Abstract
Aim: To investigate L-selectin expression and shedding in patients with and without retinopathy and to determine if any observed changes are reflected by a functional change in the adhesion of leucocytes to an endothelial monolayer.
Methods: Age matched diabetic patients (26 with retinopathy, 19 without retinopathy) were compared to 24 non-diabetic controls to determine L-selectin surface protein expression, L-selectin mRNA production, and serum L-selectin levels by flow cytometry, RT-PCR, and ELISA, respectively. An adhesion assay was used to determine the binding of lymphocytes from the respective test groups to a monolayer of human endothelial cells.
Results: Significantly reduced (p = 0.004) L-selectin expression was demonstrated on lymphocytes (CD3+) from patients with diabetes compared to controls, the lowest levels being found in those with diabetic retinopathy (p = 0.004). L-selectin mRNA levels (p = 0.007) were significantly higher in the retinopathy group than in the no retinopathy group. Serum L-selectin levels were significantly higher (p = 0.04) in those with retinopathy compared to controls. Lymphocyte adhesion relative to control (100%) was essentially unchanged (84.0% (SD 27.7%), p = 0.15) for diabetic patients with no retinopathy and was markedly increased (192% (37.6%)) for those with retinopathy (p = 0.0001).
Conclusion: Lymphocyte activation, reduced surface L-selectin, increased circulating L-selectin, and a corresponding increase in adhesion of patients’ cells using an in vitro assay, is evident in people with diabetic retinopathy. This suggests a role for lymphocyte activation in the pathogenesis of diabetic retinopathy.
Keywords: diabetic retinopathy, lymphocyte, L-selectin, adhesion
Leucocyte-endothelial cell interactions are currently thought to have a role in the pathogenesis of diabetic retinopathy (DR).1 The aetiology of retinopathy is not fully understood but evidence is accumulating that low level inflammatory processes are central to its development. Retinal ischaemia is characteristic and develops as a result of capillary occlusion.2 The processes involved include intravascular coagulation as a result of a number of rheological alterations associated with diabetes: platelets exhibit increased stickiness,3 leucocytes become activated4 and less deformable,5 and vascular endothelial cells show increased expression of adhesion molecules.6 It is suggested that trapped leucocytes may increase vascular permeability by damaging the blood-retinal barrier7 and/or by presenting a physical barrier to blood flow resulting in microaneurysm formation. Endothelial dysfunction or damage is induced by release of proteolytic enzymes and oxygen free radicals from activated leucocytes8 in response to the high levels of glucose that prevail in patients with diabetes. Circulating levels of various adhesion molecules are raised in those with progressively worsening retinopathy as a result of shedding from both activated leucocytes and abnormal endothelium.9
L-selectin (CD62L) is an adhesion molecule found on the surface of lymphocytes, granulocytes, and monocytes and is involved in the initial capture and subsequent rolling of leucocytes along the endothelium in conjunction with E-selectin and P-selectin. L-selectin is shed into the serum from leucocytes in response to activation10 and has been shown to be associated with disease activity in rheumatoid arthritis,11 adult respiratory distress syndrome,12 HIV, and sepsis10 among others.
This study examines the expression of L-selectin on lymphocytes and their adhesion to an endothelial monolayer. Patients with diabetes are compared to age matched controls. We also investigate the association of any observed changes with the presence of diabetic retinopathy.
PATIENTS AND METHODS
Sixty nine age matched subjects consisting of 45 patients with diabetes and 24 controls had fresh blood samples collected for analysis. Diabetic patients were classified into the following two groups: no clinically detectable retinopathy (NDR, n = 19), or patients with diabetic retinopathy (DR, n = 26) by dilated funduscopy using the indirect biomicroscope. The DR group consisted of eight patients with non-proliferative diabetic retinopathy and 18 with proliferative disease. Any patients undergoing laser photocoagulation therapy were excluded from the study. Not all patients in each test group were examined for each parameter owing to the practical constraints of patient involvement and the nature of the technical processes involved. Blood pressure was recorded using an automatic sphygmomanometer (Dynamapp, FL, USA). The diabetic group consisted of a greater number of men and contained more smokers. Patients were grouped into type I or II diabetes on the basis of their clinical presentation and features. A random blood glucose level was taken at the time of sampling along with biochemical measurement of renal function, lipid status, and glycosylated haemoglobin (HbA1c). The research followed the tenets of the Declaration of Helsinki and was approved by Aberdeen Royal Infirmary ethics committee.
Flow cytometry (FACS)
A fluorescent activated cell sorter (FACSCalibur, Becton Dickinson, San Jose, CA, USA) was used for the analysis. Heparinised blood samples from 15 patients with NDR, 21 with DR, and 17 non-diabetic controls were collected and stored on ice for a maximum of 30 minutes before incubation with directly conjugated fluorescent monoclonal antibodies as previously described.13 Commercially available FACS antibodies (Becton Dickinson) to L-selectin and CD3 were added followed by red blood cell lysis. Two further washes were completed before the cells were added to a fixing solution (1% paraformaldehyde) before analysis. The percentage positive cells for L-selectin were calculated using CellQuest software (Becton Dickinson) gating on cells labelled with CD3. Appropriate isotype controls were used for each experiment in order to establish baseline parameters. All procedures were identical for all of the subject groups examined.
Mononuclear cell separation
Leucocytes were separated from 30 ml whole blood samples for the reverse transcriptase polymerase chain reaction (RT-PCR) and adhesion assays (see below). The blood was added to acid citrate dextrose (Gibco BRL, Life Technologies, Paisley, UK) then mixed with equal volumes of RPMI 1640 (Gibco BRL) before layering over Histopaque (1.077 g/ml, Sigma, St Louis, MO, USA). The layered samples were centrifuged and the interface of the layers harvested and washed as previously described.14 The supernatant was discarded and the remaining pellets pooled then centrifuged to remove platelets. Remaining red blood cells were removed before a further two washes with RPMI.
RT-PCR
Samples of whole blood were taken from seven non-diabetic controls, six diabetic patients with NDR, and seven with DR and the mononuclear cell population was harvested as outlined above. RNA was extracted and purified using phenol-chloroform extraction15 and a cDNA was prepared for each patient as previously described.14 Commercially available L-selectin specific primers were used (R&D Systems, Abingdon, UK) and the β actin primers were synthesised (Oswell, Oxford, UK) using previously published sequence data (Human Genome Mapping Project). The optimal conditions for the amplification of L-selectin and the internal control (β actin) were determined using a range of cycle numbers and cDNA concentrations and quantified using the Gel Documentation System (GDS5000, UVP Systems Ltd) as previously described.14 The conditions allowing for the generation of the respective PCR products in the linear phase of their amplification were used for the test groups examined as follows: 30 cycles of 94° for 50 seconds, 55° for 60 seconds, and 72° for 90 seconds.
ELISA
Whole blood samples (10 ml) were collected in clot activator tubes (Becton Dickinson, France) from 12 non-diabetic controls, 14 patients with NDR, and 14 with DR. Serum samples were prepared and stored at −20°C. Samples were plated out in duplicate on the ELISA plate (R&D Systems, Minneapolis, MN, USA) along with controls and standards provided with the kit. A standard curve was generated with an r2 value of 0.999. The samples were read by a plate reader (Dynatech MR5000, Dynex Technologies, Chantilly, VA, USA) and analysed using computer software (Biolinx 2, Dynex Technologies).
Lymphocyte adhesion
Using a static adhesion assay the binding of fluorescently labelled lymphocyte suspensions to a monolayer of human umbilical vein endothelial cells (HUVECs) was studied using a previously described method.16 Briefly fresh blood samples (30 ml) were obtained from seven non-diabetic control subjects, four diabetic patients with NDR, and five patients with DR and the mononuclear cells prepared as described above. The cells were incubated to allow monocytes to adhere to plastic and the non-adherent cell population was collected. The cells were seeded into a microtitre plate at a concentration of 2×106 cells/ml. Cell viability was measured using trypan blue dye exclusion and was routinely in excess of 97%. Calcein AM in DMSO (Dynatech Laboratories, Chantilly, VA, USA), an intracellular fluorescent dye, was mixed in equal volumes with the lymphocyte cell suspension. The labelled cell suspension was then used at a concentration of 2×105/ml for plating. Flow cytometry confirmed that population purity was high with >99.3% purity for lymphocytes (data not shown).
The cells were seeded into a 96 well microtitre plate prepared the previous day with HUVECs at a concentration of 0.25×106/ml and allowed to grow to confluence overnight. A 150 μl aliquot of the cell suspension was added to each well and the plates incubated at 37°C for 90 minutes. Following incubation, non-adherent cells were removed by washing three times with RPMI. A fluorescent plate reader (Fluorolite 1000, Dynex Technologies) was set with an excitation wavelength of 490 nm and an emission wavelength of 530 nm.16 Samples for each experiment were plated in a minimum of six replicate wells and the fluorescent measurements averaged for analysis. Results for each adhesion assay were calculated as a percentage of the age matched control result from the non-diabetic samples collected and analysed at the same time as the diabetic samples. At least one standard control sample was used to normalise the data for each plate to minimise interplate variability. The control value for each experiment was arbitrarily set at 100% to allow for day to day variation in cell behaviour.
Statistical analysis
Pearson correlations were made between each variable measured in the respective test groups. A Mann-Whitney U test was employed for intergroup comparisons. A p value of <0.05 was taken as being statistically significant.
RESULTS
Diabetic phenotypes
Patients with DR had a significantly longer duration of diabetes (p = 0.003) than those with no retinopathy (table 1). Patients with DR had significantly higher systolic blood pressure recordings than controls (142 (SD 29) versus 120 (12) mm Hg, p = 0.02); there was no difference in diastolic blood pressure recordings between the groups.
Table 1.
Patient data
| No | Age (years) | Sex | Type | Duration of diabetes (years) | Glucose | HbA1c | |
| Controls | 24 | 49 (19) | 13F, 11M | 5.1 (1.3) | |||
| NDR | 19 | 51 (19) | 5F, 14M | 7I, 12II | 6 (4) | 11.7 (5.7)* | 9.0 (2.1) |
| DR | 26 | 49 (18) | 9F, 17M | 12I, 14II | 14 (9)ψ | 12.6 (5.3)* | 10.2 (2.4) |
Mean values (SD) indicated. Subjects with diabetes are divided into those with type I and type II diabetes. Clinical biochemistry results are given for glucose (mmol/l) and glycosylated haemoglobin (HbA1c,%). Symbols used indicate results of Mann-Whitney tests, *denotes p⩽0.02 compared to controls and ψ denotes p = 0.003 compared to the NDR group.
Patients with diabetes had random blood glucose measurements that were higher (p<0.001) than controls by definition (table 1). Patients with retinopathy did not have significantly poorer glycaemic control (p = 0.08) than those with NDR as indicated by their HbA1c values (table 1). Triglyceride levels were raised in patients with retinopathy (2.1 (0.97) mmol/l, p = 0.004) compared to controls (1.28 (0.58) mmol/l) suggesting an increased cardiovascular risk for these patients. There were no significant differences in the level of cholesterol, HDL, LDL, urea, or creatinine between any of the groups. There were three smokers each in the control and NDR groups and six in the DR group. A history of current smoking was not associated with a difference in any of the parameters studied.
L-selectin levels in diabetic patients
FACS analysis showed significantly lower expression of L-selectin on CD3 positive lymphocytes from patients with diabetes (63.94% (11.34%)) compared to controls (74.17% (6.51%)), p = 0.004 (table 2). However, the levels of L-selectin mRNA (table 2) were not significantly different (p = 0.8) for diabetic patients (1.086 (0.303)) compared to controls (1.246 (0.700)). Serum L-selectin levels (table 2) were not significantly elevated (987 (243) ng/ml, p = 0.06) compared to controls (828 (177) ng/ml).
Table 2.
L-selectin detection and lymphocyte adhesion in subjects with diabetes and controls
| Method | Controls | Diabetics | p v control |
| L-selectin on CD3+(FACS) | 74.17% (6.51) (n = 17) | 63.94% (11.34) (n = 36) | 0.004 |
| L-selectin mRNA (RT-PCR) | 1.246 (0.7) (n = 7) | 1.086 (0.303) (n = 13) | 0.8 |
| Serum L-selectin (ELISA) | 828 ng/ml (177) (n = 12) | 987 ng/ml (243) (n = 28) | 0.06 |
| Lymphocyte adhesion | 100% (n = 7) | 144% (65) (n = 9) | 0.1 |
Results of surface L-selectin on CD3+ cells detected by flow cytometry (% positive cells), nuclear mRNA detected by RT-PCR (L-selectin: β actin ratio), shed L-selectin in the serum (ng/ml) detected by ELISA and lymphocyte adhesion to a HUVEC monolayer (% of control value, which has arbitrarily been set at 100%) are tabled for patients with diabetes and controls. Mean values (SD) are indicated.
Pearson correlations indicated an association between HDL and both L-selectin mRNA levels (p = 0.008) and serum L-selectin (p = 0.049). Significantly higher serum L-selectin levels were found in those with type I diabetes (1101 (183) ng/ml) compared to those with type II diabetes (873 (247) ng/ml, p = 0.02).
L-selectin levels in diabetic retinopathy
The presence of diabetic retinopathy was associated with the lowest levels of L-selectin expression on CD3+ cells (63.47% (10.37%), p = 0.004) compared to controls (74.17% (6.51%)), (fig 1A). Serum L-selectin levels (fig 1B) were significantly higher in those with DR (992 (184) ng/ml, p = 0.04) compared to controls (828 (177) ng/ml). No significant difference between L-selectin on CD3+ cells or on serum L-selectin levels was found comparing the NDR and DR groups. Significantly higher L-selectin mRNA levels were found in the DR group (1.298 (0.202), p = 0.007) compared to the NDR group (0.874 (0.232)), (fig 2B). There was no significant difference comparing the DR or NDR groups with controls.
Figure 1.
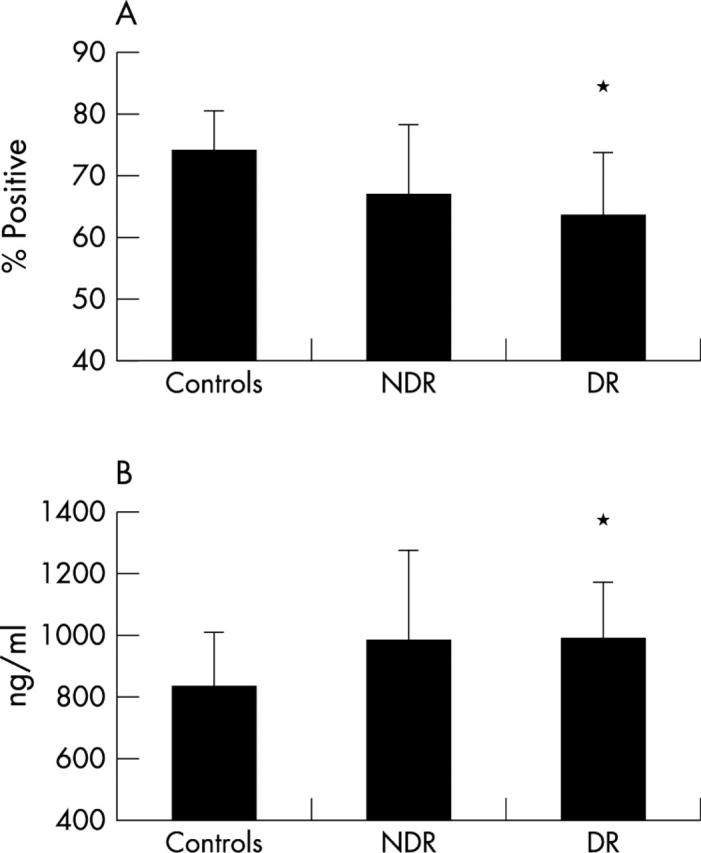
L-selectin levels on CD3+ cells and in serum. (A) Result from FACS analysis of 17 non-diabetic controls, 15 subjects with NDR and 21 with DR to show surface expression of L-selectin on CD3+ lymphocytes. Mean percentage values and standard deviations are indicated. *Denotes p = 0.004 compared to controls (Mann-Whitney test). (B) Shed levels of L-selectin detected by ELISA in the serum of 12 controls, 14 subjects with NDR, and 14 with DR. Mean values in ng/ml. *Denotes p = 0.04 compared to controls (Mann-Whitney test).
Figure 2.
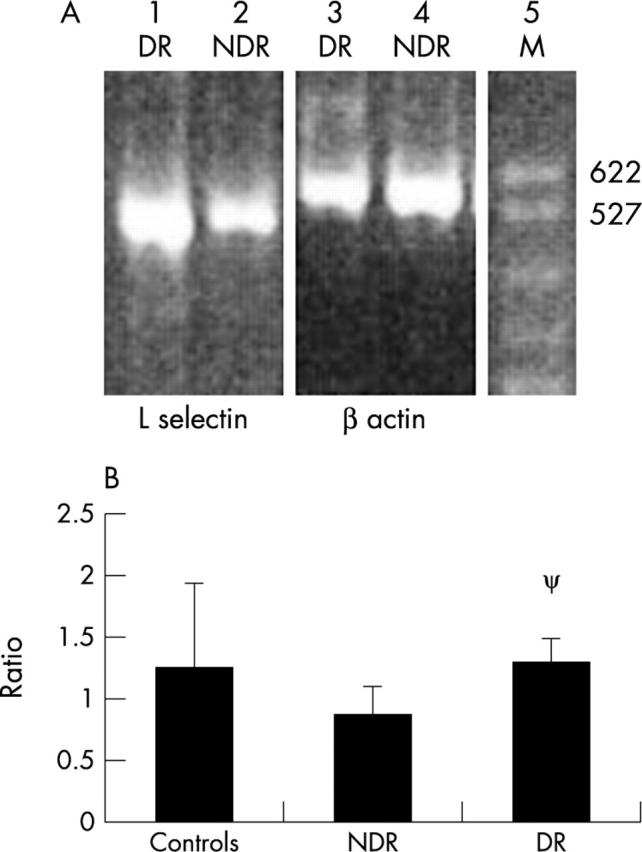
Representative gel of PCR products and result of L-selectin: β actin ratios. (A) Agarose gel visualised under ultraviolet light using the fluorescent properties of ethidium bromide. PCR products for L-selectin (528 base pairs) from a subject with DR in lane 1 and with NDR in lane 2, with their respective β actins (548 base pairs) in lanes 3 and 4 are shown. A DNA marker (DM) is shown in lane 5 with the base pairs of the first two bands indicated. (B) L-selectin: β actin ratios for mononuclear cells from seven controls, six subjects with NDR, and seven with DR. Mean values and standard deviations are indicated. ψ denotes p = 0.007 compared to NDR (Mann-Whitney test).
Lymphocyte adhesion
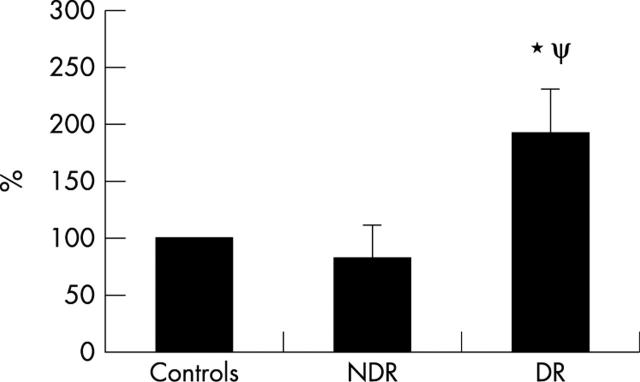
There was no significant difference in lymphocyte adhesion to a HUVEC monolayer (table 2) for diabetic patients as a group (144% (65%), p = 0.1) relative to controls (100%). When patients with DR were compared to non-diabetic individuals or to the patients with NDR, there was a significant increase in their lymphocyte adhesiveness to an endothelial monolayer. The mean value for adhesion relative to control (100%) was essentially unchanged (84% (28%), p = 0.15) for those in the NDR group but was markedly increased (192% (38%), p = 0.0001) for those in the DR group (fig 3). The significance level when comparing those in the DR group with the NDR group was p = 0.002 for those with retinopathy.
Figure 3.
Lymphocyte adhesion to HUVEC monolayer. Calcein AM labelled lymphocyte adhesion to a HUVEC monolayer using cells from seven controls, four patients with NDR, and five with DR. For samples from diabetic subjects, mean values and standard deviations have been calculated and are expressed as a percentage of the control result, which has arbitrarily been set a value of 100%. Symbols used indicate results of unpaired t tests, *denotes p = 0.0001 compared to controls and ψ denotes p = 0.002 compared to the NDR group.
DISCUSSION
This study has demonstrated lymphocyte activation in patients with diabetes as revealed by a significant reduction in the expression of L-selectin on CD3+ cells. The changes in L-selectin expression were restricted to those patients who had observable signs of diabetic retinopathy and were not correlated with duration of diabetes, HbA1c level, or circulating glucose concentration, although these factors have recently been linked with serum L-selectin levels.17 Presence of DR was also associated with elevated L-selectin mRNA levels using semiquantitative RT-PCR which paralleled the increased shedding of L-selectin in the serum. In addition, an increase in the adhesion of leucocytes to an endothelial monolayer for patients with diabetic retinopathy was identified. This is probably due to stimulation of lymphocytes through interaction with a diabetes induced activated endothelium with upregulation of adhesion molecules such as VCAM and E-selectin.9
Leucocytes from patients with diabetes (and those with preclinical type 1 diabetes) have been shown to shed adhesion molecules into the circulation.18,19 Furthermore endothelial cell activation has been demonstrated in association with DR.9 Our findings now extend these observations and associate leucocyte activation more specifically with the onset of microvascular retinal disease. There was no evidence of any difference between the type I and type II patients with respect to CD3+ cell expression. However, serum L-selectin levels were found to be higher in patients with type 1 diabetes. This may reflect differences in the level of pro-inflammatory cytokines in this patient group and differing disease aetiology between the two types of diabetes. This elevated level of soluble L-selectin does not correlate with the changes observed in the DR and NDR groups.
Our current understanding of capillary occlusion as a major component of diabetic retinopathy,4,20 and therefore our finding of increased adhesiveness to human endothelial cells of lymphocytes from those with DR, is in keeping with current views on the pathogenesis of diabetic retinopathy. Importantly, it extends previous clinical and experimental data relating to neutrophil and monocyte adhesion in diabetic retinopathy to lymphocytes. Lymphocytes have greater potential for survival and proliferation in situ than monocytes (neutrophils have no proliferative capacity) and therefore the entrapment of lymphocytes in the retinal vasculature may provide a sustainable source of growth factor14 for neovascularisation as originally suggested by Schroder et al4 for monocytes.
It is difficult to state if the changes in lymphocyte activation and adhesion are the direct consequence of the onset of retinal changes or if they are associated with the duration of diabetes as these two events are so closely intertwined. The correlation between duration of diabetes and microvascular complications has already been clearly demonstrated.21 The practical process of lymphocyte isolation may also have contributed artefactually to cell activation although we believe we have controlled for this since both diabetic and control samples were collected concurrently and treated identically. Further investigation of the mechanisms involved in L-selectin shedding and lymphocyte adhesion to determine whether adhesion can be blocked or reduced were unfortunately not feasible within this study. This information would have the potential to indicate therapeutic manipulation aimed at preventing diabetic vasculopathy.
Different sites of glycosylation have been shown to influence L-selectin mediated adhesion22 and the increase in glycosylation that is associated with an increased duration of diabetes may influence this parameter. However, given the specificity of the interaction that is required for binding of lymphocyte and endothelium it is more likely that we should see a decrease in adhesion if glycosylation of the L-selectin was the prime reason for the differences observed. The increased adhesion that is observed implies that inappropriate glycosylation is not a prime reason for these differences although a more detailed examination of the nature of this process and its influence on binding would require to be carried out to confirm this. This is further demonstrated by the increased adhesion of the lymphocytes to the endothelium despite the diabetic groups in this experiment being matched for both random glucose and HbA1c measurements. The sample numbers for the adhesion assay are small but show a clear statistical difference between those with and without retinopathy. HUVECs were chosen as the endothelial cell monolayer because a large homogeneous population of endothelial cells was required that could be depended on achieving confluence at the time of isolation of the lymphocyte sample. Ideally, retinal endothelial cells would have been used but practical constraints exclude this approach.
Previously published studies have reported on leucocyte adherence to vascular endothelial cells in relation to diabetes or hyperglycaemia.23,24 Morigi et al23 found that incubating HUVECs in 30 mM glucose conditions significantly increased adherence of control leucocytes. Increased binding of monocytes from type I diabetic patients to endothelial cells, has also been shown, particularly when the endothelium has been incubated in hyperglycaemic conditions.25 Similarly, neutrophils from diabetic versus non-diabetic animals have been reported as being more adherent to rat endothelial cell monolayers.26 However, we believe our study is the first to demonstrate a clear association between changes in L-selectin expression, the adhesive properties of human lymphocytes and DR. The data presented introduce new understanding to the potential mechanism(s) of diabetic retinopathy and the effective development of a specific therapeutic target for microvascular disease associated with diabetes.
Further investigation is required to clarify the relation between L-selectin expression and the onset and progression of diabetic retinopathy, the extent to which this also contributes to other microvascular and macrovascular complications of diabetes, and the potential of L-selectin as a therapeutic target.
Acknowledgments
We are grateful to The Wellcome Trust and Grampian University Hospitals NHS Trust Endowment for their financial support, and Hector Williams for statistical advice.
Abbreviations
DR, diabetic retinopathy
FACS, fluorescent activated cell sorter
HbA1c, glycosylated haemoglobin
HUVECs, human umbilical vein endothelial cells
NDR, no diabetic retinopathy
RT-PCR, reverse transcriptase polymerase chain reaction
REFERENCES
- 1.Joussen AM, Murata T, Tsujikawa A, et al. Leukocyte-mediated endothelial cell injury and death in the diabetic retina. Am J Pathol 2001;158:147–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Forrester JV, Knott RM, McIntosh LC. Pathogenesis of proliferative diabetic retinopathy and maculopathy. Diabetes Annual 1993;7 (Chapter 10) :178–90. [Google Scholar]
- 3.Collier A, Watson HH, Patrick AW, et al. Effect of glycaemic control, metformin and gliclazide on platelet density and aggregability in recently diagnosed type 2 (non-insulin dependent) diabetic patients. Diabetes and Metabolism 1989;15:420–5. [PubMed] [Google Scholar]
- 4.Schroder S, Palinski W, Schmid-Schonbein GW. Activated monocytes and granulocytes, capillary nonperfusion, and neovascularisation in diabetic retinopathy. Am J Pathol 1991;139:81–100. [PMC free article] [PubMed] [Google Scholar]
- 5.Pecsvarady Z, Fisher TC, Darwin CH, et al. Decreased polymorphonuclear leukocyte deformability in NIDDM. Diabetes Care 1994;17:57–63. [DOI] [PubMed] [Google Scholar]
- 6.McLeod DS, Lefer DJ, Merges C, et al. Enhanced expression of intracellular adhesion molecule-1 and p-selectin in the diabetic human retina and choroid. Am J Pathol 1995;147:642–53. [PMC free article] [PubMed] [Google Scholar]
- 7.Miyamoto K, Khosrof S, Bursell SE, et al. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci USA 1999;96:10836–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wierusz-Wysocka B, Wysocki H, Siekierka H, et al. Evidence of polymorphonuclear neutrophils (PMN) activation in patients with insulin-dependent diabetes mellitus. J Leuk Biol 1987;42:519–23. [DOI] [PubMed] [Google Scholar]
- 9.Olson JA, Whitelaw CM, McHardy KC, et al. Soluble leucocyte adhesion molecules in diabetic retinopathy stimulate retinal endothelial cell migration. Diabetologia 1997;40:1166–71. [DOI] [PubMed] [Google Scholar]
- 10.Spertini O, Schleiffenbaum B, White-Owen C, et al. ELISA for quantification of L-selectin shed from leukocytes in vivo. J Immunol Methods 1992;156:115–23. [DOI] [PubMed] [Google Scholar]
- 11.Bond A, Hay FC. L-selectin expression on the surface of peripheral blood leucocytes from rheumatoid arthritis patients is linked to disease activity. Scand J Immunol 1997;46:312–16. [DOI] [PubMed] [Google Scholar]
- 12.Donnelly SC, Haslett C, Dransfield I, et al. Role of selectins in development of adult respiratory distress syndrome. Lancet 1994;344:215–19. [DOI] [PubMed] [Google Scholar]
- 13.Kilmartin DJ, Fletcher ZJ, Almeida JA, et al. CD69 expression on peripheral CD4+T cells parallels disease activity and is reduced by mycophenolate mofetil therapy in uveitis. Invest Ophthalmol Vis Sci 2001;42:1285–92. [PubMed] [Google Scholar]
- 14.Knott RM, Pascal MM, Ferguson C, et al. Regulation of transforming growth factor-beta (TGF-β), basic fibroblast growth factor (bFGF) and vascular endothelial cell growth factor (VEGF) messenger RNA in peripheral blood leucocytes in patients with diabetic retinopathy. Metabolism: Clinical & Experimental 1999;48:1–8. [DOI] [PubMed] [Google Scholar]
- 15.Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 1987;162:156–9. [DOI] [PubMed] [Google Scholar]
- 16.Braut-Boucher F, Pichon J, Rat P, et al. A non-isotopic, highly sensitive, fluorimetric, cell-cell adhesion microplate assay using calcein AM-labeled lymphocytes. J Immunol Methods 1995;178:41–51. [DOI] [PubMed] [Google Scholar]
- 17.Karadayi K, Top C, Gulecek O. The relationship between soluble L-selectin and the development of diabetic retinopathy. Ocul Immunol Inflamm 2003;11:123–9. [DOI] [PubMed] [Google Scholar]
- 18.Lampeter ER, Kishimoto TK, Rothlein R, et al. Elevated levels of circulating adhesion molecules in IDDM patients and in subjects at risk for IDDM. Diabetes 1992;41:1668–71. [DOI] [PubMed] [Google Scholar]
- 19.Toivonen AM, Kulmala P, Savola K, et al. Soluble adhesion molecules in pre-clinical Type 1 diabetes: a prospective study. Diabetologia 2003;46:492–5. [DOI] [PubMed] [Google Scholar]
- 20.Miyamoto K, Hiroshiba N, Tsujikawa A, et al. In vivo demonstration of increased leukocyte entrapment in retinal microcirculation of diabetic rats. Invest Ophthalmol Vis Sci 1998;39:2190–4. [PubMed] [Google Scholar]
- 21.DCCT Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin dependent diabetes mellitus. N Engl J Med 1993;329:977–1034. [DOI] [PubMed] [Google Scholar]
- 22.Fieger CB, Emig-Vollmer S, Petri T, et al. The adhesive properties of recombinant soluble L-selectin are modulated by its glycosylation. Biochim Biophys Acta 2001;1524:75–85. [DOI] [PubMed] [Google Scholar]
- 23.Morigi M, Angioletti S, Imberti B, et al. Leukocyte-endothelial interaction is augmented by high glucose concentrations and hyperglycaemia in a NF-kB-dependent fashion. J Clin Invest 1998;101:1905–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fortes ZB, Farsky SP, Oliveira MA, et al. Direct vital microscopic study of defective leukocyte-endothelial interaction in diabetes mellitus. Diabetes 1991;40:1267–73. [DOI] [PubMed] [Google Scholar]
- 25.Kunt T, Forst T, Fruh B, et al. Binding of monocytes from normolipidemic hyperglycaemic patients with type I diabetes to endothelial cells is increased in vitro. Exp Clin Endocrinol Diabetes 1999;107:252–6. [DOI] [PubMed] [Google Scholar]
- 26.Barouch FC, Miyamoto K, Allport JR, et al. Integrin-mediated neutrophil adhesion and retinal leukostasis in diabetes. Invest Ophthalmol Vis Sci 2000;41:1153–8. [PubMed] [Google Scholar]