Abstract
Aim: To describe the angiographic features after photodynamic therapy (PDT) with verteporfin in choroidal neovascularisation (CNV) associated both with age related macular degeneration (AMD) and pathological myopia (PM).
Methods: 36 patients affected by subfoveal CNV in AMD and 25 patients with subfoveal CNV in PM underwent an ophthalmological examination including fluorescein angiography (FA) and indocyanine green angiography (ICGA) using the IMAGEnet System. Post-PDT examinations were performed 7, 30, and 90 days later.
Results: The typical angiographic aspect after PDT for AMD related CNV was a round hypofluorescence visible both on FA and on ICGA, which included both CNV and the surrounding tissues and corresponded to the area exposed to laser light. In PM the CNV appeared hypofluorescent during the early phases and gradually became hyperfluorescent during the late phases on FA, whereas on ICGA it was detectable in its whole extension as a hyperfluorescent lesion since the early phases. Differently from AMD, there was no round hypofluorescence surrounding the CNV on FA or on ICGA. Moreover, five patients in the AMD group showed hot spots on ICGA, which spontaneously disappeared during the follow up. Classic and occult components of the AMD related CNV revealed a different angiographic response to PDT, showing with the latter only a partial closure 1 week after PDT followed by a complete reopening at the first month in 100% of cases.
Conclusion: The post-PDT hypofluorescence typical of AMD related CNV, especially visible on FA, might be secondary to a combination of choriocapillary occlusion and masking effect due to swelling of retinal pigment epithelium cells. Hot spots in the AMD affected patients could be interpreted as the expression of a non-thermal choroidal vasculitis secondary to PDT.
Keywords: choroidal neovascularisation, age related macular degeneration, myopia, photodynamic therapy
Randomised clinical trials have demonstrated the beneficial effects of photodynamic therapy (PDT) with verteporfin in patients with subfoveal predominantly classic choroidal neovascularisation (CNV) in age related macular degeneration (AMD) and in subfoveal CNV in pathological myopia (PM).1–4
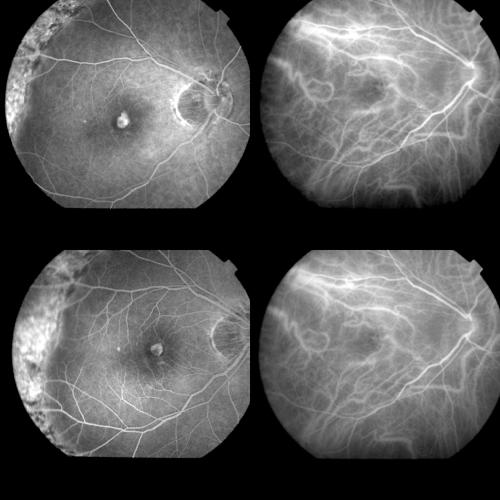
Even though histological examination showed obliteration of CNV after PDT both in animal models and in humans,5–9 the exact mechanism of PDT is not completely clear yet. The study of the post-PDT angiographic features in subfoveal CNV could provide information regarding the complex biochemical processes involved.
The present study describes the angiographic features after PDT in CNV associated both with AMD and PM and discusses the different angiographic behaviour.
METHODS
Patients referred to the angiographic centre of the eye clinic in Trieste with diagnosis of CNV in PM and AMD between December 2000 and August 2001 were considered.
Inclusion criteria were CNV under the geometric centre of the foveal avascular zone, greatest linear dimension not more than 5400 μm, and best corrected visual acuity (BCVA) of 20/200 or better on ETDRS charts. The diagnosis of AMD was based on the presence of drusen in either eye, detachment of the sensory retina, and/or retinal haemorrhages, and/or exudates.
Eyes with PM should have a distance correction of at least −6.0 dioptres, spherical equivalent, or less myopic than −6.0 dioptres with retinal abnormalities consistent with PM, such as lacquer cracks, and an axial length of at least 26.5 mm.
Exclusion criteria were features of any condition other than PM associated with CNV, any significant ocular disease that had compromised or could compromise vision in the study eye, active hepatitis or clinically significant liver disease, porphyria or other porphyrin sensitivity, pregnancy, intraocular surgery within the past 2 months, or capsulotomy within the past month in the study eye and previous laser photocoagulation.
Each patient underwent an ophthalmological examination including BCVA on standard ETDRS charts. Fluorescein angiography (FA) and indocyanine green angiography (ICGA) were carried out using the IMAGEnet System (Topcon Corp).
PDT with verteporfin was performed according to TAP guidelines.1 In particular, an additional 1000 μm was added to this dimension to provide a 500 μm safety margin around the lesion.1
Post-PDT examinations consisted of BCVA, fundus biomicroscopy, FA and ICGA, performed 7, 30, and 90 days after each PDT session.
Angiographic leakage at the 3 month follow up was classified following the method used in the TAP study.1 Two authors (MBP, SDP) independently evaluated the angiographic features with an interobserver concordance of 96.5%. The third author (GR) judged the uncertain cases.
Each patient provided informed consent for the study.
RESULTS
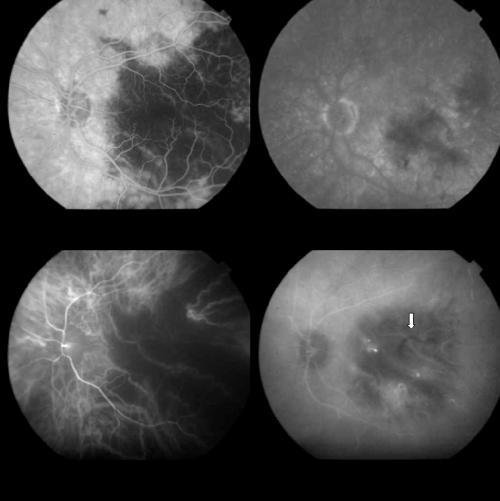
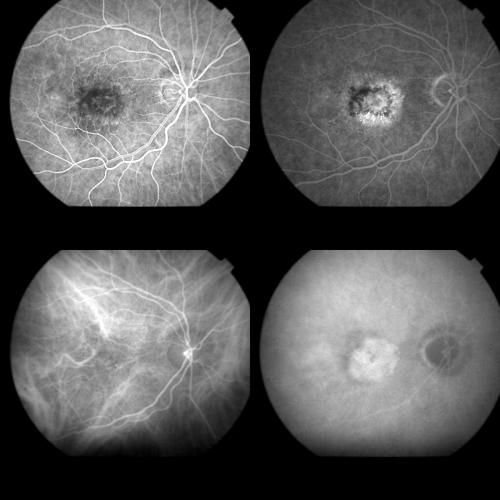
Thirty six patients affected by subfoveal CNV in AMD were recruited. CNV was predominantly classic and minimally classic on FA in 22 and in 14 eyes, respectively (Fig 1). Subretinal haemorrhage was visible in 32 eyes.
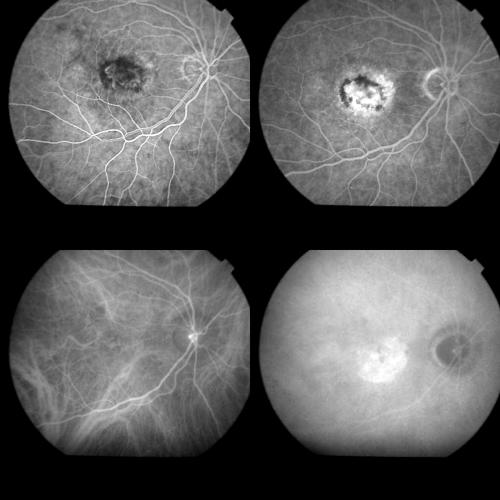
Figure 1.
Top left: Early FA phase (37 seconds) showing a predominantly classic AMD related CNV. Top right: Late FA phase revealing the extension of the occult component. Bottom left: Early ICGA image (1 minute) with regular filling of the CNV net. Bottom right: Late ICGA image (40 minutes) with the whole staining of the lesion.
Twenty five patients affected by subfoveal CNV in PM were collected. All the cases showed classic CNV on FA. Subretinal haemorrhage was present in all eyes.
Baseline BCVA in AMD eyes ranged from 20/125 to 20/50 and in PM eyes from 20/200 to 20/40.
The baseline CNV size is shown in the Table 1. The mean area was 5.86 (SD 3.26) mm2 (range 1.61–13.5; median 4.81) for the AMD related CNV, and 0.82 (0.68) mm2 (range 0.24–2.80; median 0.48) for the PM related CNV.
Table 1.
Classification of baseline CNV area for AMD and PM eyes
| Eyes | ||
| CNV area (mm2) | AMD related CNV | PM related CNV |
| ≤2.5 | 6 | 21 |
| >2.5–≤5.0 | 10 | 4 |
| >5.0 | 20 | – |
One week control
The biomicroscopic examination revealed no changes with respect to the baseline aspect. BCVA ranges were unmodified.
Concerning CNV in AMD, FA revealed an early round hypofluorescence covering the whole area of laser light exposure in all eyes and including both CNV area and the 500 μm safety margin surrounding CNV. However, the angiographic behaviour of the classic and occult components of the CNV was different. In early stages of the examination, the classic component appeared hypofluorescent in all cases, showing only a faint staining in the late phases in 21 eyes (58.3%). In 17 cases (47.2%) some portions of the occult component disclosed a slight hyperfluorescence, which appeared within the hypofluorescent round area correlated with the laser exposition and had been detectable since the early phases. This early hyperfluorescence could be related to retinal pigment epithelium (RPE) changes, which improved the visibility of the underlying choroidal vasculature, rather than to a dye leakage or staining. A dye staining became visible in all the cases by prolonging the FA up to late phases (even more than 10 minutes), with no evidence of leakage (Fig 2).
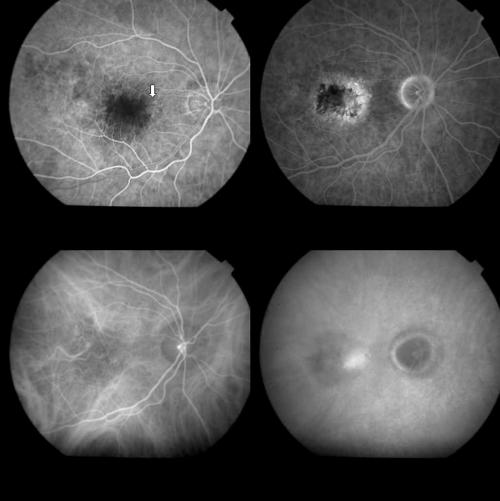
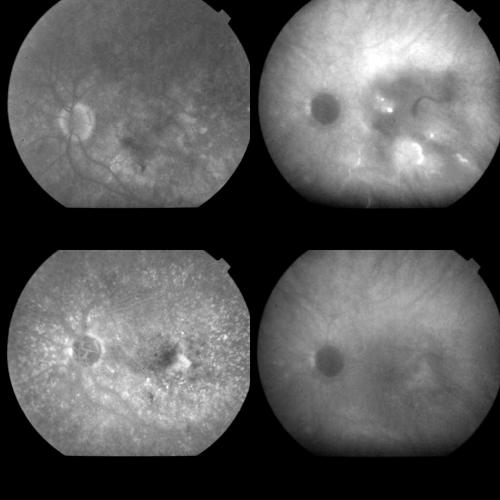
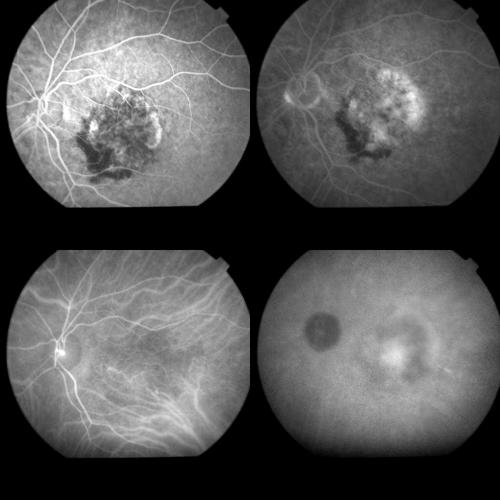
Figure 2.
Same case as Figure 1, 1 week after PDT. Top left: Early FA phase (38 seconds). The round hypofluorescence corresponding to the treatment spot is visible. The arrow indicates an early hyperfluorescence probably related to RPE changes. Top right: A dye staining, without leakage is visible during the late FA phases (14 minutes). Bottom left: Early ICGA phase (1 minute) disclosing better than on FA, the round hypofluorescence corresponding to the laser irradiated area. Bottom right: Late ICGA phases (41 minutes) showing an irregular hyperfluorescence which appears reduced in size with respect to the baseline lesion, and corresponds to part of the original classic component and, more visible, to part of the original occult component of the CNV.
As regards ICGA findings, the early phases of the examination revealed more clearly the round hypofluorescence in the laser exposed area, corresponding to that visible on FA.
During the intermediate and late phases, a gradually progressive hyperfluorescence was detectable within the area corresponding to the original CNV. Such a hyperfluorescence appeared in any case remarkably reduced in size with respect to the baseline lesion, and corresponded to part of the original classic component of the CNV in 16 eyes (45%), and to part of the original occult component in all the cases (100%). Thus, 20 eyes (55%) showed a complete closure of the classic component, disclosing no hyperfluorescence throughout the examination. Moreover, the halo surrounding the CNV appeared slightly less hypofluorescent in the late phases compared to the early phases (Fig 2).
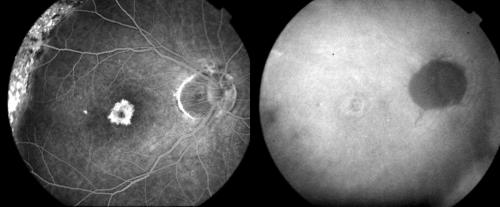
Examining carefully the vascular net in the early ICGA phases, some portions of the classic component of CNV vasculature turned out to be occluded in seven (19.4%) eyes (Fig 3). These eyes presented predominantly classic CNV at baseline.
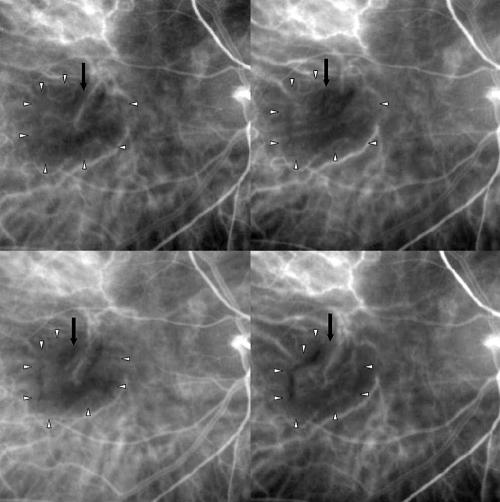
Figure 3.
Top left: Magnification of ICGA early phase (35 seconds) of CNV in AMD before PDT. The black arrow indicates a main CNV vessel. The small white arrows delineate the CNV boundaries. Top right: Magnification of ICGA early phase (35 seconds) of the same case 1 week after PDT showing the closure of the CNV vessel (black arrow). The CNV has the same size (small white arrows). Bottom left: Magnification of ICGA early phase (35 seconds) 1 month after PDT, with partial reopening of previously occluded CNV vessel (black arrow) and initial reduction in size of the CNV (small white arrows). Bottom right: Magnification of ICGA early phase (35 seconds) 3 months after PDT. Reopening of the choroidal vessels with their remodelling (black arrow). The CNV is reduced in size showing a surrounding RPE reaction (small white arrows).
Moreover, three cases (two with minimally classic and one with predominantly classic CNV) showed hot spots, which appeared 7 minutes after dye injection and were located within the treated area, along the course of the choroidal vessels. A late ICG dye leakage was detected in the three cases. It was notable that the hot spots were not detectable on FA, where only an irregular dye staining was visible in the late phases, involving an area greater than that of the hot spots detected in ICGA, possibly due to residual CNV portions.
One of the three cases with hot spots showed a marked choroidal delayed perfusion at the posterior pole both on FA and on ICGA, extending beyond the laser spot, especially towards the temporal sector. Analysis of the early ICGA phases showed the occlusion of a posterior ciliary artery. On FA, choroidal filling was severely delayed, reaching a complete filling only 25 minutes from injection, but on ICGA a wide hypofluorescence extending beyond the limit of the laser treatment was still visible 55 minutes after injection. Three hot spots along with some choroidal vessel staining were detectable on ICGA from the 10th minute (Figs 4, 5, and 6). BCVA was unchanged.
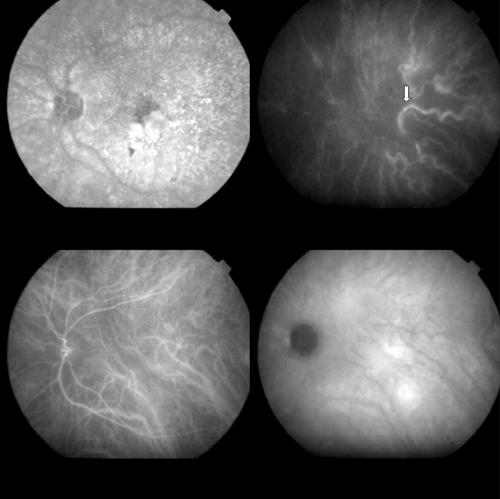
Figure 4.
Top left: FA (1 minute) showing a predominantly classic subfoveal CNV before PDT. Top right: Early ICGA image (15 seconds) with the filling of a ciliary artery (arrow). Bottom left: Early ICGA phase (1 minute) revealing the regular aspect of the choroidal vessels. Bottom right: Late ICGA phases (40 minutes) with the staining of the CNV.
Figure 5.
Same case as Figure 4, 1 week after PDT. Top left: FA at 2 minutes showing the marked choroidal delayed perfusion at the posterior pole, extending beyond the laser spot, especially towards the temporal sector. Top right: Late FA phases (12 minutes) showing the delayed filling of the choroid. Bottom left: Early ICGA phase (5 minutes) showing the delayed choroidal perfusion. Bottom right: ICGA at 10 minutes. The occlusion of the ciliary artery (arrow) is still detectable whereas three hot spots along with some choroidal vessel staining appear.
Figure 6.
Same case as Figures 4 and 5. Top left: Late FA phases (25 minutes) 1 week after PDT showing the filling of the choroid at this very late phase. Top right: Late ICGA phases (55 minutes) with incomplete filling of the choroid. Bottom left: FA image (2 minutes) 1 month after PDT disclosing the regular filling of the choroid and the reduction in size of the CNV. Bottom right: Late ICGA phases (40 minutes) 1 month after PDT showing a slight hyperfluorescence corresponding to the original CNV. The choroid fills regularly.
On FA, CNV in PM showed an early hypofluorescence, which gradually subsided, giving way to an increasing hyperfluorescence in the late phases. A subtle hypofluorescent margin surrounding the CNV, and corresponding to RPE, was visible in all the cases, but the area surrounding the CNV corresponding to the 500 μm safety margin, which was also exposed to the laser light, did not show hypofluorescence in any case (Figs 7 and 8).
Figure 7.
Top left: Early FA phase (35 seconds) of a PM related CNV (arrow). Top right: Late FA phase (11 minutes) with the dye leakage. Bottom left: Early ICGA image (41 seconds) showing the hyperfluorescence corresponding to the CNV. Bottom right: Late ICGA image (40 minutes) with the staining of the lesion.
Figure 8.
Same case as Figure 7, 1 week after PDT. Top left: Early FA phase (35 seconds) showing the slight hyperfluorescence of the CNV. No round hypofluorescent halo corresponding to the area exposed to laser light around the CNV was visible. Top right: Late FA phase (11 minutes) revealing the staining of the lesion. Bottom left: Early ICGA image (40 seconds) with regular filling of the CNV net. Bottom right: Late ICGA image (40 minutes) with a reduced staining.
Angiographic behaviour turned out to be different on ICGA. Indeed, even though it was with difficulty, and only after careful analysis, the CNV limits were detectable since the early phases, showing a slight hyperfluorescence, with a normal, non-delayed, filling of the CNV lesion. No round hypofluorescent halo corresponding to the area exposed to laser light around the CNV was visible. CNV was delineated by a subtle hypofluorescent margin corresponding to the RPE (Figs 7 and 8). Five eyes (20%) showed occlusion of some choroidal vessels. The late ICGA phases showed a greater staining of CNV.
In no case was a hot spot detectable.
We should underline that the angiographic pattern was the same in all the CNV of the two groups and the size of the lesion did not influence angiographic appearance.
One month control
BCVA in AMD eyes ranged from 20/200 to 20/25 and in PM eyes from 20/100 to 20/32. Biomicroscopically subretinal haemorrhage was partially reabsorbed in 29 AMD eyes and in all the 25 PM eyes.
In the AMD group the round hypofluorescence corresponding to the laser light exposure was still present in 10 (27.7%) and 11 (30.5%) cases respectively on FA and on ICGA, whereas it was absent in all the PM cases.
FA in 31 cases (86%) of the AMD group showed an early hyperfluorescence which corresponded to the classic component of the original CNV, and appeared smaller in size compared to the pretreatment lesion. This hyperfluorescence disclosed staining in the late phases without dye leakage.
A staining corresponding to the occult portion of the CNV was identifiable in all the eyes during the mid to late phases. Some dye leakage (difficult to be graded following the TAP definitions,1 was visible in 23 cases (63.8%) during the late phases (Fig 9).
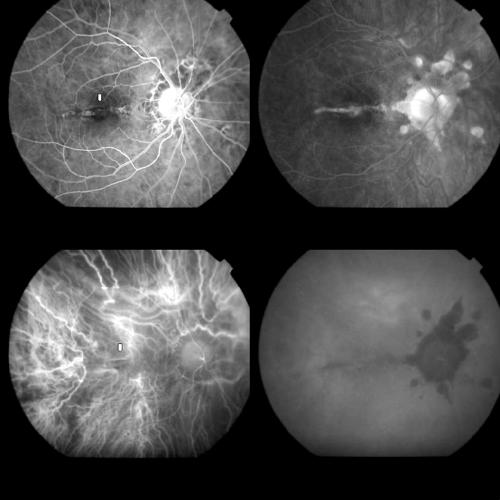
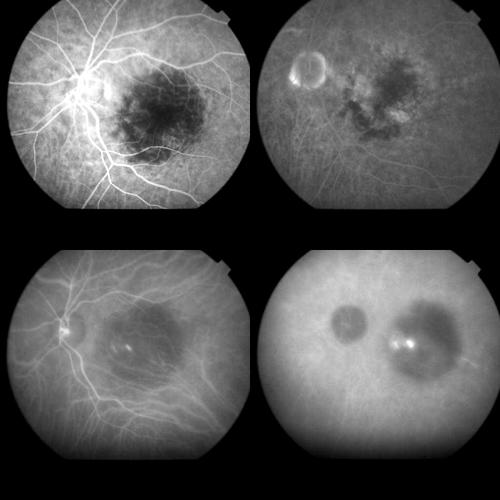
Figure 9.
Same case as Figures 1 and 2 at the 1 month control. Top left: Early FA phase (37 seconds) showing an early hyperfluorescence which corresponded to the classic component of the original CNV and appeared smaller in size compared to the pre-treatment lesion. Top right: Late FA phases (15 minutes) revealing staining, but no leakage, from the classic component and staining together with some leakage from the occult component. Bottom left: Early ICGA phase (1 minute) showing the permanence of the round hypofluorescent spot. Bottom right: Late ICGA phases (40 minutes) with the staining of the whole CNV.
ICGA showed a hyperfluorescence corresponding to the classic portion of the CNV in the same 31 eyes, but revealed a reopening of the whole occult component in all the cases, disclosing a lesion similar to that visible before PDT (Fig 9).
In the 25 eyes affected by PM related CNV, both FA and ICGA revealed a hyperfluorescence related to CNV, which appeared more clearly visible than at the 1 week examination.
The partial occlusion of the CNV vessels detected at 1 week in seven eyes affected by AMD and in five eyes affected by PM was less visible (Fig 3).
Whereas the three hot spots visible on ICGA at the 1 week control disappeared spontaneously in the AMD group, two additional cases occurred, detectable at 8 and 10 minutes from dye injection respectively (Figs 10, 11, and 12).
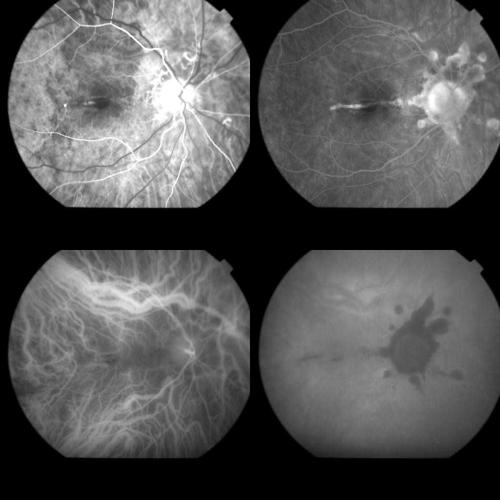
Figure 10.
Top left: FA image (59 seconds) of minimally classic AMD related CNV. Top right: Late FA phases (8 minutes) disclosing the extension of the occult and the classic components of the lesion. Bottom left: Early ICGA phase (1 minute) showing the filling of the CNV net. Bottom right: Late ICGA phases (42 minutes) revealing the staining of both the component of the CNV.
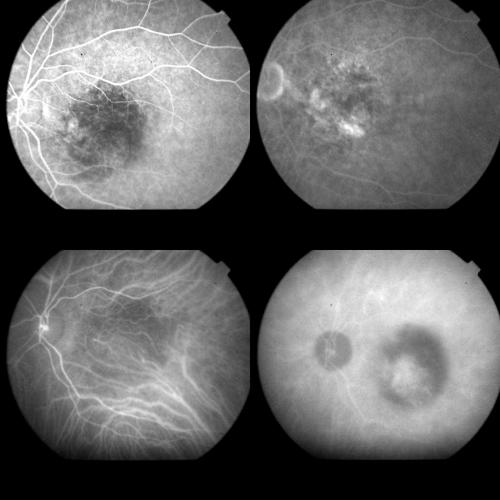
Figure 11.
Same case as Figure 10 1 month after PDT. Top left: FA (56 seconds) with the typical round hypofluorescence corresponding to the laser spot. Top right: Late FA phases (10 minutes). The classic component reveals only a mild staining, whereas the occult component shows both staining and leakage. Bottom left: Early ICGA phase (8 minutes). Two hot spots appear within treated area, along the course of choroidal vessels. Bottom right: Late ICGA phases (43 minutes). A profuse leakage from the hot spots is visible together with staining of the occult component of the CNV.
Figure 12.
Same case as Figures 10 and 11 3 month after PDT. Top left: FA (59 seconds). The round hypofluorescence is less pronounced. Top right: Late FA phases (11 minutes). The classic component discloses a faint staining, whereas the occult component shows more dye staining and leakage. Bottom left: Early ICGA phase (8 minutes). The two hot spots have spontaneously resolved. Bottom right: Late ICGA phases (43 minutes). The occult component shows a greater staining.
The delayed choroidal perfusion detected in the above mentioned case, resolved completely along with the hot spots (Fig 6).
Three month control
BCVA in AMD eyes ranged from 20/200 to 20/25 and in PM eyes from 20/200 to 20/32. Biomicroscopic examination revealed the whitish appearance of the CNV in the seven eyes of AMD group and in the five eyes of the PM group, which had shown a partial occlusion of the CNV vasculature at the 1 week control (Fig 3). These lesions appeared as quiescent neovascular membranes, with late staining both on FA and on ICGA.
The hypofluorescent halo around the CNV was detectable in seven AMD cases (19.4%) only by means of ICGA.
The occluded choroidal vessels turned out to be reopened in all cases, even though a remodelling of the choroidal vascular pattern was also evident. The two hot spots that occurred at the 1 month control on ICGA cases disappeared completely.
Considering the AMD related CNV, overall, according to the TAP definition,1 16 cases showed CNV progression on FA, 11 moderate leakage, seven minimal leakage, and two no leakage. The grading of the fluorescein leakage extent for classic and occult component is listed in Table 2.
Table 2.
Leakage grading at 12 weeks from PDT and classification of its source
| Source of leakage | ||||
| Grading of leakage | From classic component | From occult component | From both components | Total eyes |
| Progression | 9 | 3 | 4 | 16 |
| Moderate | 4 | 2 | 5 | 11 |
| Minimal | 4 | 2 | 1 | 7 |
| No leakage | – | – | 2 | 2 |
As for the PM related CNV, eight eyes showed CNV progression, six moderate leakage, eight minimal leakage, and three no leakage.
Of considerable significance is the finding that the 12 cases (seven AMD cases and five PM cases) which showed a partial choroidal occlusion at the first week post-PDT control evolved towards subretinal fibrosis.
CNV progression was observed overall in 24 eyes which presented a CNV area bigger than the baseline. However, the primary CNV—that is, the CNV area at baseline, presented as isofluorescent in the late phases, especially on ICGA, and was surrounded by a brighter hyperfluorescence corresponding to the new neovascular proliferation. In this way the margins of the original CNV were still detectable within the entire CNV lesion (Figs 13 and 14).
Figure 13.
Top left: FA (40 seconds) of subfoveal CNV in PM before PDT. Top right: Early ICGA phase (40 seconds). The CNV appears as a slightly hyperfluorescent lesion. Bottom left: FA (42 seconds) of the same case 1 week after PDT. The CNV appears reduced in size, without evidence of hypofluorescent halo in the laser exposed area. Bottom right: Early ICGA phase (40 seconds) of the same case 1 week after PDT showing the regular filling of the CNV.
Figure 14.
Same patient as in Figure 13 at the 3 month post-PDT control. Left: FA (1 minute) showing the CNV progression. Right: Late ICGA phases (40 minutes). The original CNV shows a isofluorescence and is surrounded by a slightly brighter fluorescent lesion which corresponds to the new neovascular proliferation.
From the biomicroscopic point of view, the isofluorescent area corresponding to the original CNV appeared as flatter than the remaining CNV portion.
DISCUSSION
The present study highlights some differences between the post-PDT angiographic behaviour in AMD and PM related CNV that need interpretation.
We are aware that our research can only suggest speculative hypotheses regarding how PDT acts on the strength of the simple angiographic findings. It is thus intended to provide a basis for a future in-depth evaluation on the complex processes primed by PDT. This study offers a detailed analysis of pre-PDT and post-PDT angiographic features both on FA and on ICGA. A possible drawback of the study could be the awkwardness of visualising the CNV in the early ICGA phases, especially in myopic CNV. In any case, in our experience it is essential to acquire good quality images, in order to ensure the detection of the CNV limits even in the early phases.
A typical aspect of AMD on FA is the presence of a round hypofluorescence corresponding to the area exposed to laser energy and including the CNV and the 500 μm safety margin around it. In PM only the CNV is hypofluorescent on FA, the surrounding hypofluorescent halo being absent. Moreover, the hyperfluorescence related to the CNV is detectable on ICGA from the early phases in PM and from the intermediate phases in AMD.
The histological features of AMD and PM are to be borne in mind in any attempt to explain the angiographic pattern and the discrepancy between FA and ICGA findings.
In particular, AMD occurs in elderly people and it is characterised by thicker RPE-Bruch’s membrane, with basal laminar deposits and lipofuscin accumulation in the RPE cells. Moreover, CNV is thicker and generally type 1, which is located beneath the RPE.10–14
CNV in PM occurs in younger people, with thinner RPE-Bruch’s membrane complex, depigmented RPE cells, and thin and rarefied choriocapillaris.11,15–19 Moreover, the CNV typical of PM is type 2 CNV, which is located between the sensory retina and RPE, and is coated by RPE, being characterised by an RPE layer on its inner surface and an RPE inverted layer on its outer surface, creating a double RPE layer.8
It is on the basis of the above mentioned histopathological features that we could hypothesise a different angiographic pattern after PDT between AMD an PM.
The round hypofluorescence detectable on FA after PDT for AMD associated CNV might be secondary to a combination of choriocapillaris occlusion and masking effect due to the swelling of RPE cells. Such a hypofluorescence may develop only if choriocapillaris and RPE are sufficiently present to show their alterations angiographically.
Therefore, owing to the extreme thinning of both choriocapillaris and the RPE-Bruch’s membrane complex in the areas outside the CNV in eyes affected by PM, no visible hypofluorescence might result from choriocapillaris occlusion and RPE swelling.
The situation may be different when the PM related CNV lesion is considered, where RPE is in a double layer and the neovascular net is thicker than the surrounding choroid. The early hypofluorescence limited to the CNV area detectable on FA, but not on ICGA, might be explained by considering that the RPE swelling has a masking effect on a partially occluded CNV.
Another important angiographic feature is represented by the hot spots occurrence only in AMD related CNV. Hot spots could be interpreted as the expression of a non-thermal choroidal vasculitis secondary to the release of the inflammatory mediators primed by PDT. Observing the correlation between hot spots and the course of the choroidal vessels, the hot spots detected on ICGA might represent a transient fibrin staining, due to the great affinity of the ICG molecule for fibrin secondary to the PDT induced vasculitis.
It is of particular interest that the difference in timing of hot spots in different eyes might hint that the vascular response to PDT is a dynamic process which may vary from patient to patient.20
Even though CNV obliteration after PDT has been histologically proved in animal models and in humans,5–9 conventional ICGA does not always reveal choroidal vessel occlusion. In particular Schmidt-Erfurth, using a continuous recording system, the Heidelberg retinal angiograph, reported the detection of partial occlusion of CNV in half of the PDT treated eyes on ICGA.21 In their experiments on monkeys Flower and Snyder detected no difference in the choroidal flow in the area exposed to laser light, in their comparison of pre-PDT and post-PDT high speed ICGA.22
A temporary occlusion of some portions of the classic component of CNV vasculature on ICGA was detectable among our patients only in seven AMD and five PM eyes using the IMAGEnet system.
This finding might also indicate that PDT leads to variable vascular reactions. In particular, the whitish appearance of the CNV visible at the third month in the eyes that had showed such an occlusion, suggests a quiescence of the CNV, and could be of great importance for the prognosis.
Lastly, the peculiar pattern of fluorescence detectable in the 24 eyes (16 in AMD and eight in PM) with CNV progression 3 months after PDT is noteworthy. In these cases the baseline portion of the CNV was isofluorescent in comparison with the fluorescence background, whereas the new proliferation appeared to be hyperfluorescent. This aspect may be the expression of the endothelial changes due to PDT: the CNV portion previously treated by PDT could show a reduced vascular permeability, which biomicroscopically correlates with a flatter portion of the CNV, whereas the new proliferation has a greater permeability with more dye leakage.
Previous trials have demonstrated that the composition of the CNV in AMD has an impact on the outcome of the treatment,1,2,4 predominantly classic CNV lesions having a significant treatment benefit. In our series of patients the classic and the occult components of the CNV revealed a different angiographic response to PDT. In particular, on ICGA the occult component showed only a partial closure 1 week after PDT followed by a complete reopening at the first month in 100% of cases.
These findings may contribute to explain the less favourable outcome of PDT in occult CNV.
Overall, PDT involves a number of cellular, vascular, and immunological phenomena which can express variably on the basis of the different individual conditions and on the basis of the different underlying diseases. Future histopathological investigations will provide a more thorough analysis of the mechanism of action of PDT.
Acknowledgments
The authors wish to thank Professors RW Flower and U Schmidt-Erfurth for their helpful comments.
Abbreviations
AMD, age related macular degeneration
BCVA, best corrected visual acuity
CNV, choroidal neovascularisation
FA, fluorescein angiography
ICGA, indocyanine green angiography
PDT, photodynamic therapy
PM, pathological myopia
REFERENCES
- 1.Treatment of Age-related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularisation in age-related macular degeneration with verteporfin. One-year results of 2 randomised clinical trials-TAP report 1. Arch Ophthalmol 1999;117:1329–45. [PubMed] [Google Scholar]
- 2.Treatment of Age-related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularisation in age-related macular degeneration with verteporfin. Two-year results of 2 randomised clinical trials-TAP report 2. Arch Ophthalmol 2001;119:198–207. [PubMed] [Google Scholar]
- 3.Verteporfin in Photodynamic Therapy. Photodynamic therapy of subfoveal choroidal neovascularisation in pathologic myopia with verteporfin. 1-year results of a randomised clinical trial—VIP report 1. Ophthalmology 2001;108:841–52. [DOI] [PubMed] [Google Scholar]
- 4.Verteporfin in Photodynamic Therapy. Verteporfin therapy of subfoveal choroidal neovascularisation in age-related macular degeneration: two-year results of a randomised clinical trial including lesions with occult with no classic choroidal neovascularisation—verteporfin in photodynamic therapy report 2. Am J Ophthalmol 2001;131:541–60. [DOI] [PubMed] [Google Scholar]
- 5.Schmidt-Erfurth U, Bauman W, Gragoudas E, et al. Photodynamic therapy of experimental choroidal melanoma using lipoprotein-delivered benzoporphyrin. Ophthalmology 1994;101:89–99. [DOI] [PubMed] [Google Scholar]
- 6.Schmidt-Erfurth U, Hasan T, Flotte T, et al. Photodynamic therapy of experimental intraocular tumors with benzoporphyrin-lipoprotein. Ophthalmologe 1994;91:348–56. [PubMed] [Google Scholar]
- 7.Schmidt-Erfurth U, Flotte TJ, Gragoudas ES, et al. Benzoporphyrin-lipoprotein-mediated photodestruction of intraocular tumours. Exp Eye Res 1996;62:1–10 [DOI] [PubMed] [Google Scholar]
- 8.Schnurrbusch UEK, Welt K, Horn L-C, et al. Histological findings of surgically excised choroidal neovascular membranes after photodynamic therapy. Br J Ophthalmol 2001;85:1086–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ghazi NG, Jabbour NM, De La Cruz ZC, et al. Clinicopathologic studies of age-related macular degeneration with classic subfoveal choroidal neovascularization treated with photodynamic therapy. Retina 2001;21:478–86. [DOI] [PubMed] [Google Scholar]
- 10.Green WR, McDonnell PJ, Yeo JH. Pathologic features of senile macular degeneration. Ophthalmology 1985;92:615–627. [PubMed] [Google Scholar]
- 11.Grossniklaus HE, Hutchinson AK, Capone A Jr, et al. Clinicopathologic features of surgically excised choroidal neovascular membranes. Ophthalmology 1994;101:1099–111. [DOI] [PubMed] [Google Scholar]
- 12.Grossniklaus HE, Green WR. Histopathologic and ultrastructural findings of surgically excised choroidal neovascularization. Submacular Surgery Trials Research Group. Arch Ophthalmol 1998;116:745–9. [DOI] [PubMed] [Google Scholar]
- 13.Grossniklaus HE, Gass JDM. Clinicopathologic correlations of surgically excised type 1 and type 2 submacular choroidal neovascular membranes. Am J Ophthalmol 1998;126:59–69. [DOI] [PubMed] [Google Scholar]
- 14.Delori FC, Goger DG, Dorey CK. Age-related accumulation and spatial distribution of lipofuscin in RPE of normal subjects. Invest Ophthalmol Vis Sci 2001;42:1855–66. [PubMed] [Google Scholar]
- 15.Lutty G, Grunwald J, Majji AB, et al. Changes in choriocapillaris and retinal pigment epithelium in age-related macular degeneration. Mol Vis 1999;3:5–35. [PubMed] [Google Scholar]
- 16.Green JL, Rabb MF. Degeneration of Bruch’s membrane and retinal pigment epithelium. Int Ophthalmol Clin 1981;21:27–50. [DOI] [PubMed] [Google Scholar]
- 17.Green WR, McDonnell PJ, Yeo JH. Pathologic features of senile macular degeneration. Ophthalmology 1985;92:615–27. [PubMed] [Google Scholar]
- 18.Okabe S, Nobuhiko M, Okamoto S, et al. Electron microscopic studies on retinochoroidal atrophy in the human eye. Acta Med Okayama 1982;36:11–21. [DOI] [PubMed] [Google Scholar]
- 19.Rabb MF, Garoon I, LaFranco FP. Myopic macular degeneration. Int Ophthalmol Clin 1981;21:51–69. [DOI] [PubMed] [Google Scholar]
- 20.Schmidt-Erfurth U, Miller J, Sickenberg M, et al. Photodynamic therapy of subfoveal choroidal neovascularization: clinical and angiographic examples. Graefes Arch Clin Exp Ophthalmol 1998;236:365–74. [DOI] [PubMed] [Google Scholar]
- 21.Schmidt-Erfurth U. Indocyanine green angiography and retinal sensitivity after photodynamic therapy of subfoveal choroidal neovascularization. Sem Ophthalmol 1999;14:35–44. [DOI] [PubMed] [Google Scholar]
- 22.Flower RW, Snyder WJ. Expanded hypothesis on the mechanism of photodynamic therapy action on choroidal neovascularization. Retina 1999;5:365–9. [DOI] [PubMed] [Google Scholar]