Abstract
Background: Chronic pancreatitis is characterised clinically by early exocrine insufficiency, with diabetes mellitus occurring as a late phenomenon. This is mirrored pathologically by extensive acinar cell destruction and islet preservation. The mechanisms underlying this differential rate of cellular destruction are unknown.
Aims: To test the hypothesis that acinar loss and islet preservation in chronic pancreatitis occurs due to differential epithelial kinetics and investigate the role of inflammatory cells and cell cycle associated molecules.
Methods: Archival tissue from six chronic pancreatitis cases was compared with six normal controls using TUNEL and immunohistochemistry for CD3, CD20, CD68, MIB-1, Bcl-2, Bax, Fas, Fas ligand, retinoblastoma protein (Rb), and tissue inhibitor of metalloproteinases 1 (TIMP-1) and 2 (TIMP-2).
Results: The acinar cell apoptotic index (AI) and proliferation index were higher in chronic pancreatitis than controls. T lymphocytes diffusely infiltrated fibrous bands and acini but rarely islets. Acinar Bcl-2 expression exceeded islet expression in chronic pancreatitis and controls while Bax was strongly expressed by a subset of islet cells and weakly by centroacinar cells. Islet Fas and Fas ligand expression exceeded acinar expression in chronic pancreatitis and controls. Acinar Rb expression was higher in chronic pancreatitis than in controls. Islets in chronic pancreatitis and controls showed intense TIMP-1 and TIMP-2 expression.
Conclusion: Apoptosis plays a significant role in acinar loss in chronic pancreatitis. Acinar Bcl-2 and islet Bax expression indicates complex AI control. Increased acinar Rb expression in chronic pancreatitis may differentially promote acinar loss. Fas ligand expression may be restricted to islet cell membranes through TIMP-1 expression and inhibit islet damage by promoting apoptosis of cytotoxic T lymphocytes.
Keywords: chronic pancreatitis, apoptosis, proliferation, cell cycle, Fas/Fas ligand
Chronic pancreatitis is defined, by the Marseille-Rome classification of 1988, as “a continuing inflammatory disease of the pancreas characterised by the irreversible destruction of exocrine parenchyma and fibrosis and at least in the later stages, the destruction of the endocrine parenchyma”.1 Clinically, this condition typically causes pain and after some years there is exocrine insufficiency, manifest as steatorrhoea. Endocrine insufficiency, due to the loss of pancreatic islet cells, eventually occurs in 60% of patients with chronic pancreatitis but is a late manifestation of the disease.2
The aetiology of chronic pancreatitis is variable but the majority of cases in western countries are associated with a long history of high alcohol intake. The mechanism by which high alcohol intake and other aetiological factors lead to chronic pancreatitis is not clear but two theories have been proposed. The duct obstruction hypothesis of Sarles et al is based on the observation of protein precipitates within interlobular ducts, associated with a patchy lobular pattern of acinar cell loss and inflammatory infiltrate.1 The necrosis/fibrosis hypothesis of Kloppel and Maillet seeks to explain the acinar cell loss and associated fibrosis by postulating localised acute pancreatitis and subsequent necrosis of acinar cells, leading to proliferation of fibroblasts.3
The histopathological features of chronic pancreatitis include dilated pancreatic ducts, variable chronic inflammatory cell infiltration, acinar cell loss, and replacement of the functional parenchyma with fibrosis.4 However, islet cells are often well preserved despite extensive acinar cell damage.
Apoptosis or programmed cell death was first described by Kerr et al in 19725 and is now a well recognised process by which individual cells are lost in normal and diseased tissues. There are characteristic morphological features of apoptotic cell death, including cell shrinkage, nuclear chromatin condensation and margination, and nuclear fragmentation. Finally, the cell separates into discrete membrane bound bodies known as apoptotic bodies.5 Within the gastrointestinal tract, epithelial and mesenchymal cells of the liver and epithelial cells of the small and large intestine and pancreas are now known to undergo cell death by the process of apoptosis.6–8 In the rat model, a copper deficient diet, ethionine administration, and pancreatic duct ligation have all led to pancreatic acinar cell apoptosis resulting in pancreatic atrophy.9–11 In a model more pertinent to human chronic pancreatitis, acinar cell atrophy occurred in rodents fed ethanol and a low protein diet.12 Islet cells have also been shown to undergo apoptosis during their destruction in the non-obese diabetic mouse.13 Acinar cell loss may occur due to apoptosis.11 However, as yet the mechanism for the loss of exocrine parenchyma and the relative preservation of endocrine epithelial cells in chronic pancreatitis is unclear.
In this study, we hypothesised that the loss of pancreatic acinar cells in human chronic pancreatitis is associated with an increased apoptotic index within this cellular compartment, compared with both islet cells and acinar cells in the normal pancreas. We then investigated the role of inflammatory cells and cell cycle influencing molecules in the control of pancreatic epithelial cell kinetics.
MATERIALS AND METHODS
Selection of cases and staining
Formalin fixed and paraffin wax embedded tissue blocks from six cases of chronic pancreatitis and six normal controls were retrieved from the histopathology database at Southampton General Hospital and haematoxylin and eosin (H&E) stained sections were prepared from each block. Of the cases of chronic pancreatitis, three specimens were resections (one proximal, two distal) performed for painful alcohol related chronic pancreatitis. One specimen was an open pancreatic biopsy of a nodular pancreas found at cholecystectomy. One was a proximal resection of a symptomatic pancreatic mass (idiopathic chronic pancreatitis) and one was from an area of obstructive chronic pancreatitis above a pancreatic adenocarcinoma. The normal tissue was obtained from two Whipples' resections for duodenal adenocarcinoma and an adenocorticotrophic hormone secreting neuroendocrine tumour of the pancreas. Three specimens were derived from patients undergoing resection of a gastro-oesophageal adenocarcinoma and in which the tumour extended close to, or focally into, the pancreas. One sample was an open biopsy for a suspicious nodule in the pancreas found at cholecystectomy. In each of these six cases there was no evidence, in the blocks chosen for the study, of tumour involvement, pancreatic duct dilatation, or any other evidence of chronic pancreatitis.
Labelling of apoptotic cells
A paraffin section (5 μm) from each case under investigation was examined for the presence of fragmented DNA in apoptotic cells by the terminal deoxynucleotidyl transferase UTP nick end labelling (TUNEL) technique, using the Roche TUNEL staining kit (Roche, Lewes, UK), according to the manufacturer's instructions (modified from that described by Gavrieli and colleagues14). Briefly, after dewaxing and two five minute washes in sterile water, DNAse solution was applied to the positive and negative control slides for one hour at room temperature. These control sections underwent two further five minute rinses in sterile water. Proteinase K solution (Roche) was then applied to all sections, together with a coverslip, and incubated for 15 minutes at room temperature. Specimens were washed twice in Tris buffered saline at this stage and between each subsequent stage. The TUNEL solution (Roche TUNEL kit; fluorescein labelled nucleotides and terminal deoxynucleotidyl transferase) was applied to all sections except the negative controls, a coverslip was applied, and the sections were incubated for one hour at 37°C. Endogenous peroxide blocker (59 μl fresh 30% H2O2 in 5.9 ml analar methanol) was applied for 15 minutes at room temperature after which converter solution (Roche TUNEL kit; antifluorescein antibody conjugated with horseradish peroxidase) was applied for 30 minutes at 37°C (100 μl per section). Finally, diaminobenzidine (DAB) solution (50 μl of 15% sodium azide solution per 2.5 ml DAB; Menarini, Wokingham, UK) was applied for five minutes. After a two minute wash in running tap water and a rinse in 70% alcohol, the sections were counterstained with Harris's haematoxylin for 1–2 minutes, rinsed for one minute in running tap water, differentiated in 1% acid alcohol for five seconds, and blued in running tap water for five minutes before dehydration and coverslip mounting.
Immunohistochemistry
Tissue sections (5 μm) from each case under investigation underwent immunohistochemical staining with the proliferation marker MIB-1 (Immunotech, Marseille, France) and primary antibodies to CD3 (monoclonal) (Vector, Peterborough, UK), CD20 (L26) (Dako, Ely, UK), CD68 (PGM-1) (Dako), Bcl-2 (Dako), Bax (Biogenex (A Menarini Diagnostics), Finchampstead, UK), Fas (Calbiochem-Oncogene Research Products, Cambridge, UK), Fas ligand (Santacruz (AutoGenBioclear), Calne, UK), retinoblastoma protein (Biogenex (A Menarini Diagnostics), Finchampstead, UK), tissue inhibitor of metalloproteinase 1 (TIMP-1; Chemicon International Inc., Temecula, California, USA), and tissue inhibitor of metalloproteinase 2 (TIMP-2; Chemicon International Inc.). The pretreatment regimens and primary antibody dilutions are given in table 1 ▶. Microwave and pressure cooker treatment were performed using a 0.01 M citrate buffer (pH 6.0) for 25 minutes at medium power (microwave) or two minutes at 15 psi (pressure cooker).
Table 1.
Pretreatment regimens and primary antibody concentrations for immunohistochemistry
| Primary antibody | Pretreatment | Dilution |
| CD3 (monoclonal) | Pressure cooker | 1:200 |
| CD20 (L26) | No treatment | 1:1000 |
| CD68 (PGM-1) | Microwave | 1:200 |
| MIB-1 | Microwave | 1:400 |
| Bcl-2 | Pressure cooker | 1:200 |
| Bax | None | 1:50 |
| Fas | Microwave | 1:50 |
| Fas ligand | Pressure cooker | 1:80 |
| Retinoblastoma protein | Microwave | 1:200 |
| TIMP-1 | Pronase | 1:200 |
| TIMP-2 | Pronase | 1:400 |
TIMP, tissue inhibitor of metalloproteinase.
Characterisation of inflammatory cell infiltrate
Descriptive analysis was used to characterise the distribution of B lymphocytes (CD20 positive) and T lymphocytes (CD3 positive) in normal pancreas and chronic pancreatitis. Fully quantitative analysis was used to identify the number of T lymphocytes, identified using immunohistochemistry for monoclonal CD3, infiltrating acinar and islet epithelium. This comprised formal counting of the number of CD3 positive cells present within 20 random high power fields of acinar or islet cells (area of each field 0.07 mm2). The results for each case were expressed as the total number of CD3 cells/mm2 of acinar or islet tissue.
Assessment of apoptotic index
The number of apoptotic bodies was determined and expressed relative to the total number of cells within 20 random high power fields of acinar or islet cells (area of each field 0.07 mm2). Strict criteria were used to define a cell as apoptotic on the H&E and TUNEL stained sections. These were: the presence of a condensed often fragmented nucleus together with a surrounding halo. Cells containing weakly to moderately TUNEL positive nuclei in the absence of these additional morphological features were not assessed as apoptotic. The apoptotic index (AI) was calculated as the percentage of acinar cell apoptotic bodies per total number of acinar cells counted for each case. A similar index was calculated for islet cells.
Assessment of proliferation index
For assessment of the proportion of epithelial cells within the active portion of the cell cycle, the same counting method was used to determine the number of MIB-1 positive cells per high power field. A proliferation index (PI) was likewise calculated for MIB-1 positive cells in each case, in the same way as for AI.
Assessment of Bcl-2, Bax, Fas ligand, and retinoblastoma protein labelling
Descriptive analysis was used to assess the distribution of labelling achieved for Bcl-2, Bax, Fas, and Fas ligand. Fully quantitative analysis identical to that used for measurement of AI and PI was performed for retinoblastoma protein.
Statistics
All statistical analyses were performed using SPSS for Windows version 10.0. Outcome measures were summarised using median (range). Comparisons between groups were investigated using the Mann-Whitney U test while comparisons within groups were examined using the Wilcoxon signed rank test. As the number of tests was large, p<0.01 was taken to represent a statistically significant result.
RESULTS
All cases of chronic pancreatitis in the study showed typical histological features, with intra-acinar and periacinar patterns of fibrosis associated with destruction of exocrine epithelial glandular tissue and often surrounding the surviving islets. All of the control sections showed a normal histological appearance, with no evidence of tumour infiltration, inflammation, or fibrosis.
Distribution of inflammatory cells in chronic pancreatitis
The acinar cell destruction in chronic pancreatitis was paralleled by a chronic inflammatory cell infiltrate comprising lymphocytes and macrophages. The lymphocytic infiltrate comprised both aggregates of CD20 cells (B lymphocytes) surrounded by CD3 cells (T lymphocytes) and a diffuse CD3 cell infiltrate of variable density. This diffuse infiltrate was present within the periacinar and intra-acinar fibrous tissue and was also intimately associated with acinar epithelium. The CD3 cell counts for all cases and controls are listed in table 2 ▶. Normal pancreatic tissue contained small numbers of CD3 cells within acinar tissue but only an occasional CD3 cell within islets. In chronic pancreatitis there was an increase in CD3 cell numbers within acinar tissue (p=0.002) but a statistically significant increase was not found in CD3 cell numbers within islets (p=0.394).
Table 2.
CD3 cell numbers within acinar cells and islet cells, stained with CD3 immunohistochemistry
| CD3 cell count/mm2 | ||
| Case No | Acinar tissue | Islet tissue |
| CP 1 | 183 | 2 |
| CP 2 | 58 | 0 |
| CP 3 | 40 | 1 |
| CP 4 | 56 | 3 |
| CP 5 | 41 | 0 |
| CP 6 | 190 | 1 |
| Median (range) | 57 (40–190) | 1 (0–3) |
| N 1 | 15 | 0 |
| N 2 | 5 | 0 |
| N 3 | 21 | 1 |
| N 4 | 11 | 0 |
| N 5 | 20 | 1 |
| N 6 | 19 | 1 |
| Median (range) | 17 (5–21) | 0.5 (0–1) |
CP, chronic pancreatitis; N, normal.
Apoptotic and proliferation indices in pancreatic epithelial cells
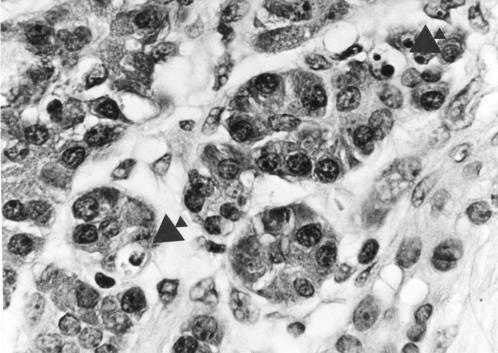
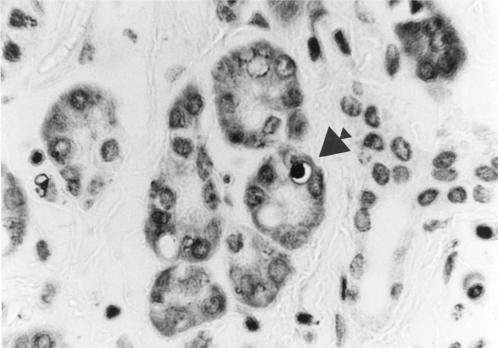
The positive control sections used for the TUNEL method exhibited widespread nuclear TUNEL labelling, including labelling of islet cell nuclei. AI and PI values for all cases and controls are shown in tables 3 and 4 ▶ ▶. Assessment of AI using both H&E (fig 1 ▶) and TUNEL (fig 2 ▶) staining yielded similar results. Therefore, for clarity, comparisons and probability values given here relate to the results obtained with the H&E method. In chronic pancreatitis the AI within acinar cells was increased compared with controls (p=0.002) but a statistically significant increase in AI was not found within islet cells (p=0.065).
Table 3.
Apoptotic index within acinar cells and islet cells, stained with haematoxylin and eosin (H&E) and the TUNEL method
| Apoptotic index (AI) (%) | ||||
| Case No | Acini AI (H&E) | Islets AI (H&E) | Acini AI (TUNEL) | Islets AI (TUNEL) |
| CP 1 | 0.18 | 0.048 | 0.26 | 0 |
| CP 2 | 0.17 | 0 | 0.091 | 0 |
| CP 3 | 0.28 | 0.14 | 0.20 | 0 |
| CP 4 | 0.24 | 0 | 0.13 | 0 |
| CP 5 | 0.44 | 0.11 | 0.21 | 0 |
| CP 6 | 0.21 | 0.075 | 0.17 | 0 |
| Median (range) | 0.23 (0.17–0.44) | 0.062 (0–0.14) | 0.19 (0.091–0.26) | 0 |
| N 1 | 0.025 | 0 | 0 | 0 |
| N 2 | 0.041 | 0 | 0.022 | 0 |
| N 3 | 0 | 0 | 0 | 0 |
| N 4 | 0 | 0 | 0.024 | 0 |
| N 5 | 0.027 | 0 | 0 | 0 |
| N 6 | 0.020 | 0 | 0 | 0 |
| Median (range) | 0.023 (0–0.041) | 0 | 0 (0–0.024) | 0 |
CP, chronic pancreatitis; N, normal.
Table 4.
Proliferation index within acinar cells and islet cells, stained with MIB-1 immunohistochemistry
| Proliferation index (PI) (%) | ||
| Case No | Acini PI | Islets PI |
| CP 1 | 0.58 | 0.15 |
| CP 2 | 2.6 | 0.044 |
| CP 3 | 2.5 | 0.11 |
| CP 4 | 1.5 | 0.20 |
| CP 5 | 2.4 | 0.20 |
| CP 6 | 1.9 | 0.21 |
| Median (range) | 2.2 (0.58–2.6) | 0.18 (0.044–0.21) |
| N 1 | 0.28 | 0 |
| N 2 | 0.13 | 0.15 |
| N 3 | 0.10 | 0 |
| N 4 | 0.86 | 0.071 |
| N 5 | 0.44 | 0 |
| N 6 | 0.22 | 0.086 |
| Median (range) | 0.25 (0.10–0.86) | 0.036 (0–0.15) |
CP, chronic pancreatitis; N, normal.
Figure 1.
Haematoxylin and eosin stained section of pancreas (chronic pancreatitis) showing two apoptotic bodies (arrowed) within acinar epithelial cells. Magnification ×500.
Figure 2.
Section of pancreas (chronic pancreatitis) stained using the TUNEL technique showing darkly labelled nuclear material within an apoptotic acinar epithelial cell (arrowed). Magnification ×500.
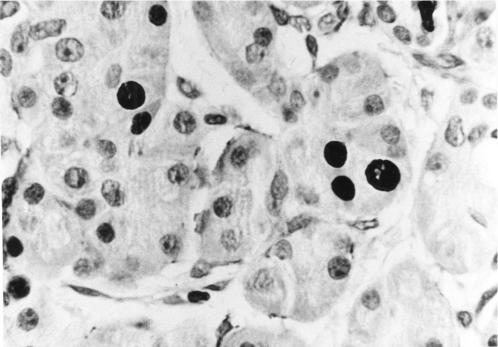
A basal level of cell proliferation, as assessed via the PI, was observed within both acinar and islet cells in controls (fig 3 ▶). In chronic pancreatitis PI was increased within acinar cells (p=0.004) compared with controls. Statistically significant differences in PI were not found within islet cells in chronic pancreatitis compared with controls (p=0.026) or between islet and acinar cells either in chronic pancreatitis (p=0.031) or controls (p=0.22).
Figure 3.
Section of pancreas (chronic pancreatitis) stained using MIB-1 immunohistochemistry showing several positively labelled nuclei within acinar epithelial cells. Magnification ×500.
Expression of cell cycle modulating and related molecules
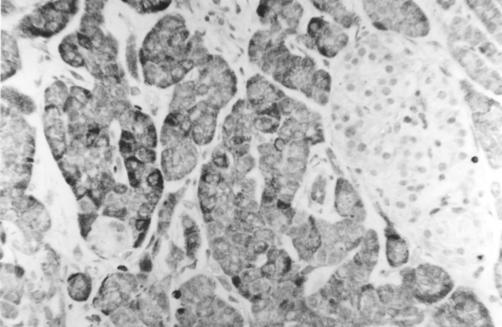
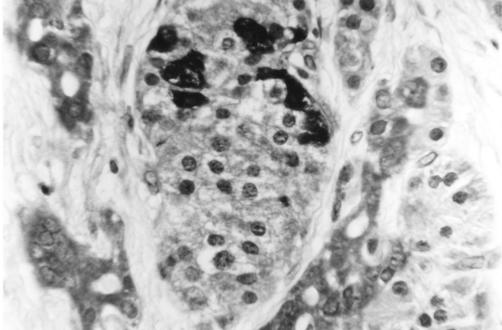
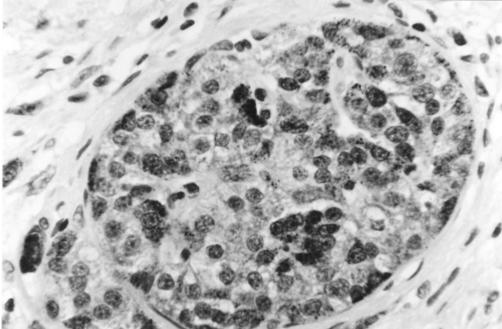
Bcl-2 was widely and strongly expressed within acinar cell cytoplasm but only weakly within islet cell cytoplasm, in both chronic pancreatitis and controls (fig 4 ▶). Bax labelling showed the opposite pattern, again within both chronic pancreatitis and controls, with a subset of islet cells showing intense cytoplasmic expression (fig 5 ▶). The outer secretory cells of the individual acini showed little or no Bax expression. However, Bax was expressed weakly within the cytoplasm of centroacinar cells (the small ductular cells at the centre of each individual acinus and continuous with the exocrine duct system) (fig 5 ▶).
Figure 4.
Section of pancreas (normal) stained using immunohistochemistry for Bcl-2 protein, showing cytoplasmic Bcl-2 expression within acinar cells but not within islets. Magnification ×250.
Figure 5.
Section of pancreas (chronic pancreatitis) stained using immunohistochemistry for Bax protein, showing intense cytoplasmic Bax expression within a subset of islet cells as well as within centroacinar cells (on either side of the islet). Labelling of the latter cells is accentuated within this section of chronic pancreatitis since many of the outer acinar cells have been destroyed. Magnification ×500.
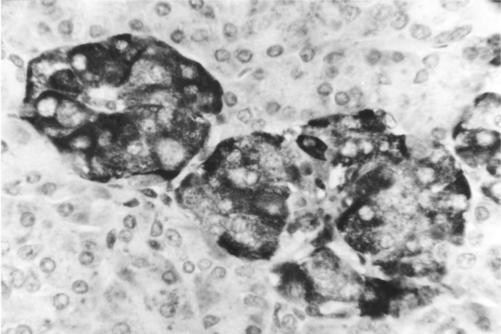
Fas was expressed within the cytoplasm of a subset of islet cells in both chronic pancreatitis and controls, within the cytoplasm of neutrophils, and the cytoplasm of a subset of lymphocytes. Within some islets, Fas expression was maximal at the periphery of the islet (fig 6 ▶). Fas ligand was expressed very strongly within islet cell cytoplasm in both chronic pancreatitis and controls but generally only weakly by acinar cells (fig 7 ▶). Fas ligand was also expressed strongly within the cytoplasm of the vast majority of lymphocytes.
Figure 6.
Section of pancreas (chronic pancreatitis) stained using immunohistochemistry for Fas protein, showing cytoplasmic Fas expression within islet cells. Magnification ×500.
Figure 7.
Section of pancreas (normal) stained using immunohistochemistry for Fas ligand protein, showing intense cytoplasmic Fas ligand expression within islet cells. Magnification ×500.
The retinoblastoma labelling index for all cases and controls is shown in table 5 ▶. Retinoblastoma protein was expressed within the nucleus of a small proportion of acinar and islet cells in both chronic pancreatitis and controls. No statistically significant difference in retinoblastoma labelling index was observed between islet and acinar cells in chronic pancreatitis (p=0.69) or controls (p=0.69). An increase in retinoblastoma labelling index was found within acinar cells compared with controls in chronic pancreatitis (p=0.004) but a statistically significant increase was not observed within islet cells (p=0.41).
Table 5.
Retinoblastoma labelling index within acinar cells and islet cells, stained with retinoblastoma protein immunohistochemistry
| Retinoblastoma labelling index (%) | ||
| Case No | Acinar tissue | Islet tissue |
| CP 1 | 0.84 | 0.39 |
| CP 2 | 2.1 | 1.3 |
| CP 3 | 0.61 | 0.70 |
| CP 4 | 1.2 | 2.2 |
| CP 5 | 1.6 | 1.5 |
| CP 6 | 2.3 | 1.1 |
| Median (range) | 1.4 (0.61–2.3) | 1.2 (0.39–2.2) |
| N 1 | 0.46 | 0.67 |
| N 2 | 0.083 | 0.13 |
| N 3 | 0.098 | 0.57 |
| N 4 | 0.18 | 0.77 |
| N 5 | 0.026 | 0.0 |
| N 6 | 0.73 | 0.61 |
| Median (range) | 0.14 (0.026–0.73) | 0.59 (0–0.77) |
CP, chronic pancreatitis; N, normal.
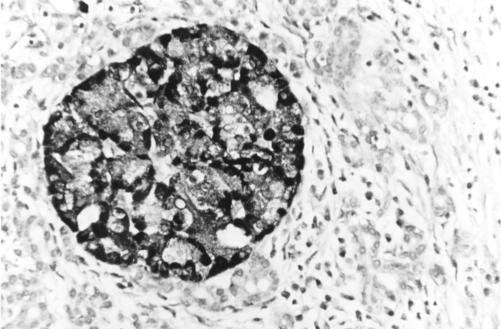
TIMP-1 and TIMP-2 were both strongly expressed within islet cell cytoplasm in chronic pancreatitis and controls (fig 8 ▶).
Figure 8.
Section of pancreas (chronic pancreatitis) stained using immunohistochemistry for tissue inhibitor of metalloproteinase (TIMP)-1 protein, showing intense cytoplasmic TIMP-1 expression within islet cells. A very similar expression pattern was identified for TIMP-2. Magnification ×250.
DISCUSSION
We have shown that in chronic pancreatitis there is a significantly higher AI and PI within acinar cells compared with this cellular compartment in the normal pancreas.
The results of our study suggest that epithelial cell turnover is increased in chronic pancreatitis. Indices of cellular apoptosis and proliferation, as measured solely by counting cells within tissue sections, cannot be translated directly into rates of cell death and division. These methods provide only a “snapshot” of proliferative and apoptotic activity within the tissue at the time of biopsy or resection. Nevertheless, these indices act as a guide to the degree of cellular proliferation and apoptosis within a tissue. Indeed, because individual apoptotic events can take as little as 30 minutes, modest changes in AI (for example, from 1% to 2%) in the presence of constant proliferation are associated with dramatic changes in overall cell numbers in 24–48 hours.15
Our data are consistent with a model in which, in chronic pancreatitis, an imbalance occurs between the processes of apoptosis and proliferation. As a result, cell proliferation, although increased in comparison with the normal pancreas, is insufficient to prevent net exocrine cell loss.
The increase in AI within acinar cells but not islet cells observed in chronic pancreatitis is consistent with the clinical observation of preservation of endocrine function until late in the disease process and with the concept of chronic pancreatitis as a disease of disordered function of, or injury to, acinar cells. These differences are paralleled by histological evidence of acinar cell destruction and islet cell preservation in chronic pancreatitis.
The cases of normal pancreas and chronic pancreatitis were randomly selected from the pathology files. Pancreatic resection for chronic pancreatitis in the absence of pancreatic neoplasia is unusual and tends to be reserved for patients with intractable pain. However, our tissue archive contains cases such as this together with examples of less severe or focal disease identified incidentally or during the investigation of suspected pancreatic neoplasia. Our archive also includes chronic pancreatitis associated with pancreatic tumours. The cases of chronic pancreatitis used in this study therefore represented a spectrum of clinical presentation, from an incidental pancreatic nodule identified at laparotomy to pancreatic resections for severe painful chronic pancreatitis and a case of chronic pancreatitis distant to but associated with a pancreatic tumour. We believe that these cases were therefore representative of the patient population with chronic pancreatitis, including examples of both asymptomatic and clinically severe disease. Surgical specimens comprising normal pancreatic tissue are also unusual but most of the cases in this study were derived from partial pancreatectomies for gastric or duodenal tumours (in which tumour extended close to but not into the pancreas) together with one small and non-obstructing pancreatic neuroendocrine tumour. None of these cases showed histopathological evidence of chronic pancreatitis and were therefore entirely representative of the normal population.
We used two techniques (H&E staining and in situ end labelling) to identify cells undergoing apoptosis as it is believed that it may be difficult to identify all apoptotic cells within a tissue section using light microscopy and morphology alone. The “gold standard” is recognition of the morphological changes of apoptosis by electron microscopy.16 However, when the AI of a cell population is low (that is, only 1–2% of the total number of cells), it is impractical to use electron microscopy as a method of measuring this index. In situ end labelling techniques such as the TUNEL method highlight cleaved DNA within apoptotic bodies although false negative and false positive labelling can occur and therefore we only assessed TUNEL positive cells as apoptotic if they also showed the morphological features of apoptosis. Using these methods we obtained consistent and reproducible results. Moreover, the results generated with each individual technique (that is, TUNEL and H&E) gave comparable data. We used immunohistochemistry with MIB-1 as the primary antibody to determine relative indices of cellular proliferation in different cell compartments. The MIB-1 antibody identifies the Ki67 epitope, expressed by proliferating cells in the active portion of the cell cycle. Staining with this antibody gave consistent results and nuclear labelling which was easy to interpret. Identification and quantification of cell numbers defined as proliferating or apoptotic was determined by a single observer (SMT). To ensure that our data were accurate and not skewed by geographical variation in cell proliferation and apoptosis (as might occur adjacent to a focus of inflammation, for example), each tissue section was examined “blind” and quantification of proliferating and apoptotic cells was determined over 20 random high power fields.
We understand that the small sample sizes used in this study mean that future work should be performed to confirm our findings. However, we believe that our samples are representative of the diseased and normal populations and that the striking differences in acinar cell apoptotic and proliferation index between normal and disease states, statistically significant even at the 1% level, suggest that important changes in epithelial cell kinetics occur during the pathogenesis of chronic pancreatitis.
To investigate the processes underlying our observations of pancreatic epithelial cell turnover, we defined the pancreatic expression characteristics of a range of molecules that are each known to influence control of the cell cycle. Members of the Bcl-2 family interact as homodimers and heterodimers either promoting or inhibiting apoptosis.17 Bcl-2 classically inhibits apoptosis18 and overexpression of Bcl-2 occurs in a range of neoplasms, including follicular lymphoma.19 Bax, another member of the Bcl-2 family, promotes or facilitates apoptosis.20 From these characteristics we predicted a model of chronic pancreatitis in which antiapoptotic Bcl-2 was expressed by islets and proapoptotic Bax by acinar epithelium. Paradoxically, we found that Bcl-2 was expressed more strongly by acinar epithelium than islet cells while Bax expression showed the opposite pattern, with strong expression occurring within a subset of islet cells but only weakly within acinar epithelium, and in particular, centroacinar cells. These expression patterns were similar within both normal pancreas and chronic pancreatitis. Taken together, our data indicate that complex interactions between Bcl-2 family molecules must be occurring within both normal pancreas and chronic pancreatitis, possibly via heterodimer formation between Bcl-2, Bax, and other Bcl-2 family members.21
In our study, islet cells in both normal pancreas and chronic pancreatitis strongly expressed Fas and Fas ligand. Expression of both molecules has been demonstrated within other cell types, including the testicular Leydig cell,22 natural killer cells,23 and colonic intraepithelial T lymphocytes.24 Fas/Fas ligand binding usually induces apoptosis within the Fas expressing cells.25 This suggests that pancreatic islet cells must possess an additional mechanism for inhibiting “self-induced” Fas/Fas ligand triggered apoptosis. Current evidence suggests that the extracellular domain of the Fas ligand molecule is cleaved from expressing cells by a metalloproteinase dependent process to yield a product that is released from the membrane. Once liberated, Fas ligand has the propensity to induce apoptosis in any Fas expressing cell.26 The metalloproteinase dependence of the release process is of particular interest in the context of the islets because they strongly express TIMP-1 and TIMP-2. We propose that expression of these powerful metalloproteinase inhibitors provides a mechanism which inhibits autoinduction of apoptosis in islets. This hypothesis is supported by the recent observation that TIMP-1 inhibits cytokine induced apoptosis in a rat islet cell culture model.27
Membrane bound Fas ligand expression by islet cells may provide a defence mechanism for islets against immune mediated attack in chronic pancreatitis via induction of apoptosis in islet infiltrating T lymphocytes. This hypothesis is supported by our observation that T lymphocyte infiltrates in chronic pancreatitis are widespread within acinar tissue but rare within islets. Fas/Fas ligand induced apoptosis is a known mechanism for modulation of the immune response within both health and disease. For example, Fas ligand expression is a characteristic feature of immunological “sanctuary site” tissues such as retinal pigment epithelium, acting via induction of T lymphocyte apoptosis.28 Furthermore, Fas ligand expression by tumour cells (for example, breast cancer) may lead to Fas mediated T lymphocyte apoptosis and therefore protect developing tumours from T lymphocyte induced killing.29
We found that islet cell apoptosis occurred at a relatively low level in chronic pancreatitis despite Fas and Fas ligand expression by islet cells and T lymphocytes, respectively. Fas/Fas ligand mediated islet cell destruction by Fas ligand bearing cytotoxic T lymphocytes is believed to represent an important mechanism in the development of autoimmune diabetes mellitus30 but does not occur to the same extent in chronic pancreatitis. The reason for this increase in islet cell susceptibility in autoimmune diabetes mellitus is unclear but may relate to a cytokine (for example, tumour necrosis factor α, interferon γ) and nitric oxide mediated increase in islet cell Fas expression and the presence of increased numbers of islet cell specific activated cytotoxic T lymphocytes in this condition.31,32
Retinoblastoma protein was observed at a low level in normal pancreas but at increased levels within acinar cells in chronic pancreatitis. Retinoblastoma protein interacts with p53 and MDM-2, controlling progression of the cell cycle, mainly by binding to and inactivation of the E2F transcription factors.33 Under certain forms of cellular stress retinoblastoma protein promotes apoptosis.34 p53 induced apoptosis is usually inhibited by MDM-2 binding to p53. However, binding of retinoblastoma protein to MDM-2 may abrogate the inhibitory effect of MDM-2 on p53 and thereby allow apoptosis to occur even in the presence of MDM-2.34 Therefore, our results suggest that retinoblastoma protein may act as a promoter of acinar cell apoptosis under the cellular stress induced during the development and progression of chronic pancreatitis.
In summary, our study suggests that differential indices of apoptosis within acinar and islet epithelium in chronic pancreatitis may play a significant role in determining the histological and clinical pattern of disease progression. Fas ligand expression by islets may affect the relative distribution of T lymphocytes within acini and islets and thereby act as a survival factor for islet cells. Retinoblastoma protein overexpression may represent a further mechanism of apoptosis induction within acinar cells in chronic pancreatitis. By understanding the processes regulating the pathogenesis of chronic pancreatitis at a molecular level we may ultimately identify a potential avenue for future therapy.
Acknowledgments
We thank Lorraine Low for her statistical advice during the preparation of this manuscript.
Abbreviations
H&E, haematoxylin and eosin
TUNEL, terminal deoxynucleotidyl transferase UTP nick end labelling
DAB, diaminobenzidine
AI, apoptotic index
PI, proliferation index
TIMP, tissue inhibitor of metalloproteinase
REFERENCES
- 1.Sarles H, Bernard JP, Gullo L. Pathogenesis of chronic pancreatitis. Gut 1990;31:629–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Di Magno EP, Holtmann G. Chronic pancreatitis. Curr Opin Gastroenterol 1992;8:824–9. [Google Scholar]
- 3.Kloppel G, Maillet B. Chronic pancreatitis: evolution of the disease. Hepatogastroenterology 1991;8:408–12. [PubMed] [Google Scholar]
- 4.Matthias PA, Ebert MD, Ademmer MD, et al. CD8+ CD103+ T-cells analogous to intestinal intraepithelial lymphocytes infiltrate the pancreas in chronic pancreatitis. Am J Gastroenterol 1998;93:2141–7. [DOI] [PubMed] [Google Scholar]
- 5.Kerr JFR, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972;26:239–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Armends MJ, Wyllie AH. Apoptosis: mechanisms and roles in pathology. Int Rev Exp Pathol 1991;32:223–54. [DOI] [PubMed] [Google Scholar]
- 7.Shibihara T, Sato N, Waguri S, et al. The fate of effete epithelial cells at the villus tip of the human intestine. Arch Histol Cytol 1995;58:205–19. [DOI] [PubMed] [Google Scholar]
- 8.Iredale JP, Benyon RC, Pickering J, et al. Mechanisms of spontaneous resolution of rat liver fibrosis. J Clin Invest 1998;10:538–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kishimoto S, Iwamoto S, Matstani R, et al. Apoptosis of acinar cells in the pancreas of rats fed on a copper-deficient diet. Exp Toxicol Pathol 1994;45:489–95. [DOI] [PubMed] [Google Scholar]
- 10.Walker NI, Winterford CM, Kerr JFR. Ultrastructure of the rat pancreas after experimental duct ligation. II. Duct and stromal proliferation, differentiation, and deletion. Pancreas 1992;7:420–34. [DOI] [PubMed] [Google Scholar]
- 11.Walker NI, Winterford CM, Williamson RM, et al. Ethionine-induced atrophy of rat pancreas involves apoptosis of acinar cells. Pancreas 1993;8:443–9. [DOI] [PubMed] [Google Scholar]
- 12.Jones BA, Gores GJ. Physiology and pathophysiology of apoptosis in epithelial cells of the liver, pancreas and intestine. Am J Physiol 1997;273:1174–88. [DOI] [PubMed] [Google Scholar]
- 13.Augstein P, Stephens LA, Allison J, et al. Cell apoptosis in an accelerated model of autoimmune diabetes. Mol Med 1998;4:495–501. [PMC free article] [PubMed] [Google Scholar]
- 14.Gavrieli Y, Sherman Y, Ben-Sasson SA. Identification of programmed cell death in situ via specific labelling of nuclear DNA fragmentation. J Cell Biol 1992;119:493–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mooney A, Baker AJ, Hughes D, et al. Mesangial cell apoptosis: the major mechanisms for resolution of glomerular hypercellularity in experimental mesangial proliferative nephritis. J Clin Invest 1994;94:105–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kerr JFR, Searle J, Harmon BV, et al. Apoptosis. In: Potton CS, editor. Perspectives on mammalian cell death. Oxford: Oxford University Press, 1987:93–128.
- 17.Kernohan NM, Cox LS. Regulation of apoptosis by Bcl-2 and its related proteins: immunochemical challenges and therapeutic implications. J Pathol 1996;179:1–3. [DOI] [PubMed] [Google Scholar]
- 18.Hale AJ, Smaith CA, Sutherland LC, et al. Apoptosis: molecular regulation of cell death. Eur J Biochem 1996;235:1–26. [DOI] [PubMed] [Google Scholar]
- 19.Lai R, Arber DA, Chang KL, et al. Frequency of bcl-2 expression in non-Hodgkin's lymphoma: a study of 778 cases with comparison of marginal zone lymphoma and monocytoid B-cell hyperplasia. Mod Pathol 1998;11:864–9. [PubMed] [Google Scholar]
- 20.Fadeel B, Zhivotovsky B, Orrenius S. All along the watchtower: on the regulation of apoptosis regulators. FASEB 1999;13:1647–54. [DOI] [PubMed] [Google Scholar]
- 21.Hengarter MO. The biochemistry of apoptosis. Nature 2000;407:770–6. [DOI] [PubMed] [Google Scholar]
- 22.Francavilla S, D'Abrizio P, Rucci N et al. Fas and Fas ligand expression in fetal and adult human testis with normal or deranged spermatogenesis. J Clin Endocrinol Metab 2000;85:2692–700. [DOI] [PubMed] [Google Scholar]
- 23.Leite-De Moraes MC, Herbelin A, Gouarin C, et al. Fas/Fas ligand interactions promote activation-induced cell death of NK T lymphocytes. J Immunol 2000;165:4367–71. [DOI] [PubMed] [Google Scholar]
- 24.Hongo T, Morimoto Y, Iwagaki H, et al. Functional expression of Fas and Fas ligand on human colonic intraepithelial T lymphocytes. J Int Med Res 2000;28:132–42. [DOI] [PubMed] [Google Scholar]
- 25.Shurin GV, Gerein V, Lotze MT, et al. Apoptosis induced in T cells by human neuroblastoma cells: Fas ligand. Nat Immun 1998;16:263–74. [DOI] [PubMed] [Google Scholar]
- 26.Mariani SM, Matiba B, Baumler C, et al. Regulation of cell surface APO-1/Fas(CD95) ligand expression by metalloproteinases. Eur J Immunol 1995;25:2303–7. [DOI] [PubMed] [Google Scholar]
- 27.Han X, Sun Y, Scott S, et al. Tissue inhibitor of metalloproteinase-1 prevents cytokine-mediated dysfunction and cytotoxicity in pancreatic islets and beta-cells. Diabetes 2001:50:1047–55. [DOI] [PubMed] [Google Scholar]
- 28.Wenkel H, Streilein JW. Evidence that retinal pigment epithelium functions as an immune-privileged tissue. Invest Opthalmol Vis Sci 2000;41:3467–73. [PubMed] [Google Scholar]
- 29.Mor G, Kohen F, Garcia-Velasco J, et al. Regulation of fas ligand expression in breast cancer cells by functional differences between estradiol and tamoxifen. J Steroid Biochem Mol Biol 2000;73:185–94. [DOI] [PubMed] [Google Scholar]
- 30.Kreuwel HT, Morgan DJ, Krahl T, et al. Comparing the relative role of perforin/granzyme versus Fas/Fas ligand cytotoxic pathyways in CD8+ T cell-mediated insulin-dependent diabetes mellitus. J Immunol 1999;163:4335–41. [PubMed] [Google Scholar]
- 31.Amrani A, Verdaguer J, Thiessen S, et al. IL-1 alpha, IL-1 beta, and IFN-gamma mark beta cells for Fas dependent destruction by diabetogenic CD4(+) T lymphocytes. J Clin Invest 2000;105:459–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stassi G, De Maria R, Trucco G, et al. Nitric oxide primes pancreatic beta cells for Fas-mediated destruction in insulin-dependent diabetes mellitus. J Exp Med 1997;186:1193–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chellappan SP, Hiebert S, Mudryj M, et al. The E2F transcription factor is a cellular target for the Rb protein. Cell 1991;65:1053–61. [DOI] [PubMed] [Google Scholar]
- 34.Yap DBS, Hsieh J-K, Chan FSG, et al. mdm-2: a bridge over the two tumour suppressors, p53 and Rb. Oncogene 1999;18:7681–9. [DOI] [PubMed] [Google Scholar]