Abstract
Background: Survival at the intermediate stage of colorectal cancer (CRC) is less predictable than in the early and advanced stages. Several genetic markers possibly involved in growth and progression of CRC can be used for prognosis.
Aims: This study investigated the proportion of allelic loss (loss of heterozygosity (LOH)) at the BRCA1 locus in sporadic CRC and its value in patient prognosis.
Patients and methods: A total of 314 patients were investigated for LOH at the BRCA1 locus using polymerase chain reaction by means of three intragenic polymorphic microsatellite markers. Allelic losses were compared with clinicopathological characteristics of patients, recurrence rate, disease free survival (DFS), and overall survival.
Results: Twenty six patients were excluded because of microsatellite instability. Of the remaining 288 cases, 244 (84.7%) were informative, with 97 (39.8%) patients bearing BRCA1 LOH. Recurrence rate was higher in patients with LOH (p = 0.0003), and DFS was 73.3% (SEM 5.7) at five years in patients without LOH, and 49.2% (7.1) in cases with positive allelic loss (p = 0.0004). Retention of alleles at the BRCA1 locus was associated with a favourable DFS in stages I and II (p<0.05). The presence of LOH was also significantly associated with short overall survival (p = 0.02). Multivariate analysis in the complete series showed that stage (p = 0.006) and lymph node metastases (⩾4 nodes, p = 0.0001; 1–3 nodes, p = 0.038) were independent prognostic factors. However, multivariate study by stages revealed that BRCA1 LOH was an independent prognostic factor in stages I and II (p = 0.001).
Conclusions: BRCA1 LOH is a molecular alteration present in CRC, with unfavourable repercussions for overall survival, that could be considered as an outstanding independent prognostic factor in stages I and II.
Keywords: colorectal cancer, BRCA1 locus, loss of heterozygosity, disease free survival, survival
Colorectal cancer (CRC) is one of the most frequent malignancies in Western countries and remains associated with a high mortality rate.1 Today, pathological staging is still the most useful prognostic factor.2 Presentation of CRC is most commonly sporadic but a significant number of cases have hereditary influences.3
Numerous CRC studies have provided evidence for the involvement of tumour suppressor genes in its tumorigenesis, including those demonstrating allelic loss. Some chromosomal regions with a high rate of deletions have been found:4–6 loss of heterozygosity (LOH) has been detected in chromosomal arm 5q in 20% and 50% of CRCs,4,7,8 allelic losses at 18q in approximately 70% of CRCs,9 and LOH at 17p in more than 75% of CRCs.10 The target genes located in these regions possibly involved in tumour growth and progression are APC, DCC, and TP53, respectively. Furthermore, LOH status has been used as a factor in disease prognosis. Several reports analysing the direct relationship between LOH and clinicopathological parameters in some types of tumours have demonstrated the existence of significant associations with phenotypic characteristics accepted as indicative of poor disease prognosis and shorter patient survival.11–15
BRCA1 is a large gene with many functional domains, each with different biological features. The C terminal region is related to the transactivation region of the protein16 and residues 758–1064 to the domain binding to Rad51,17 thus working as a complex to repair double stranded DNA breaks.18 In relation to its repair role, BRCA1 is also related to coactivation of p53.19 The relationship of truncating germline mutations in the BRCA1 gene and breast and ovarian cancers is established. Mutations in this gene are responsible in part for the inherited predisposition to breast and ovarian cancers, and probably for one third of all site specific breast cancer kindreds.20 However, whether these mutations are a factor in sporadic forms of these tumours remains unclear. It has been suggested that members of families linked to the BRCA1 gene have a higher than normal risk of CRC and prostate cancer, with relative risks of 3.3 and 2.95, respectively.21
In a previous study, we found a high percentage (49%) of LOH in the 17q21 region in sporadic CRC cases. Seven polymorphic markers of this region were studied and no cases displayed loss of the all informative markers, suggesting that losses in this region were not due to loss of the entire chromosome.22 In this series we also studied the LOH status of locus TP53, to determine the status of chromosome 17 and the relation between concomitant LOH at the two loci.
The present study investigated LOH at the 17q21 region using three microsatellite markers intragenic to the BRCA1 gene in a series of sporadic CRCs with unknown status with respect to mutations in germline BRCA1. The clinical relevance of 17q21 allelic losses was analysed in a study comparing them with clinicopathological characteristics, to determine their value as a prognostic factor and influence on disease free survival (DFS) and overall survival.
PATIENTS AND METHODS
Patients, tumour samples, and DNA extraction
The present study, approved by the research ethics boards of the three participating hospitals, was based on a consecutive series of 314 patients undergoing surgery for colorectal cancer between January 1986 and May 2001. All patients were considered sporadic cases because those with familial adenomatous polyposis and clinical criteria for hereditary non-polyposis colorectal cancer (Amsterdam criteria) were excluded. Tumour samples and corresponding normal tissues consisted of 63 paired samples that were formalin fixed and paraffin embedded, and 251 snap frozen tissues.
All tumours were examined histologically by two pathologists to: (a) confirm the diagnosis of adenocarcinoma, (b) confirm the presence of tumour and evaluate the percentage of tumour cells in these samples, and (c) carry out pathological staging. All samples had at least 75% tumour cells. To ascertain whether contamination of the tumour samples by normal cells could affect allelic signal intensity in terms of the value considered to indicate the presence of LOH, 30 tumour samples were randomly selected and any normal tissue was removed by micro dissection. Subsequently, DNA was extracted from microdissected and non-microdissected paired tumour samples, and the LOH analysis described below was performed to assess the possible correlation between the two sets of results.
DNA from tumour and corresponding normal tissues was extracted from formalin fixed paraffin embedded tissues using chelatin resin, or from snap frozen tissues using a non-organic method (S-4520 Kit; Oncor Inc., Gaithersburg, Maryland, USA).
Clinicopathological characteristics
The medical records of the 314 patients provided birth date and sex, and the following parameters: tumour location, tumour size, lymph node metastases, pathological stage, vascular invasion, tumour differentiation, and carcinoembryonic antigen (CEA) levels. Pathological stage was assessed using the tumour, node, and metastases classification system. All tumours were graded for percentage glandular appearance. CEA levels were determined by a microparticle enzyme immunoassay method (AxSYM CEA; Abbott, Wiesbaden, Germany) and the range 0–5 ng/ml was considered normal.
Follow up was based on regular (every three months during the first year, every six months during the second year, and then yearly until relapse) clinical, biochemical (including CEA levels), and radiological examination, and yearly endoscopy. The starting point for DFS time was the date of surgery, and the end was the date of relapse. Overall survival was the interval between the date of surgery and the last visit or death.
PCR conditions, primers, and LOH assay
BRCA1 locus
Allelic deletions were detected by LOH analysis using the microsatellite markers D17S1323, D17S1322, and D17S855, which localise to introns 12, 19, and 20, respectively.23 Polymerase chain reaction (PCR) was performed in 25 μl volumes using 0.2 units of Taq Gold DNA polymerase and 1× PCR buffer (Perkin-Elmer, Roche Molecular Systems, Inc., Branchburg, New Jersey, USA), 200 μM dNTPs, 30 pmol of each primer, and different concentrations of KCl and MgCl2 depending on the polymorphic marker used. PCR was performed in a thermocycler (Perkin-Elmer, Cetus, Foster City, California, USA). Each sample was denatured at 94°C for 11 minutes and subjected to 30 cycles (denaturation at 94°C for 30 seconds, annealing at 56°C for 40 seconds, and elongation at 72°C for 30 seconds) followed by a final 12 minute extension at 72°C. The amplified products were mixed with 6 μl of loading buffer (0.02% xylene cyanol, 0.02% bromophenol blue) and run on non-denaturing 8% polyacrylamide gels for four hours at 500 V. Following gel electrophoresis, allelic band intensity was detected by a non-radioisotope technique using a commercially available silver staining method.24
TP53 region, 17p13
Using two polymorphic markers of the 17p13 region, TP53.6,25 intragenic to PT53, and D17S786,26 centromeric to TP53, a LOH study was performed in an LOH assay similar to the one above, with specific annealing temperatures of 59°C and 58°C for each marker.
Analysis of LOH
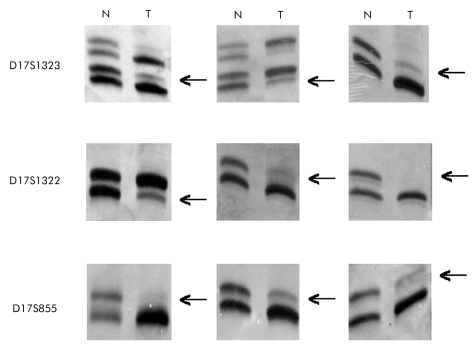
Allele intensities were analysed by densitometry. The gel image was captured using a GS-690 Imaging Densitometer (Bio-Rad Laboratories, Hercules, California, USA), digitised at 400 dpi and analysed using Multi-Analyst/PC (Bio-Rad Laboratories). In this study, LOH was considered to exist when the allele signal was over 75% less than in the normal counterpart DNA (fig 1 ▶). In the 30 tumours studied after microdissection, there were no determinable decreases in allelic signal intensity to below our 75% cutoff point. All samples maintained a range of signal intensity similar to that exhibited prior to microdissection. Microsatellite instability (MI) was considered to be present when there were mobility shifts in tumour bands in at least one marker; as only five markers were used some cases could be microsatellite stable tumours. Samples with MI were excluded from the studies.
Figure 1.
Photographs of gels taken under normal light after staining with an (NO3)Ag method, showing examples of loss of heterozygosity at different loci studied in the BRCA1 gene locus (17q21 region). N, normal tissue; T, tumour. Arrows indicate allelic loss in tumour DNA.
Statistical analyses
Variables analysed were compared using the χ2 test with Yates’ correction, or Fisher’s exact test when any of the expected frequencies was less than 5. Two tailed p values <0.05 were considered statistically significant. Univariate survival analyses were performed to determine which independent variables were predictors of recurrence. The survival distributions were calculated using the Kaplan-Meier product limit method. Overall survival time was defined as the interval between the date of diagnosis and tumour related death, or the last visit if the patient survived. Hypotheses on the differences between groups were tested using the log rank test. Multivariate analysis used Cox’s model to identify the factors involved in the outcome. Statistical analysis used the SPSS v10.0 for Windows (computer software package; SPSS Inc., USA). Relative risk was calculated for all factors, with 95% confidence interval (CI).
RESULTS
In this series of 314 patients, 26 (8.3%) showed MI in at least one of the polymorphic markers studied and were eliminated from the study to avoid misinterpretation of the results (table 1 ▶); 244 (84.7%) of the 288 eligible cases were informative.
Table 1.
Distribution of microsatellite instability (MI) for BRCA1 and TP53 loci
| Marker | MI (%) |
| D17S855 | 7 (27) |
| D17S1322 | 5 (19) |
| D17S1323 | 8 (31) |
| TP53.6 | 0 (0) |
| D17S786 | 6 (23) |
LOH rate and clinicopathological characteristics
Of the 244 informative cases, 97 patients (39.8%) showed LOH in at least one of the markers used. Results for each locus are summarised in table 2 ▶.
Table 2.
Informativity and loss of heterozygosity (LOH) for each marker at the BRCA1 gene locus
| Marker | Informative cases | LOH (%) |
| D17S855 | 178 | 62 (34.8) |
| D17S1322 | 167 | 55 (32.9) |
| D17S1323 | 124 | 44 (35.5) |
| Overall I* | 244 | 97 (39.8) |
| Overall II† | 103 | 41 (39.8) |
| Overall III‡ | 52 | 18 (34.6) |
*Patients with at least one informative marker; †patients with two informative markers; ‡patients with three informative markers.
The sex distribution in our series of informative patients was as follows: 146 (60%) were male, of whom 58 (39.7%) showed LOH; and 98 (40%) were female, with 39 (39.7%) showing LOH. Median age was 70 (11) years for the entire series, and 71 (11) years and 70 (10) years for patients with and without LOH, respectively. CEA levels and histopathological tumour characteristics and their distribution are summarised in table 3 ▶. When the incidence of LOH in each of the subgroups was analysed in terms of pathological characteristics, no significant differences were observed (tables 3 ▶, 4 ▶).
Table 3.
Relationship between allelic loss at the TP53 gene locus and clinicopathological characteristics
| Characteristic | Informative tumours | With LOH (%) | Without LOH (%) | p Value |
| Patients (n) | 159 | 78 (49) | 81 (51) | — |
| Age (y)* | 71 (9.8) | 70.5 (9.8) | 71 (10) | — |
| Sex | ||||
| Male | 96 (60.5) | 51 (53) | 45 (47) | |
| Female | 63 (39.5) | 27 (43) | 36 (57) | 0.26 |
| Tumour side | ||||
| Right | 39 (23.9) | 20 (53) | 18 (47) | |
| Left | 60 (37.8) | 32 (53) | 28 (47) | |
| Rectum | 61 (38.3) | 26 (43) | 35 (57) | 0.44 |
| Stage | ||||
| I | 13 (7.9) | 3 (23) | 10 (77) | |
| II | 77 (48.7) | 44 (57) | 33 (43) | |
| III | 59 (36.8) | 26 (44) | 33 (56) | |
| IV | 10 (6.6) | 5 (50) | 5 (50) | 0.10 |
| Vascular invasion | ||||
| Yes | 79 (49.5) | 33 (42) | 46 (58) | |
| No | 80 (50.5) | 45 (56) | 35 (44) | 0.09 |
| Polyps | ||||
| + | 52 (32.7) | 29 (56) | 23 (44) | |
| − | 107 (67.3) | 49 (48) | 58 (52) | 0.31 |
| CEA | ||||
| 0–5 ng/ml | 76 (48.1) | 37 (49) | 39 (51) | |
| > 5 ng/ml | 28 (17.7) | 14 (50) | 14 (50) | 0.09 |
| Unknown | 55 (34.2) | 27 (49) | 28 (51) | |
| Lymph node metastases | ||||
| Negative | 98 (61.5) | 50 (51) | 48 (49) | |
| 1–3 | 41 (25.9) | 21 (51) | 20 (49) | 0.40 |
| 4 | 20 (12.6) | 7 (35) | 13 (65) | |
| Tumour differentiation | ||||
| Well | 56 (35) | 26 (46) | 30 (54) | |
| Moderate | 76 (48) | 42 (55) | 34 (45) | 0.23 |
| Poor | 27 (17) | 10 (37) | 17 (63) | |
*Values are median (SEM). All other values are number (%).
LOH, loss of heterozygosity; CEA, carcinoembryonic antigen.
Table 4.
Relationship between allelic loss at the BRCA1 gene locus and clinicopathological characteristics
| Characteristic | Informative tumours | With LOH (%) | Without LOH (%) | p Value |
| Patients (n) | 244 | 97 (39.8) | 147 (60.2) | — |
| Age (y)* | 70 (11) | 71 (11) | 70 (10) | — |
| Sex | ||||
| Male | 146 (60) | 58 (39.7) | 88 (60.2) | |
| Female | 98 (40) | 39 (39.7) | 59 (60.3) | 0.84 |
| Tumour side | ||||
| Right | 55 (22.5) | 18 (32.7) | 37 (67.3) | |
| Left | 100 (41) | 45 (45) | 55 (55) | |
| Rectum | 89 (36.5) | 34 (38.3) | 55 (61.7) | 0.30 |
| Stage | ||||
| I | 30 (12.3) | 11 (36.7) | 19 (63.3) | |
| II | 102 (41.8) | 36 (35.3) | 66 (64.7) | |
| III | 94 (38.5) | 40 (42.6) | 54 (57.4) | |
| IV | 18 (7.4) | 10 (55.6) | 8 (44.4) | 0.37 |
| Vascular invasion | ||||
| Yes | 118 (48.3) | 46 (39) | 72 (61) | |
| No | 126 (51.7) | 51 (40.5) | 75 (59.5) | 0.91 |
| Polyps | ||||
| + | 75 (30.8) | 29 (39) | 46 (61) | |
| − | 169 (69.2) | 69 (40.5) | 100 (59.5) | 0.81 |
| CEA | ||||
| 0–5 ng/ml | 113 (46.3) | 44 (40) | 69 (60) | |
| >5 ng/ml | 46 (18.9) | 18 (39) | 28 (61) | 0.94 |
| Unknown | 85 (34.8) | 35 (40) | 50 (60) | |
| Lymph node metastases | ||||
| Negative | 137 (56.1) | 53 (38.7) | 84 (61.3) | |
| 1–3 | 71 (29.1) | 30 (42.3) | 41 (57.7) | 0.87 |
| ⩾4 | 36 (14.8) | 14 (38.9) | 22 (61.1) | |
| Tumour differentiation | ||||
| Well | 76 (31.1) | 27 (35.5) | 49 (64.5) | |
| Moderate | 124 (50.8) | 49 (39.5) | 75 (60.5) | 0.42 |
| Poor | 44 (18.1) | 21 (47.7) | 23 (52.3) | |
*Values are median (SEM). All other values are number (%).
LOH, loss of heterozygosity; CEA, carcinoembryonic antigen.
Relation to TP53 LOH status
After screening of the tumour samples with the two polymorphic markers of the TP53 gene region, 159 (50.8%) cases were informative, of which 78 (49%) tumours showed LOH. In these cases a correlation study between clinicopathological characteristics and LOH at the locus of this gene, similar to that performed for BRCA1, was done. No significant differences were observed (table 3 ▶). Informativity was detected in 130 (41.4%) samples for both regions, BRCA1 and TP53, but no significant association was demonstrated between allelic deletions in the two regions, suggesting that chromosome 17 is not lost in the majority of cases (table 5 ▶).
Table 5.
Relationships between loss of heterozygosity (LOH), and status of BRCA1 and TP53 gene regions
| Informative cases: 130 | TP53 region | p Value | ||
| With LOH | Without LOH | |||
| BRCA1 region | With LOH | 23 | 20 | 0.66 |
| Without LOH | 43 | 44 | ||
LOH, loss of heterozygosity.
Recurrence, disease free survival, and overall survival analyses for BRCA1 LOH
As no significant association between LOH and tumour pathological characteristics was found, the relationship of allelic loss with disease relapse, DFS, and overall survival was studied.
Adjuvant chemotherapy with combinations of 5-fluorouracil plus levamisole or 5-fluorouracil plus leucovorin was administered to 38 (39.6%) of the informative stage III patients. In these cases the recurrence rate was 12 (31.5%), five cases with LOH and seven without LOH. No adjuvant chemotherapy was dispensed to any patient with stage II disease. In contrast, a high proportion of stage IV patients received systemic chemotherapy.
The recurrences observed among the informative cases with complete follow up (n = 214) for stages I, II, and III were 51 (23.8%), with a distribution for LOH as follows: 31 (60.7%) showed LOH and 20 (39.3%) no LOH (p = 0.0003). Analysis of recurrences showed significant differences in the presence of LOH in stages I and II, which disappeared at stage III (table 6 ▶).
Table 6.
Recurrences overall and by stage (I, II, and III) compared with the status of BRCA1 loss of heterozygosity (LOH)
| Informative cases (%) | With LOH (%) | Without LOH (%) | p Value | |
| Overall recurrences | ||||
| Yes | 51 (23.8) | 31 (60.8) | 20 (39.2) | 0.0003 |
| No | 163 (76.2) | 53 (32.5) | 110 (67.5) | |
| Stage I | ||||
| Yes | 5 (17.2) | 5 (100) | 0 | 0.008 |
| No | 24 (82.8) | 6 (25) | 18 (7.5) | |
| Stage II | ||||
| Yes | 14 (14.6) | 10 (71.4) | 4 (28.6) | 0.01 |
| No | 82 (85.4) | 26 (31.7) | 56 (68.3) | |
| Stage III | ||||
| Yes | 32 (36) | 16 (50) | 16 (50) | 0.32 |
| No | 57 (64) | 21 (36.8) | 36 (73.2) | |
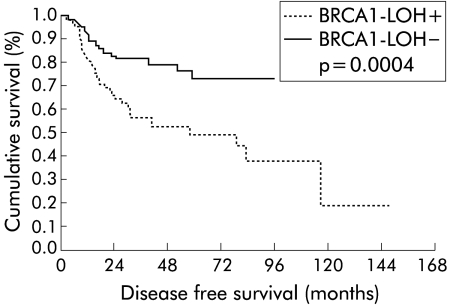
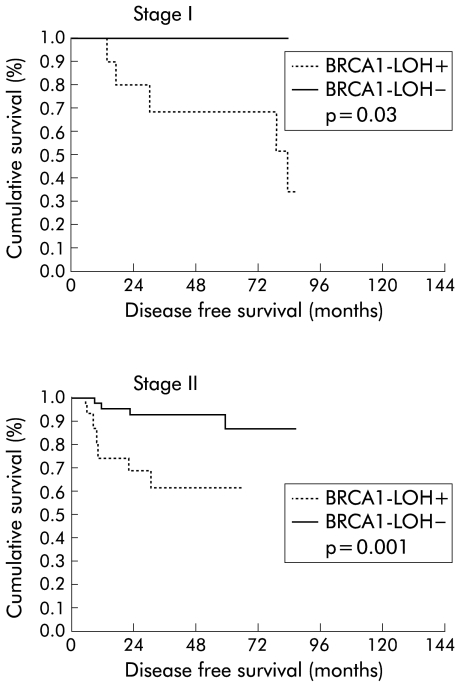
DFS of patients was 63.3% (SEM 4.6) at five years. The presence of LOH had a statistically significant impact on DFS (p = 0.0004), with a DFS rate of 73.3% (SEM 5.7) at five years in patients without LOH and 49.2% (SEM 7.1) in cases with positive allelic loss (fig 2 ▶). In the study by stages of DFS and LOH status, the Kaplan-Meier curves established the following results; in stage I, a difference between patients with LOH and patients without allelic loss (p = 0.03) was observed; in stage II, a clearer difference than in stage I was determined, with shorter DFS in patients with LOH (p = 0.001); and in stage III, these differences disappeared (p = 0.38) (fig 3 ▶).
Figure 2.
Kaplan-Meier disease free survival curves for the entire series, in relation to BRCA1 loss of heterozygosity (LOH).
Figure 3.
Kaplan-Meier disease free survival curves for patients with stage I and II disease, with reference to allelic loss (LOH) at the BRCA1 locus.
When DFS was analysed for pathological tumour characteristics, tumour differentiation, vascular invasion, stage, lymph node metastases, and tumour location, stage III (p = 0.007) and lymph node metastases (p = 0.001) were found, as expected, to affect DFS.
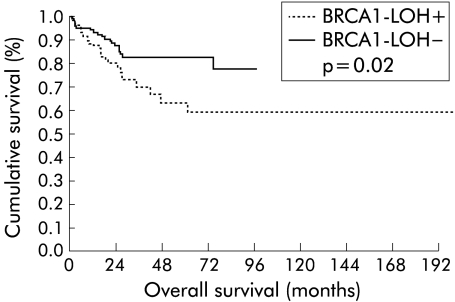
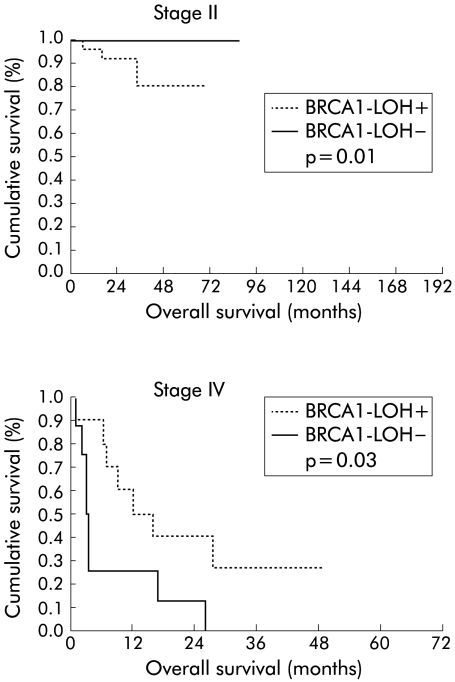
Overall survival of patients was 75% at five years (95% CI 67%–83%). When this survival was analysed for the status of LOH at the BRCA1 locus, we found a significant difference between cases with LOH and without allelic loss (p = 0.02) of 83% (95% CI 75%–91%) and 64% (95% CI 50%–78%), respectively (fig 4 ▶). Analysis of survival by stages revealed that in stages I and III there were no differences for the presence or absence of LOH, but that LOH was present in stage II (p = 0.01). In stage IV, the significance was inverted (p = 0.03) in favour of cases without BRCA1 LOH; of 15 patients who died, eight had no allelic loss. Median survival was 3.17 (0.3) months in patients without BRCA1 LOH and 12.2 (5) months in cases with BRCA1 LOH (fig 5 ▶).
Figure 4.
Kaplan-Meier curves of overall survival of the entire series for retention of the BRCA1 alleles. LOH, loss of heterozygosity.
Figure 5.
Kaplan-Meier overall survival curves for patients with stage II and IV disease, with the presence or absence of BRCA1 loss of heterozygosity (LOH) in their tumours.
Univariate and multivariate analyses
A univariate analysis was performed to determine the influence of the molecular and pathological variables on DFS. It revealed that of the six variables analysed, only pathological stage, BRCA1 LOH, and lymph node metastases had a significant prognostic impact (table 7 ▶).
Table 7.
Disease free survival (univariate Cox’s regression model)
| Variable | Category | RR | 95% CI | p Value |
| BRCA1 LOH | Positive v negative | 2.54 | 1.45–4.48 | 0.001 |
| Vascular invasion | Yes v no | 1.33 | 0.76–2–36 | 0.31 |
| Tumour side | Left v right | 1.35 | 0.6–3 | 0.46 |
| Rectum v right | 1.31 | 0.66–3 | 0.52 | |
| Stage | II v I | 1.07 | 0.38–3 | 0.88 |
| III v I | 2.43 | 0.94–6.26 | 0.05 | |
| Tumour differentiation | Moderate v well | 1.09 | 0.57–2.08 | 0.78 |
| Poor v well | 1.64 | 0.75–3.60 | 0.21 | |
| Lymph node metastases | 1–3 v 0 | 1.90 | 1.02–3.54 | 0.04 |
| ⩾4 v 0 | 3.44 | 1.63–7.25 | 0.001 |
RR, risk ratio; CI, confidence interval; LOH, loss of heterozygosity.
In the multivariate Cox’s regression model for DFS, the variables that showed an independent prognostic factor were as follows: stage (p = 0.006); ⩾4 positive lymph nodes (p = 0.0001); and 1–3 lymph nodes affected (p = 0.038) (table 8 ▶). However, when we performed specifically the multivariate analysis stratified by stages, the only variable observed with independent prognostic value for DFS was BRCA1 LOH in stages I and II. Patients with BRCA1 LOH had a risk ratio of recurrence of 7.04 (95% CI 2.3–21.3; p = 0.001).
Table 8.
Factors influencing recurrence (multivariate Cox’s regression model)
| Variable | Category | RR | 95% CI | p Value |
| Stage | III v I | 4.6 | 1.56–13.56 | 0.006 |
| Lymph node metastases | ⩾4 v 0 | 3.83 | 1.81–8.11 | 0.0001 |
| 1–3 v 0 | 0.44 | 0.21–0.95 | 0.038 |
RR, risk ratio; CI, confidence interval; LOH, loss of heterozygosity.
DISCUSSION
The current prognostic factor in CRC, of proven practical clinical use, is pathological staging. Consequently, five year survival of stage I patients, close to 90%, contrasts with less than 10% for stage IV patients. However, between 50% and 80% of patients in stage II can reach five year survival, with a disease rate recurrence of 20%–30%.1 In patients with stage III disease, five year survival decreases to 30%–50%, although it is possible to achieve a significant improvement in survival with adjuvant chemotherapy based on 5-fluorouracil and levamisole27 or 5-fluorouracil and leucovorin.28
The controversy about adjuvant treatment in stage II CRC is currently unresolved. The IMPACT B2 study pooled results from five trials in Dukes’ B2 colon cancer patients. These did not support any benefit in five year overall survival; 80% in the control group and 82% in the 5-fluorouracil and leucovorin group.29 However, the four NSABP adjuvant studies showed a decreased risk of death with adjuvant treatment similar to the benefit obtained in stage C.30 Another Dutch study reported a beneficial effect of 5-fluorouracil and levamisole adjuvant therapy in stage II patients, similar to the expected benefit in stage III.31 In stage II patients with high risk clinicopathological characteristics (intestinal obstruction, perforation, tumour adherence, poor differentiation, vascular or lymphatic invasion), adjuvant therapy can reasonably be offered.
The 1994 communication by the Breast Cancer Linkage Consortium21 of an increased risk of CRC and prostate cancer in members of linked BRCA1 families led to new research into BRCA1 gene alterations in these malignancies. As a result, it has been found that some loci on 17q21, including the BRCA1 locus, are lost in human prostate cancer.32,33 In a similar study we detected22 a high proportion of allelic deletions in the 17q21 region in a series of CRCs. However, loss of BRCA1 function, if it exists, may be caused by other genetic or epigenetic mechanisms, such as somatic mutations of BRCA1, which are unlikely, or hypermethylation of BRCA1 promoter, as has been described in breast and ovarian cancers.34
The overall LOH observed in 97 (39.8%) cases, although lower than in a previous study using seven polymorphic markers,22 is consistent with the percentage of LOH reported by some studies of breast cancer,35,36 which is considered the prototype tumour for BRCA1 gene analyses. Considering that the three polymorphic markers used are intragenic to BRCA1, it is impossible to ensure that other genes surrounding the BRCA1 locus are not involved in CRC, as has been suggested in other reports on LOH on chromosome 17 in CRC.37,38 Because the molecular target is located at chromosome 17, BRCA1 locus, we judged that TP53 could be the gene functionally most related to CRC. We thus attempted to assess the level of association with LOH at the BRCA1 locus. It was important to establish that the loss at locus TP53 or even the loss of the entire chromosome 17 concomitant with the loss at BRCA1 was not a statistically significant association.
Analysis of DFS and overall survival showed the most interesting results. The difference in the entire series for DFS (except stage IV cases) between patients with BRCA1 LOH and patients without allelic losses was significant, a difference that was also conserved in the individual analysis by stage, except for stage III. It is possible that at stage III, the biological effect of loss of alleles at the BRCA1 locus, if it occurred, was surmounted by other molecular changes. The results of the univariate analysis support this possible influence of LOH at BRCA1, because this molecular alteration displayed a risk ratio for DFS similar to that expressed by the recognised prognostic parameters of colon cancer, such as advanced stage and lymph node metastases.1 It is important to emphasise that although the multivariate study of the whole series did not show LOH in BRCA1 to be a determining factor in DFS, the study by stages showed that this variable was an independent prognostic factor in stages I and II.
Our BRCA1 LOH status results for survival, overall and by stage, were in parallel with the DFS results, except for stage IV. At this stage, the improved survival of cases with LOH at BRCA1 appears paradoxical, and could be secondary to other mechanisms. Stage IV tumours may be the last stage in tumour progression, with accumulation of molecular alterations,39 or some stage IV tumours may be different forms of the disease with a specific molecular signature, as observed in comparative genomic hybridisation studies.40 In both situations it is conceivable that the particular molecular status may influence treatment response and patient survival.
Studies analysing the impact of several molecular alterations on DFS and overall survival in CRC suggest that genes mapped at regions 18q, 17p, and 5q can be considered potential molecular prognostic indicators.14,41–44 Some authors have documented that stage II tumours have a similar prognosis to stage III when they present T4 lesions, a high S phase, or 18q deletions.41 The 18q region can represent an important genetic cluster in CRC because three genes located in this region are currently potential gene targets: DCC, SMAD4, and SMAD2.45 Thus knowledge of the functional significance of BRCA1 protein is increasing,17 and some of its demonstrated functions, such as regulation of p53 responsive genes (p21Waf1),46 control of recombination and genome integrity,16 or coactivator for p53,18 if lost or decreased, could be involved in processes of tumorigenesis in general, not only in breast carcinomas.
Adjuvant chemotherapy is one of the main factors affecting DFS in stage III. Although no significant difference in DFS in stage III arose from BRCA1 LOH status, if adjuvant chemotherapy is given, such a difference cannot be ruled out. Nevertheless, among the 38 stage III patients receiving adjuvant chemotherapy, the proportion of recurrences detected was not significantly different from the remaining stage III patients not receiving adjuvant treatment, although it is possible that the small number of patients analysed may have influenced this result. However, other studies have claimed a possible influence of allelic deletions on the chemotherapy effect although results have not been conclusive.14,42,44 Further studies are required.
These results on the proportion of recurrences, and the improvement in DFS and overall survival in patients with retention of BRCA1 alleles, suggest that BRCA1 LOH may be an independent prognostic factor in patients with CRC, in stages I and II. It may well have a use in stage II to identify patient subgroups at high risk with a poor prognosis who might benefit from adjuvant chemotherapy.
Acknowledgments
We are grateful to Robin Rycroft for his assistance with the English, and E Fernandez, MC Farelo, and PC Zalallos for help with the collection of tissue samples. Supported by grants from Fundación Banco Santander Central Hispano, SEOM, and Bristol-Myers, SA.
Abbreviations
CRC, colorectal cancer
LOH, loss of heterozygosity
DFS, disease free survival
CEA, carcinoembryonic antigen
PCR, polymerase chain reaction
MI, microsatellite instability
REFERENCES
- 1.Cohen AM, Minsky BD, Schilsky RL. Cancer of the colon. In: Devita VT jr, Hellman S, Rosemberg SA, eds. Cancer principles and practice of oncology. Philadelphia: Lippincott-Raven, 1997:1144–97.
- 2.Deans GT, Parks TG, Rowlands BJ, et al. Prognostic factors in colorectal cancer. Br J Surg 1992;79:608–13. [DOI] [PubMed] [Google Scholar]
- 3.Silverberg E, Boring CE, Squires TS. Cancer statistics. CA Cancer J Clin 1990;40:9–26. [PubMed] [Google Scholar]
- 4.Vogelstein B, Fearon ER, Hamilton SR, et al. Genetics alterations during colorectal-tumor development. N Engl J Med 1988;319:525–32. [DOI] [PubMed] [Google Scholar]
- 5.Delattre O, Olschwang S, Law DJ, et al. Multiple genetics alterations in distal and proximal colorectal cancer. Lancet 1989;12:353–6. [DOI] [PubMed] [Google Scholar]
- 6.Vogelstein B, Fearon ER, Kern SE, et al. Allelotype of colorectal carcinoma. Science 1989;244:207–11. [DOI] [PubMed] [Google Scholar]
- 7.Solomon E, Voss R, Hall V, et al. Chromosome 5 allele loss in human colorectal carcinomas. Nature 1987;328:616–19. [DOI] [PubMed] [Google Scholar]
- 8.Ashton-Rickardt PG, Dunlop MG, Nakamura Y, et al. High frequency of APC loss in sporadic colorectal carcinoma due to breaks clustered in 5q21–22. Oncogene 1989;4:1169–74. [PubMed] [Google Scholar]
- 9.Fearon ER, Cho KR, Nigro JM, et al. Identification of a chromosome 18q gene that is altered in colorectal carcinoma. Science 1990;247:49–56. [DOI] [PubMed] [Google Scholar]
- 10.Baker SJ, Fearon ER, Nigro JM, et al. Chromosome 17 deletions and p53 gene mutations in colorectal carcinoma. Science 1989;244:217–21. [DOI] [PubMed] [Google Scholar]
- 11.Kellsell DP, Spurr NK, Barnes DM, et al. Combined loss of BRCA1/BRCA2 in grade 3 breast carcinomas. Lancet 1996;347:1554–5. [DOI] [PubMed] [Google Scholar]
- 12.Backmann MW, Picard F, An HX, et al. Clinical impact of loss of heterozygosity of BRCA1 and BRCA2 markers in sporadic breast cancer. Br J Cancer 1996;73:1220–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Silva JM, Gonzalez R, Provencio M, et al. Loss of heterozygosity in BRCA1 and BRCA2 markers and high-grade malignancy in breast cancer. Breast Cancer Res Treat 1999;53:9–17. [DOI] [PubMed] [Google Scholar]
- 14.Kern SE, Fearon ER, Tersmette KW F, et al. Clinical and pathological associations with allelic loss in colorectal carcinoma. JAMA 1989;261:3099–103. [DOI] [PubMed] [Google Scholar]
- 15.Martínez-López E, Abad A, Monzó M, et al. Allelic loss on chromosome 18q as a prognostic marker in stage II colorectal cancer. Gastroenterology 1998;114:1180–7. [DOI] [PubMed] [Google Scholar]
- 16.Chapman MS, Verma MI. Transcriptional activation by BRCA1. Nature 1996;382:678–9. [DOI] [PubMed] [Google Scholar]
- 17.Scully R, Chen J, Plug A, et al. Association of BRCA1 and Rad51 in mitotic and meiotic cells. Cell 1997;88:265–75. [DOI] [PubMed] [Google Scholar]
- 18.Scully R, Livingston DM. In search of the tumor-suppressor functions of BRCA1 and BRCA2. Nature 2000;408:429–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ouchi T, Monteiro AN A, August A, et al. BRCA1 regulates p53-dependent gene expression. Proc Natl Acad Sci U S A 1998;95:2302–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Newman B, Mu H, Butler LM, et al. Frequency of breast cancer attributable to BRCA1 in a population-based series of American women. JAMA 1998;279:915–21. [DOI] [PubMed] [Google Scholar]
- 21.Ford D, Easton D, Bishop D, et al. Risk of cancer in BRCA1-mutation carriers. Lancet 1994;343:692–5. [DOI] [PubMed] [Google Scholar]
- 22.García-Patiño E, Gomendio B, Lleonart M, et al. Loss of heterozygosity in the region including the BRCA1 gene on 17q in colon cancer. Cancer Genet Cytogen 1998;104:19–23. [DOI] [PubMed] [Google Scholar]
- 23.Smith TM, Lee MK, Szabo CI, et al. Complete genomic sequence and analysis of 117 kb of human DNA containing the gene BRCA1. Genome Res 1996;6:1029–49. [DOI] [PubMed] [Google Scholar]
- 24.Oto M, Miyake S, Yuasa Y. Optimization of nonradioisotopic single strand conformation polymorphism analysis with a conventional minislab gel electrophoresis apparatus. Ann Biochem 1993;213:19–22. [DOI] [PubMed] [Google Scholar]
- 25.Deng G, Lu Y, Zlotnikov G, et al. Loss of heterozygosity in normal tissue adjacent to breast carcinomas. Science 1996;274:2057–9. [DOI] [PubMed] [Google Scholar]
- 26.Werness BA, Parvatiyar P, Ramus SJ, et al. Ovarian carcinoma in situ with germline BRCA1 mutation and loss of heterozygosity at BRCA1 and TP53. J Nat Cancer Inst 2000;92:1088–91. [DOI] [PubMed] [Google Scholar]
- 27.NIH consensus conference. Adjuvant therapy for patients with colon and rectal cancer. JAMA 1990;264:1444–50. [PubMed] [Google Scholar]
- 28.International Multicentre Pooled Analysis of Colon Cancer Trials (IMPACT) investigators. Efficacy of adjuvant fluorouracil and folinic acid in colon cancer. Lancet 1995;345:939–44. [PubMed] [Google Scholar]
- 29.International Multicentre Pooled Analysis of B2 Colon Cancer Trials (IMPACT B2) Investigators. Efficacy of adjuvant fluorouracil and folinic acid in B2 colon cancer. J Clin Oncol 1999;17:1356–63. [PubMed] [Google Scholar]
- 30.Mamounas E, Wieand S, Wolmark N, et al. Comparative efficacy of adjuvant chemotherapy in patients with Dukes’ B versus Dukes’ C colon cancer: Results from four National Surgical Adjuvant Breast and Bowel Project adjuvant studies (C-01, C-02, C-03, and C-04). J Clin Oncol 1999;17:1349–55. [DOI] [PubMed] [Google Scholar]
- 31.Taal BG, Van Tinteren H, Zoetmulder FA. Adjuvant 5FU plus levamisole in colonic or rectal cancer: Improved survival in stage II and III. Br J Cancer 2001;85:1437–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gao X, Zacharek A, Salkowski A, et al. Loss of heterozygosity of the BRCA1 and other loci on chromosome 17p in human prostate cancer. Cancer Res 1995;55:1002–5. [PubMed] [Google Scholar]
- 33.Gao X, Zacharek A, Grignon DJ, et al. Localization of potential tumor suppressor loci to a <2 Mb region on chromosome 17q in human prostate cancer. Oncogene 1995;11:1241–7. [PubMed] [Google Scholar]
- 34.Esteller M, Silva JM, Domingez G, et al. Promoter hypermethylation and BRCA1 inactivation in sporadic breast and ovarian tumors. J Natl Cancer Inst 2000;92:564–9. [DOI] [PubMed] [Google Scholar]
- 35.Cropp CS, Nevanlinna HA, Pyrhönen S, et al. Evidence for involvement of BRCA1 in sporadic breast carcinoma. Cancer Res 1994;54:2548–51. [PubMed] [Google Scholar]
- 36.Gonzalez R, Silva JM, Dominguez G, et al. Detection of loss of heterozygosity at Rad51, Rad52, Rad54 and BRCA1 and BRCA2 loci in breast cancer: pathological correlations. Br J Cancer 1999;81:503–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leggett B, Young J, Buttenshaw R. Colorectal carcinomas show frequent allelic loss on the long arm of chromosome 17 with evidence for a specific target region. Br J Cancer 1995;71:1070–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Campo E, Miquel R, Jares P, et al. Prognostic significance of the loss of heterozygosity of NM23-H1 and p53 genes in human colorectal carcinomas. Cancer 1994;73:2913–21. [DOI] [PubMed] [Google Scholar]
- 39.Claij N, de Riele H. Microsatellite instability in human cancer: A prognostic marker for chemotherapy? Exp Cell Res 1999;246:1–10. [DOI] [PubMed] [Google Scholar]
- 40.Al-Mulla F, Keith WN, Pickford IR, et al. Comparative genomic hybridization analysis of primary colorectal carcinomas and their synchronus metastases. Genes Chromosom Cancer 1999;24:306–14. [DOI] [PubMed] [Google Scholar]
- 41.Jen J, Kim H, Piantadosi S, et al. Allelic loss of chromosome 18q and prognosis in colorectal cancer. N Engl J Med 1994;331:213–21. [DOI] [PubMed] [Google Scholar]
- 42.Watanabe T, Wu TT, Catalano PJ, et al. Molecular predictors of survival after adjuvant chemotherapy for colon cancer. N Engl J Med 2001;344:1196–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cohn KH, Ornstein DL, Wang F, et al. The significance of allelic deletions and aneuploidy in colorectal carcinoma. Cancer 1997;79:233–44. [PubMed] [Google Scholar]
- 44.Carethers JM, Hawn JK, Greenson JK, et al. Prognostic significance of allelic loss at chromosome 18q21 for stage II colorectal cancer. Gastroenterology 1998;114:1188–95. [DOI] [PubMed] [Google Scholar]
- 45.Eppert K, Scherer SW, Ozcelik H, et al. MADR2 maps to 18q21 and encodes a TGF-beta-regulated SMAD-related protein that is functionally mutated in colorectal carcinoma. Cell 1996;86:543–52. [DOI] [PubMed] [Google Scholar]
- 46.Somasundaram K, Zhang H, Zeng Y- X, et al. Arrest of the cell cycle by the tumor-suppressor BRCA1 requires the CDK-inhibitor p21WAF1/CIP. Nature 1997;389:187–90. [DOI] [PubMed] [Google Scholar]