Abstract
Background and aims: Invoked peristaltic contractions and movement of solid content have not been attempted in normal canine colon. The purpose of this study was to determine if movement of solid content through the colon could be produced by microprocessor controlled sequential stimulation.
Methods: The study was performed on six anaesthetised dogs. At laparotomy, a 15 cm segment of descending colon was selected, the proximal end closed with a purse string suture, and the distal end opened into a collecting container. Four sets of subserosal stimulating electrodes were implanted at 3 cm intervals. The segment of bowel was filled with a mixture of dog food and 50 plastic pellets before each of 2–5 random sessions of non-stimulated or stimulated emptying. Propagated contractions were generated using microprocessor controlled bipolar trains of 50 Hz rectangular voltage having 20 V (peak to peak) amplitude, 18 second stimulus duration, and a nine second phase lag between stimulation trains in sequential electrode sets.
Results: Electrical stimulation using the above mentioned parameters resulted in powerful phasic contractions that closed the lumen. By phase locking the stimulation voltage between adjacent sets of electrodes, propagated contractions could be produced in an aboral or orad direction. The number of evacuated pellets during the stimulation sessions was significantly higher than during the non-stimulated sessions (p<0.01).
Conclusions: Microprocessor controlled electrical stimulation accelerated movement of colonic content suggesting the possibility of future implantable colonic stimulators.
Keywords: gastric emptying, electrical control activity, electrical pacing, electrical response activity
In 1963, Bilgutay and colleagues1 described accelerated recovery from postoperative ileus when the stomach was stimulated at high electrical frequency (HF). Although others could not confirm these results, there have been several subsequent attempts to use electrical stimulation as an alternative treatment for gastrointestinal motility disorders. Three different methods of stimulation have been described.
The first method consists of electrical stimulation at similar or slightly higher frequencies than the electrical control activity (ECA) of the studied organ.2–10 As the basic intrinsic oscillation of the membrane potential controls the occurrence of contractions in the intestinal smooth muscle, this approach is aimed to entrain and synchronise ECA.2,3 For the stomach, this type of stimulation was labelled gastric electrical pacing by Sarna and colleagues.4 Although entrainment of ECA has been achieved using electrical pacing (EP) in different segments of the gastrointestinal tract,4–6 its effect on contractions and movement of content is questionable. In normal canine stomachs, no significant improvement in gastric emptying has been documented with the use of EP. Sarna and colleagues4 showed that proximal gastric EP entrained pacesetter potentials but did not improve gastric emptying. However, distal antral stimulation caused orad propagation of ECA and delayed gastric emptying. Kelly and Code5 confirmed these findings applying EP to the proximal duodenum with no effect on gastric emptying. Distal duodenal EP produced orad propagation of ECA causing duodenal-gastric reflux and delaying gastric emptying. Eagon and Kelly7 studied the effect of gastric EP at frequencies of 6 and 30 cycles/minute on gastric motility and emptying, reporting an increase in the frequency of pacesetter potentials but no significant effect on gastric contractions and emptying. However, Bellahsene and colleagues,8 in a model of gastroparesis in five vagotomised dogs with glucagon invoked dysrhythmia, showed acceleration of gastric emptying using gastric EP but no effect was documented without the artificially created dysrhythmia. In humans, the effect of gastric EP on emptying is controversial. In 17 postoperative patients, Hocking and colleagues9 reported no improvement in emptying using gastric EP. In contrast, in a recent study by McCallum and colleagues10 performed in nine patients with gastroparesis, gastric EP improved emptying and diminished symptoms.
A second method of stimulation has been described by Familoni and colleagues.11 They stimulated the canine stomach with frequencies 4–10 times the normal ECA (12–30 cycles/minute). The amplitude and frequency of contractions using this technique were significantly higher than those obtained using electrical stimulation at physiological frequencies.11 Improvement in symptoms and gastric emptying was documented in one patient with refractory diabetic gastroparesis using electrical stimulation at 12 cycles/minute.12
The third method uses electrical stimulation at HF (10–40 Hz).13–15 It has been used successfully to improve colonic transit in a cat model with spinal cord injury,13 to empty ileal pouches in dogs,14 and to create an electrically stimulated neoanal sphincter in a canine model.15
In our previous studies of the stomach,16,17 we have proposed a new fourth stimulation technique based on sequential electrical stimulation at HF (50 Hz) utilising sets of circumferentially implanted electrodes.17 The contractions elicited by this method16 were abolished by atropine and therefore can be considered under cholinergic control. The effectiveness of this method was illustrated by accelerated microprocessor controlled gastric emptying of liquids16 and solids17 in acute dogs. Microprocessor controlled stimulation is facilitated by theoretical mathematical modelling of the smooth muscle electromechanical dynamics in order to develop a quantitative description of the synchronisation and propagation contraction patterns in the colon for the purpose of their recreation. We have described such computer models for the stomach18 and colon.19
The purpose of this study was to determine if movement of solid colonic content could be achieved using microprocessor controlled sequential electrical stimulation in the colon.
MATERIALS AND METHODS
Six healthy dogs (four males, two females; weight 28.6 (7.8) kg; length 114 (19) cm; body mass index 22.12 (3.39) kg/m2) underwent laparotomy under Pentothal anaesthesia (Abbott, Montreal, Quebec, Canada). The initial dosage of anaesthesia was 30 mg/kg, supplemented with 3 mg/kg as needed based on the corneal reflex. Artificial ventilation was given during all experiments. At the end of the experiment the animal was sacrificed with an anaesthetic overdose.
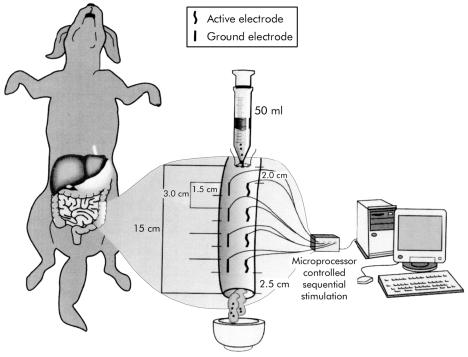
A 15 cm segment of the descending colon was chosen preserving intact the mesenteric innervation and blood supply. In this segment, four sets of locally designed bipolar stainless steel wire electrodes (15×0.25 mm) were implanted longitudinally into the subserosa and sutured in place with 3-0 silk at 3 cm intervals. Each set consisted of one active and one reference electrode. All active electrodes were placed along the longitudinal axis of the colon, with the reference counterparts implanted parallel and diametrically opposite on the other side (fig 1 ▶). Usually, the last set of electrodes was positioned at 25–30 cm from the anus.
Figure 1.
Schematic diagram of the implantation of four sets of longitudinal subserosal electrodes (15×0.25 mm) at 3 cm intervals. The electrode sets were attached to a multichannel stimulator controlled by specially designed software on a personal computer. A 15 cm segment of descending colon was filled with 40–50 ml of viscous material plus 50 pellets through a transversal opening made 2.0 cm proximal to the first set of electrodes and maintained closed with a purse string suture. The colon was transected 2.5 cm distal to the last set of electrodes. The resulting distal stoma was positioned in a metallic dish to collect eventual emptied material during stimulation and non-stimulation sessions.
The four electrode sets were attached to a custom designed electronic multichannel stimulator controlled by specially designed software20 on an IBM personal computer (133 MHz).
A 2 cm transverse opening was made in the colon 2.0 cm proximal to the first set of electrodes. Through this opening, 40–50 ml of a viscous material obtained from a mixture of commercially available dog food (Friskies Petcare, North York, Ontario, Canada) and water (3:1 in volume) plus 50 plastic cylindrical pellets (radius 0.75 mm, height 1.5 mm; Hythe, Kent, UK) were introduced into the colon to quantify eventual movement of solid contents.
Prior to starting the experimental sessions, the colon was transected 2.5 cm distal to the tip of the last electrode. The resulting distal stoma was set on a metal dish to collect the emptied material from the stimulated segment (see fig 1 ▶).
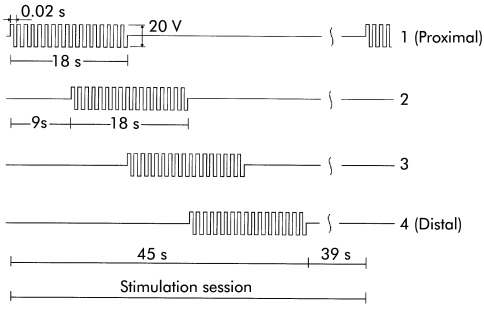
The following stimulation patterns, suggested by our previous dynamic parametric model of the colon,20 were chosen: bipolar voltage trains of 50 Hz, 20 V (peak to peak) rectangular voltage, 18 second stimulus duration, nine second phase lag between the stimuli applied to successive electrode sets, and 18 second pause between successive stimulation sessions (fig 2 ▶ shows the stimulation sequence for the four channels).
Figure 2.
Time characteristics of the stimuli applied to the electrode sets that produced aboral propagation of contractions (starting with the most proximal pair).
The effectiveness of microprocessor controlled movement of solid content using sequential neural stimulation was evaluated in separate random sessions by comparing the number of pellets evacuated spontaneously without stimulation to the number of pellets evacuated after stimulation. Each experimental session consisted of one period of 30 minutes without stimulation (spontaneous emptying) and one period of two consecutive stimulation trains (emptying after stimulation) (see fig 2 ▶). After each stimulation period, the colonic segment was filled again with the original amount of viscous material plus 50 additional pellets.
Between two and five repetitions of each experimental session were performed in each dog. Altogether 25 experimental sessions were completed.
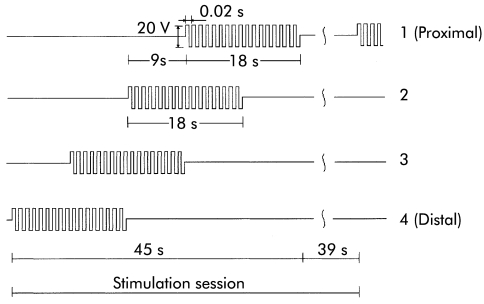
At the end of the experiment, the order of administering stimulation sequences to the electrode sets was reversed to evaluate the possibility of orad movement of content (fig 3 ▶ shows the stimulation sequence that produced orad movement).
Figure 3.
Time characteristics of the stimuli applied to the electrode sets that produced orad propagation of contractions (starting with the most distal pair).
In three dogs, an additional and final stimulation session was performed. The colon was filled with the standard amount of viscous material and pellets, and atropine 0.6 mg intravenously was given prior to electrical stimulation
The number of pellets obtained as a result of spontaneous and stimulated emptying during each experimental session was analysed and documented using mean (SD) values and the paired Student's t test for statistical significance.
The experiments were approved by the Animal Welfare Committee at the Faculty of Medicine, University of Alberta.
RESULTS
Motility patterns
Stimulation of a single set of electrodes using rectangular trains of 50 Hz, 20 V (peak to peak) amplitude, and 18 second duration resulted in a powerful phasic ring-like contraction that totally closed the lumen with a deflection of the colonic wall starting at about 1.5 cm proximal to each electrode and ending at about 1.5 cm distally from it, defining a contraction scope of 4.5 cm (including the electrode length of 1.5 cm). The produced contraction did not propagate. However, by sequential stimulation of adjacent electrodes with a time lag of nine seconds, the contraction propagated in peristaltic fashion. The direction of the peristalsis depended on the chronological organisation of the stimulus. If the time lag was oriented from proximal to distal adjacent electrode sets, peristalsis followed the caudad direction (see fig 2 ▶). Conversely, when the order of the electrodes was reversed, peristalsis followed the orad direction (see fig 3 ▶).
As the electrode sets were positioned at about 3 cm intervals (see fig 1 ▶), the effective distance between the metal wires was about 1.5 cm, giving a length of the stimulated segment between the most remote electrode sets of about 15 cm. A contraction produced by a given electrode set could overlap with a subsequently produced contraction by the adjacent electrode set, providing there was an overlap in the electrical stimuli, which in our experiments was 4.5 seconds (see for example fig 2 ▶). This overlapping between adjacent single contractions was important in avoiding the formation of pouches of immobile content between the adjacent electrode sets.
Emptying measurements
The microprocessor controlled sequential stimulation produced propagated powerful contractions that closed the lumen of the stimulated colonic segment, propelling and emptying its solid content through the stoma synchronously with the periods of active stimulation. The average number of pellets emptied after a given stimulation session was 39, accounting for 78% of the content.
Although we performed two consecutive stimulation trains in each experimental session, most of the content was expelled in the first train of stimulation. We observed a similar outcome in all experimental sessions. No signs of muscular fatigue, accommodation, or tissue damage were observed.
The number of pellets expelled after stimulation was significantly higher than the number of pellets expelled spontaneously (p<0.01) (table 1 ▶).
Table 1.
Effect of electrical stimulation on colonic emptying of solids
| No of pellets evacuated after 30 min of spontaneous emptying | No of pellets emptied after stimulation | |
| Dog 1, session 1 | 0 | 41 |
| Dog 1, session 2 | 1 | 44 |
| Dog 2, session 3 | 0 | 31 |
| Dog 2, session 4 | 0 | 27 |
| Dog 2, session 5 | 0 | 38 |
| Dog 3, session 6 | 0 | 38 |
| Dog 3, session 7 | 0 | 46 |
| Dog 3, session 8 | 0 | 50 |
| Dog 3, session 9 | 0 | 30 |
| Dog 3, session 10 | 0 | 50 |
| Dog 4, session 11 | 0 | 31 |
| Dog 4, session 12 | 1 | 30 |
| Dog 4, session 13 | 1 | 40 |
| Dog 4, session 14 | 0 | 33 |
| Dog 5, session 15 | 0 | 35 |
| Dog 5, session 16 | 1 | 39 |
| Dog 5, session 17 | 0 | 50 |
| Dog 5, session 18 | 0 | 41 |
| Dog 5, session 19 | 0 | 43 |
| Dog 6, session 20 | 0 | 41 |
| Dog 6, session 21 | 0 | 31 |
| Dog 6, session 22 | 0 | 46 |
| Dog 6, session 23 | 0 | 48 |
| Dog 6, session 24 | 0 | 38 |
| Dog 6, session 25 | 0 | 31 |
Administration of atropine abolished the generation and propagation of ring-like contractions elicited by electrical stimulation. As a result, movement of solid content and evacuation were not observed.
DISCUSSION
This study demonstrates that it is possible to accelerate the transit of solid content in the colon using sequential microprocessor based stimulation. In these experiments, we used the same 50 Hz frequency bipolar rectangular voltage that proved to be effective in our previous study of solid gastric emptying.17 Synchronising the stimuli, combined with appropriate electrode set positioning derived from a parametric electromechanical model,20 resulted in artificial “massaging” of the colon and immediate movement of colonic content. The direction of this movement depended on the synchronisation pattern of the stimulating voltage.
The two conceptually different approaches of electrical stimulation for promoting gastrointestinal motility, electrical pacing and neuroelectrical stimulation, aim at modulating intrinsic physiological phenomena.
The use of EP attempts to organise and entrain normal ECA, thus restoring the physiological balance in patients with electrical dysrhythmias. This so-called “physiological” electrical stimulation has proved its effectiveness invoking entraining of normal pacesetter potentials. However, its effect on contractions and transit of solid content remains controversial. We are surprised by the optimistic report from McCallum and colleagues10 that showed significant improvement in symptoms and gastric retention of gastroparetic patients using gastric EP because entraining of a physiological pacesetter potential does not generate contractions by itself. When the membrane potential reaches a certain threshold,21,22 it generates a different type of electrical activity known as electrical response activity (ERA). Simultaneously with ERA the muscle contracts. Intrinsic ECA depolarisations do not exceed the threshold potential under resting conditions. The membrane potential changes necessary for the genesis of contractions require neural control.
In contrast, the use of neuroelectrical stimulation aims at promoting ERA by stimulation of the cholinergic excitatory system. Although mechanical activity of the intestinal smooth muscle depends on changes in the membrane potential of the basic cyclic depolarisations (ECA), initiation of these changes is achieved by neural stimulation. Significant evidence supports this hypothesis: (a) acetylcholine increases the amplitude and duration of the plateau potential beyond the ERA threshold21; (b) atropine significantly decreases the amplitude and duration of the plateau potential increasing ERA threshold and as a result blocking the contractions21; (c) extrinsic autonomic denervation of the pelvic parasympathetic plexus produced abnormalities in colonic motility and defecation23; and (d) extrinsic stimulation of anterior sacral roots using HF produced colonic contractions.24
Neural electrical stimulation does not have any effect on the organisation of physiological pacesetter potentials but is effective in invoking powerful contractions which, when combined with multiple sequentially activated electrode sets under microprocessor control, result in movement of gastrointestinal content that can be precisely timed and controlled.
Our previous experiments in the stomach,17 and the results obtained in the present study clearly show a dramatic acceleration in the transit of solid content (p<0.01). In the current experiments, an identical response of powerful ring-like contractions that propagated in peristaltic fashion and moved solid contents with each stimulation was achieved without signs of muscular fatigue or tissue damage.
Physiological organised contractions have been described resembling our artificially invoked propagated contractions. They are called giant migrating contractions22,25 and are responsible for mass movement in the intestine.
The use of a microprocessor controlled system with flexible patterns of stimulation makes this method individually adjustable, depending on the physiological requirements of the organ. The method suggested in this article has been tested only in acute experiments in dogs. Evaluation of the long term effects of this technique on the tissue surrounding the stimulating electrodes, accommodation, and absorption calls for future investigation on chronic animal models. The development of possible secondary effects such as pain must also be studied before applying this method to humans.
In conclusion, a method for neural electrical stimulation is proposed using a microprocessor controlled sequential stimulation with flexible and adjustable parameters to artificially produce and propagate contractions in peristaltic fashion. The technique accelerated significantly the movement of solid content thus demonstrating the feasibility of future implantable microelectronic stimulators for the colon.
Acknowledgments
This study was sponsored in part by the Natural Sciences and Engineering Research Council of Canada, Canada Foundation for Innovation, Alberta Software Engineering Research Consortium, and Gastrointestinal Motility Laboratory (Edmonton, Alberta, Canada).
Abbreviations
ECA, electrical control activity
EP, electrical pacing
ERA, electrical response activity
HF, high electrical frequency
Note: The technology described in this paper is protected by a pending patent.
REFERENCES
- 1.Bilgutay AM, Wingrove R, Griffen GO, et al. Gastrointestinal pacing. A new concept in the treatment of ileus. Ann Surg 1963;158:338–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Quiang L, Lin X, Chen JD. Normalization of atropine induced dysrhythmias with gastric pacing. Am J Physiol 1999;276:G387–92. [DOI] [PubMed] [Google Scholar]
- 3.Lin ZV, McCallum RW, Schirmer BD, et al. Effects of pacing parameters on entrainment of gastric slow waves in patients with gastroparesis. Am J Physiol 1998;274:G186–91. [DOI] [PubMed] [Google Scholar]
- 4.Sarna SK, Bowes KL, Daniel EE. Gastric pacemakers. Gastroenterology 1976;70:226–31. [PubMed] [Google Scholar]
- 5.Kelly KA, Code CF. Duodenal-gastric reflux and slowed gastric emptying by electrical pacing of the canine duodenal pacesetter potential. Gastroenterology 1997;72:429–33. [PubMed] [Google Scholar]
- 6.Sarr MG, Kelly KA, Gladen HE. Electrical control of canine jejunal propulsion. Am J Physiol 1981;240:G355–60. [DOI] [PubMed] [Google Scholar]
- 7.Eagon JC, Kelly KS. Effect of electrical stimulation on gastric electrical activity, motility, and emptying. Neurogastroenterol Motil 1995;7:39–45. [DOI] [PubMed] [Google Scholar]
- 8.Bellahsene B, Lind CD, Schirmer BD, et al. Acceleration of gastric emptying with electrical stimulation in a canine model of gastroparesis. Am J Physiol 1992;276:G826–34. [DOI] [PubMed] [Google Scholar]
- 9.Hocking MP, Vogel SB, Sninsky CH. Human gastric myoelectrical activity and gastric emptying following gastric surgery and with pacing. Gastroenterology 1992;103:1811–16. [DOI] [PubMed] [Google Scholar]
- 10.McCallum RW, Chen JD, Lin Z, et al. Gastric pacing improves emptying and symptoms in patients with gastroparesis. Gastroenterology 1998;114:456–61. [DOI] [PubMed] [Google Scholar]
- 11.Familoni B, Abell TL, Nemoto D, et al. Efficacy of electrical stimulation at frequencies higher than basal rate in canine stomach. Dig Dis Sci 1997;42:892–7. [DOI] [PubMed] [Google Scholar]
- 12.Familoni B, Abell TL, Nemoto D, et al. Electrical stimulation at frequencies higher than the basal rate in canine stomach. Dig Dis Sci 1997;42:895–1. [DOI] [PubMed] [Google Scholar]
- 13.Bruninga K, Riedy L, Keshaversian A, et al. The effect of electrical stimulation on colonic transit following spinal cord injury in cats. Spinal Cord 1998;36:847–53. [DOI] [PubMed] [Google Scholar]
- 14.Grundfest-Broniakowski S, Moritz A, Ilyes L, et al. Voluntary control of an ileal pouch by coordinated electrical stimulation, a pilot study in the dog. Dis Colon Rectum 1988;31:261–7. [DOI] [PubMed] [Google Scholar]
- 15.Hughes SF, Scott SM, Pilot MA, et al. Electrically stimulated smooth muscle neosphincter. Br J Surg 1996;82:1321–6. [PubMed] [Google Scholar]
- 16.Mintchev MP, Sanmiguel CP, Otto SJ, et al. Microprocessor controlled movement of liquid gastric content using sequential neural electrical stimulation. Gut 1988;43:607–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mintchev MP, Sanmiguel CP, Amaris M, et al. Microprocessor controlled movement of solid gastric content using sequential neural electrical stimulation. Gastroenterology 2000;118:259–63. [DOI] [PubMed] [Google Scholar]
- 18.Mintchev MP, Bowes KL. Computer model of gastric electrical stimulation. Ann Biomed Eng 1997;25:726–30. [DOI] [PubMed] [Google Scholar]
- 19.Rashev PZ, Mintchev Mp, Bowel KL. Three-dimensional static parametric model of phasic contractions for the purpose of microprocessor controlled functional stimulation. J Med Eng Technol 2001;25:85–96. [DOI] [PubMed] [Google Scholar]
- 20.Rashev PZ, Amaris M, Bowes KL, et al. Microprocessor-controlled colonic peristalsis. Dynamic parametric modeling in dogs. Dig Dis Sci 2002; (in press). [DOI] [PubMed]
- 21.Szurszewski JH. Electrical basis for gastrointestinal motility. In: Johnson LR, ed. Physiology of the gasytrointestinal tract, vol 2. New York: Raven Press, 1981:1435–66.
- 22.Sarna SK. Physiology and pathophysiology of colonic motor activity (part one of two). Dig Dis Sci 1991;36:827–62. [DOI] [PubMed] [Google Scholar]
- 23.Ishihawa M, Mibu R, Iwamoto T, et al. Change in colonic motility after extrinsic autonomic denervation in dogs. Dig Dis Sci 1997;42:1950–6. [DOI] [PubMed] [Google Scholar]
- 24.Varma JS, Binnie N, Smith AN, et al. Different effects of anterior root stimulation on anal sphincter and colorectal motility in spinally injured man. Br J Surg 1986;73:478–82. [DOI] [PubMed] [Google Scholar]
- 25.Sarna SK. Giant migrating contractions and their myoelectric correlates in the small intestine. Am J Physiol 1987;253:G679–705. [DOI] [PubMed] [Google Scholar]