Abstract
The diagnostic features and clinical course of three children (aged 1 month to 15 years) with severe functional intestinal obstruction and inflammation of the colonic lamina propria and myenteric plexus are described. The myenteric inflammatory infiltrate was eosinophil predominant with none of the immunological characteristics of lymphocytic ganglionitis. Neurones in the myenteric ganglia expressed the potent eosinophil chemoattractant interleukin 5. None responded to dietary exclusion but all three responded symptomatically to immunosuppression/anti-inflammatory treatments. Eosinophilic ganglionitis is associated with a pseudo-obstructive syndrome which is amenable to anti-inflammatory treatment.
Keywords: eosinophilic gastroenteropathy, interleukin 5, myenteric ganglionitis, intestinal obstruction
Chronic intestinal pseudo-obstruction results from primary or secondary abnormalities of the enteric neuromusculature,1–3 Acquired (secondary) disorders are most often due to paraneoplastic syndromes4 or to autoimmune diseases affecting visceral nerves and muscle.5–7 Autoimmune neuropathic disorders are characterised by lymphocytic infiltration of the myenteric plexus and loss of rectoanal inhibitory reflexes. We describe three children with functional gastrointestinal obstruction in association with an inflammatory process affecting the lamina propria and the myenteric plexus, in which eosinophils were predominant and T lymphocytes absent.
MATERIALS AND METHODS
Patients
Three girls (aged one month, 11 years, and 15 years 11 months) were studied over a period of 20 months having presented with severe functional gastrointestinal obstruction.
Motility investigations
Where indicated, whole bowel transit was measured using a well validated radio-opaque pellet method. Antroduodenal and anorectal manometry was performed using water perfused catheters and a previously validated data acquisition and analysis system (Synectics, Stockholm, Sweden).8
Histological methods
Full thickness small or large bowel biopsies were obtained in all patients (see below). All underwent mucosal biopsy of the foregut and hindgut. Histological sections were examined by routine methods and inflammatory infiltrates characterised by immunocytochemistry using monoclonal antibodies against neutrophil elastase, leucocyte common antigen, CD45Ro (UCHL1), CD3, CD5, CD20, and CD79α for the presence of neutrophils, white cells, memory cells, and T and B cells, respectively. Interleukin 5 (IL-5), a potent eosinophil chemoattractant, was detected using a polyclonal antibody and revealed with Duet (Dako, Glostrup, Denmark). Antigen retrieval by microwaving was required for all antigens except neutrophil elastase. Negative controls were performed for each specimen by omitting the primary antibody and positive controls using tonsillar tissue.
CASE REPORTS
Patient No 1
A full term female infant aged one month presented with vomiting, constipation, and marked abdominal distension having deteriorated from the age of nine days. She had passed meconium in the first 24 hours of life and left the postnatal ward with no signs of abdominal distension. Her subsequent clinical presentation was suggestive of Hirschsprung’s disease with no further bowel openings and massive abdominal distension.
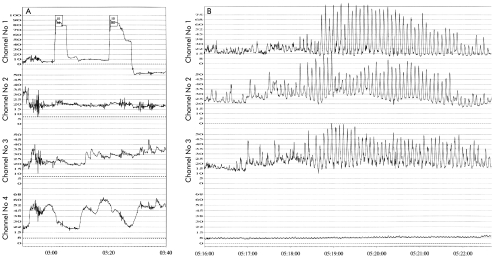
Barium enema demonstrated a dilated large bowel from the hepatic flexure to the anal canal and anorectal manometry demonstrated a raised internal anal sphincter (IAS) pressure of 46 mm Hg (age related norm=15–25 mm Hg). Ninety five per cent IAS relaxation followed rectal balloon insufflation to 10 ml (fig 1A ▶).
Figure 1.
(A) Anorectal manometry of patient No 1 showing preservation of rectoanal inhibitory responses and a raised internal anal sphincteric pressure. (B) Antroduodenal manometry of patient No 2 showing abnormal propagation of normal amplitude contractile activity.
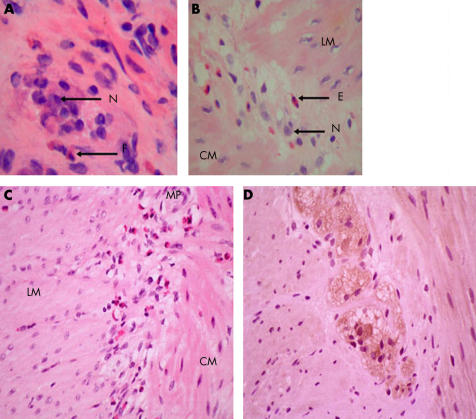
Full thickness and suction rectal biopsies demonstrated the presence of normal ganglia and neurones within the submucosal and myenteric plexus and a normal acetylcholinesterase staining pattern, excluding Hirschsprung’s disease. An inflammatory infiltrate consisting of macrophages, lymphocytes, and eosinophils was present in the mucosa. Scattered eosinophils were present in the submucosa and circular muscle coat. Degranulating eosinophils infiltrated the myenteric ganglia (fig 2A ▶). Immunostaining demonstrated that the inflammatory cells in the myenteric plexus infiltrate consisted of a mixed inflammatory infiltrate in which T cells were absent (LCA, CD45Ro, CD3 stains). Mucosal biopsies from the right and left colon were chronically inflamed with an eosinophilic infiltrate. Stomach and small bowel mucosal biopsies were normal.
Figure 2.
(A) Haematoxylin/eosin (H/E) stained frozen section of full thickness rectal biopsy from patient No 1 demonstrating an inflammatory infiltrate within the myenteric plexus. N, neurone; E, eosinophil. (B) H/E stained section of a full thickness colonic biopsy from patient No 2 demonstrating eosinophils in the myenteric plexus. LM, longitudinal muscle; CM, circular muscle. (C) H/E stained section of the myenteric plexus and muscularis propria in the colon of patient No 3. Note eosinophils in the myenteric plexus and muscularis. MP, myenteric plexus. (D) Colonic seromuscular biopsy of patient No 2 immunostained for interleukin 5 showing intense positive staining of myenteric neurones.
She had a peripheral blood eosinophilia (1.15×109/l), an elevated age related IgE concentration (32.2kU/l), and weakly positive smooth muscle and antinuclear autoantibodies. A panel of specific IgE RASTS was negative. She was initially treated with an amino acid based infant formula feed (Neocate, SHS) and sulphasalazine (40 mg/kg/day) but failed to improve over an eight week period with defecation remaining dependent on rectal washouts. Addition of systemic steroids (oral beclomethasone 200 μg three times daily) resulted in a brisk improvement with return of daily spontaneous defecation. Aged one year her bowel habit has remained normal while adhering to a diet free of wheat, cow’s milk, egg, andsoya. Steroids were withdrawn but sulphasalazine continued because of persistent (although much improved) colonic mucosal inflammation. Follow up biopsy of the submucus or myenteric plexus was not undertaken.
Patient No 2
A girl aged 11 years presented with intractable constipation, nausea, and abdominal pain. She opened her bowels with difficulty every 2–3 weeks and subsequently developed intestinal obstruction with bilious vomiting and abdominal distension. Abdominal x ray confirmed dilated bowel loops and multiple fluid levels, compatible with distal bowel obstruction. At laparotomy no mechanical cause was found. The acute obstructive episode settled after three weeks of intravenous fluids. She continued to suffer slow intestinal transit and chronic constipation.
Prior investigation had shown a high total IgE concentration (2445 kU/l) and negative specific IgE RASTs to a wide variety of allergens. Contrast studies had excluded anatomical abnormality. Orocaecal transit time was four hours.
Stomach and small bowel mucosal biopsies were normal. Colonoscopic mucosal biopsies revealed severe patchy mucosal eosinophilia with eosinophil degranulation and cryptitis, affecting the entire colon. Eosinophils were found trafficking through the epithelium and the vascular endothelium. Suction rectal biopsy had a normal acetylcholinesterase staining pattern and ganglion cells were visible in the submucosal plexus, thus excluding Hirschsprung’s disease. An eosinophil predominant inflammatory infiltrate was present within the myenteric ganglia of a full thickness rectal biopsy. Colonic seromuscular biopsy revealed eosinophil rich inflammation around neurones within the myenteric plexus (fig 2B ▶). T and B lymphocytes were inconspicuous. Immunostaining for the eosinophil chemoattractant IL-5 demonstrated expression in both neurones and inflammatory cells within the myenteric plexus (fig 2D ▶). Intense IL-5 immunoreactivity was also noted within the lamina propria (data not shown).
Anorectal manometry showed elevation of anal sphincteric pressure (60 mm Hg) and preservation of the rectoanal inhibitory reflex. Antroduodenal manometry was performed to gain insight into the effects of the inflammatory process on programmed intestinal motor activity. This revealed phase 3 motor activity of normal amplitude but with poor propagation, suggestive of an underlying enteric neuropathy (fig 1B ▶).8
Dietary exclusion (cow’s milk, soya, and wheat), using an amino acid based formula (E028, SHS) as a milk substitute, sodium cromoglycate, and Sennokot were without clinical benefit. When treated with prednisolone (60 mg/day) and azathioprine (2–3 mg/kg/day), normal bowel habit returned (one spontaneous bowel motion each day). Radio-opaque pellet transit studies pre and post treatment confirmed a reduction in oroanal transit from >96 hours to <48 hours after three months of treatment with azathioprine at which time prednisolone had been weaned off. Azathioprine was continued for 12 months. Intestinal transit times lengthened and severe ileocolonic inertia returned following its withdrawal.
Patient No 3
A girl aged 15 years and 11 months presented with acute small intestinal obstruction. She had undergone proctocolectomy five years previously because of a non-specific colonic inflammatory process which had caused intractable abdominal and hip pain. These symptoms had been steroid responsive although steroid dependent (≥20 mg prednisolone/day). Stomach and small bowel mucosal biopsies had been histologically normal. Latterly she had received maintenance anti-inflammatory treatment (5 aminosalicylate 40 mg/kg/day) for recurrent pouchitis. Autoimmune markers (antineutrophil cytoplasmic antibodies, antinuclear antibodies, anti-smooth muscle antibodies, and antienterocyte antibodies) were consistently negative. Serum IgE was within the age related normal range and specific IgE RASTS were negative.
At presentation she was noted to have a tender distended, although quiet, abdomen and dilated bowel loops on plain abdominal x ray. An emergency laparotomy was performed during which no evidence of adhesion or mechanical obstruction was found. Full thickness distal ileal biopsies taken during surgery showed an increase in inflammatory cells, in particular eosinophils, in the mucosa and submucosa. Degranulating eosinophils were also present in the circular muscle coat and the myenteric plexus (fig 2C ▶). Immunostaining with neutrophil elastase, CD3, CD5, CD20, and CD79α showed very few neutrophils or T and B cells within the mucosa. The finding of eosinophil predominant inflammation of the mucosa and myenteric plexus (“eosinophilic epiganglionitis”) led to the use of large doses (600 mg) of intravenous methylprednisolone to treat her continued postoperative signs of intestinal ileus/obstruction. She recovered within 24 hours. Maintenance treatment with azathioprine and a reducing dose of prednisolone has continued. Twelve months on she has had no further episodes of severe functional obstruction although she has persisting ileal mucosal inflammation.
DISCUSSION
We have described three children with different presentations of an acquired and reversible functional intestinal obstructive syndrome in association with an eosinophilic ganglionitis and eosinophilic colitis. They differed from classical submucosal eosinophilic gastroenteropathy as the functional intestinal obstruction was closely associated with an eosinophilic ganglionitis rather than bowel wall thickening.
Functional gastrointestinal obstruction (often referred to as intestinal pseudo-obstruction) arises as a consequence of a variety of congenital or acquired disorders of the enteric neuromusculature. The clinical presentation is highly variable depending on both the site and severity of the intestinal disease.1,9 Inflammation within the myenteric ganglia is a recognised cause of this syndrome. A severe progressive acquired neuropathic pseudo-obstructive syndrome is seen in conjunction with autoimmune disease4,5 and circulating antienteric neuronal antibodies (which were absent in the patients described here). There is an associated lymphocytic ganglionitis in which the inflammatory infiltrate consists predominantly of CD3+ and CD4+ T lymphocytes. Lymphocytic ganglionitis resulting in functional gastrointestinal obstruction is described in paraneoplastic syndromes4,10,11 in association with destruction of enteric neurones and acquired aganglionosis. Functional manometric correlates of this denervation include loss of small intestinal phase III fasting motor activity and absence of anorectal inhibitory responses. In contrast, eosinophilic ganglionitis is characterised by an absence of demonstrable lymphocytes within the inflamed enteric ganglia and is not associated with either morphological or functional evidence of enteric denervation. Small intestinal phase III motor activity and reflex anorectal inhibitory responses are preserved in eosinophilic ganglionitis. Retrograde small intestinal contractile motor activity, a feature of enteric ganglionitis,8 is seen in both lymphocytic and eosinophilic variants. The motor abnormality associated with enteric neuropathy is highly variable8—a manifestation of both severity and stage of denervation.
The immunopathogenesis of lymphocytic ganglionitis and eosinophilic ganglionitis has many similarities to T lymphocyte driven enteropathies and eosinophilic gastroenteropathy, respectively. In lymphocytic ganglionitis the immune response is T helper lymphocyte 1 (Th1) skewed whereas that in eosinophilic ganglionitis is Th2 skewed. In none of our patients with eosinophilic ganglionitis could we detect lymphocytes within the myenteric ganglia with a variety of specific immunocytochemical methods. Interestingly, we observed expression of IL-5 by enteric neurones of our patients. Hence the presence of eosinophils within the ganglia might be a consequence of IL-5 secretion by enteric neurones rather than by Th2 polarised lymphocytes. The concept of neuronal expression of cytokines in inflammatory neuropathies is well established, particularly with regard to interleukin 1β.12,13 To our knowledge, IL-5 expression has not previously been described in enteric ganglia. Recent data have demonstrated in transgenic animals that over expression of IL-5 results in both peripheral blood eosinophilia and compartmentalised distribution of eosinophils within the gastrointestinal tract. These findings are in keeping with the observations in our patients.14,15
IL-5 expression is a recognised characteristic of delayed type hypersensitivity to food allergen.16 There was no clinical improvement in the gastrointestinal dysmotility syndrome in the two patients treated for 6–8 weeks with an oligoantigenic amino acid based liquid feed (Neocate or elemental 028, SHS) as a sole source of nutrition. All three patients did, however, respond briskly to steroids.
Intestinal obstruction is reported in seromuscular eosinophilic gastroenteropathy. Hitherto this has been considered a predominantly mechanical phenomenon secondary to bowel wall thickening17,18 as a consequence of eosinophilic infiltration of the intestinal musculature.19–21 Our patients with eosinophilic ganglionitis lacked these features having no evidence of bowel wall thickening or narrowing of the intestinal lumen. None had evidence of eosinophilic infiltration of the stomach or duodenum, as seen in mucosal eosinophilic gastroenteropathy.
In summary, we have described a variant of eosinophilic gastroenteropathy in which functional gastrointestinal obstruction was associated with a myenteric eosinophilic ganglionitis rather than a transmural inflammatory process. Ganglionitis was immunologically distinct from that of lymphocytic ganglionitis. This pseudo-obstructive syndrome can affect children of all ages and is steroid responsive.
Abbreviations
EG, eosinophilic gastroenteropathy
Th, T helper lymphocyte
IL-5, interleukin 5
IAS, internal anal sphincter
REFERENCES
- 1.Smith VV, Milla PJ. Histological phenotypes of enteric smooth muscle disease causing functional intestinal obstruction in childhood. Histopathology 1997;31:112–22. [DOI] [PubMed] [Google Scholar]
- 2.Smith VV, Lake BD, Kamm MA, et al. Intestinal pseudo-obstruction with deficient smooth muscle alpha-actin. Histopathology 1992;21:535–42. [DOI] [PubMed] [Google Scholar]
- 3.Milla PJ, Smith VV. Aganglionosis, hypoganglionosis and hyperganglionosis: clinical presentation and histopathology. In: Kamm MA, Lennard-Jones MA, eds. Constipation. Wrightson Biomedical Publishing Ltd, 1994:183–92.
- 4.Lennon VA, Sas DF, Busk MF, et al. Enteric neuronal autoantibodies in pseudoobstruction with small-cell lung carcinoma. Gastroenterology 1991;100:137–42. [DOI] [PubMed] [Google Scholar]
- 5.Smith VV, Gregson N, Foggensteiner L, et al. Acquired intestinal aganglionosis and circulating autoantibodies without neoplasia or other neural involvement. Gastroenterology 1997;112:1366–71. [DOI] [PubMed] [Google Scholar]
- 6.Lindberg G, Glia A, Nyberd B. Lymphocytic ganglionitis—a new entity causing severe motility disorders of the gut. Gastroenterology 1999;116:A1030. [Google Scholar]
- 7.Ruuska TH, Karikoski R, Smith VV, et al. Acquired myopathic intestinal pseudo-obstruction may be due to autoimmune enteric leiomyositis. Gastroenterology 2002;122:1133–9. [DOI] [PubMed] [Google Scholar]
- 8.Fell JM, Smith VV, Milla PJ. Infantile chronic idiopathic intestinal pseudo-obstruction: the role of small intestinal manometry as a diagnostic tool and prognostic indicator. Gut 1996;39:306–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Milla PJ. Intestinal motility during ontogeny and intestinal pseudo-obstruction in children. Pediatric Gastroenterology II. Pediatric Clinics of North America. Philadelphia: WB Saunders, 1996:511–32. [DOI] [PubMed]
- 10.De Giorgio R, Barbara G, Stanghellini V, et al. Idiopathic myenteric ganglionitis underlying intractable vomiting in a young adult. Eur J Gastroenterol Hepatol 2000;12:613–16. [DOI] [PubMed] [Google Scholar]
- 11.Mitomi H, Tanabe S, Igarashi M, et al. Autoimmune enteropathy with severe atrophic gastritis and colitis in an adult: proposal of a generalized autoimmune disorder of the alimentary tract. Scand J Gastroenterol 1998;33:716–20. [DOI] [PubMed] [Google Scholar]
- 12.Skundric DS, Lisak RP, Rouhi M, et al. Schwann cell-specific regulation of IL-1 and IL-1Ra during EAN: possible relevance for immune regulation at paranodal regions. J Neuroimmunol 2001;116:74–82. [DOI] [PubMed] [Google Scholar]
- 13.Conti G, Scarpini E, Baron P, et al. Macrophage infiltration and death in the nerve during the early phases of experimental diabetic neuropathy: a process concomitant with endoneurial induction of IL-1beta and p75NTR. J Neurol Sci 2002;195:35–40. [DOI] [PubMed] [Google Scholar]
- 14.Zou LP, Abbas N, Volkmann I, et al. Suppression of experimental autoimmune neuritis by ABR-215062 is associated with altered Th1/Th2 balance and inhibited migration of inflammatory cells into the peripheral nerve tissue. Neuropharmacology 2002;42:731–9. [DOI] [PubMed] [Google Scholar]
- 15.Mishra A, Hogan SP, Brandt EB, et al. Enterocyte expression of the eotaxin and interleukin-5 transgenes induces compartmentalized dysregulation of eosinophil trafficking. J Biol Chem 2002;277:4406–12. [DOI] [PubMed] [Google Scholar]
- 16.Vandezande LM, Wallaert B, Desreumaux P, et al. Interleukin-5 immunoreactivity and mRNA expression in gut mucosa from patients with food allergy. Clin Exp Allergy 1999;29:652–9. [DOI] [PubMed] [Google Scholar]
- 17.Hummer-Ehret BH, Rohrschneider WK, Oleszczuk-Raschke K, et al. Eosinophilic gastroenteritis mimicking idiopathic hypertrophic pyloric stenosis. Pediatr Radiol 1998;28:711–13. [DOI] [PubMed] [Google Scholar]
- 18.Hoefer RA, Ziegler MM, Koop CE, et al. Surgical manifestations of eosinophilic gastroenteritis in the pediatric patient. J Pediatr Surg 1977;12:955–62. [DOI] [PubMed] [Google Scholar]
- 19.Kelly KJ. Eosinophilic gastroenteritis. J Pediatr Gastroenterol Nutr 2000;30(suppl):S28–35. [DOI] [PubMed] [Google Scholar]
- 20.Olson AD, Fukui-Miner K. Eosinophilic mucosal infiltrate in infants with congenital gastrointestinal obstruction. Am J Gastroenterol 1994;89:934–6. [PubMed] [Google Scholar]
- 21.Talley NJ, Shorter RG, Phillips SF, et al. Eosinophilic gastroenteritis: a clinicopathological study of patients with disease of the mucosa, muscle layer, and subserosal tissues. Gut 1990;31:54–8. [DOI] [PMC free article] [PubMed] [Google Scholar]