Abstract
A 40 year old man presented with abdominal pain, jaundice, weight loss, and hepatosplenomegaly. Liver function tests revealed cholestatic jaundice and a computed tomography scan showed an enlarged liver, with a normal biliary tree. Liver biopsy showed diffuse infiltration by neutrophils, monocytoid cells, and blasts. Peripheral blood film and bone marrow were consistent with acute myeloid leukaemia. After treatment with chemotherapy using an acute myeloid leukaemia protocol (UK Medical Research Council AML-12), there was complete resolution of jaundice and the patient went into complete molecular remission.
Keywords: acute myeloid leukaemia, cholestasis, hepatic failure
Jaundice is rarely a presenting feature of acute myeloid leukaemia. More often, it is treatment related. Obstructive jaundice has been reported on rare occasions in acute myeloid leukaemia (AML).1 We report a patient with AML who presented with cholestatic jaundice as a result of hepatic sinusoidal infiltration, which regressed with chemotherapy.
CASE REPORT
A 40 year old man presented in January 2000 with a history of abdominal pain of one month’s duration with some preceding cough, coryzal symptoms, and loss of appetite. He was suffering from longstanding depression and had been on antidepressants (paroxetine) for one year. Physical examination showed pyrexia cachexia, jaundice, few palpable cervical lymph nodes, and hepatosplenomegaly.
Laboratory investigations showed a haemoglobin concentration of 114 g/litre, a white blood cell count of 23.4 × 109/litre, and a platelet count of 75 × 109/litre. The differential count showed: neutrophils, 6.3 × 109/litre; lymphocytes, 3.7 × 109/litre; monocytes, 12.4 × 109/litre; myelocytes 0.2 109/litre; eosinophils, 0.5 109/litre; and blasts, 0.2 × 109/litre.
Examination of the blood film showed many dysplastic monocytes with occasional monoblasts. Bone marrow aspirate showed 22% monocytes, 17% blasts, 0.7% promonocytes, and 20% dysplastic eosinophils. Bone marrow trephine showed an increased number of myeloid and monocytoid cells, with few blasts.
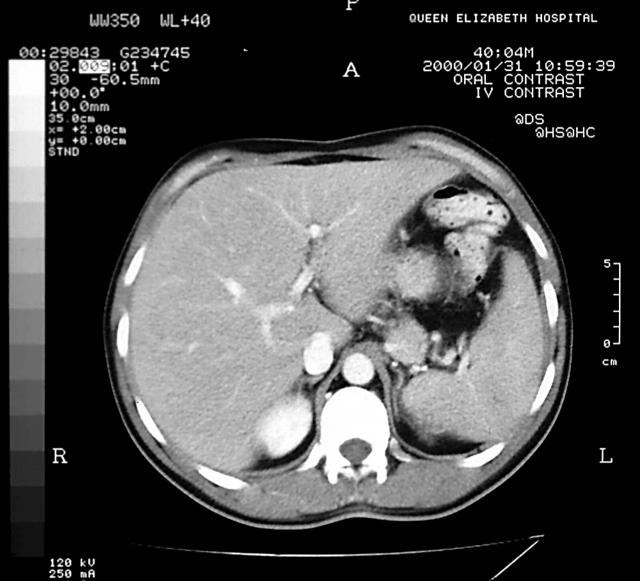
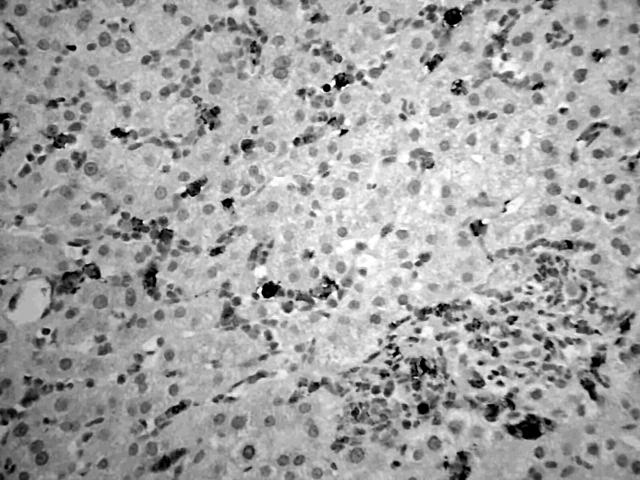
Immunophenotyping of the bone marrow sample showed that 85% of cells were positive for CD13 and CD33; 10% of these cells were CD34 positive, 38% CD11b positive, 46% CD11c positive, and 29% CD14 positive. Cytogenetics on the bone marrow sample showed inversion of chromosome 16 and an additional chromosome 2 (46, XY, add (2)(q37), inv (16) (p13q22)). Molecular analysis of the bone marrow by polymerase chain reaction confirmed the presence of the CBFB–MYH11 fusion gene. These results were consistent with the diagnosis of AML with abnormal bone marrow eosinophils (inv (16) (p13q22), CBFB–MYH11) according to the World Health Organisation classification.2 Serum bilirubin was 80 μmol/litre (normal range, 3–17), alkaline phosphatase was 1258 U/litre (normal range, 30–150), and aspartate aminotransferase was 142 IU/litre (normal range, 5–35). Serological tests for hepatitis viruses were negative. A computed tomography scan showed enlarged liver and spleen with altered echogenicity of the liver but normal biliary ducts (fig 1). Liver biopsy showed diffuse sinusoidal infiltration by a pleomorphic population of cells, mostly consisting of neutrophils and monocytes, with some immature blasts. There was moderate perivenular cholestasis. A similar cellular infiltrate was also present in the portal tracts. Immunohistochemistry showed that most of the cells infiltrating the portal tracts and sinusoids expressed the myeloid marker CD15 and macrophage/monocyte marker CD68, whereas a few sinusoidal cells were positive for CD34, a marker for haemopoeitic precursor cells (fig 2). The patient became progressively unwell with pyrexia, hypoxia, and a rise in bilirubin (413 μmol/litre), aspartate aminotransferase (172 IU/litre), and alkaline phosphatase (1442 U/litre). He was treated according to the UK Medical Research Council (UK MRC-12) protocol and showed a very good response to chemotherapy (dose adjusted for liver dysfunction), with liver function tests resolving back to normal. He went on to receive four courses of chemotherapy on this protocol and is currently (four years after diagnosis) in complete cytogenetic and molecular remission with normal liver function tests.
Figure 1.
Computed tomography scan of abdomen showing hepatomegaly with normal biliary tree.
Figure 2.
A liver biopsy showing sinusoidal infiltration by CD15 positive myeloid cells. A similar infiltrate is present in a small portal tract. Immunoperoxidase staining for CD15.
DISCUSSION
Jaundice is rarely seen as a presenting feature in AML. It can occur as a result of drug induced hepatocellular damage, obstruction to the flow of bile, post transfusion viral hepatitis, or infiltration of liver by the leukaemic process or granulocytic sarcoma.3 Most case reports of jaundice in AML are the result of obstructive jaundice secondary to granulocytic sarcoma.3–5 Our patient is atypical in that he had interesting hepatic histology with diffuse sinusoidal infiltration of the liver by leukaemic cells. Although our patient was on longterm antidepressant treatment (paroxetine), which can cause liver dysfunction, we confirmed leukaemic infiltration of the liver by liver biopsy. He showed a good response to chemotherapy and went into complete molecular remission, with normal liver function. This case also illustrates that liver dysfunction secondary to leukaemic infiltration can respond to successful treatment (with appropriate dose adjustment of the chemotherapy drugs) of the underlying haematological condition.
Take home messages.
We report a rare case of acute myeloid leukaemia presenting as jaundice
Chemotherapy resulted in complete resolution of the jaundice and complete molecular remission
Thus, liver dysfunction secondary to leukaemic infiltration can respond to successful treatment of the underlying haematological condition
Abbreviations
AML, acute myeloid leukaemia
REFERENCES
- 1.Verma N, Prakash D, Marwah N, et al. Granulocytic sarcoma preceding acute myeloid leukaemia. Indian J Pediatr 1989;56:775–6. [DOI] [PubMed] [Google Scholar]
- 2.Haris LH, Jaffe SE, Diebold J, et al. World Health Organisation classification of neoplastic diseases of the haematopoietic and lymphoid tissues: report of the clinical advisory committee meeting—Airlie House, Virginia, November 1997. J Clin Oncol 1999;17:3835–49. [DOI] [PubMed] [Google Scholar]
- 3.Mclord RG, Gilbert EF, Jod PJ. Acute leukaemia presenting as jaundice with acute liver failure. Clin Pedriatr (Phila) 1973;12:17–20. [DOI] [PubMed] [Google Scholar]
- 4.Abe Y, Takatsuki H, Okada Y, et al. Mucosa-associated lymphoid tissue type lymphoma of the gall bladder associated with acute myeloid leukaemia. Intern Med 1999;38:442–4. [DOI] [PubMed] [Google Scholar]
- 5.King DJ, Ewen SWB, Sewell HF, et al. Obstructive jaundice: an unusual presentation of granulocytic sarcoma. Cancer 1987;60:114–17. [DOI] [PubMed] [Google Scholar]