Colorectal cancer develops in approximately 5–6% of the adult population and is one of the leading causes of cancer death in Europe and the USA.1,2 Screening colonoscopy is the widely accepted gold standard for early diagnosis of colorectal cancer and should be offered to patients older than 50 years.3–5 However, colonoscopy is increasingly in competition with computed tomographic virtual endoscopy.6 Therefore, it is essential to understand whether total colonoscopy rather than virtual endoscopy enables the detection of all relevant premalignant and malignant lesions as only early diagnosis of neoplasias offers the possibility for curative endoscopic or surgical resection.
Polypoid lesions are easy to detect by endoscopy. In contrast, non-polypoid lesions are often overlooked.7 The endoscopist must be aware of flat and depressed lesions because the primarily visible mucosal changes of such lesions are often very discrete. In this context, the endoscopist should look for slight colour changes, interruption of the capillary network pattern, slight deformation of the colonic wall, spontaneously bleeding spots, shape changes of the lesion with insufflation or deflation of air, and interruption of the innominate grooves.7
An important diagnostic tool for detection of non-polypoid colorectal lesions consists of chromoendoscopy.8 In contrast with ulcerative colitis where pan-chromoendoscopy is favourable,9 targeted staining with indigo carmine or methylene blue in a selective fashion is sufficient for non-polypoid colorectal lesions.10 Chromoendoscopy helps to unmask such lesions in the colon and to delineate its borders. By the help of magnifying endoscope, the stained surface and crypt architecture can be analysed, and differentiation between non-neoplastic and neoplastic lesions becomes possible with high accuracy.11 Recently, it was shown in a prospective randomised trial that magnifying endoscopy is superior to standard videoendoscopy because of the possibility of analysing stained colorectal lesions at higher magnifications.12 Differentiation between non-neoplastic and neoplastic lesions was possible however when using high magnification chromoendoscopy to differentiate between non-invasive and invasive neoplasias but sensitivity decreased to 50%.10
Initially, knowledge about non-polypoid colorectal lesions was based on observations in Japanese patients.16 Similar observations have since been made in Western populations using magnifying chromoendoscopy. In fact, introduction of newly developed magnifying and high resolution endoscopes, as well as the renaissance of chromoendoscopy, have led to a significant increase in the detection of non-polypoid lesions in the Western world. In recent prospective chromoendoscopic studies, 7–31% of diagnosed lesions were flat or depressed.8,13–15 Furthermore, Saitoh and colleagues8 observed that 62% of flat and depressed lesions could be judged correctly by the help of intravital staining. Thus Western endoscopists have to recognise these newly discovered lesions. For this reason, it is essential to discuss the malignant potential, screening, and therapeutic strategies of such non-polypoid lesions.
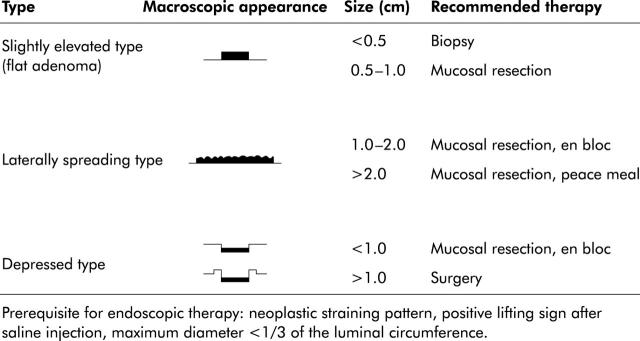
Macroscopic appearance of colorectal lesions can be classified according to the Japanese classification of colorectal cancer (see fig 1 ▶).17 However, Kudo et al have recommended a more practical classification.7 Here, early non-polypoid colorectal neoplasias can be classified as slightly elevated (small flat adenoma <1 cm), laterally spreading tumours (large flat adenoma >1 cm) or depressed (see fig 2 ▶). Specifically, laterally spreading tumours (LST) of the colorectum are defined as lesions of more than 10 mm in diameter with a low vertical axis which extends laterally along the interior luminal wall. Such LSTs can be further subdivided into granular and non-granular types.7
Figure 1.
Diagnostic classification17 and malignant potential of early colorectal cancer.
Figure 2.
Classification of non-polypoid colorectal lesions7 and recommended therapy.
In this issue of Gut, Hurlstone and colleagues18 report their experience with colonoscopic resection of 82 LSTs of the colorectum (see page 1334). Firstly, identification of these lesions was made with the help of dye spraying, underlining the high impact of targeted chromoendoscopy during screening colonoscopy. Using this approach, a total of 26 non-granular and 56 granular LSTs were identified. The non-granular type of LSTs was more often associated with invasive disease and was frequently localised to the right colon.
The theory of the adenoma-carcinoma sequence is well established for polypoid adenomas.19 Some authors have advocated a new theory of cancer development with special attention to the superficial forms of cancer that exhibit non-polypoid growth. They postulated a higher risk of malignant invasion compared with polypoid neoplasms.20,21 Hurlstone and colleagues15 observed areas of high grade dysplasia in 25% of flat adenomas whereas high grade dysplasia was observed in only 12% of protuberant adenomas, consistent with the idea that such flat lesions have a high risk for malignancy. However, previous studies suggested that among non-polypoid lesions, only depressed lesions have a higher risk of malignant infiltration. Up to a size of 2 cm, the rate of malignant infiltration is 40% for depressed lesions compared with only 1.3% for polypoid lesions.7 Surprisingly, the rate of lymph node metastasis is similar for polypoid (11%) and non-polypoid lesions (11%). Even for depressed lesions with the tendency to grow towards the muscularis propria, the rate of lymph node metastasis has been reported as only 6–7%.22
Early colorectal carcinoma is defined by its depth of infiltration (the carcinoma is invading the submucosal layer). The degree of submucosal invasion is classified into three types based on depth of invasion: when less than one third of the submucosa is invaded the stage is called sm1 and if more than two thirds are invaded the stage is sm3. Stage sm2 is intermediate. Sm1 early colorectal cancers without lymphatic or venous invasion have almost no risk of lymph node metastasis.7 Thus they can be removed endoscopically.
Severe dysplastic epithelium confined to the mucosa is called severe dysplasia or intramucosal carcinoma, although there is some controversy as to the terminology for this particular lesion. Japanese pathologists tend to use the term “intramucosal or mucosal carcinoma” whereas Western pathologists tend to avoid this term to prevent unnecessary major surgery. However, these lesions are ideally suited for endoscopic resection.22
Selection of suitable lesions for endoscopic resection is of crucial clinical importance. Hurlstone et al have developed essential inclusion and exclusion criteria.18 According to these criteria, 56 of 82 identified LSTs underwent endoscopic mucosal resection and high ”cure” rates were achieved. The results show impressively that endoscopic management for LST is possible and justified. However, the question remains how the endoscopic or surgical management of non-polypoid lesions should be performed? Depressed lesions are often malignant and have been shown to rapidly invade the submucosal layer. Therefore, this group of lesions should always be treated. Those confined to the mucosa or that only slightly invade the submucosa (without vessel infiltration) can be completely removed and cured by endoscopic resection. If histology shows that the cancer has massively invaded the submucosa or permeates the vessels, additional surgery is required.
Signs of advanced carcinoma are depressed lesions >10 cm and unstructured crypt architecture after staining, as well as lacking the lifting sign after saline injection. These lesions should undergo surgery primarily.
Flat lesions up to a size of 5 mm without suspected staining pattern or depression can be removed by biopsy. Lesions up to 2 cm should be resected en bloc whereas lesions larger than 2 cm can be treated by peacemeal resection.7,18 Peacemeal resection harbours the risk of local recurrence, as shown by Hurlstone and colleagues,18 and an intense follow up programme should therefore be added (see fig 2 ▶).
Complete resection should always be intended. However, chromoendoscopy after resection helps to identify remnant islands of neoplastic tissue which can be removed by argon beam coagulation.18,23 Hurlstone and colleagues18 reported a local recurrence rate of 17% and recurrent disease in 10%. In patients with local recurrence, eight of 10 underwent further endoscopic resection without residual disease present at 24 months of follow up. These data are comparable with previous Japanese experience.24,25 In the current study, Hurlstone and colleagues18 thus showed that neither the diagnosis nor the treatment of non-polypoid lesions is a Japanese phenomenon or expertise. Western endoscopists should be aware of these lesions and need to include mucosal resection for flat and depressed colorectal lesions into their therapeutic strategies.
In conclusion, introduction of magnifying endoscopy in combination with intravital staining has led to a significant improvement in the diagnosis of non-polypoid neoplasms. Flat and depressed lesions are now increasingly diagnosed in the Western world during colonoscopy using this new technique. In contrast, the diagnostic yield of alternative approaches such as computed tomography virtual endoscopy decreases for small lesions.6,26 Differentiated knowledge about non-polypoid lesions is essential to identify suitable lesions for endoscopic resection. Endoscopic mucosal resection is the treatment of choice for defined non-polypoid lesions and offers a sufficient and curative therapy. Because of the possible higher malignant potency of non-polypoid lesions, an intense endoscopic follow up programme should be initiated and offers the possibility of repeat endoscopic treatment for local recurrence.
REFERENCES
- 1.Grady WM. Genetic testing for high-risk colon cancer patients. Gastroenterology 2003;124:1574–94. [DOI] [PubMed] [Google Scholar]
- 2.Burt RW. Colon cancer screening. Gastroenterology 2000;119:837–53. [DOI] [PubMed] [Google Scholar]
- 3.Gatta G , Ciccolallo L, Capocaccia R, et al. Differences in colorectal cancer survival between European and US populations: the importance of sub-site and morphology. Eur J Cancer 2003;39:2214–22. [DOI] [PubMed] [Google Scholar]
- 4.Weir HK, Thun MJ, Hankey BF, et al. Annual report to the nation on the status of cancer, 1975–2000, featuring the uses of surveillance data for cancer prevention and control. J Natl Cancer Inst 2003;95:1276–99. [DOI] [PubMed] [Google Scholar]
- 5.Winawer S , Fletcher R, Rex D, et al. Colorectal cancer screening and surveillance: clinical guidelines and rationale—Update based on new evidence. Gastroenterology 2003;124:544–60. [DOI] [PubMed] [Google Scholar]
- 6.Pickhardt PJ, Choi JR, Hwang I, et al. Computed tomographic virtual colonoscopy to screen for colorectal neoplasia in asymptomatic adults. N Engl J Med 2003;349:2191–200. [DOI] [PubMed] [Google Scholar]
- 7.Kudo S , Kashida H, Tamura T, et al. Colonoscopic diagnosis and management of nonpolypoid colorectal cancer. World J Surg 2000;24:1081–90. [DOI] [PubMed] [Google Scholar]
- 8.Saitoh Y , Waxman I, West AB, et al. Prevalence and distinctive biologic features of flat colorectal adenomas in a North American population. Gastroenterology 2001;120:1657–65. [DOI] [PubMed] [Google Scholar]
- 9.Kiesslich RJ, Fritsch M, Holtmann M, et al. Methylene blue-aided chromoendoscopy for the detection of intraepithelial neoplasia and colon cancer in ulcerative colitis. Gastroenterology 2003;124:880–8. [DOI] [PubMed] [Google Scholar]
- 10.Hurlstone DP, Cross SS, Adam I, et al. Efficacy of high magnification chromoscopic colonoscopy for the diagnosis of neoplasia in flat and depressed lesions of the colorectum: a prospective analysis. Gut 2004;53:284–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kudo S , Tamura S, Nakajima T, et al. Diagnosis of colorectal tumorous lesions by magnifying endoscopy. Gastrointest Endosc 1996;44:8–14. [DOI] [PubMed] [Google Scholar]
- 12.Konishi K , Kaneko K, Kurahashi T, et al. A comparison of magnifying and nonmagnifying colonoscopy for diagnosis of colorectal polyps: A prospective study. Gastrointest Endosc 2003;57:48–53. [DOI] [PubMed] [Google Scholar]
- 13.Rembacken BJ, Fujii T, Cairns A, et al. Flat and depressed colonic neoplasms: a prospective study of 1000 colonoscopies in the UK. Lancet 2000;8:1211–14. [DOI] [PubMed] [Google Scholar]
- 14.Kiesslich R , von Bergh M, Hahn M, et al. Chromoendoscopy with indigocarmine improves the detection of adenomatous and nonadenomatous lesions in the colon. Endoscopy 2001;33:1001–6. [DOI] [PubMed] [Google Scholar]
- 15.Hurlstone DP, Cross SS, Adam I, et al. A prospective clinicopathological and endoscopic evaluation of flat and depressed colorectal lesions in the United Kingdom. Am J Gastroenterol 2003;98:2543–9. [DOI] [PubMed] [Google Scholar]
- 16.Matsui T , Yao T, Iwashita A. Natural history of colorectal cancer. World J Surg 2000;24:1022–8. [DOI] [PubMed] [Google Scholar]
- 17. Research Society for Cancer of the Colon and Rectum.General rules for clinical and pathological studies on cancer of the colon, rectum and anus. Part II. Histological classification. Jpn J Surg 1983;23:574. [DOI] [PubMed] [Google Scholar]
- 18.Hurlstone DP, Sanders DS, Cross SS, et al. Colonoscopic resection of lateral spreading tumours: a prospective analysis of endoscopic mucosal resection. Gut 2004;53:1334–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Muto T , Bussey HJR, Morson BC. The evaluation of cancer of the colon and rectum. Cancer 1975;36:2251. [DOI] [PubMed] [Google Scholar]
- 20.Kuramoto S , Oohara T. Flat early cancers of the large intestine. Cancer 1989;15:950–5. [DOI] [PubMed] [Google Scholar]
- 21.Minamoto T , Sawaguchi K, Ohta T, et al. Superficial-type adenomas and adenocarcinomas of the colon and rectum: a comparative morphological study. Gastroenterology 1994;106:1436–43. [DOI] [PubMed] [Google Scholar]
- 22.Ajioka Y , Watanabe H, Kazama S, et al. Early colorectal cancer with special reference to the superficial nonpolypoid type from a histopathologic point of view. World J Surg 2000;24:1075–80. [DOI] [PubMed] [Google Scholar]
- 23.Brooker JC, Saunders BP, Shah SG, et al. Treatment with argon plasma coagulation reduces recurrence after piecemeal resection of large sessile colonic polyps: a randomized trial and recommendations. Gastrointest Endosc 2002;55:371–5. [DOI] [PubMed] [Google Scholar]
- 24.Tanaka S , Haruma K, Oka S, et al. Clinicopathologic features and endoscopic treatment of superficially spreading colorectal neoplasms larger than 20 mm. Gastrointest Endosc 2001;54:62–6. [DOI] [PubMed] [Google Scholar]
- 25.Tamura S , Nakajo K, Yokoyama Y, et al. Evaluation of endoscopic mucosal resection for laterally spreading rectal tumors. Endoscopy 2004;36:306–12. [DOI] [PubMed] [Google Scholar]
- 26.Laghi A , Iannaccone R, Carbone I, et al. Detection of colorectal lesions with virtual computed tomographic colonography. Am J Surg 2002;183:124–31. [DOI] [PubMed] [Google Scholar]