Abstract
Aim: Cohen syndrome is an uncommon autosomal recessive condition comprising a characteristic facial appearance, mental retardation, benign neutropenia, and retinal dystrophy. This study aimed to identify patients with Cohen syndrome from across the United Kingdom in order to define the variability of ophthalmic manifestations.
Methods: Ophthalmic assessment was undertaken and past ophthalmic records reviewed in 22 patients with classic features of Cohen syndrome.
Results: All patients had visual problems which commonly started in the preschool years. 82% developed strabismus or refractive error during the first 5 years of life. 70% developed high myopia by the second decade. By contrast with the findings of others, early onset retinal dystrophy was common, occurring in 80% of study patients under age 5 years. 35% of patients were registered partially sighted or blind.
Conclusion: The ophthalmic abnormalities associated with Cohen syndrome, including high myopia and a generalised, severe retinal dystrophy, are of early onset and frequently result in severe visual handicap. Cohen syndrome should be considered in the young, developmentally delayed child who presents with severe myopia and nyctalopia.
Keywords: Cohen syndrome, myopia, pigmentary retinopathy
Cohen syndrome was first described in 1973 and is an uncommon autosomal recessive syndrome comprising mental retardation, joint hyperextensibility, truncal obesity, myopia and retinal dystrophy, and a benign neutropenia.1 There is a characteristic facial appearance with a short, upturned philtrum, a grimacing smile, and prominent upper incisors. Over 100 cases have been described.2–14,17–20 Owing to a strong founder effect, the condition is relatively common in Finland and in this patient population a locus for Cohen syndrome (COH1) has been mapped to 8q23, by means of linkage analysis, with a common haplotype shared by the majority.15,16
Ophthalmic abnormalities have been suggested to be typical of the Cohen syndrome. In the three patients originally reported by Cohen et al,1 two siblings had mottling of the retinal pigment, while the third patient had microphthalmia with iris/retinochoroidal colobomata. Subsequent case reports have described a wide range of ophthalmic abnormalities including strabismus, myopia, and microcornea,7,10,17–19 Ocular examination in the Finnish patients demonstrated a high frequency of retinal dystrophy in association with high myopia. Visual problems were progressive with significant visual handicap frequent among adult patients,14,20 However, the strong founder effect among the Finnish patients implies that the majority carry an identical mutation which therefore reduces the impact of the clinical homogeneity identified. Until now, no similar patient cohort from outside Finland has been assessed. We have identified a large group of patients with Cohen syndrome from across the United Kingdom and, through analysis of data from previous and current ophthalmic assessments in these patients, determined the clinical variability of the ophthalmic phenotype in Cohen syndrome.
MATERIALS AND METHODS
Over a 2 year period (1999–2001) 22 patients with Cohen syndrome, referred from clinical geneticists throughout the United Kingdom, were assessed by the authors and their clinical phenotype characterised. A diagnosis of Cohen syndrome was based on the presence of the at least seven of the following typical clinical features as originally reported by Cohen et al,1 and further delineated by Norio et al14:
mental retardation
microcephaly
characteristic facial appearance (thick eyebrows and eyelashes, down slanting and wave-shaped palpebral fissures, prominent nose, short and upturned philtrum with an open mouthed expression revealing the upper central incisors)
slim tapering extremities with relative truncal obesity
hypotonia
joint laxity
neutropenia
ophthalmic abnormalities.
The authors recorded a detailed ophthalmic history from the parents of all patients. During the study period, nine patients underwent formal assessment (visual acuity, visual field assessment, full clinical examination, funduscopy, electroretinography, and visual evoked potential testing). In addition, past ophthalmic notes were reviewed on 15 patients. Although undertaken in different centres, electroretinograms (ERGs) were performed with skin electrodes in all cases and the results were graded as normal, attenuated, or extinguished.
RESULTS
The study patients’ ranged in age from 3–53 years. The spectrum of ophthalmic abnormalities are summarised in Table 1. The age at onset of visual problems, as noted by parents, ranged from 6 months to 15 years, with a median age of 3 years; 18/22 (82%) parents recalled their child’s visual difficulties as starting before the age of 5 years.
Table 1.
Ophthalmic findings in 22 UK Cohen syndrome patients
| Latest | ||||||||||
| Patient | Age (years) | Onset of visual problems | VR | VL | Myopia | Refraction (D) (if known) | Anterior segment | Fundus | Onset of nyctalopia | ERG |
| 1 | 3 years | 1 year | 6/9 | 6/9 | Y | – | NAD | Bull’s eye maculopathy | – | Normal age 1.5 years |
| 2 | 4 years | 2 years | 6/6 | 6/6 | Y | – | NAD | Bull’s eye maculopathy | 4 years | Attenuated age 3 years |
| 3 | 4 years | 2 years | 6/9 | 6/9 | Y | −7.5/−3.5 × 180 −7.5/−2.5 × 150 | NAD | Myopic fundus | – | Extinguished age 4 years |
| 4 | 4 years | 1 year | – | Y | −9 | NAD | Generalised pigmentary retinopathy | – | Not done | |
| 5 | 5 years | 6 months | 6/9 | 6/9 | Y | −1/+0.75 × 100 −2.25/+1 × 80 | NAD | Bull’s eye maculopathy | 2 years | Markedly attenuated age 2 years |
| 6 | 6 years | 18 months | 6/12 | 6/12 | Y | – | NAD | Bull’s eye maculopathy+ disc pallor | 4 years | Extinguished in dark adapted state age 3.5 years |
| 7 | 7 years | 18 months | 6/12 | 6/12 | Y | −2/−4.25 × 110 −2.5/−4 × 180 | NAD | Bull’s eye maculopathy+ disc pallor | 5 years | Extinguished age 4 years |
| 8 | 7 years | 6 months | 6/18 | 6/18 | Y | −12.5/−2 × 180 −12.5/−2 × 180 | NAD | Generalised pigmentary retinopathy Optic nerve pallor | 3 years | Markedly attenuated age 5 years |
| 9 | 9 years | 4 years | 6/18 | 6/18 | Y | −5/−4.5 × 180 −4.5/−3 × 180 | NAD | Generalised pigmentary retinopathy | 4 years | Not done |
| 10 | 10 years | <5 years | 6/9 | 6/9 | Y | – | NAD | Generalised pigmentary retinopathy Optic nerve pallor | – | Markedly attenuated |
| 11 | 15 years | 10 years | 6/24 | 6/24 | Y | −11.5/+5 × 90 −11/+5 × 97 | NAD | Generalised pigmentary retinopathy | – | Markedly attenuated age 15 years |
| 12 | 15 years | 3 years | 6/36 | 6/24 | Y | −3/+0.25 × 90 −0.25 | NAD | Retina within normal limits Optic nerve pallor | – | Extinguished age 15 years |
| 13 | 16 years | 15 years | 6/24 | 6/9 | Y | – | NAD | Retina within normal limits Optic nerve pallor | – | Markedly attenuated age 15 years |
| 14 | 16 years | 3 years | 6/12 | 6/36 | Y | −7/−4.5 × 10 −8/−4 × 165 | NAD | Generalised pigmentary retinopathy + disc pallor | 5 years | Extinguished age 7 years |
| 15 | 17 years | 4 years | 6/18 | 6/24 | Y | NAD | Patchy pigmentary retinopathy | 6 years | Extinguished age 17 years | |
| 16 | 17 years | 7 years | 6/12 | 6/12 | Y | – | NAD | Generalised pigmentary retinopathy | 7 years | Extinguished age 16 years |
| 17 | 19 years | 4 years | 6/60 | 6/60 | Y | −8.5/+3.5 × 120 −10/+4.5 × 75 | NAD | Generalised pigmentary retinopathy Bilateral retinal coloboma | Extinguished age 16 years | |
| 18 | 22 years | 2 years | 6/24 | 6/60 | Y | – | – | Generalised pigmentary retinopathy Optic nerve pallor | 10 years | Extinguished age 9 years |
| 19 | 34 years | <3 years | 6/18 | 6/36 | Y | – | Raised intraocular pressure age 34 years | Generalised pigmentary retinopathy Retinal detachment age 34 | – | Extinguished age 27 years |
| 20 | 42 years | 18 months | – | Y | – | Atrophic irides | Generalised pigmentary retinopathy Optic nerve pallor | – | Extinguished age 35 years | |
| 21 | 46 years | 15 years | 6/36 | 6/60 | Y | (Pre op) | Lens subluxation | Generalised pigmentary retinopathy | – | Extinguished age 30 years |
| −17/+4.5 × 85 | Keratoconus | |||||||||
| −18/+2.5 × 85 | Bilateral cataracts age 42 years | |||||||||
| 22 | 53 years | <5 years | 6/36 | 6/36 | Y | (Pre op) | Bilateral cataracts age 50 years | Generalised pigmentary retinopathy | 20 years | Not done |
| −14.50/+4.25 × 110 | ||||||||||
| −14/+3 × 80 | ||||||||||
All patients were myopic with refractive errors ranging from −0.25D to −18D. It was frequently of early onset. Myopia was documented before 5 years of age in 15/22 patients. Three patients had not undergone a formal ophthalmic assessment before their teenage years and so myopia was not detected until then. In four patients, the age at which refractive error was first present was not remembered by the parents nor documented in the past ophthalmic notes. Of the four myopic patients aged 5 years or less for whom refraction details were available, one had low myopia (<−3D), and three had high myopia (>−7D). In the five myopic patients aged between 5 and 10 years, three were high and two were moderate myopes. Finally, in the 10 patients aged over 10 years, eight had high myopia and two had moderate myopia.
In the majority of patients abnormal ocular findings were confined to the posterior segment. However, presenile posterior subcapsular cataracts were identified in the two oldest patients, aged over 40 years. One of these patients also had bilateral keratoconus and lens subluxation.
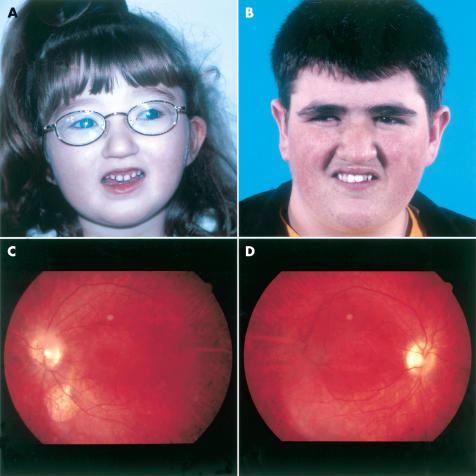
Retinal examination had been performed on all 22 study patients. Nine were children examined aged under the age of 10 years. Of these, five were noted to have isolated bull’s eye maculopathy while the remaining three had more generalised changes with diffuse retinal pigment disturbance and arteriolar attenuation (Fig 1A). One child, aged 4 years, had fundal changes in keeping with her high myopia but no evidence of a pigmentary retinopathy. Of the 13 adolescent and adult patients examined, 11 had widespread, severe retinopathy with classic bone spicule pigment deposition, optic pallor, and vessel attenuation. Two siblings had a normal retinal appearance except for early onset optic nerve pallor. A single patient had bilateral and visually insignificant retinal colobomata (Fig 1B).
Figure 1.
(A, B) Two patients, aged 4 and 19 years, with Cohen syndrome illustrating the characteristic facial appearance. Note the short philtrum, prominent incisors, and grimace-like appearance on smiling. (C) Bilateral incomplete inferior coloboma and pigmentary retinopathy in a young man with Cohen syndrome, (patient 17). (D) Pigmentary retinopathy typically seen in Cohen syndrome: note bone spicule changes in the lower retina.
Electrodiagnostic tests had been carried out on 19 of the study patients. Ten were children aged less than 10 years, of which nine, including two siblings, had markedly attenuated or extinguished responses. In the nine older patients tested, seven had extinguished ERGs, the remainder were severely attenuated.
Nyctalopia was recorded in 18/22 (82%) of patients. Many parents were unable to pinpoint a particular time when they became aware of symptoms of poor night vision in their child. However, in the 11 cases when an age of onset of nyctalopia was recalled, the median age was 5 years (range 2–20 years).
DISCUSSION
Our study of a large group of Cohen syndrome patients illustrates the range, progression, and severity of ophthalmic abnormalities typically associated with Cohen syndrome. Review of the literature reveals marked clinical heterogeneity among the reported patients and as a result the range and severity of associated ocular anomalies with Cohen syndrome is unclear. Microphthalmia, microcornea, colobomata, strabismus, and myopia have all been reported. In Finnish patients with Cohen syndrome, specific visual problems have been delineated and high myopia and chorioretinal dystrophy have been proposed as a hallmark of Cohen syndrome.14,20 Kivitie-Kallio et al’s recent, long term follow up study in 22 Finnish patients,20 described severe and progressive myopia with chorioretinal dystrophy and extinguished ERG results in all but the youngest patients. However, their retrospective data collection identified nyctalopia and symptoms of progressive visual field loss in most patients before the age of 7 years.20
Our study patients are from a geographically diverse population in whom haplotype analysis does not suggest the presence of a common founder mutation (unpublished observations). Therefore, they represent a valuable patient resource for assessment of the variability of ophthalmic abnormalities found in Cohen syndrome, particularly when compared with the Finnish patient group.
Our results confirm that the visual problems are of early onset: 82% of our patients developed squint or refractive errors during the first 5 years of life. Myopia occurred in all of our study patients, was early in onset (under 5 years) and progressive: 63% of patients had high myopia. The severity of the myopia in our patients was comparable to that seen in other studies with a median refraction of −8 dioptres (range −0.25 to −18D).
Patients with Cohen syndrome develop a progressive pigmentary retinopathy.14,17–20 In our study, this was identified in all our patients and was frequently early in onset. All of the study children aged under 5 years had evidence of a retinopathy. Symptoms of nyctalopia were reported from a median age of 5 years. The rate of progression of visual symptoms varied between individual patients, even within the same families. However, by the second decade nyctalopia, reduced visual acuity, and peripheral visual field loss secondary to retinal degeneration were evident in 85% of patients. The pattern of progression of the retinal dystrophy is comparable to that described by Kivitie-Kallio et al.20
The majority of our study patients had severe visual handicap and 35% were registered as partially sighted or blind. This contrasts with the Finnish group of Cohen syndrome patients in which marked deterioration of visual function occurred over the age of 50 years but no patient was totally blind.20
Analysis of the range of visual problems seen in this group of children and adults with Cohen syndrome highlights the important contribution ophthalmic abnormalities make to the condition, not only in terms of handicap and morbidity but also in confirming the diagnosis. Ophthalmologists should consider Cohen syndrome in a young, developmentally delayed child who presents with severe myopia and nyctalopia. Similarly, for paediatricians and clinical geneticists considering a diagnosis of Cohen syndrome, evidence of an early onset, and progressive myopia in association with a pigmentary retinopathy should be sought. Early identification of a visual impairment in a child with Cohen syndrome and referral to the appropriate agencies within a multidisciplinary team are of vital importance to the family.
Acknowledgments
We would like to thank the families who agreed to take part in this study and their clinicians and ophthalmologists for their help in gathering clinical and ophthalmological information. GB is a Wellcome clinician scientist fellow (ref 51390/Z).
REFERENCES
- 1.Cohen MM Jr, Hall BD, Smith DW, et al. A new syndrome with hypotonia, obesity, mental deficiency and facial, oral, ocular and limb anomalies. J Pediatr 1973;83:280–4. [DOI] [PubMed] [Google Scholar]
- 2.Carey JC, Hall BD. Confirmation of the Cohen syndrome. J Pediatr 1978;93:239–44. [DOI] [PubMed] [Google Scholar]
- 3.Kousseff BG. Cohen syndrome: further delineation and inheritance. Am J Med Genet 1981;9:25–30. [DOI] [PubMed] [Google Scholar]
- 4.North C, Patton MA, Baraitser M, et al. The clinical features of the Cohen syndrome: further case reports. J Med Genet 1985;22:131–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sack J, Friedman E. The Cohen syndrome in Israel. Israel J Med Sci 1986;22:766–70. [PubMed] [Google Scholar]
- 6.Young I D, Moore J R. Intrafamilial variation in Cohen syndrome. J Med Genet 1987;24: 488–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Steinlein O, Tariverdian G, Boll HU, et al. Tapetoretinal degeneration in brothers with apparent Cohen syndrome: nosology with Mirrhosseini-Holmes-Walton syndrome. Am J Med Genet 1991;41:196–200. [DOI] [PubMed] [Google Scholar]
- 8.Partington M, Anderson D. Mild growth retardation and developmental delay, microcephaly and a distinctive facial appearance. Am J Med Genet 1994;49:247–50. [DOI] [PubMed] [Google Scholar]
- 9.North KN, Fulton AB, Whiteman DAH. Identical twins with Cohen syndrome. Am J Med Genet 1995;58:54–8. [DOI] [PubMed] [Google Scholar]
- 10.Fryns JP, Legius E, Devriendt K, et al. Cohen syndrome: the clinical symptoms and stigmata at a young age. Clin Genet 1996;49:237–41. [DOI] [PubMed] [Google Scholar]
- 11.Okamoto N, Hatsukawa Y, Arai H, et al. Cohen syndrome with high urinary excretion of hyaluronic acid. Am J Med Genet 1998;76:387–8. [PubMed] [Google Scholar]
- 12.Thomaidis L, Fryssira H, Katsarou E, et al. Cohen syndrome: two new cases in siblings. Eur J Pediatr 1999;158:838–41. [DOI] [PubMed] [Google Scholar]
- 13.Horn D, Krebsova A, Kunze J, et al. Homozygosity mapping in a family with microcephaly, mental retardation, and short stature to a Cohen syndrome region on 8q21.3–8q22.1: redefining a clinical entity. Am J Med Genet 2000;92:285–92. [PubMed] [Google Scholar]
- 14.Norio R, Raitta C, Lindahl E. Further delineation of the Cohen syndrome; report on chorioretinal dystrophy, leukopenia and consanguinity. Clin Genet 1984;25:1–14. [DOI] [PubMed] [Google Scholar]
- 15.Tahvanainen E, Norio R, Karila E, et al. Cohen syndrome gene assigned to the long arm of chromosome 8 by linkage analysis. Nat Genet 1994;7:201–4. [DOI] [PubMed] [Google Scholar]
- 16.Kolehmainen J, Norio R, Kivitie-Kallio S, et al. Refined mapping of the Cohen syndrome gene by linkage disequilibrium. Eur J Hum Genet 1997;5:206–13 [PubMed] [Google Scholar]
- 17.Resnick K, Zuckerman J, Cotlier E. Cohen syndrome with bull’s eye macular lesion. Ophthal Paediatr Genet 1986;7:1–8. [DOI] [PubMed] [Google Scholar]
- 18.Kondo I, Nagataki S, Miyagi N. The Cohen syndrome: does a mottled retina separate a Finnish and a Jewish type? Am J Med Genet 1990;37:109–13. [DOI] [PubMed] [Google Scholar]
- 19.Warburg M, Pedersen SA, Horlyk H. The Cohen syndrome. Retinal lesions and granulocytopenia. Ophthal Paediatr Genet 1990;11:7–13. [DOI] [PubMed] [Google Scholar]
- 20.Kivitie-Kallio S, Summanen P, Raitta C, et al. Opthalmologic findings in Cohen syndrome—a long term follow up. Ophthalmology 2000;107:1737–45. [DOI] [PubMed] [Google Scholar]