Abstract
Despite advances in the treatment of neuroblastoma (NBL), recurrence and metastases continue to pose major problems in clinical management. The relation between micrometastases and the development of secondary disease is not fully understood. However, accurate methods to detect low numbers of tumour cells may allow the evaluation of their role in the disease process, and by implication the possible benefits of eliminating them. Although there is substantial evidence for the increased sensitivity of current molecular methods for the detection of NBL cells compared with more conventional cytology, the clinical relevance and usefulness of detecting this disease remain controversial. The primary goal of current translational research must be to evaluate the clinical relevance of micrometastatic disease detected by these methods in multicentre prospective clinical outcome studies. Only then can the clinical usefulness of these methods be defined so that they may be introduced into relevant clinical practice.
Keywords: micrometastases, neuroblastoma, clinical significance, minimal residual disease, immunocytology, reverse transcriptase polymerase chain reaction
Neuroblastoma (NBL) is the most common extracranial solid tumour of childhood, comprising between 8% and 10% of all childhood cancers. Its incidence is about 1/10 000 live births in the USA,1 occurring in children less than 10 years old, and usually in those between 1 and 5 years.2 The median age at diagnosis is 22 months. NBL has a broad spectrum of clinical behaviour, varying from aggressive malignant disease (stage 4) to spontaneous maturation and even regression (stage 4s)(table 1).
Table 1.
The international neuroblastoma staging system
| Stage | Tumour characteristics |
| Stage 1 | Localised tumour with complete gross excision, with or without microscopic residual disease; representative ipsilateral lymph nodes negative for tumour microscopically (nodes attached to and removed with the primary tumour may be positive) |
| Stage 2A | Localised tumour with incomplete gross excision: representative ipsilateral non-adherent lymph nodes negative for tumour microscopically |
| Stage 2B | Localised tumour with or without complete gross excision, with ipsilateral non-adherent lymph nodes positive for tumour. Enlarged contralateral lymph nodes must be negative microscopically |
| Stage 3 | Unresectable unilateral tumour, infiltrating across the midline, with or without regional lymph node involvement; or localised unilateral tumour with contralateral regional lymph node involvement; or midline tumour with bilateral extension by infiltration or lymph node involvement |
| Stage 4 | Any primary tumour with dissemination to distant lymph nodes, bone, bone marrow, liver, and/or other organs (except as defined for stage 4s) |
| Stage 4s | Localised primary tumour (as defined for stage 1, 2A, or 2B), with dissemination limited to liver, skin, and/or bone marrow (limited to infants <1 year of age) |
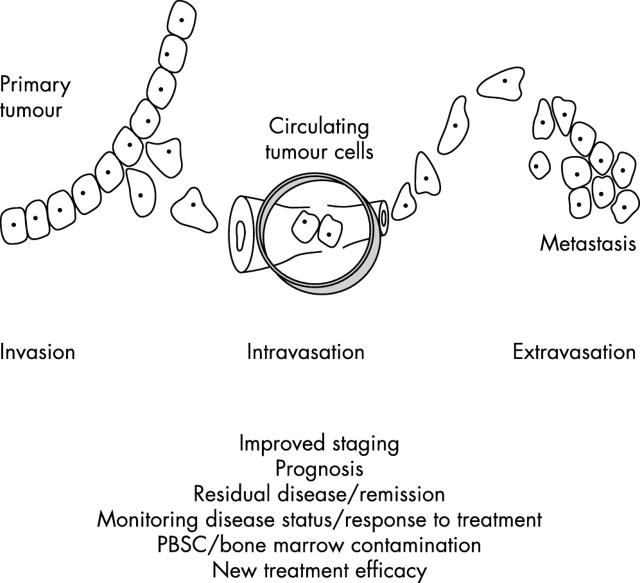
Most children over 1 year of age present with disseminated disease, identified in bone marrow (BM), distant lymph nodes, bone, liver, and/or other organs by imaging and/or histological examination using light microscopy (international neuroblastoma staging system stage 4; table 1; figs 1 and 2A.).3 Metastasis to the BM is a hallmark of high risk in NBL, and predictive of poor prognosis for most children, although children with stage 4s disease may present with BM metastases but still have a good outcome. Children with stage 1 or 2 disease—that is, no BM metastases detected by conventional cytology—have a much better prognosis. The presence of NBL cells in the BM can be used to evaluate response to treatment, although for more than half of those children with stage 4 disease prognosis remains poor, even with the use of dose intensive chemotherapy regimens. Although the presence of NBL cells detected in the BM by cytology is one of the most powerful markers of poor prognosis in NBL, some children with apparently localised disease at diagnosis have rapidly progressing disease of which they subsequently die. This suggests that these children may have micrometastatic disease at diagnosis that is not detected by current routine methods; more sensitive methods for the detection of disseminated disease may improve stratification for treatment and potentially outcome (fig 1). In addition, such methods may be applied to monitor disease course and detect relapse before overt metastases occur, resulting in improved staging and the application of more intensive treatment regimens or earlier clinical intervention for some children. Because the stage of the disease is an important prognostic indicator at diagnosis, and treatment strategies are determined by staging, the accurate assessment of clinically relevant disseminated disease in children with NBL is essential.
Figure 1.
Potential advantages of molecular detection of micrometastases. Metastatic disease, detected by imaging and/or light microscopy, is a feature of high risk (stage 4) neuroblastoma, and an important indicator of poor prognosis. For approximately 10% of children with stage 4 disease, those with limited stage disease (stages 1, 2, and 3), and the rare cases of stage 4s disease with no apparent bone marrow involvement, these methods are suboptimal. For these children, immunocytology or reverse transcriptase polymerase chain reaction may detect clinically significant disease that could improve outcome in several different ways. PBSC, peripheral blood stem cells.
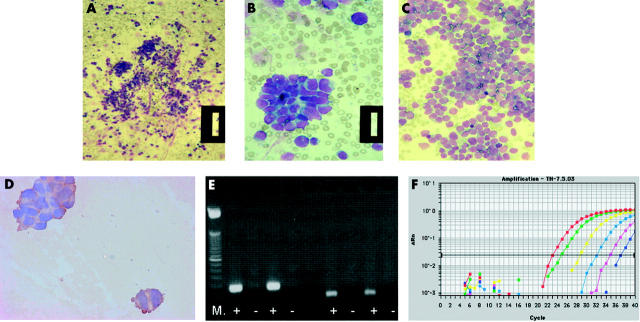
Figure 2.
Methods for the detection of micrometastases in bone marrow (BM), peripheral blood (PB), and peripheral blood stem cells from children with neuroblastoma (NBL). (A–C) Most children present with overt metastatic disease (stage 4), which can be detected by cytological assessment of BM smears for infiltrating NBL. Cytological features of infiltrating BM typically include clumps of NBL cells, readily identified from normal haemopoietic cells as blue stained round cells (A, B: patient 1; original magnification, ×16 and ×64, respectively). In some cases, the bone marrow infiltration can be so extensive that it can be difficult to identify normal BM cells (C: patient 2; original magnification, ×40). (B) Immunocytology (IC) with anti-GD2 antibodies can be used to help identify NBL cells: the cytoplasm of NBL cells is brown after staining with anti-GD2 antibodies and detection with diaminobenzidine. However, when there are less than 10 contaminating NBL cells, IC can be unreliable, unless combined with histological examination by an experienced pathologist or haematologist. (E) Reverse transcriptase polymerase chain reaction (RT-PCR) for tyrosine hydroxylase (TH) mRNA can detect down to one NBL cell in 1 × 106 normal cells. After amplification for TH mRNA, PCR products are separated on an agarose gel, stained with ethidium bromide, and visualised under ultraviolet light as a bright band. Results of RT-PCR for TH mRNA (left of the gel; product size, 180 bp) and the housekeeping gene β2 microglobulin (right of gel; product size, 136 bp) in two PB samples are shown. Relevant positive and negative controls must be included in any analysis, including amplification for a housekeeping gene to confirm the quality of isolated RNA (right of gel). M, molecular weight markers; +, RT present; −, RT absent; both PB samples were positive for TH mRNA and β2 microglobulin. (F) Amplification of TH mRNA by RT real time PCR is increasingly used to identify the number of target gene transcripts in each sample; using this method it is possible to detect a single transcript of TH mRNA/cell. Amplification curves for a set of serially diluted total RNA extracted from an NBL cell line are shown: red line, 250 ng total RNA; green, 25 ng; yellow, 2.5 ng; light blue, 250 pg; pink, 25 pg; blue, 2.5 pg. Change in fluorescence value, on the y axis, is the normalised target specific fluorescence signal, plotted against the number of PCR cycles on the x axis. Each dilution is usually carried out in triplicate; only a single plot for each RNA concentration is shown for clarity.
“Metastasis to the bone marrow (BM) is a hallmark of high risk in neuroblastoma, and predictive of poor prognosis for most children, although children with stage 4s disease may present with BM metastases but still have a good outcome”
Currently, staging of disease in NBL typically depends on imaging (computed tomography or MIBG (I-123 metaiodobenzylguanidine) scan) and assessment of BM for infiltrating NBL by Romanovsky stained BM smears and BM trephines stained with haematoxylin and eosin or Giemsa (table 1; fig 2A). Cytological features of infiltrating BM include clumps of NBL cells, rosettes, syncytia, and cytoplasmic/stromal characteristics. At least two posterior iliac crest BM aspirates and trephine (core) biopsies are examined at initial staging.3 These methods are informative for the assessment of disease in most children at diagnosis, because most present with overt metastatic disease. They are equally informative at the time of relapse when extensive disease is present.4 However for approximately 10% of children with stage 4 disease,5 those with limited stage disease (stages 1, 2, and 3), and the rare cases of stage 4s with no apparent BM involvement, these methods may be suboptimal. For these children the development of more sensitive methods for the detection of NBL cells and the evaluation of their clinical relevance is vital.
METHODS FOR DETECTING MICROMETASTATIC NBL
Nearly two decades ago, light microscopy coupled with immunocytological detection (IC) was shown to detect low numbers of tumour cells in BM from children with NBL.6–9 This technique has subsequently been used to detect NBL cells in peripheral blood stem cell harvest (PBSC)10 and peripheral blood (PB).11,12 This technique is dependent on the availability of antibodies to tumour associated cell surface antigens that are expressed on all target NBL cells, but not in the cells of the compartment to be studied—that is, normal haemopoietic cells. Because NBL is a tumour of neural origin, some groups have evaluated antibodies raised against human neural proteins; of these, UJ13A was reported to be the most useful.8,9 However, anti-GD2 antibodies appear to be the most specific and sensitive for the detection of NBL in BM.7,12 This is a robust approach to identify NBL when there are clumps of NBL cells within the BM (fig 2D). However, when there are less than 10 contaminating NBL cells on each slide, particularly if single or doublets of cells are stained, IC can be unreliable. Interpretation of PB is more difficult because NBL cells are often present as doublets or triplets—clumps comprising more than 10 cells do not pass into the peripheral vasculature. However, the technique is reported by some to detect a single NBL cell in 105 normal cells.11,12 Although IC combined with light microscopy may reliably detect 10 or more NBL cells clumped in a BM smear, this approach has not been introduced into clinical practice, probably reflecting the ambiguity of analyses and because clumps of 10 or more NBL cells can be identified by cytology alone. Some studies have used antibodies to detect NBL cells using flow cytometry, although the sensitivity of this approach alone is low.13–15
More recently, amplification of tissue specific mRNA by reverse transcriptase polymerase chain reaction (RT-PCR) has been used to detect minimal disease (fig 2E).16,17 This approach assumes that non-haemopoietic cells are not normally present in the compartment to be studied, and requires the identification of a target mRNA expressed in tumour cells but not normal haemopoietic cells. Because catecholamines are produced by NBL cells, the first enzyme in the catecholamine synthesis pathway, tyrosine hydroxylase (TH), has most frequently been used as a target for the detection of NBL by RT-PCR. Although other targets for the detection of NBL cells by RT-PCR have been evaluated, TH mRNA is the single most useful target.18–20 Using this methodology, a single NBL cell may be specifically detected in 1 × 106 normal cells, with no reports of false positives.21,22 However, some studies have shown that multiplex PCR for a panel of target genes expressed by NBL may increase the sensitivity of tumour cell detection, overcoming tumour heterogeneity,22,23 although this is very target dependent, and in some cases may result in reduced specificity.19 Furthermore, the choice of target may depend on the compartment to be studied—that is, targets may be expressed in normal BM but not in normal PB.16,17 Reflecting these challenges, some studies have combined RT-PCR with flow cytometry15 or IC24 in an attempt to increase sensitivity and specificity.
“Because catecholamines are produced by neuroblastoma cells, the first enzyme in the catecholamine synthesis pathway, tyrosine hydroxylase, has most frequently been used as a target for the detection of neuroblastoma by reverse transcriptase polymerase chain reaction”
Although RT-PCR may detect low numbers of NBL cells with an increased sensitivity and specificity compared with other methods, it does not measure absolute tumour cell numbers. This will be a limitation if a critical number of tumour cells in the BM or PB are prognostically important, and not purely the presence or absence of disease (see below). The development of semiquantitative methods, comparing amounts of TH mRNA with those of a housekeeping gene, such as β2 microglobulin, has allowed potentially useful evaluation of changes in tumour cell burden within the PB of a child during the disease course (fig 3A).25 However, target mRNA expression must be interpreted carefully because it is influenced by many factors, such as suboptimal conditions for collection and storage of samples, inappropriate sample processing, low sensitivity RT-PCR techniques, and changes in the transcription of target genes. More recent studies have described real time PCR that can determine target gene transcript numbers in NBL clinical samples (fig 2D).26–29 Using this method it is possible to detect a single TH29 or GD226 transcript. Some groups have suggested that this technique may be used for the accurate assessment of tumour cell number within a sample, although variability in transcript number in each NBL cell will limit this. Further studies of transcript numbers in NBL and normal cells are required to define the parameters for unambiguous interpretation of real time RT-PCR to detect circulating tumour cells. However, real time PCR can be used to monitor changes in tumour cell burden throughout the disease course in the same way as more conventional PCR (fig 3C).29 Whether the increased sensitivity of real time PCR compared with more traditional PCR methods will be an advantage for the assessment of clinically relevant disease in children with NBL remains to be seen. The ability to detect absolute transcript numbers accurately is certainly valuable for the evaluation of optimal methods for collection, storage, and preparation of clinical samples (B Kågedal and SA Burchill, unpublished, 2003).
Figure 3.
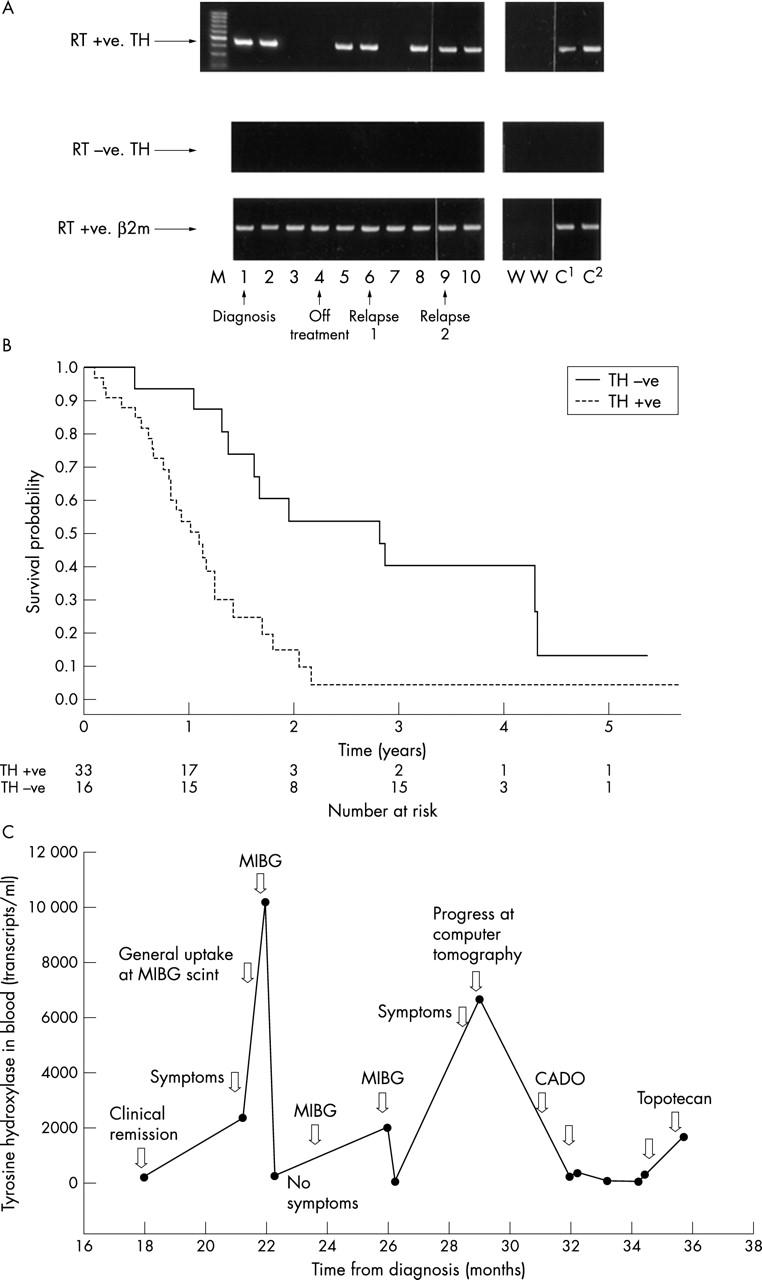
Assessment of disease status by reverse transcriptase polymerase chain reaction (RT-PCR) in children with neuroblastoma (NBL). RT-PCR and RT real time PCR are the most sensitive and specific methods for the detection of micrometastases, although their clinical usefulness is unclear. Using PCR based methods it is not possible to measure absolute NBL cell numbers, but changes in disease can be monitored throughout the disease course by RT-PCR (A) or RT real time PCR (C). (A) Detection of tyrosine hyroxylase (TH) mRNA (NBL cells) in sequential blood samples taken from a child with stage 4 NBL. Blood samples were positive at diagnosis (1) and four weeks into treatment (2), but negative five (3) and seven (4) weeks after the start of treatment. Peripheral blood (PB) was positive for TH mRNA at 13 weeks (5) and when the child was clinically disease free at 12 months (6), but negative at 13 months (7). Before clinical relapse the PB was positive (8; 14 months), and remained positive (9; 17 months) until relapse 19 months after initial diagnosis (10). The child did not respond to further treatment and died of disease 22 months after initial diagnosis. RT+ve, RT present; RT-ve, RT absent (negative control); β2m, β2 microglobulin mRNA (housekeeping gene used to check quality of RNA); M, molecular weight markers; W, negative control; C1 and C2, positive controls. (B) The presence of TH mRNA in PB from children with high risk (stage 4) NBL greater than 1 year old at diagnosis is associated with an increased risk of death; p = 0.002 (95% confidence interval, 1.49 to 6.66), log rank test. (C) TH mRNA detected by RT real time PCR in PB from a child with stage 4 NBL taken 18–36 months after diagnosis. TH mRNA increased in PB at the time of relapse and dropped after further treatment. (A, B) Reproduced from Burchill et al;25 (C) reproduced from Träger et al.29 CADO, vincristinecyclo-phosphamide-doxorubicin; MIBG, I-125 metaiodobenzylguanidine.
Additional methods to assess clinical samples for contaminating tumour cells are the tumour clonogenic assay30 and genetic profiling.31 Both these methods have the potential advantage that they not only detect circulating tumour cells that may go on to form metastases, but they also assess their potential neoplastic nature by evaluating the growth potential or the biological genotype or phenotype of the cells. Using an automatic immunofluorescence and fluorescence in situ hybridisation device it is possible to quantify disseminating tumour cells and characterise the genetic profile of these cells; this is a powerful method that can confirm the malignant identity of cells detected by IC and potentially can more accurately predict their prognostic impact by evaluating the genotype of the cell.31 Therefore, more extensive prognostic information may be achieved by combining sensitive methods for the detection of micrometastases with methods to characterise biological markers of viability, growth, invasion, and multidrug resistance. This approach might allow the discrimination of disseminating tumour cells with unfavourable biology that go on to form metastases from those that do not metastasise. For example, although most children with stage 4s disease have a good prognosis, despite the presence of BM metastases, 10–25% have a poor prognosis. The identification of those children with disseminating tumour cells having an unfavourable biology and poor outcome should improve clinical management and potentially survival for this group.
“Additional methods to assess clinical samples for contaminating tumour cells are the tumour clonogenic assay and genetic profiling”
CLINICAL RELEVANCE OF NBL MICROMETASTASES
Using sensitive methods for the detection of NBL it is now clear that micrometastases frequently occur in children with NBL.16,17,32,33 Although the detection of NBL cells in BM or PB is not sufficient for the development of overt metastatic lesions (fig 1), the presence of such cells is necessary for disease dissemination. The most important challenge is to determine whether the detection of disease using these sensitive methods provides a more reliable tool for the identification of children with high risk NBL than current routine methods.
AT DIAGNOSIS
The presence of NBL cells detected by IC4,12,34 or RT-PCR22,35,36 in BM taken at diagnosis identifies those children with an unfavourable prognosis by association with disease stage. The association with disease stage is good evidence that using these sensitive methods for the detection of disease produces data of clinical relevance. More interestingly, the frequency of NBL cell contamination, detected by RT-PCR, of BM taken at diagnosis from children with stage 4 disease is reported to be 95–100%,20,35,37 compared with a reported frequency of 90% by conventional cytology.5 Similarly, increased frequency of tumour cell detection by IC in BM from children with widespread disease (stage 4) has been described.34 These observations suggest that RT-PCR and IC are more sensitive methods than conventional cytology to detect NBL cells in BM; however, whether or not these tumour cells are prognostically relevant remains to be seen. The identification of BM micrometastases in children diagnosed with high risk stage 4 disease is currently unlikely to have an effect on outcome because these children are already high risk and treated accordingly; some may benefit from more intensive therapy, but for most novel therapeutic strategies are needed. In this group of children, these sensitive methods may be most useful to identify those for whom current treatments would not be useful.
Although there is limited information on the frequency of NBL detection in BM from children with stage 1, 2, 3, or 4s disease, NBL cells have been detected in BM from children with low stage disease analysed by RT-PCR (28% positive BM)37 or IC (34% positive BM).34 This is consistent with the hypothesis that these children have disseminated disease that is not detected by cytology. It is this group of children in whom it is most important to establish whether the detection of NBL cells by more sensitive techniques is clinically important, and whether they might benefit from upstaging and more aggressive treatment. Whether the detection of micrometastases by PCR correlates with other biological markers of poor outcome (for example, MYC-N amplification in children with stage 2 or 3 disease) remains to be seen; in children with high risk disease the detection of NBL cells by RT-PCR in BM is associated with other unfavourable biological features.37
RT-PCR of PB taken at diagnosis from children with NBL also identifies those with an unfavourable prognosis by association with stage; that is, tumour cells are frequently detected in PB from children with stage 4 disease, but rarely in those with stage 1, 2, or 3 disease.21,38 More importantly, the detection of circulating tumour cells in PB by RT-PCR in a small number of children diagnosed with stage 3 disease appears to correlate with poor outcome,21 again supporting the restaging for risk of some children with stage 3 disease. Further studies are essential in this group of children.
In children with advanced stage disease (in this case, stage 4 over 1 year of age) the detection of NBL micrometastases in PB is predictive of rapidly progressing disease (fig 3B).25 However, as is the case for low level disease detected in BM (see above), there remains a need for new treatment modalities before the treatment of children with advanced disease guided by molecular detection results in improved outcome and survival benefit for many. The frequency of NBL detection in PB taken at diagnosis is considerably less than that in BM from the same population taken at diagnosis; using RT-PCR in PB, approximately 50% of blood samples from children with high stage disease are positive, compared with 95–100% of BM samples (SA Burchill, personal observation, 1999).21 Similarly, a two log difference in detection of disease between PB and BM is reported by IC.12 The absence of NBL cells in PB from children with high risk disease has been reported by some as “false” negatives, although this over simplifies the situation. The “false” negatives will reflect biological features such as intermittent tumour cell shedding, dilution of tumour cells in PB, and technical factors such as frequency and volume of samples analysed. The different distribution of NBL cells in PB compared with BM suggests that circulating NBL cells do not necessarily “seed” in the BM, and NBL cells in the BM do not inevitably disseminate into the PB. The high frequency of positive BM analysed using PCR based methods reflects the detection of overt metastases, in addition to NBL cells that may be micrometastases or disseminating through the BM but not forming metastases. In contrast, the detection of NBL cells in PB will reflect only disseminating disease, which may or may not develop clinically relevant micrometastases or overt metastases. The presence of NBL cells in these two compartments might therefore have altered prognostic power for different stages of disease. An accurate assessment of the clinical value of RT-PCR and IC in these two compartments is essential to determine their place in clinical practice, and to understand the metastatic disease process in NBL.
MONITORING DISEASE: DURING, AT THE END OF TREATMENT, AND ON FOLLOW UP
Currently, for most children diagnosed with NBL (those with high risk disease), detection of micrometastases will probably have its biggest impact as a tool to monitor disease course and response to treatment. The most informative studies evaluating the power of micrometastatic disease detection throughout the clinical course have been made using RT-PCR. The analysis of BM20,35 or PB25,29,39 samples taken sequentially from children suggests that RT-PCR may be used to monitor response to treatment (fig 3A, C). Although the measurement of catecholamine metabolites in the urine has been used to monitor disease in children, this reflects turnover of tumour mass. Detection of disease by RT-PCR may be a more informative measure of disease status because it identifies disseminating NBL cells that have the potential to form overt metastases.16,17 Although the presence of tumour cells in PB at the end of treatment is rare, such cells are associated with rapidly progressing disease and a high relapse rate.25 This may be useful to help determine whether children might benefit from further treatment, and might result in a redefinition of disease free. Similarly, the detection of NBL cells in PB from clinically disease free children is potentially a powerful tool to identify micrometastases before clinical relapse. Preliminary studies have shown that NBL cells can be detected in children up to 11 months before urinary catecholamine concentrations increase and clinical relapse occurs,21 potentially providing an opportunity for earlier clinical intervention that may improve outcome for some children.
“Currently, for most children diagnosed with neuroblastoma (those with high risk disease), detection of micrometastases will probably have its biggest impact as a tool to monitor disease course and response to treatment”
The superior response of children with stage 4 NBL to high dose chemotherapy has stimulated greater use of autologous BM or stem cell transplantation, to improve tolerance to the intensive treatment. Although these more intensive treatments have improved response rates, in many children this does not result in an increased overall survival. This may be explained by drug resistant disease or poor immune response. Alternatively, the correlation between tumour cell detection in PBSCs and poor post-transplant clinical outcome10,40 supports a role for reinfused tumour cells in relapse. Although there is no evidence that cytokines, such as granulocyte colony stimulating factor, used to mobilise stem cells during the preparation of PBSCs increase tumour cell contamination,40a there is substantial risk of tumour cell contamination in PBSC harvests, when analysed by RT-PCR40 or IC.32 The detection of tumour cells in PBSC harvests might equally reflect the advanced disease status of these children—tumour cells might be preferentially detected in PBSC harvests from those children with more aggressive disease who are more likely to relapse, although the presence of gene marked reinfused NBL cells at sites of disease relapse41,42 strongly supports a role for these reinfused NBL cells in the development of clinical relapse. The role of such cells may ultimately result in a need to reduce tumour burden in harvested peripheral blood or bone marrow stem cells before infusion.32 This might be achieved by purging to remove tumour cells (negative selection), enriching grafts for stem cell progenitors (positive selection), or the collection of stem cells at a time when they can be shown to be tumour free.40a,43–46
Take home messages.
Recurrence and metastasis are still common in neuroblastoma and need to be monitored to predict outcome and help in treatment decisions
Accurate methods to detect micrometastatic disease, such as reverse transcriptase polymerase chain reaction (RT-PCR), RT real time PCR, and genetic profiling, have been developed
However, the clinical relevance and usefulness of detecting micrometastatic disease are still unclear, emphasising the need for multicentre prospective clinical outcome studies
Although the capacity to detect NBL micrometastases does not inevitably translate into improved outcome for children with the disease, the ability to detect circulating tumour cells accurately may allow the evaluation of new strategies targeting this disease. Consequently, as new treatments evolve, so molecular information may be useful in monitoring their impact. For example, in the current European Société Internationale d’Oncologie Pédiatrique high risk neuroblastoma study, in the final randomisation children are treated for minimal residual disease using 13 cis-retinoic acid,47,48 alone and in combination with anti-GD2 antibody.36,49 By analysing BM and PB taken before and after this treatment for NBL cells, it will be possible to evaluate and compare the relative efficacy of these two treatments. A second crucial aim of this prospective international study is to evaluate the clinical relevance of NBL cells detected using RT-PCR or IC.
The detection of clinically relevant NBL micrometastases may provide improved risk information for children with low stage NBL at diagnosis, and throughout the disease course provide a method to monitor metastatic disease status in all children. This will probably result in a redefinition of what constitutes disease free and relapse. However, despite the literature on micrometastatic disease in NBL, the small numbers of children studied thus far and inconsistent reporting emphasise the need for large, multicentre, quality controlled, prospective clinical outcome studies. Such studies will provide information on the clinical usefulness and best clinical practice to maximise the benefit children with NBL may obtain from more sensitive methods for the detection of disease.
Acknowledgments
Thanks to Dr S Kinsey for providing images of bone marrow cytology, and to Professor B Kågedal for permission to reproduce fig 3C.
Abbreviations
BM, bone marrow
IC, immunocytology
NBL, neuroblastoma
PB, peripheral blood
PBSC, peripheral blood stem cells
PCR, polymerase chain reaction
RT, reverse transcriptase
TH, tyrosine hydroxylase
REFERENCES
- 1.Bolande RP. The spontaneous regression of neuroblastoma. Experimental evidence for a natural host immunity. Pathol Annu 1991;26:187–99. [PubMed] [Google Scholar]
- 2.Wilson LM. Neuroblastoma, its natural history and prognosis: a study of 487 cases. BMJ 1974;3:301–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brodeur GM, Pritchard J, Berthold F, et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment. J Clin Oncol 1993;11:1466–77. [DOI] [PubMed] [Google Scholar]
- 4.Cheung NK, Heller G, Kushner BH, et al. Detection of metastatic neuroblastoma in bone marrow: when is routine marrow histology insensitive? J Clin Oncol 1997;8:2807–17. [DOI] [PubMed] [Google Scholar]
- 5.Reid MM, Pearson ADJ. Bone marrow infiltration in neuroblastoma. Lancet 1991;337:681–2. [DOI] [PubMed] [Google Scholar]
- 6.Favrot MC, Frappaz D, Maritax O, et al. Histological, cytological and immunological analyses are complementary for the detection of neuroblastoma cells in bone marrow. Br J Cancer 1986;54:637–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheung NK, Von Hoff DD, Strandjord SE, et al. Detection of neuroblastoma cells in bone marrow using GD2 specific monoclonal antibodies. J Clin Oncol 1986;4:363–9. [DOI] [PubMed] [Google Scholar]
- 8.Rogers DW, Treleaven JG, Kemshead JT, et al. Monoclonal antibodies for detecting bone marrow invasion by neuroblastoma. J Clin Pathol 1989;42:422–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carey PJ, Thomas L, Buckle G, et al. Immunocytochemical examination of bone marrow in disseminated neuroblastoma. J Clin Pathol 1990;43:9–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moss TJ, Cairo M, Santana VM, et al. Clonogenicity of circulating neuroblastoma cells: implications regarding peripheral blood stem cell transplantation. Blood 1994;83:3085–9. [PubMed] [Google Scholar]
- 11.Moss TJ, Sanders DG. Detection of neuroblastoma cells in blood. J Clin Oncol 1990;8:736–40. [DOI] [PubMed] [Google Scholar]
- 12.Faulkner LB, Tintori V, Tamburini A, et al. High-sensitivity immunocytologic analysis of neuroblastoma cells in paired blood and marrow samples. J Hematother 1998;4:361–6. [DOI] [PubMed] [Google Scholar]
- 13.Komada Y, Zhang XL, Zhou YW, et al. Flow cytometric analysis of peripheral blood and bone marrow for tumor cells in patients with neuroblastoma. Cancer 1998;82:591–9. [PubMed] [Google Scholar]
- 14.Nagai J, Ishida Y, Koga N, et al. A new sensitive and specific combination of CD81/CD56/CD45 monoclonal antibodies for detecting circulating neuroblastoma cells in peripheral blood using flow cytometry. J Pediatr Hematol Oncol 2000;22:20–6. [DOI] [PubMed] [Google Scholar]
- 15.Sze Tsang K, Kong Li C, Chiu Tsoi W, et al. Detection of micro-metastases of neuroblastoma to bone marrow and tumor dissemination to hematopoietic autografts using flow cytometry and reverse transcriptase-polymerase chain reaction. Cancer 2003;97:2887–97. [DOI] [PubMed] [Google Scholar]
- 16.Burchill SA, Selby P. Early detection of small volume disease using molecular technology. Cancer Topics 1999;10:1–4. [Google Scholar]
- 17.Burchill SA, Selby PJ. Molecular detection of low-level disease in patients with cancer. J Pathol 2000;190:6–14. [DOI] [PubMed] [Google Scholar]
- 18.Kuroda T, Saeki M, Nakano M, et al. Clinical application of minimal residual neuroblastoma cell detection by reverse transcriptase-polymerase chain reaction. J Pediatr Surg 1997;32:69–72. [DOI] [PubMed] [Google Scholar]
- 19.Gilbert J, Norris MD, Marshall GM, et al. Low specificity of PGP9.5 expression for detection of micro-metastatic neuroblastoma. Br J Cancer 1997;75:1779–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Miyajima Y, Kato K, Numata S, et al. Detection of neuroblastoma cells in bone marrow and peripheral blood at diagnosis by the reverse transcriptase-polymerase chain reaction for tyrosine hydroxylase mRNA. Cancer 1995;75:2757–61. [DOI] [PubMed] [Google Scholar]
- 21.Burchill SA, Bradbury FM, Lewis IJ. Early clinical evaluation of reverse transcriptase-polymerase chain reaction (RT-PCR) for tyrosine hydroxylase. Eur J Cancer 1995;31:553–6. [DOI] [PubMed] [Google Scholar]
- 22.Cheung IY, Cheung NK. Detection of microscopic disease: comparing histology, immunocytology, and RT-PCR of tyrosine hydroxylase, GAGE, and MAGE. Med Pediatr Oncol 2001;36:210–12. [DOI] [PubMed] [Google Scholar]
- 23.Cheung IY, Barber D, Cheung N. Detection of microscopic neuroblastoma in marrow by histology, immunocytology and reverse transcription-PCR of multiple molecular markers. Clin Cancer Res 1998;4:2801–5. [PubMed] [Google Scholar]
- 24.Lode HN, Handgretinger R, Schuermann U, et al. Detection of neuroblastoma cells in CD34+ selected peripheral stem cells using a combination of tyrosine hydroxylase nested RT-PCR and anti-ganglioside GD2 immunocytochemistry. Eur J Cancer 1997;33:2024–30. [DOI] [PubMed] [Google Scholar]
- 25.Burchill SA, Lewis IJ, Abrams KR, et al. Circulating neuroblastoma cells detected by reverse transcriptase polymerase chain reaction for tyrosine hydroxylase mRNA are an independent poor prognostic indicator. J Clin Oncol 2001;19:1795–801. [DOI] [PubMed] [Google Scholar]
- 26.Cheung IY, Cheung NK. Quantitation of marrow disease in neuroblastoma by real-time reverse transcription-PCR. Clin Cancer Res 2001;7:1698–705. [PubMed] [Google Scholar]
- 27.Pession A, Libri V, Sartini R, et al. Real-time RT-PCR of tyrosine hydroxylase to detect bone marrow involvement in advanced neuroblastoma. Oncol Rep 2003;10:357–62. [PubMed] [Google Scholar]
- 28.Lambooy LHJ, Gidding CEM, van den Heuvel LP, et al. Real-time analysis of tyrosine hydroxylase gene expression: a sensitive and semiquantitative marker for minimal residual disease detection of neuroblastoma. Clin Cancer Res 2003;9:812–19. [PubMed] [Google Scholar]
- 29.Träger C, Kogner P, Lindskog M, et al. Quantitative analysis of tyrosine hydroxylase mRNA for sensitive detection of neuroblastoma cells in blood and bone marrow. Clin Chem 2003;49:104–12. [DOI] [PubMed] [Google Scholar]
- 30.Hamburger AW, Salmon SE. Primary bioassay of human tumor stem cells. Science 1977;197:461–3. [DOI] [PubMed] [Google Scholar]
- 31.Mehes G, Luegmayr A, Kornmuller R, et al. Detection of disseminated tumor cells in neuroblastoma: 3 log improvement in sensitivity by automatic immunofluorescence plus FISH (AIPF) analysis compared with classical bone marrow cytology. Am J Pathol 2003;163:393–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ross AM. Minimal residual disease in solid tumour malignancies: a review. J Hematother 1998;7:9–18. [DOI] [PubMed] [Google Scholar]
- 33.Moss TJ. Clinical relevance of minimal residual cancer in patients with solid malignancies. Cancer Metastasis Rev 1999;18:91–100. [DOI] [PubMed] [Google Scholar]
- 34.Moss T, Rynolds C, Sather H, et al. Prognostic value of immunocytologic detection of bone marrow metastases in neuroblastoma. N Engl J Med 1991;324:219–26. [DOI] [PubMed] [Google Scholar]
- 35.Horibe K, Fukuda M, Miyajima Y, et al. Outcome prediction by molecular detection of minimal residual disease in bone marrow for advanced neuroblastoma. Med Pediatr Oncol 2001;36:203–4. [DOI] [PubMed] [Google Scholar]
- 36.Fukuda M, Horibe K, Furukawa K. Enhancement of in vitro and in vivo anti-tumor activity of anti-GD2 monoclonal antibody 220–51 against human neuroblastoma by granulocyte–macrophage colony-stimulating factor and granulocyte colony-stimulating factor. Int J Mol Med 1998;2:471–5. [DOI] [PubMed] [Google Scholar]
- 37.Shono K, Tajiri T, Fujii Y, et al. Clinical implications of minimal disease in the bone marrow and peripheral blood in neuroblastoma. J Pediatr Surg 2000;35:1415–20. [DOI] [PubMed] [Google Scholar]
- 38.Gao Y, Li G, Zhang X, et al. Detection of neuroblastoma cells in blood by reverse transcriptase-polymerase chain reaction. Chin Med J (Engl) 1997;110:341–5. [PubMed] [Google Scholar]
- 39.Miyajima Y, Horibe K, Fukuda M, et al. Sequential detection of tumor cells in the peripheral blood and bone marrow of patients with stage IV neuroblastoma by the reverse transcription-polymerase chain reaction for tyrosine hydroxylase mRNA. Cancer 1996;77:1214–19. [DOI] [PubMed] [Google Scholar]
- 40.Burchill SA, Kinsey SE, Roberts P, et al. Minimal residual disease at the time of peripheral blood stem cell harvest in patients with advanced neuroblastoma. Med Pediatr Oncol 2001;36:213–19. [DOI] [PubMed] [Google Scholar]
- 40a.Burchill SA, Picton S, Wheeldon J, et al. Reduced tumour load in peripheral blood after treatment with G-CSF and chemotherapy in children with tumours of the Ewing sarcoma family but not neuroblastoma. Blood 2003;102:1–2. [DOI] [PubMed] [Google Scholar]
- 41.Brenner MK, Rill DR, Holladay MS, et al. Gene marking to determine whether autologous marrow infusion restores long-term haemopoiesis in cancer patients. Lancet 1993;342:1134–7. [DOI] [PubMed] [Google Scholar]
- 42.Rill DR, Santana VM, Roberts WM, et al. Direct demonstration that autologous bone marrow transplantation for solid tumours can return a multiplicity of tumorigenic cells. Blood 1994;84:380–3. [PubMed] [Google Scholar]
- 43.Atra A, Pinkerton R. Autologous stem cell transplantation in solid tumours of childhood. Ann Med 1996;28:159–64. [DOI] [PubMed] [Google Scholar]
- 44.Handgretinger R, Greil J, Schurmann U, et al. Positive selection and transplantation of peripheral CD34+ progenitor cells: feasibility and purging efficacy in pediatric patients with neuroblastoma. J Hematother 1997;6:235–42. [DOI] [PubMed] [Google Scholar]
- 45.Tchirkov A, Kanold J, Giollant M, et al. Molecular monitoring of tumor cell contamination in leukapheresis products from stage IV neuroblastoma patients before and after positive CD34 selection. Med Pediatr Oncol 1998;30:228–32. [DOI] [PubMed] [Google Scholar]
- 46.Leung W, Chen AR, Klann, et al. Frequent detection of tumor cells in hematopoietic grafts in neuroblastoma and Ewing’s sarcoma. Bone Marrow Transplant 1998;22:971–9. [DOI] [PubMed] [Google Scholar]
- 47.Matthay KK, Villablanca JG, Seeger RC, et al. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-CIS-retinoic acid. N Engl J Med 1999;341:1165–73. [DOI] [PubMed] [Google Scholar]
- 48.Villablanca JG, Khan AA, Avramis VI, et al. Phase I trial of 13-cis-retinoic acid in children with neuroblastoma following bone marrow transplantation. J Clin Oncol 1995;13:894–901. [DOI] [PubMed] [Google Scholar]
- 49.Manzke O, Russello O, Leenen C, et al. Immunotherapeutic strategies in neuroblastoma: antitumoral activity of deglycosylated ricin A conjugated anti-GD2 antibodies and anti-CD3xanti-GD2 bispecific antibodies. Med Pediatr Oncol 2001;36:185–9. [DOI] [PubMed] [Google Scholar]