Abstract
Aim: To determine the effectiveness of treatment with immunosuppressive drugs and monoclonal antibodies (mAb) after penetrating keratoplasty in two different models of high risk mouse recipients.
Methods: Corneas were grafted orthotopically in mouse models of high risk recipients with either neovascularisation of the graft bed or presensitisation to graft donor antigens. Recipients were treated with mAb against CD4+ or CD8+ cells or against T cells, or were treated with cyclosporin A (CsA) or mycophenolate mofetil (MMF), or a combination of both drugs.
Results: Control untreated recipients with neovascularised graft bed or presensitised to the graft donor antigens rejected corneal allografts in 12.5 (SD 2.3) and 9.9 (1.6) days, respectively. Treatment of graft recipients with a neovascularised graft bed with mAb anti-CD4 or anti-T cells, but not with mAb anti-CD8 or with immunosuppressive drugs, resulted in a significant prolongation of graft survival; 75% and 28.5%, respectively, of grafts survived for more than 45 days after grafting. However, none of the treatments were successful in presensitised recipients.
Conclusions: Treatment of high risk recipients with mAb anti-CD4 is more effective in preventing corneal allograft rejection than the treatment with mAb anti-CD8 or the immunosuppressive drugs MMF and CsA. However, the effectiveness of the treatment depends on the recipients’ pretransplantation risk type.
Keywords: high risk recipients, corneal allografts, immunosuppression, graft survival, mouse model
Corneal transplantation is the most common form of solid tissue transplantation in humans and is characterised by an unusually high success in graft survival. The first year graft survival rate under cover of only local immunosuppression reaches 90%.1 Whereas graft survival in low risk conditions (keratoconus, corneal dystrophies) remains over 90% at 10 years after grafting, the 10 year success rate in high risk recipients (with a history of anterior segment inflammation, corneal neovascularisation, etc) is much lower, achieving less than 35%.1,2 The most common cause of the graft failure is immunological rejection. To obtain the long term survival of corneal grafts, especially in high risk recipients, it is necessary to search for new and effective immunosuppressive approaches with minimal undesirable side effects.
Immunohistochemical analysis of cells infiltrating rejected corneal allografts in humans and in experimental animals demonstrated large numbers of CD4+ T cells and macrophages.3–6 The role of macrophages as effector cells in corneal allograft rejection was demonstrated in experiments which showed indefinite graft survival in rats after the selective depletion of macrophages with dichloromethylene diphosphonate.7 CD4+ T cells are important for the activation of macrophages, which are consequently able to kill allogeneic cells.8 The importance of CD4+ T cells during corneal allograft rejection was demonstrated in experimental models by a long term graft survival after systemic or topical treatment with anti-CD4 antibody.9–12 Several other monoclonal antibodies (mAb), such as anti-T cells administered intracamerally13,14 and anti-Campath-1H or anti-CD25 in a systemic treatment,15–17 have already been tested for immunosuppression in human corneal transplantation. Systemic treatment with immunosuppressive drugs such as cyclosporine A (CsA) or mycophenolate mofetil (MMF) has been used after penetrating keratoplasty in high risk patients.18–21
Despite the fact that various immunosuppressive treatments have been used in high risk patients, there are no published reports comparing the effectiveness of individual immunosuppressive protocols in experimental models of high risk recipients. In this study we therefore attempted to find a method of preventing high risk rejection and compared the effectiveness of treatment with immunosuppressive drugs or mAb in preventing corneal allograft rejection in two different experimental models of orthotopic high risk corneal transplantation in mice.
MATERIALS AND METHODS
Animals
Mice of the inbred strain BALB/c (H-2d) were used as recipients and mice of the strain C57BL/10Sn (B10) (H-2b) as graft donors. Mice of both sexes were aged from 12 to 16 weeks. All animals were obtained from the breeding colony of the Institute of Molecular Genetics, Prague. The experiments were performed in accordance with the law governing the protection of animals and were approved by the local animal ethics committee.
Preparation of high risk recipients
Prevascularisation
Mice were anaesthetised by intramuscular injection with a mixture of ketamine (Calypsol, Gedeon Richter Ltd, Budapest, Hungary) and xylazine (Rometar, Spofa, Prague, Czech Republic) diluted in balanced salt solution (BSS, Alcon Laboratories Inc, Fort Worth, TX, USA). Four intrastromal sutures in four quadrants of the recipient cornea were inserted surgically 14 days before transplantation. These sutures were removed with recipients’ button during the transplantation.
Presensitisation
BALB/c recipients were presensitised to the graft donor antigens by intraperitoneal injection of 2×107 spleen cells from B10 donors 14 days before the transplantation.
Technique of orthotopic corneal transplantation
The procedure was adapted from the technique described previously by She et al22 and modified by Plšková et al.23 The donor cornea (B10 mice) was marked with 2 mm trephine, the anterior chamber was penetrated using a sharp needle and the cornea was then cut out with Vannas scissors. The donor endothelium was protected during dissection by instilling a high molecular weight hyaluronic acid (Provisc, Alcon Canada Inc, Mississauga, Ontario, Canada) into the anterior chamber. The donor cornea was placed in BSS. The recipient mouse was anaesthetised as described previously. To dilate the pupil tropicamid drops (Mydrum, Chauvin Ankepharm GmBh, Rudolstadt, Germany) were used. The recipient right cornea was marked with a 1.5 mm trephine and removed by the same method as the donor cornea. The donor graft was sutured into the recipient bed using a running suture (11-0 Ethilon, a 50 µm diameter needle, Ethicon, St-Stevens-Woluve, Belgium). After the transplantation the eye was covered with antibiotic ointment, a combination of bacitracin and neomycin (Ophthalmo-Framykoin, Léčiva, Prague, Czech Republic). The sutures were not removed.
Clinical evaluation of rejection
The mice were examined every other day using the operating microscope. The opacity grade (from 0 to 4) of the grafted cornea was used as the indicator of graft rejection.24 Opacity grade 2 or more was considered as graft rejection. Mice with complications such as cataract, inflammation, or haemorrhage were excluded from the study.
Monoclonal antibodies
The first group of high risk recipients were treated with mAb anti-Thy1.2 (clone F7D5),25 anti-CD4 (clone GK1.5),26 and anti-CD8 (clone TIB 150).27 Antibodies were prepared in the form of ascites in nu/nu mice and were injected intraperitoneally at a dose of 200 µg/mouse/day. The treatment started the day after transplantation and continued every other day until the rejection. FACS analysis performed 1 week after the beginning of the treatment revealed that the number of cells of a particular cell subpopulation in lymph nodes and spleens was already reduced to less than 1.5% of the value in normal animals and this selective T cell lymphopenia was sustained for the duration of the treatment (data not shown).
Immunosuppressive drugs
The second group of recipients were treated with MMF (Cellcept, F Hoffmann-La Roche Ltd, Basel, Switzerland) at a dose of 25 mg/kg/day diluted in a buffer solution or with CsA (Ciclosporinum, Sandimmun Neoral, Novartis Pharma, Pharma AG, Basel, Switzerland) at a dose of 25 mg/kg/day diluted in a citrate solution or with the combination of MMF/CsA, at a dose of 12.5 mg/kg/day of each compound. The orally administered treatment started the day after transplantation and continued daily until rejection occurred.
Statistical analysis
The statistical differences in graft rejection rate between experimental and control groups were evaluated by Mann-Whitney U test. The day of the onset of rejection was used for this statistical analysis and to calculate the mean survival time (MST) plus or minus SD. In the case of some animals no rejection had occurred by the time the experiment was terminated. In these cases for statistical purposes we assigned the day after termination as the day of rejection.
RESULTS
Survival of corneal allografts in low risk and high risk control untreated recipients
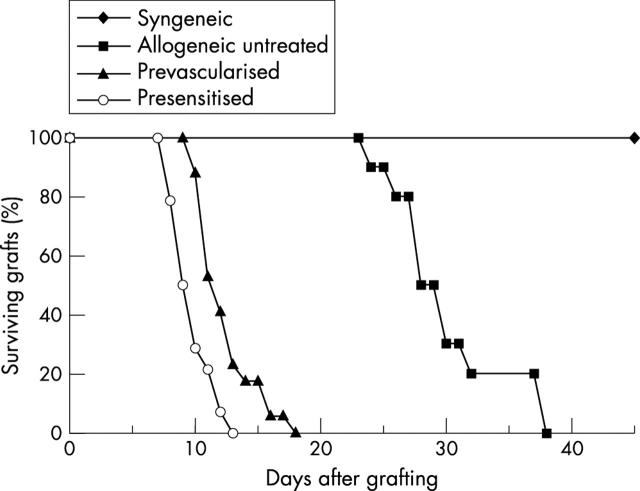
Corneal allografts in low risk recipients were rejected at the MST 30.2 (4.7) days (10 mice), and all allografts were rejected by day 38. Corneal allografts in recipients with prevascularised graft bed or presensitised to the graft donor antigens were rejected with MST 12.5 (2.3) days (17 mice) and 9.9 (1.6) days (14 mice); respectively. All syngeneic grafts (eight mice) remained clear without sign of rejection (fig 1).
Figure 1.
Survival of syngeneic corneal grafts (eight mice) or corneal allografts in control (unmodified) recipients (10 mice), recipients with prevascularised graft bed (17 mice), or recipients presensitised to the graft donor antigens (14 mice).
Survival of corneal allografts in recipients with prevascularised graft bed and treated with mAb or immunosuppressive drugs
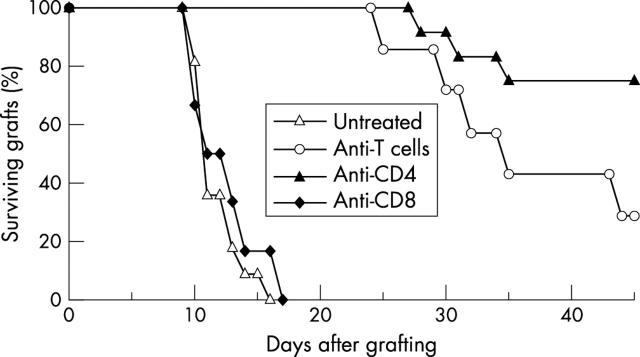
The recipients with prevascularised graft bed treated with mAb anti-CD8 rejected their grafts in MST of 12.5 (2.7) days (six mice) and there was no significant difference from the untreated control group (p = 0.44). On the other hand, corneal graft survival was significantly prolonged by mAb anti-T cells (MST 36.9 (8.5) days, seven mice, p<0.001) and anti-CD4 cells (MST 42.3 (6.8) days, 12 mice, p<0.001). In these two groups, 28.6% and 75% of grafts, respectively, remained unrejected at 45 days, the point when the experiment was terminated (fig 2).
Figure 2.
Survival of corneal allografts in recipients with prevascularised graft bed and treated with mAb. The recipients were untreated (11 mice) or were treated with mAb anti-CD4 (12 mice), anti-CD8 (six mice), or anti-T cells (seven mice).
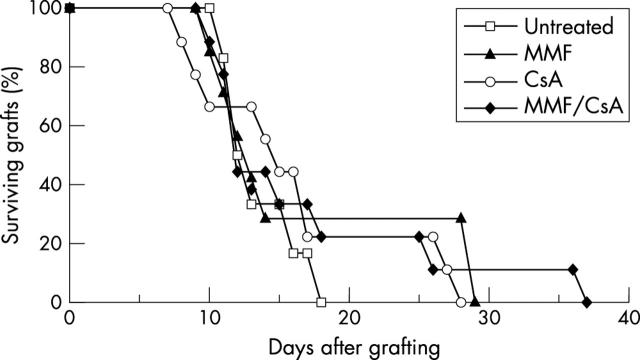
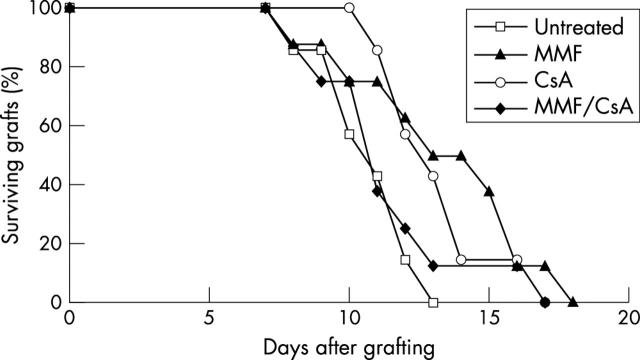
Another group of recipients with prevascularised graft beds were treated with immunosuppressive drugs. In comparison to the control group, there was a short but statistically insignificant prolongation of graft survival after treatment with MMF (MST 16.9 (8.4) days, seven mice, p = 0.47), CsA (MST 16.1 (7.3) days, nine mice, p = 0.39), and a combination of MMF/CsA (MST 17.0 (9.0) days, nine mice, p = 0.48). In these four groups all animals had undergone graft rejection by day 37 (fig 3).
Figure 3.
Survival of corneal allografts in recipients with prevascularised graft bed and treated with immunosuppressive drugs. The recipients were untreated (six mice) or were treated with MMF (seven mice), or CsA (nine mice), or with a combination of MMF/CsA (nine mice).
Survival of corneal allografts in recipients presensitised to the graft donor antigens and treated with mAb or immunosuppressive drugs
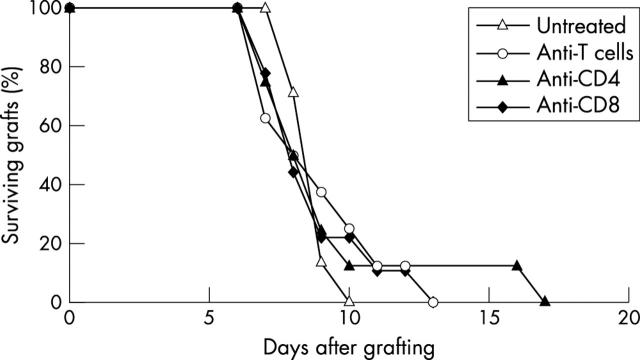
In comparison with the control group there was no significant prolongation of corneal graft survival after treatment with mAb anti-T cells (MST 9.0 (2.2) days, eight mice, p = 0.48), anti-CD4 cells (MST 9.4 (3.3) days, eight mice, p = 0.32), or anti-CD8 cells (MST 8.9 (2.0) days, nine mice, p = 0.26). In these four groups all animals rejected by day 17 after grafting (fig 4).
Figure 4.
Survival of corneal allografts in presensitised recipients and treated with mAb. The recipients were untreated (seven mice) or were treated with mAb anti-CD4 (eight mice), anti-CD8 (nine mice), or anti-T cells (eight mice).
Treatment with CsA significantly delayed corneal graft rejection (MST 13.3 (2.0) days, seven mice, p<0.05) but the graft survival times after treatment with MMF (MST 13.5 (3.4) days, eight mice, p = 0.06) and with a combination of MMF/CsA (MST 11.5 (2.7) days, eight mice, p = 0.41) were not significantly different from those in the control group. In these three groups all animals rejected by day 18 after the transplantation (fig 5)
Figure 5.
Survival of corneal allografts in presensitised recipients and treated with immunosuppressive drugs. The recipients were untreated (seven mice) or were treated with MMF (eight mice), CsA (seven mice), or with a combination of MMF/CsA (eight mice).
DISCUSSION
Although various immunosuppressive treatments have been used in high risk patients with keratoplasty for several years, there have been no comparative studies on experimental models of high risk recipients. In the present work, we used two models of mouse high risk recipients. The first group had their graft bed prevascularised by sutures (strong neovascularisation in all four quadrants of the cornea). The second group was presensitised to the alloantigens of the graft donor genotype. Both groups of high risk mice had markedly reduced corneal graft survival and they became a convenient model for evaluating the efficacy of treatment. We used these mice to compare the effect of immunosuppressive drugs and mAb against T cell subpopulations on corneal allograft survival. Significant and long term corneal allograft survival was obtained after treatment with mAb against CD4+ cells or against T cells in recipients with a prevascularised graft bed. However, the same treatment failed in recipients presensitised to alloantigens of the graft donor. There was no statistically significant delay of a corneal graft rejection in either model of high risk mice when we used systemic treatment with MMF and with the MMF/CsA combination. Treatment with CsA significantly prolonged corneal graft survival only in presensitised mice but this effect was only moderate and all treated mice rejected their grafts by day 18 after grafting.
The doses of CsA and MMF used for oral treatment in our study were comparable to those used for systemic administration in other experimental models. Reis et al tested MMF and CsA in experimental corneal transplantation in rats but they used naive recipients without risk.28,29 In their studies, immunosuppressive drugs usually significantly prolonged corneal graft survival time, but the immunosuppressive effects were not too strong, especially in groups treated with MMF when rejection had already started during the treatment period, and most of the treated animals rejected their grafts during the 2 week treatment period. Treatment with CsA and with the combination of CsA/MMF significantly prolonged corneal graft survival in low risk rats.28 Our findings in high risk mice are different from the observations of Reis et al28 in rats or from observations of other authors in rabbits, where systemic treatment with CsA was effective in the prevention of corneal allograft rejection in naive recipients,30,31 in recipients with corneal neovascularisation induced before keratoplasty,32 or even where the recipients had heavily vascularised corneas following alkali burns.33 Although in the majority of the above studies CsA was administered intramuscularly in a dose of 25 mg/kg/day, we treated the animals orally to closely mimic the way of drug administration in clinical treatment. Unfortunately, the oral treatment with immunosuppressive drugs in high risk mice was less successful, though we used higher doses of the drugs than those administered in the clinical treatment protocols, where CsA is usually used at doses of about 5 mg/kg/day.
Although drug based immunosuppression is generally effective in high risk vascularised grafts in humans and in experimental models, the effectiveness of the treatment is much lower in high risk corneal transplantation (see above) and failed in both our models of high risk corneal transplantation in mice. This failure occurred in spite of the fact that we used the highest oral doses of CsA, which were without detectable side effects, and that we used oral administration mimicking clinical treatment. The reasons for the failure to inhibit corneal graft rejection in mice are unknown but they can involve a lower uptake of the drugs from the gastrointestinal tract of the mouse or a high risk corneal rejection may involve effector cell populations which are not so sensitive to the drug treatment as those responsible for rejection of vascularised grafts. The exact causes of the failure of the treatment remain to be determined.
Next, we used the same models of high risk mice to compare the effectiveness of treatment with mAb on corneal graft survival. Treatment with mAb anti-CD4 induced the long term survival of corneal allografts in a high proportion of prevascularised recipients. This is in agreement with previous reports that CD4+ T cells have an essential role in the rejection of corneal grafts.10,34,35 Surprisingly, treatment with the same mAb in presensitised mice failed. This suggests that different cell populations and distinct mechanisms may be involved in the rejection of corneal allografts in prevascularised and presensitised recipients. These differences may explain different effectiveness of anti-T cell therapy that we observed in our models. The production of IFN-γ by remaining CD8+ T cells and activation of graft infiltrating macrophages by IFN-γ to produce nitric oxide may be a reason for observed graft rejections in anti-CD4 treated recipients and especially in the recipients presensitised to the graft donor alloantigens.
In accordance with the recent observations that CD8+ T cells do not have a significant role in a corneal graft rejection,36,37 mAb anti-CD8 had no effect on corneal graft survival in our models either.
In conclusion, our results indicate that treatment of high risk recipients with mAb anti-CD4 is more effective in preventing corneal allograft rejection than treatment with mAb anti-CD8 or with immunosuppressive drugs MMF and CsA. However, the effectiveness of the treatment depends on the type of recipient’s risk before the transplantation.
Acknowledgments
This work was supported by grants NR/7816-3 and NI/7531-3 from the grant agency of the Ministry of Health of the Czech Republic, The Wellcome Trust grant 061347/Z/000/Z, grants 310/02/D162 and 310/03/H147 from the grant agency of the Czech Republic, and projects LN00A026 and MSM 113100003 from the Ministry of Education of the Czech Republic.
Abbreviations
CsA, cyclosporin A
mAb, monoclonal antibodies
MMF, mycophenolate mofetil
MST, mean survival time
REFERENCES
- 1.Williams KA, Muehlberg SM, Lewis RF, et al. The Australian corneal graft registry 1996 report. Adelaide: Mercury Press, 1997;11:47–50. [Google Scholar]
- 2.Williams KA, Muehlberg SM, Lewis RF, et al. Long-term outcome in corneal allotransplantation. Transplant Proc 1997;29:983. [DOI] [PubMed] [Google Scholar]
- 3.Kuffová L, Holáň V, Lumsden L, et al. Cell subpopulations in failed human corneal grafts. Br J Ophthalmol 1999;83:1364–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Larkin DF, Alexander RA, Cree IA. Infiltrating inflammatory cell phenotypes and apoptosis in rejected human corneal allografts. Eye 1997;11:68–74. [DOI] [PubMed] [Google Scholar]
- 5.Kuffová L, Lumsden L, Veselá V, et al. Kinetics of leukocyte and myeloid cell traffic in the murine corneal allograft response. Transplantation 2001;72:1292–8. [DOI] [PubMed] [Google Scholar]
- 6.Larkin DF, Calder VL, Lightman SL. Identification and characterization of cells infiltrating the graft and aqueous humour in rat corneal allograft rejection. Clin Exp Immunol 1997;107:381–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Veen GV, Broersma L, Dijkstra CD, et al. Prevention of corneal allograft rejection in rats treated with subconjunctival injections of liposomes containing dichloromethylene diphosphonate. Invest Ophthalmol Vis Sci 1994;35:3505–15. [PubMed] [Google Scholar]
- 8.Krulová M, Zajícová A, Frič J, et al. Alloantigen-induced, T-cell-dependent production of nitric oxide by macrophages infiltrating skin allografts in mice. Transplant Int 2002;15:108–16. [DOI] [PubMed] [Google Scholar]
- 9.Ayliffe W, Alam Y, Bell EB, et al. Prolongation of rat corneal graft survival by treatment with anti-CD4 monoclonal antibody. Br J Ophthalmol 1992;76:602–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.He YG, Ross J, Niederkorn JY. Promotion of murine orthotopic corneal allograft survival by systemic administration of anti-CD4 monoclonal antibody. Invest Ophthalmol Vis Sci 1991;32:2723–8. [PubMed] [Google Scholar]
- 11.Thiel MA, Takano T, Hawksworth N, et al. Low-dose, short-term treatment with anti-CD4 monoclonal antibody prolongs corneal allograft survival. Transplant Proc 2001;33:635–6. [DOI] [PubMed] [Google Scholar]
- 12.Pleyer U, Milani JK, Dukes A, et al. Effect of topically applied anti-CD4 monoclonal antibodies on orthotopic corneal allografts in a rat model. Invest Ophthalmol Vis Sci 1995;36:52–61. [PubMed] [Google Scholar]
- 13.Ippoliti G, Fronterre A. Use of locally injected anti-T monoclonal antibodies in the treatment of acute corneal graft rejection. Transplant Proc 1987;19:2579–80. [PubMed] [Google Scholar]
- 14.Ippoliti G, Fronterre A. Usefulness of CD3 or CD6 anti-T monoclonal antibodies in the treatment of acute corneal graft rejection. Transplant Proc 1989;21:3133–4. [PubMed] [Google Scholar]
- 15.Newman DK, Isaacs JD, Watson PG, et al. Prevention of immune-mediated corneal graft destruction with the anti-lymphocyte monoclonal antibody, Campath-1H. Eye 1995;9:564–9. [DOI] [PubMed] [Google Scholar]
- 16.Dick AD, Meyer P, James T, et al. Campath-1H therapy in refractory ocular inflammatory disease. Br J Ophthalmol 2000;84:107–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schmitz K, Hitzer S, Behrens-Baumann W. Immune suppression by combination therapy with basiliximab and cyclosporin in high risk keratoplasty. A pilot study. Ophthalmology 2002;99:38–45. [DOI] [PubMed] [Google Scholar]
- 18.Reis A, Reinhard T, Voiculescu A, et al. Mycophenolate mofetil versus cyclosporin A in high risk keratoplasty patients: prospectively randomised clinical trial. Br J Ophthalmol 1999;83:1268–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Reinhard T, Reis A, Bohringer D, et al. Systemic mycophenolate mofetil in comparison with systemic cyclosporin A in high-risk keratoplasty patients: 3 years’ results of a randomized prospective clinical trial. Graefes Arch Clin Exp Ophthalmol 2001;239:367–72. [DOI] [PubMed] [Google Scholar]
- 20.Soares-Wulf A, Aboalchamat B, Krüger R, et al. Long-term results of immunosuppressive therapy with mycophenolate mofetil (CellCept) for keratoplasty in high-risk patients. Invest Ophthalmol Vis Sci 2002;43 E-Abstract 2241.
- 21.Poon AC, Forbes JE, Dart JKG, et al. Systemic cyclosporin A in high risk penetrating keratoplasties: a case-control study. Br J Ophthalmol 2001;85:1464–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.She SC, Steahly LP, Moticka EJ. A method for performing full-thickness, orthotopic, penetrating keratoplasty in the mouse. Ophthalmic Surg 1990;21:781–5. [PubMed] [Google Scholar]
- 23.Plšková J, Duncan L, Holáň V, et al. The immune response to corneal allograft requires a site-specific draining lymph node. Transplantation 2002;73:210–15. [DOI] [PubMed] [Google Scholar]
- 24.Plšková J, Kuffová L, Holáň V, et al. Evaluation of corneal graft rejection in a mouse model. Br J Ophthalmol 2002;86:108–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lake P, Clark EA, Khorshidi M, et al. Production and characterization of cytotoxic Thy-1 antibody-secreting hybrid cell lines. Detection of T cell subsets. Eur J Immunol 1979;9:875–86. [DOI] [PubMed] [Google Scholar]
- 26.Dialynas DP, Wilde DB, Marrack P, et al. Characterization of the murine antigenic determinant designated L3T4a, recognized by monoclonal antibody GK1.5: expression of L3T4a by functional T-cell clones appears to correlate primarily with class II MHC antigen-reactivity. Immunol Rev 1983;74:29–56. [DOI] [PubMed] [Google Scholar]
- 27.Gottlieb PD, Marshak-Rothstein A, Auditore-Hargreaves K, et al. Construction and properties of new Lyt-congeneic strains and anti-Lyt-2.2 and anti-Lyt-3.1 monoclonal antibodies. Immunogenetics 1980;10:545–52. [DOI] [PubMed] [Google Scholar]
- 28.Reis A, Reinhard T, Sundmacher R, et al. Effect of mycophenolate mofetil, cyclosporin A, and both in combination in a murine corneal graft rejection model. Br J Ophthalmol 1998;82:700–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reis A, Megahed M, Reinhard T, et al. Coadministration of the new macrolide immunosuppressant RAD and mycophenolate mofetil in experimental corneal transplantation. Transplantation 2000;70:1397–407. [DOI] [PubMed] [Google Scholar]
- 30.Shepherd WF, Coster DJ, Fook TC, et al. Effect of cyclosporine A on the survival of corneal grafts in rabbits. Br J Ophthalmol 1980;64:148–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Coster DJ, Shepard WFI, Chin Fook T, et al. Prolonged survival of corneal allografts in rabbits treated with cyclosporine A. Lancet 1979;2:688–9. [DOI] [PubMed] [Google Scholar]
- 32.Hill JC, Maske R. An animal model for corneal graft rejection in high risk keratoplasty. Transplantation 1988;46:26–30. [DOI] [PubMed] [Google Scholar]
- 33.Rehany U, Waisman M. Suppression of corneal allogtaft rejection by systemic cyclosporine-A in heavily vascularized rabbit corneas following alkali burns. Cornea 1994;13:447–53. [DOI] [PubMed] [Google Scholar]
- 34.Yamada J, Kurimoto I, Streilein W. Role of CD4+ T cells in immunobiology of orthotopic corneal transplants in mice. Invest Ophthalmol Vis Sci 1999;40:2614–21. [PubMed] [Google Scholar]
- 35.Boisgérault F, Liu Y, Anosova N, et al. Role of CD4+ and CD8+ T cells in allorecognition: Lessons from corneal transplantation. J Immunol 2001;167:1891–9. [DOI] [PubMed] [Google Scholar]
- 36.Hedge S, Niederkorn JY. The role of cytotoxic T lymphocytes in corneal allograft rejection. Invest Ophthalmol Vis Sci 2000;41:3341–7. [PubMed] [Google Scholar]
- 37.Yamada J, Ksander BR, Streilein JW. Cytotoxic T cells play no essential role in acute rejection of orthotopic corneal allografts in mice. Invest Ophthalmol Vis Sci 2001;42:386–92. [PubMed] [Google Scholar]