysans (GenBank accession number L14326) (fig 1). Therefore, gemella accounted for 0.7% of bacteraemia caused by α haemolytic streptococci other than S pneumoniae.
Table 1.
Characteristics of patients with gemella bacteraemia
| Patients | ||
| Characteristics | 1 | 2 |
| Year of isolation | 1997 | 2000 |
| Age/sex | 66/M | 41/F |
| Underlying disease | Abdominal aortic aneurysm | Non-Hodgkin lymphoma, autologous bone marrow transplant |
| Diagnosis | Infective endocarditis with septic thromboemboli | Primary bacteraemia during the neutropenic period |
| Community/hospital acquired | Community | Hospital |
| Number of positive blood cultures | 3 | 1 |
| Monomicrobial/polymicrobial bacteraemia | Monomicrobial | Polymicrobial (with Streptococcus mitis and Escherichia coli) |
| Positive cultures from other specimens | None | None |
| Identification by 16S rRNA sequencing | Gemella morbillorum | Gemella haemolysans |
| Antibiotic susceptibility | ||
| Penicillin | Sensitive | Resistant |
| Cefalothin | Sensitive | Sensitive |
| Erythromycin | Sensitive | Resistant |
| Clindamycin | Sensitive | Resistant |
| Vancomycin | Sensitive | Sensitive |
| Treatment | Penicillin G+netilmicin | Imipenem |
| Outcome | Remission | Remission |
Figure 1.
Phylogenetic tree showing the relation of the two gemella isolates to other species. The tree was inferred from 16S rRNA sequencing data by the neighbour joining method. The scale bar indicates the estimated number of substitutions for each 100 bases using the Jukes-Cantor correction. Names and accession numbers are given as cited in the GenBank database.
Phenotypic characterisation and identification of gemella by commercial systems
Gram smears of the two isolates showed Gram positive cocci in pairs. The Vitek System (GPI) identified the G morbillorum and G haemolysans strains as 63% G morbillorum/S agalactiae/S acidominimus, 22% S pneumoniae, and 68% G morbillorum/S agalactiae/S acidominimus, 24% S pneumoniae, respectively. The API system (20 STREP) identified the G morbillorum and G haemolysans strains as 60% Leuconostoc sp., 28% Streptococcus mitis, and 94% G haemolysans, 6% G morbillorum, respectively. The ATB expression system (ID32 STREP) identified the G morbillorum and G haemolysans strains as “unidentified” and 99% G haemolysans, respectively.
Patient characteristics
Table 1 summarises the characteristics of the two patients with gemella bacteraemia. Briefly, the first patient, a 66 year old man, was admitted because of occasional abdominal and low back pain for one month. He was afebrile. Examination revealed a collapsing pulse, an early diastolic murmur of grade 3/6 at the left lower sternal border, and a pulsatile mass in the abdomen. Computer tomography of the abdomen confirmed an infrarenal abdominal aortic aneurysm of 7.5 cm in diameter, without signs of rupture. Transthoracic echocardiogram showed severe aortic regurgitation and vegetations on both the aortic and mitral valves. Three sets of blood cultures all recovered the same strain of G morbillorum. He was treated for community acquired infective endocarditis with intravenous penicillin G and netilmicin. He developed sudden retrograde amnesia 17 days after admission. Magnetic resonance imaging of the brain showed multiple subacute infarcts at both occipito–temporal regions, compatible with septic thromboembolism. Intravenous antibiotics were continued for a total of six weeks with residual memory loss. He subsequently underwent an elective endoaneurysmectomy with insertion of an aortic graft and was discharged two months from admission.
The second patient, a 41 year old woman with non-Hodgkin’s lymphoma, was admitted because of autologous bone marrow transplantation. On day two postmarrow infusion, she developed neutropenic fever. Examination did not reveal a heart murmur or other focus of infection. Blood cultures were performed and recovered G haemolysans, S mitis, and Escherichia coli. She responded to empirical intravenous imipenem and subsequent blood cultures were negative. Her bone marrow engrafted on day 15.
DISCUSSION
The identification of infrequently encountered bacterial species in clinical microbiology laboratories has always been a problem. Because the number of reference strains used for building up databases in commercial kits is usually small for rare bacteria, it is not uncommon to encounter clinical isolates of these rare bacterial species with ambiguous biochemical profiles. Furthermore, even if they are “successfully” identified by the commercial kits, the low prevalence rate would imply a low positive predictive value. In our study, we described our experience in using 16S rRNA gene sequencing to characterise two strains of a rarely encountered bacterial genus, gemella, recovered from blood cultures of our patients in the past six years.
The genus gemella consists of six species. Before 1998, there were only two known gemella species, G haemolysans and G morbillorum. Gemella haemolysans was originally classified as Neisseria haemolysans,28 and was subsequently reclassified as G haemolysans in 1960; whereas G morbillorum was originally classified as Diplococcus morbillorum,29 subsequently as Peptostreptococcus morbillorum and Streptococcus morbillorum, and was finally reclassified as G morbillorum in 1988.30 In the past five years, four additional Gemella species have been discovered. Gemella bergeriae and G sanguinis were recovered from human clinical specimens,31,32 whereas G palaticanis was recovered from the oral cavity of a dog,33 and G cunicula from an abscess of a rabbit.34
The identification of “gemella-like” species by the commercial kits was unsatisfactory, especially when the Vitek system (GPI) was used. In the present series, the Vitek system (GPI) was not able to identify a single isolate successfully, whereas the API system (20 STREP) and the ATB expression system (ID32 STREP) were only able to identify one of the two gemella species correctly. This poor accuracy in identifying gemella species by commercial kits was also demonstrated in a recent study, in which none of the three gemella species was successfully identified by the rapid phenotypic identification systems.35 Interestingly, among the nine isolates of Granulicatella adiacens and A defectiva that we reported recently, the Vitek system (GPI) incorrectly identified seven of them as gemella, and the API system (20 STREP) and the ATB expression system (ID32 STREP) incorrectly identified one of them as gemella.23 Therefore, we think that 16S rRNA gene sequencing should be used as the method of choice for identifying gemella and “gemella-like” species.
Species of gemella are important causes of infective endocarditis. Among the reported cases of gemella infections, infective endocarditis is the most common diagnosis. Over 25 cases of gemella endocarditis have been reported in the literature, with the incidence of G morbillorum and G haemolysans endocarditis being approximately the same.36 In our present report, one of the two patients had gemella endocarditis. However, further studies using 16S rRNA gene sequencing need to be carried out to ascertain the true incidence of gemella endocarditis, because gemella species are frequently misidentified as species of streptococcus, leuconostoc, and abiotrophia, and granunicatella and abiotrophia species are frequently misidentified as species of gemella.
“We think that 16S rRNA gene sequencing should be used as the method of choice for identifying gemella and gemella-like species”
We report the first case of gemella bacteraemia during the pre-engraftment period in a bone marrow transplant recipient. Viridans streptococci are commonly isolated from the blood cultures of bone marrow transplant recipients during the pre-engraftment period.37 As members of the oral flora, it is not unexpected that gemella species would also cross the mucosal barrier and cause bacteraemia in those patients with mucositis. The concomitant isolation of S mitis and E coli was in line with the hypothesis that the oral flora was the source of the bacteraemia in case 2 (in immunocompromised patients, part of the normal oral flora is replaced by enterobacteriaceae and non-fermentative Gram negative bacilli).
The emergence of penicillin and macrolide resistance in gemella species is of great concern. In the past, all gemella species isolated from clinical specimens were highly sensitive to penicillin G and ampicillin.38 In 1993, a strain of glycopeptide resistant G haemolysans that showed reduced susceptibility to penicillin (minimum inhibitory concentration (MIC) of 0.5 μg/ml) was recovered from the blood culture of a 20 month old boy.39 In 1996, another strain of G morbillorum that was resistance to penicillin (MIC of ≥ 4 μg/ml) was recovered from the blood culture of an 11 year old girl with nasopharyngeal Burkitt’s lymphoma.40 In our present series, one of the two gemella strains showed reduced susceptibility to penicillin (MIC of 0.5 μg/ml). Furthermore, it was also resistant to erythromycin, clarithromycin, and azithromycin. Because amoxicillin or macrolides are used as prophylaxis for infective endocarditis, the rising incidence of penicillin and macrolide resistance may mean the failure of these agents in the prophylaxis of infective endocarditis in a considerable proportion of these patients undergoing dental or other invasive procedures.
Take home messages.
Gemella bacteraemia is rare and 16S rRNA gene sequencing should be the method of choice for identification of gemella and gemella-like isolates
The identification of “gemella-like” species by the commercial kits was unsatisfactory, especially when the Vitek system (GPI) was used
Species of gemella are important causes of infective endocarditis and one of the two patients reported here had gemella endocarditis
The other patient was a bone marrow transplant recipient who had gemella bacteraemia during the pre-engraftment period and the oral flora was the source of the bacteraemia in this case
Acknowledgments
This work is partly supported by the University Development Fund, University Research Grant Council, and the Committee for Research and Conference Grant, The University of Hong Kong.
Abbreviations
MIC, minimum inhibitory concentration
PCR, polymerase chain reaction
REFERENCES
- 1.Olsen GJ, Woese CR. Ribosomal RNA: a key to phylogeny. FASEB J 1993;7:113–23. [DOI] [PubMed] [Google Scholar]
- 2.Woo PCY, Leung PKL, Leung KW, et al. Identification by 16S ribosomal RNA gene sequencing of an Enterobacteriaceae species from a bone marrow transplant recipient. Mol Pathol 2000;53:211–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Woo PCY, Leung ASP, Leung KW, et al. Identification of slide-coagulase positive, tube-coagulase negative Staphylococcus aureus by 16S ribosomal RNA gene sequencing. Mol Pathol 2001;54:244–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Woo PCY, Cheung EYL, Leung KW, et al. Identification by 16S ribosomal RNA gene sequencing of an Enterobacteriaceae species with ambiguous biochemical profile from a renal transplant recipient. Diagn Microbiol Infect Dis 2001;39:85–93. [DOI] [PubMed] [Google Scholar]
- 5.Woo PCY, Fung AMY, Lau SKP, et al. Diagnosis of pelvic actinomycosis by 16S ribosomal RNA gene sequencing and its clinical significance. Diagn Microbiol Infect Dis 2002;43:113–18. [DOI] [PubMed] [Google Scholar]
- 6.Woo PCY, Fung AMY, Lau SKP, et al. Identification by 16S ribosomal RNA gene sequencing of Lactobacillus salivarius bacteremic cholecystitis. J Clin Microbiol 2002;40:265–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Woo PCY, Fung AMY, Wong SSY, et al. Isolation and characterization of a Salmonella enterica serotype Typhi variant and its clinical and public health implications. J Clin Microbiol 2001;39:1190–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Woo PCY, Leung KW, Tsoi HW, et al. Thermo-tolerant Campylobacter fetus bacteraemia identified by 16S ribosomal RNA gene sequencing: an emerging pathogen in immunocompromised patients. J Med Microbiol 2002;51:740–6. [DOI] [PubMed] [Google Scholar]
- 9.Woo PCY, Teng JLL, Lau SKP, et al. Analysis of a viridans group strain reveals a case of bacteremia due to Lancefield group G alpha-hemolytic Streptococcus dysgalactiae subsp. equisimilis in a patient with pyomyositis and reactive arthritis. J Clin Microbiol 2003;41:613–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lau SKP, Woo PCY, Tse H, et al. Invasive Streptococcus iniae infections outside North America. J Clin Microbiol 2003;41:1004–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Woo PCY, Tsoi HW, Leung KW, et al. Identification of Mycobacterium neoaurum isolated from a neutropenic patient with catheter-related bacteremia by 16S rRNA sequencing. J Clin Microbiol 2000;38:3515–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Woo PCY, Chong KTK, Leung KW, et al. Identification of Arcobacter cryaerophilus isolated from a traffic accident victim with bacteraemia by 16S ribosomal RNA gene sequencing. Diagn Microbiol Infect Dis 2001;40:125–7. [DOI] [PubMed] [Google Scholar]
- 13.Lau SKP, Woo PCY, Chan BYL, et al. Haemophilus segnis polymicrobial and monomicrobial bacteremia identified by 16S ribosomal RNA gene sequencing. J Med Microbiol 2002;51:635–40. [DOI] [PubMed] [Google Scholar]
- 14.Lau SKP, Woo PCY, Teng JLL, et al. Identification by 16S ribosomal RNA gene sequencing of Arcobacter butzleri bacteraemia in a patient with acute gangrenous appendicitis. Mol Pathol 2002;55:182–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lau SKP, Woo PCY, Woo GKS, et al. Catheter-related Microbacterium bacteremia identified by 16S ribosomal RNA gene sequencing. J Clin Microbiol 2002;40:2681–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheuk W, Woo PCY, Yuen KY, et al. Intestinal inflammatory pseudotumor with regional lymph node involvement: identification of a new bacterium as the etiologic agent. J Pathol 2001;192:289–92. [DOI] [PubMed] [Google Scholar]
- 17.Woo PCY, Li JHC, Tang WM, et al. Acupuncture mycobacteriosis. N Engl J Med 2001;345:842–3. [DOI] [PubMed] [Google Scholar]
- 18.Woo PCY, Leung KW, Wong SSY, et al. Relative alcohol-resistant mycobacteria are emerging pathogens in patients receiving acupuncture treatment. J Clin Microbiol 2002;40:1219–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yuen KY, Woo PCY, Teng JLL, et al. Laribacter hongkongensis gen. nov., sp. nov., a novel Gram-negative bacterium isolated from a cirrhotic patient with bacteremia and empyema. J Clin Microbiol 2001;39:4227–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Woo PCY, Tam DMW, Leung KW, et al. Streptococcus sinensis sp. nov., a novel Streptococcus species isolated from a patient with infective endocarditis. J Clin Microbiol 2002;40:805–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Teng JLL, Woo PCY, Leung KW, et al. Pseudobacteraemia in a patient with neutropenic fever caused by a novel Paenibacillus species: Paenibacillus hongkongensis sp. nov. Mol Pathol 2003;56:29–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Woo PCY, Fung AMY, Lau SKP, et al. Group G beta-hemolytic streptococcal bacteremia characterized by 16S ribosomal RNA gene sequencing. J Clin Microbiol 2001;39:3147–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Woo PCY, Fung AMY, Lau SKP, et al. Granulicatella adiacens and Abiotrophia defectiva bacteraemia characterized by 16S ribosomal RNA gene sequencing. J Med Microbiol 2003;52:137–40. [DOI] [PubMed] [Google Scholar]
- 24.Woo PCY, Wong SSY, Lum PNL, et al. Cell-wall-deficient bacteria and culture-negative febrile episodes in bone-marrow-transplant recipients. Lancet 2001;357:675–9. [DOI] [PubMed] [Google Scholar]
- 25.Murray PR, Baro EJ, Pfaller MA, et al. Manual of clinical microbiology, 7th ed. Washington, DC: American Society for Microbiology, 1999.
- 26.Woo PCY, Lo CY, Lo SK, et al. Distinct genotypic distributions of cytomegalovirus (CMV) envelope glycoprotein in bone marrow and renal transplant recipients with CMV disease. Clin Diagn Lab Immunol 1997;4:515–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting position-specific gap penalties and weight matrix choice. Nucleic Acids Res 1994;22:4673–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thjota T, Boe J. Neisseria haemolysans. A haemolytic species of Neisseria Treviscan. Acta Pathol Microbiol Scand 1938;(suppl 37):527–31.
- 29.Prevot AR. Etudes de systematique bactererienne. I. Lois generales. II. Cocci anaerobies. Ann Soc Nat Zool Biol Anim 1933;15:23–6. [Google Scholar]
- 30.Kilpper-Balz R, Schleifer KH. Transfer of Streptococcus morbillorum to the genus Gemella as Gemella morbillorum comb. nov. Int J Syst Bacteriol 1988;38:442–3. [Google Scholar]
- 31.Collins MD, Hutson RA, Falsen E, et al. Gemella bergeriae sp. nov., isolated from human clinical specimens. J Clin Microbiol 1998;36:1290–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Collins MD, Hutson RA, Falsen E, et al. Description of Gemella sanguinis sp. nov., isolated from human clinical specimens. J Clin Microbiol 1998;36:3090–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Collins MD, Rodriguez JM, Foster G, et al. Characterization of a Gemella-like organism from the oral cavity of a dog: description of Gemella palaticans sp. nov. Int J Syst Bacteriol 1999;49:1523–6. [DOI] [PubMed] [Google Scholar]
- 34.Hoyles L, Foster G, Falsen E, et al. Characterization of a Gemella-like organism isolated from an abscess of a rabbit: description of Gemella cunicula sp. nov. Int J Syst Bacteriol 2000;50:2037–41. [DOI] [PubMed] [Google Scholar]
- 35.Scola BL, Raoult D. Molecular identification of Gemella species from three patients with endocarditis. J Clin Microbiol 1998;36:866–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Brouqui P, Raoult D. Endocarditis due to rare and fastidious bacteria. Clin Microbiol Rev 2001;14:177–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yuen KY, Woo PCY, Hui CH, et al. Unique risk factors for bacteraemia in allogeneic bone marrow transplant recipients before and after engraftment. Bone Marrow Transplant 1998;21:1137–43. [DOI] [PubMed] [Google Scholar]
- 38.Buu-Hoi A, Sapoetra A, Branger C, et al. Antimicrobial susceptibility of Gemella haemolysans isolated from patients with subacute endocarditis. Eur J Clin Microbiol 1982;1:102–6. [DOI] [PubMed] [Google Scholar]
- 39.Efstratiou A, D Morrison, Woodford N. Glycopeptide-resistant Gemella haemolysans from blood. Lancet 1993;342:927–8. [DOI] [PubMed] [Google Scholar]
- 40.Vasoshtha S, Isengerh HD, Sood SK. Gemella morbillorum as a cause of septic shock. Clin Infect Dis 1996;22:1084–6. [DOI] [PubMed] [Google Scholar]