Infectious diarrhoea is the most common cause of diarrhoea worldwide and is the leading cause of death in childhood. Gastrointestinal infections have their major impact in the developing world. In the developed world, despite improvements in public health and economic wealth, the incidence of intestinal infection remains high and continues to be an important clinical problem.
During the past 10 years there have been some major improvements in our knowledge base regarding the treatment of infectious diarrhoea. Oral rehydration therapy (ORT) remains central to case management but advances have been made by the introduction of hypotonic solutions and there is early evidence that resistant starch may be the substrate of the future. The search for antisecretory drugs continues, with real progress having been made by the introduction of a new class of drugs, the enkephalinase inhibitors. Other new drugs are in the early phases of development. The role of antimicrobial agents in the management of infective diarrhoea continues to be clarified with the emergence of new agents and simplified treatment regimens. Probiotics are popular with diarrhoea sufferers and have been shown to have some efficacy but further scrutiny is required to determine the magnitude of their effects.
INTRODUCTION
Infectious diarrhoea is the most common cause of diarrhoea worldwide and is responsible for more deaths than gastrointestinal cancers, peptic ulcer, or inflammatory bowel disease. Diarrhoeal disease is the leading cause of childhood death and the second most common cause of death worldwide.
Gastrointestinal infections have their major impact in the developing world: diarrhoeal diseases are responsible, directly or indirectly, for approximately three million deaths each year among children under five years of age—that is, 1 every 10 seconds. There are an estimated 1.8 billion episodes of childhood diarrhoea per year and virtually all of these acute diarrhoeal episodes are related to infectious agents. In some parts of Africa preschool children may suffer up to seven attacks of acute diarrhoea annually, although the average worldwide is approximately three episodes per year.
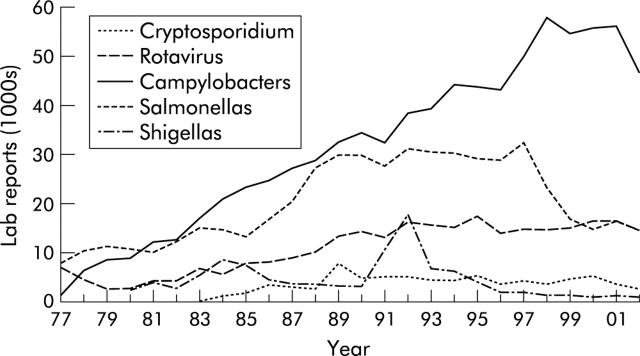
In the developed world, despite improvements in public health and economic wealth, the incidence of intestinal infection remains high and continues to be an important clinical problem, although mortality has fallen sharply in recent decades. In England, 1 in 5 people has an intestinal infection each year, of whom 1 in 6 presents to a general practitioner. Many of these cases are not reported to the Health Protection Agency that has now incorporated the Public Health Laboratory Service.1 In England and Wales, the incidence of gastrointestinal infections appears to have stabilised since the mid-1990s. Salmonella isolates have decreased by 37% since 1998, reaching the lowest recorded annual total since 1985. This may be attributed to the introduction of vaccination of chicken flocks against salmonella. Laboratory reporting of Campylobacter jejuni reached a peak in 1998 and has slowly fallen by 7.5% in 2000 (fig 1 ▶). However, intestinal infections are increasing generally in the western world, notably foodborne infections, such as Salmonella spp, Campylobacter jejuni, and enterohaemorraghic Escherichia coli(EHEC), and waterborne infections such as Giardia intestinalis and Cryptosporidium parvum. However, reductions in foodborne campylobacter, listeria, and yersinia have been recently reported by the Centers for Disease Control and Prevention in the United States.
Figure 1.
Laboratory reporting of selected gastrointestinal pathogens in England and Wales (source: Health Protection Agency).
CAUSES OF INFECTIOUS DIARRHOEA
There are vast numbers of bacteria, viruses, and parasites that can cause diarrhoeal disease.2 New enteropathogens continue to be discovered; the microorganisms listed in table 1 ▶ are the most clinically significant agents. Infectious diarrhoea presents clinically as one of three major clinical syndromes.
Table 1.
Causes of infectious diarrhoea
| Enteropathogen | Acute watery diarrhoea | Dysentery | Persistent diarrhoea |
| Viruses | |||
| Rotavirus | + | − | − |
| Enteric adenovirus (types 40, 41) | + | − | − |
| Calicivirus | + | − | − |
| Astrovirus | + | − | − |
| Cytomegalovirus | + | + | + |
| Bacteria | |||
| Vibrio cholera and other vibrios | + | − | − |
| Enterotoxigenic E coli(ETEC) | + | − | − |
| Enteropathogenic E coli(EPEC) | + | − | + |
| Enteroaggregative E coli(EAggEC) | + | − | + |
| Enteroinavsive E coli(EIEC) | + | + | − |
| Enterohaemorraghic E coli(EHEC) | + | + | − |
| Shigella spp | + | + | + |
| Salmonella spp | + | + | + |
| Campylobacter spp | + | + | + |
| Yersinia spp | + | + | + |
| Clostridium difficile | + | + | + |
| Mycobacterium tuberculosis | − | + | + |
| Protozoa | |||
| Giardia intestinalis | + | − | + |
| Cryptosporidium parvum | + | − | + |
| Microsporidia | + | − | + |
| Isospora belli | + | − | + |
| Cyclospora cayetanensis | + | − | + |
| Entamoeba histolytica | + | + | + |
| Balantidium coli | + | + | + |
| Helminths | |||
| Strongyloides stercoralis | − | − | + |
| Schistosoma spp | − | + | + |
Acute watery diarrhoea, which usually resolves within 5–10 days.
Diarrhoea with blood (dysentery).
Persistent diarrhoea with or without evidence of intestinal malabsorption; persistence is defined as diarrhoea that has continued for more than 14 days
The clinical syndromes of infectious diarrhoea can be a useful but sometimes unreliable indicator of the likely pathogen responsible. The reason for the latter is shown in table 1 ▶, as there can be considerable overlap between the major syndromes. For example, dysenteric pathogens do not always cause bloody diarrhoea; the initial phase of shigella infection can present as watery diarrhoea. The presence of blood in the stool almost always indicates an invasive enteropathogen, excluding misdiagnosis due to exacerbation of bleeding haemorrhoids.
DIARRHOEA MECHANISMS
Infectious diarrhoea occurs as a result of two major disturbances in normal intestinal physiology:
increased intestinal secretion of fluid and electrolytes, predominantly in the small intestine; and
decreased absorption of fluid, electrolytes, and sometimes nutrients that can involve the small and large intestine.
Increased intestinal secretion
Intestinal secretory processes can be activated by infection with bacteria and viruses. Secretory enterotoxins are the major cause of increased intestinal secretion in infective diarrhoea. Cholera toxin (CT) is the “prototype” enterotoxin and its mechanism of action has been extensively researched; it is the paradigm for enterotoxin mediated diarrhoea. CT switches on secretion without any macro- or microscopic damage to the enterocyte. Other secretory enterotoxins have also been well characterised and include the closely related E coli heat labile toxin (LT) and the structurally distinct E coli heat stable toxin (ST).3 Since the discovery of these toxins, other prosecretory enterotoxins have been discovered. Intracellular mediators and other accessory mechanisms of enterotoxin action are summarised in table 2 ▶.
Table 2.
Bacterial enterotoxins and their mechanisms of action
| Enterotoxin | Signal transduction | Accessory pathways |
| Cholera toxin family | ||
| Cholera toxin | cAMP | ENS, 5-HT |
| E coli heat labile toxin I (LT-I) | cAMP | ENS |
| E coli heat labile toxin II (LT-II) | cAMP | ? |
| Salmonella enterotoxin | cAMP | ? |
| Shigella enterotoxin (ShET I+II) | cAMP | ? |
| Heat stable toxin family | ||
| E coli heat stable toxin (STa) | cGMP | ENS |
| Enteroaggregative E coli heat stable toxin 1 (EAST-1) | cGMP | ? |
| Yersinia enterocolitica heat stable toxin (Y-ST) | cGMP | ? |
| V cholera non-O1 heat stable toxin (NAG-ST) | cGMP | ? |
| Other enterotoxins | ||
| Accessory cholera enterotoxin | ? | ? |
| Clostridium difficile toxin A | Ca++ | Cytoskeleton |
| Enteroinvasive E coli toxin | ? | ? |
| Plesiomonas shigelloides LT+ST | ? | ? |
| Aeromonas hydrophila enterotoxin | ? | ? |
Other more recently discovered enterotoxins have been less well characterised. Accessory cholera enterotoxin increases short circuit current in Ussing chambers, although its precise mode of action has not been defined. Zonular occludens toxin, which is produced by V cholerae O1, increases the permeability of the small intestine by interacting with the cytoskeleton and altering the structure of intercellular tight junctions.
It is now evident that secretory diarrhoea may be mediated by other mechanisms of secretion, as well as the classical enterocyte interaction. Multiple extracellular factors regulate epithelial ion transport—paracrine, immunological, neural, and endocrine factors; there is extensive overlap and interplay between these systems that a single superregulatory system has been termed PINES (paracrine-immuno-neuro-endocrine system). Secretory diarrhoea may be mediated by a variety of secretagogues, including prostaglandins, 5-hydroxytryptamine (5-HT), substance P, and vasoactive intestinal peptide (VIP). Neuronal pathways are involved in the amplification of the effects of enterotoxins.4
CT has been shown to release 5-HT from enterochromaffin cells, which is thought to then activate the afferent limb of a neuronal reflex.4–6 The effector limb of the neuronal reflex is likely to complete the neuronal pathway by releasing the neurotransmitter VIP.5 This binds to specific receptors on the basolateral membrane and activates adenylate cyclase-cAMP intracellular secretory pathways. Interneurones propagate the secretory effects of CT distally in the small intestine. The importance of 5-HT in mediating CT induced secretory diarrhoea has been confirmed by the use of 5-HT2 and 5-HT3 receptor antagonists, which decrease secretion in the rat and human intestine.6,7 Substance P antagonists also reduce CT induced fluid secretion in mammalian small intestine, suggesting that it may be a key neurotransmitter in the sensory afferent limb or interneurone of the neuronal reflex.8 Hence CT affects the epithelium directly but also recruits other components in PINES, including enteric neurones, enterochromaffin cells, and multiple mediators to produce a complex secretory response. There may also be distant effects in the small intestine9 and a reflex secretory response in the colon.10 LT and ST also activate neural secretory reflexes but 5-HT does not appear to be involved in the secretory pathway of these toxins.11
Rotavirus has been assumed to elicit diarrhoea by damaging absorptive cells but evidence is emerging that rotavirus intestinal infection can evoke fluid and electrolyte secretion by activation of the enteric nervous system.12
Decreased intestinal absorption
The other major mechanism by which enteric pathogens cause diarrhoea is impaired intestinal absorption. This is usually accompanied by macroscopic and microscopic injury to the intestine.13 Diarrhoea due to impaired intestinal absorption can be due to:(i) impaired epithelial transport processes—that is, impaired fluid, electrolyte, and nutrient absorption in the small intestine;(ii) osmotic diarrhoea due to the appearance of incompletely absorbed nutrients in the colon; or (iii) impaired water and sodium reabsorption by the colon due to direct involvement of the colonic absorptive process. Intestinal absorption is also dependent on the duration of time allowed for digestion and contact with the epithelium, and therefore any alteration in small intestinal and whole gut transit times may result in impaired absorption.
Epithelial injury in the small intestine and colon occurs in association with many enteropathogens—bacteria, parasites, and viruses. The nature of the injury can occur at many levels; from discrete damage to the microvillus membrane during the attachment of E coli and Cryptosporidium parvum, to the mucosal inflammatory response to invasive pathogens—for example, Shigella spp, Salmonella spp, and Entamoeba histolytica, usually involving the release of cytolethal cytotoxins resulting in epithelial cell loss and ulceration. Rotavirus, another invasive enteropathogen, directly invades the epithelial cells in the middle and upper portion of the villus, with rapid epithelial cell death and acute villous trophy. Invasive enteropathogens also produce an acute inflammatory response within the mucosa, recruiting proinflammatory mediators such as prostaglandins and leukotrienes, resulting in both impaired intestinal absorption and the initiation of a prosecretory state in the intestine.3 Invasive enteropathogens also promote the synthesis and release of chemokines, such as interleukin (IL)-8, by intestinal epithelial cells. IL-8 is a known potent chemoattractant for polymorphonuclear leucocytes that enhance the inflammatory cascade and produce further mucosal and epithelial damage by release of reactive oxygen species. Neutrophils also release 5′-AMP, which is a potent secretatgogue acting though the adenosine A2 receptor on the apical membrane of intestinal epithelial cells.
In the clinical setting, these two pathophysiological disturbances—secretory diarrhoea, and secondly, impairment of epithelial transport processes with enteropathogenic invasion and epithelial cell injury—often coexist. Shigella, salmonella, and campylobacter produce a secretory diarrhoea in the small intestine in the early phase of the illness, most likely as a result of enterotoxin activity, but then invade the epithelium of the distal ileum and colon to produce an inflammatory ileocolitis. At this stage there will be epithelial cell loss and impaired absorption of fluid and electrolytes.
DIAGNOSIS OF INFECTIOUS DIARRHOEA
The majority of intestinal infections are self limiting in immunocompetant individuals so one could argue that making a specific diagnosis is unnecessary. This is certainly true for most viral diarrhoeas and many bacterial diarrhoeas. From the microbiological and public health perspective, a specific diagnosis is helpful. The major challenge facing the gastroenterologist is to decide whether an episode of diarrhoea is infectious or due to another cause, such as a functional bowel disorder or inflammatory bowel disease.
Clinical history
The clinical history is valuable in deciding whether intestinal infection is a likely cause of diarrhoea. Some individuals are more susceptible to intestinal infection and can often be identified by taking a careful history (table 3 ▶). Food and water are important vehicles for infection, as previously discussed, in both the third world and the developed world. A careful history of oral intake may be crucial in identifying the source. Major outbreaks of giardiasis and cryptosporidiosis have been well documented in North America and Europe following contamination of water supplies. Swimming in seawater, freshwater, and swimming pools is also a risk factor for intestinal infection. Foodborne diarrhoeal disease occurs either as a true infection in which the enteropathogens are consumed or as ingestion of preformed toxin.
Table 3.
Special risk groups for infectious diarrhoea
| Risk factors | Groups at risk |
| Age | Infants |
| Young children | |
| The elderly | |
| Non-immune host defence-gastric acid | The elderly Hypo- and achlorhydria Patients on acid inhibitory drugs Congenital immunodeficiency |
| Immunodeficiency | HIV/AIDS |
| Cancer and cancer chemotherapy | |
| Undernutrition | |
| Increased exposure to enteropathogens | Travellers Contaminated food and water |
| Antibiotics | Especially the elderly and cancer patients |
Physical examination
Physical examination is unhelpful in forming a specific diagnosis of infectious diarrhoea. However, it is vitally important in assessing the individual’s hydration status and in identifying other causes and risk factors for diarrhoea. Assessment of hydration status is particularly important in infants, young children, and the elderly. Specific clinical criteria have been established to formally assess the hydration state in infants and young children and provide helpful clinical guidance on the replacement volume of fluid required and the most appropriate route of administration.14 Most useful indicators for assessing hydration and for monitoring of rehydration in infants are anterior fontanelle, systolic blood pressure, skin elasticity, ocular tension, and urine flow.
Painful swollen joints may accompany intestinal infection, Yersinia enterocolitica, and C jejuni as part of Reiter’s syndrome. Guillain-Barré syndrome may develop as a result of C jejuni intestinal infection, which is now known to be the commonest cause of this syndrome. The haemolytic uraemic syndrome is an important although uncommon complication of dysenteric shigellosis and EHEC infection. There is good evidence that intestinal infections may initiate a functional bowel disorder such as irritable bowel syndrome (IBS). Some patients with so-called post-infective IBS have a mild but significant increase in mucosal inflammatory cells and an increase in 5-HT containing enterochromaffin cells, both of which are thought to contribute to symptom production.
Persistent diarrhoea is usually accompanied by weight loss and possibly other clinical features of macro- and micronutrient deficiency. There are a few specific clinical stigmata of some tropical causes of persistent diarrhoea. Larvae currens is an erythematous pruritic migrating weal associated with strongyloidiasis. Hepatomegaly may accompany intestinal schistosomiasis.
Rigid sigmoidoscopy may be helpful in confirming the presence of proctocolitis; it can be extremely difficult to distinguish between proctocolitis secondary to infection or non-specific inflammatory bowel disease, and hence the specificity of sigmoidoscopic appearance is generally poor. It is important to note that a normal rectum does not exclude infective colitis or non-specific inflammatory bowel disease.
Specific investigations
Specific investigation is not normally required in the majority with acute watery diarrhoea as this is usually self limiting, and resolves without specific treatment. Patients with bloody diarrhoea (dysentery) or persistent diarrhoea do require further investigation. The general approach is to start with the simplest, least invasive, “economically competitive” test, progressing in a hierarchical way to more invasive and expensive investigations.
Stool microscopy and culture
Stool microscopy and culture is the first line investigation. Three stool samples should be examined under the light microscope for parasites by an experienced observer, and then cultured for bacterial enteropathogens. Detection of parasites with standard microscopy is labour intensive and insensitive. Special stains are required to enhance detection of cysts and spores. Microscopy is vital for the diagnosis of Entamoeba histolytica, Giardia intestinalis, Cryptosporidium parvum, and Cyclospora cayetanensis. Newer antigen detection assays have been developed that increase the sensitivity of the examination for giardia and cryptosporidium. In addition, commercially available enzyme immunoassays are able to distinguish between E histolytica and the non-pathogenic but microscopically indistinguishable E dispar. C difficile requires confirmation by detection of toxin A in faeces by enzyme linked immunosorbent assay (ELISA). Faecal antigen ELISAs are also available for rotavirus.
Serodiagnosis
Antibody testing is useful to confirm or support other tests in a limited number of infections. Specific serum antibodies are present in 80–90% of patients in invasive amoebiasis. Antibodies are useful in Y enterocolitica, but a result can take up to 10–14 days. ELISA kits are widely available for the diagnosis of strongyloides and schistosomiasis: they are often used as first line screening tests for these infections, especially in travellers returning from endemic areas.
Abdominal imaging
Plain abdominal radiograph is usually performed in those who are severely unwell with abdominal pain to exclude bowel perforation and for assessing the severity and extent of infectious colitis.
Transabdominal ultrasound can detect bowel wall thickening, enlarged lymph nodes, pneumatosis, abdominal tuberculosis, and complications such as amoebic liver abscesses.
Endoscopy
Upper gastrointestinal endoscopy is useful in the investigation of patients with persistent diarrhoea, with or without clinical features of intestinal malabsorption. Severe villous atrophy in the second part of the duodenum can occur in infections due to small intestinal protozoa—giardia, cryptosporidium, cyclospora, and the microsporidia. Changes in villous morphology can be confirmed by duodenal biopsy, which may also reveal the presence of protozoal cysts or trophozoites. Duodenal fluid can also be aspirated during the procedure—this is particularly helpful for the detection of Giardia intestinalis cysts and trophozoites and for the larvae of strongyloides.
Endoscopic examination of the colon and ileum is useful following negative stool culture and microscopy in the presence of dysentery or persistent symptoms. This may be helpful for distinguishing between infectious colitis and inflammatory bowel disease, but the pathological features are not very reliable in the acute setting. Discrete ulceration can occur in amoebiasis and colonic tuberculosis and there are few distinguishing features that reliably differentiate these infections from Crohn’s disease. Pseudomembranes in the colon are generally indicative of C difficile infection but can be also found in ischaemic colitis. Colonic biopsies can detect E histolytica, cytomegalovirus, and the ova of Schistosoma spp.
Histology
If colonic mucosal biopsies are taken within the first 24–72 hours, histological features may be indicative of infection, including mucosal oedema, straightening of the glands, and an acute inflammatory infiltrate.15,16 After this stage it can very difficult to distinguish between infectious colitis and non-specific inflammatory bowel disease. Biopsies can reveal the pseudomembranes of C difficile and the caseating granulomata of tuberculosis.
TREATMENT
During the past 10 years there have been some major improvements in our knowledge base regarding the treatment of infectious diarrhoea.
Major advances in the treatment of infectious diarrhoea (table 4 ▶)
Table 4.
Major advances in the treatment of infectious diarrhoea
| Supportive therapy |
| Hypotonic oral rehydration solutions17–19 |
| Resistant starch based ORS20 |
| Antisecretory drugs |
| Racecadotril-enkephalinase inhibitor31–34 |
| Others in development35,36 |
| Antimicrobial chemotherapy |
| Nitazoxanide, antiprotozoal agent72 |
| Ultrashort regimens: cholera44,45, traveller’s diarrhoea40,41 |
| Rifaximin, non-absorbed antibiotic42 |
| Probiotics |
| Rotavirus diarrhoea47,48 |
| Antibiotic associated diarrhoea?49 |
ORS, oral rehydration solution.
Oral rehydration therapy (ORT) remains central to case management but advances have been made by the introduction of hypotonic solutions and early evidence that resistant starch may be the substrate of the future. The search for antisecretory drugs continues, with real progress having been made by the introduction of a new class of drugs, the enkephalinase inhibitors. Other new drugs are in the early phases of development. The role of antimicrobial agents in the management of infective diarrhoea continues to be clarified with the emergence of new agents and simplified treatment regimens. The place of probiotics in the treatment and prevention of infectious diarrhoea continues to be evaluated but studies to date suggest moderate efficacy.
There are four main approaches to the treatment of infectious diarrhoea.
Supportive therapy—fluid and electrolyte replacement.
Antidiarrhoeal symptomatic treatment to reduce stool frequency and any other symptoms such as abdominal pain.
Antisecretory drug therapy aimed at reducing faecal losses.
Specific therapy such as antimicrobial chemotherapy to reduce duration and severity of the illness.
Supportive therapy
Fluid and electrolyte replacement
This is the cornerstone of treatment. Fluid and electrolyte replacement via the oral route is usually sufficient unless the person is vomiting and/or losses are very severe. Dehydration occurs more quickly in infants and young children and therefore early administration of an oral rehydration solution (ORS) is advised to prevent severe dehydration and acidosis. In severe dehydration in infants and young children, intravenous fluids are advisable. The acidosis that can occur in severe dehydration is corrected with fluid replacement alone and does not require any specific bicarbonate therapy. Food should be commenced as soon as the individual wishes to eat and drink normally. Breast feeding should be continued in infants. In most cases in adults a formal ORS is often not required but a recommended increase in oral fluids with for example salty soups (sodium), fruit juices (potassium), and a source of carbohydrates (salty crackers, rice, bread, pasta, potatoes) to provide glucose for the glucose-sodium cotransport.
Oral rehydration therapy
Recommended oral replacement fluids are glucose-electrolyte solutions known collectively as oral rehydration solutions (ORS). ORT has been a life saving therapy for many patients with severe diarrhoea. The scientific principle and rationale for this therapy is based on active carrier mediated sodium-glucose cotransport.14
The World Health Organisation (WHO) has for several decades recommended an ORS containing 90 mmol/l of sodium. There has been some concern about the widespread use of the 90 mmol/l ORS because of the small but significant risk of hypernatraemia. A lower sodium concentration of 50–60 mmol/l is as effective as the previously recommended 90 mmol/l and appears to be more efficacious in reducing faecal losses.17 The WHO in 2002 finally endorsed the use of a low osmolality ORS (245 mosmol/kg) with a sodium concentration of 75 mmol/l.
Although glucose has traditionally been the main substrate for ORS, the possibility that efficacy may be increased by using complex substrates, such as cereals or defined glucose polymers, has been explored extensively in the last few decades. Replacing glucose with a glucose polymer such as rice starch has the dual advantage of producing low osmolality solution18 while delivering an increased amount of substrate in the form of rice starch polymer along with some protein, which will also drive active sodium absorption. Cereal based ORS has only a significant advantage in cholera but not in other diarrhoeal states.19
Resistant starch is only partially hydrolysed in the small intestine and approximately 30% enters the colon where it is degraded by colonic bacteria to short chain fatty acids that promote sodium and water absorption. A randomised controlled trial in cholera diarrhoea showed that a resistant starch ORS was superior to the WHO-ORS and hypotonic glucose monomer ORS in its effectiveness in reducing faecal losses.20
Antidiarrhoeal therapy
There are two major classes of antidiarrhoeal agents useful for reducing stool frequency, abdominal cramps, and possibly stool volume.
Antimotility agents
Antisecretory agents
Antimotility agents
The most commonly used are the antimotility agents such as loperamide and a diphenoxylate-atropine combination. These agents act by increasing intestinal transit time and enhancing the potential for reabsorption of fluid and electrolytes. They have a modest effect on reducing faecal losses. Loperamide may have some antisecretory activity but this contribution to its clinical efficacy is probably marginal. Loperamide is usually the first line treatment in self therapy and no self respecting traveller is without a packet in his/her travel kit. Loperamide has been studied in various randomised controlled trials; it has failed to demonstrate any benefit over placebo in some trials,21 but a more recent trial has shown benefit.22 Loperamide combined with an antibiotic has been shown to be advantageous in some trials23,24 but of no benefit in others.25 Antimotility agents are not recommended for children and young infants due to the potential for central nervous system side effects and the theoretical possibility of respiratory depression. Antimotility agents are generally not recommended in dysentery because of the risk of colonic dilatation associated with infective colitis. However, there is limited clinical evidence for this concern. Loperamide has been shown to be safe in the treatment of bacillary dysentery if used in conjunction with an antibiotic.24 Antimotility agents have also been thought to increase the faecal carriage of gut enteropathogens but there is little evidence that this is the case.
Antisecretory agents
There is an ongoing search for the ideal antisecretory agent—that is, a drug that will directly inhibit secretory processes within the enterocyte.26,27 Intracellular signalling mechanisms were an initial pharmacological target, especially those related to calcium and the calcium binding protein calmodulin. Zaldaride maleate, a calmodulin inhibitor, has been evaluated in phase III randomised controlled trials but future development was discontinued because of no additional benefit compared with standard antidiarrhoeal agents.28,29 Recent attention has focused on the enteric nervous system (ENS). It is now well established that the ENS is involved in the promotion of intestinal secretion. A number of neurotransmitters have been identified in the ENS, and many are thought to be involved in intestinal secretion and are therefore potential pharmacological targets for the treatment of watery diarrhoea.30
Another approach has been the development of an enkephalinase inhibitor, racecadotril, which has proabsorptive activity via its ability to potentiate endogenous enkephalins in the intestine.31,32 This is an effective agent for reducing stool weight and bowel frequency, it can be safely used in children, and does not cause rebound constipation, which can be a problem with more commonly used antimotility antidiarrhoeal agents.33,34
The thiazolidinone drug-like moieties which inhibit the cystic fibrosis transmembrane regulator protein may also hold promise for the future.35 This protein is integral to the chloride channel on the apical membrane of the intestinal epithelial cell that is an essential component of the secretory process. Further clinical evaluation is required to determine whether this will be a valuable addition to the management of secretory diarrhoea.
SP 303, a naturally occurring polyphenolic polymer with chloride channel blocking activity, has been shown to have antisecretory actions and in a double blind randomised controlled trial reduced the duration of traveller’s diarrhoea by 29%.36 Further studies are required to determine whether this agent will find a place in the treatment regimens for this condition.
Bismuth salicylate has been shown to be effective in the treatment of traveller’s diarrhoea.37 It is an effective antidiarrhoeal, reducing the number of unformed stools by approximately 50%; this is attributed to the antisecretory action of its salicylate moiety but it is also thought to have antibacterial and anti-inflammatory properties.38 It is not a popular drug of choice as a large number of tablets must be taken (eight tablets), it has a delayed onset of action (up to four hours), it can interfere with the absorption of other medications such as doxycyline, and has some unpleasant side effects (tinnitus, black tongue).
Antimicrobial therapy
Antibiotic therapy for infectious diarrhoea is controversial. Those with mild symptoms and those who are clearly improving probably do not need antibiotic treatment. However, there are certain infectious diarrhoeas in which treatment is recommended: dysenteric shigellosis, cholera, pseudomembranous enterocolitis, that due to parasites, and sexually transmitted diseases. There are several diseases in which the indications are less clear but treatment is usually recommended: infection with the non-cholera vibrios, prolonged or protracted infection with yersinia, early in the course of campylobacteriosis, aeromonas and plesiomonas infections, and outbreaks of enteropathogenic E coli diarrhoea in nurseries. Patients should be treated if they are debilitated, particularly with malignancy, immunosuppressed, have an abnormal cardiovascular system, have valvular, vascular, or orthopaedic prostheses, have haemolytic anaemia (especially if salmonellosis is involved), or are extremely young or old. Treatment is also advised for those with prolonged symptoms and those who relapse.
There is a large body of evidence to show that antimicrobial agents can reduce the severity and duration of some intestinal infections, especially in those bacteria and infections that produce acute watery diarrhoea. Antimicrobials are also useful in bacterial intestinal infections that cause systemic involvement. There are numerous antibiotics that have been studied in the treatment of infectious diarrhoea, some empirical and some targeted. Intestinal infections can be regarded in different categories depending on whether antimicrobial therapy has been proven to be effective in clinical trials. Efficacy varies from being definitely effective to possible and/or doubtful efficacy. Efficacy is regarded as reduction in duration of illness, severity, and complications (see table 5 ▶).
Table 5.
Antimicrobial therapy for acute infectious diarrhoea
| Organism | Efficacy of antimicrobial therapy | Drug of choice | Alternative choice |
| Bacteria | |||
| Vibrio cholerae | Proven | Tetracycline 500 mg qds 3 days. Ciprofloxacin 1000 mg single dose44,45 | TMP-SMX, doxycyline, norfloxacin, ciprofloxacin, 3 days43 |
| ETEC | Proven | Ciprofloxacin 500 mg bd 3–5 days39 | Ciprofloxacin 500 mg single dose41 |
| Norfloxacin 400 mg bd, 3–5 days40 | |||
| EPEC | Possible | ||
| EIEC | Possible | ?Same as Shigella spp | |
| EHEC | Controversial | See text | |
| Shigella spp | Proven efficacy in dysenteric shigellosis | TMP-SMX 2 tabs bd 5 days*.50 Ciprofloxacin 500 mg bd 5 days.51 Other quinolones—norfloxacin, fleroxacin, cinoxacin | Short term quinolone.51–55 Cefixime 400 mg daily 5–7 days OR other third generation cephalosporins. Nalidixic acid 1 g qds 5–7 days |
| Salmonella spp | Doubtful efficacy in enterocolitis. Proven efficacy in severe salmonellosis (dysentery, fever) | Ciprofloxacin 500 mg bd 10–14 days. 3rd gen cephalosporins 10–14 days. Carrier state: norfloxacin 400 mg bd 28 days | TMP-SMX.53 Ampicillin, amoxycillin |
| Campylobacter spp | Possible efficacy in campylobacter enteritis. Proven efficacy in campylobacter, dysentery/sepsis | Erythromycin 250–500 mg qds 7 days56–59 | Ciprofloxacin 500 mg bd 5–7 days. Azithromycin 500 mg od 3 days |
| Yersinia spp | Doubtful efficacy in Yersinia enteritis. Proven efficacy in Yersinia septicaemia | Ciprofloxacin 500 mg bd 7–10 days60,61 | Tetracycline 250 mg qds 7–10 days60,61 |
| Clostridium difficile | Proven | Metronidazole 400 mg tds 7–10 days62 | Vancomycin 125 mg qds 7–10 days.62–64 Fusidic acid, teicoplanin65 |
| Protozoa | |||
| Cryptosporidium parvum | Possible | ||
| Isospora belli | Proven | ||
| Cyclospora cayetanensis | Proven | ||
| Entamoeba histolytica | Proven | Metronidazole 750 mg tds 5 days.66 Diloxanide furoate 500 mg tds 10 days66 | Paromomycin 25–35 mg/kg tds 7–10 days66 |
| Balantidium coli | Proven | Metronidazole 400 mg tds 10 days66,67 | Tetracycline 500 mg qds 10 days66,67 |
Antimicrobial therapy is not indicated for acute viral diarrhoea such as that due to rotavirus, enteric adenoviruses, and small round structured viruses.
*TMP/SMX is of limited value because of resistance patterns.
In cases where there is doubt about the efficacy of antibiotics, it may not be related solely to the potency of the antibiotic but also to the study design. Administration of the antibiotic may be delayed after the onset of symptoms. When given relatively late in the natural history of the illness, additional benefits of therapy could be missed.
Acute watery diarrhoea
In acute watery diarrhoea, treatment is largely supportive. Antibiotic therapy is controversial unless the illness is severe or due to cholera. It is a widely held belief that in what is generally a mild self limiting illness, antibiotic use is unnecessary; the risk of antibiotic resistance is increased and introduces the possibility of antibiotic side effects (for example, Stevens Johnson syndrome or pseudomembranous colitis).
In traveller’s diarrhoea, a major form of acute watery diarrhoea, antimicrobial therapy is unequivocally effective; this is supported by many randomised controlled trials. Traveller’s diarrhoea is mainly due to bacterial enteropathogens (approximately 80%), the most frequently isolated being enterotoxigenic E coli; broad spectrum antibiotics have been shown to be effective but there is increasing resistance to trimethoprim-sulphamethoxazole and ampicillin and therefore these are less suitable for blind therapy. Quinolone antibiotics are now the treatment of choice; standard doses for 3–5 days can reduce the severity and duration of illness by at least 50%.39,40 Similar efficacy has also been shown with single dose regimens.41 Recently, there has been renewed interest in a non-absorbed locally active antibiotic, rifaximin, for the treatment of traveller’s diarrhoea. This drug has been shown to be as effective as ciprofloxacin but with the potential advantage of only minimal systemic absorption.42
Azithromycin is also a good choice for pregnant women and children, for whom fluoroquinolones are not approved, and for patients who cannot otherwise tolerate fluoroquinolones
Cholera is treated with antibiotics—standard therapy is with tetracycline for three days but other agents are equally as effective—doxycycline, trimethoprim-sulphamethoxzole, norfloxacin, and ciprofloxacin.43,44 Single dose ciprofloxacin has been shown to be as effective as three days of doxycyline.45
Treatment of E coli O157:H7 is not recommended at present because current antibiotics do not appear to be helpful, and inconclusive data have suggested that the incidence of complications, including haemolytic uraemic syndrome, may be greater after antibiotic therapy. Antibiotics are not routinely recommended for use in children, and there is concern that their use might increase the risk of haemolytic uraemic syndrome secondary to EHEC infection.
Probiotics
In 1985, Gorbach identified a lactobacillus as a result of screening bacteria in fermented milk products thought to be beneficial to human health.46 This lactobacillus species was acid and bile resistant, adhered to human intestinal epithelial cells, and had growth characteristics necessary for commercial development. This strain, identified as LactobacillusGG, is one of several probiotics, a non-pathogenic organism, used to improve intestinal microbial balance. Following this discovery, multiple candidate microorganisms have been developed, but Lactobacillus GG remains the most common strain to be tested in controlled trials. In a multicentre trial, Lactobacillus GG was shown to reduce the duration of rotavirus episodes but had no effect on bacterial diarrhoeas.47 A recent meta-analysis would support the view that probiotics can shorten the duration of acute diarrhoeal illness in children by one day.48 Although meta-analysis also suggests that probiotics benefit antibiotic associated diarrhoea,49 further studies are required to provide a definitive answer.
Dysentery
Antibiotics are recommended for the treatment of dysentery due to most organisms50–67(table 5 ▶). However, antibiotic therapy for campylobacter56,57 and EHEC infection remains controversial.68–70 In campylobacter infection there is good evidence that antibiotics do not alter the natural course of the illness if antibiotics are started >4 days after the onset of symptoms. Randomised controlled trials are conflicting in terms of efficacy of antibiotics if started early in the course of infection. One randomised controlled trial has shown that erythromycin started early reduces the duration of illness in children58 but a second study failed to confirm these findings.59 EIEC infection, if severe, with evidence of systemic involvement can be treated with antibiotics recommended in dysenteric shigellosis, but a role for routine use has not been established. Antimicrobial therapy in EHEC infection remains controversial for two reasons:(i) antibiotics do not significantly improve outcome, especially if started well after infection established68; and (ii) there is anecdotal evidence that antibiotics can promote the development of haemolytic uraemic syndrome.69,70 Antibiotics are thought to increase the lysis of organisms and release of SLT and endotoxin.
Persistent diarrhoea
Most of the enteropathogens which cause persistent diarrhoea are treatable with antimicrobial therapy (table 6 ▶). There are randomised controlled trials for most agents to support their use; these agents reduce the duration and severity of illness. Cryptosporidium parvum is however difficult to treat and is resistant to most antimicrobial agents. Paromomycin has been shown to have some efficacy in one open study.71 Recent studies have shown that high dose albendazole or nitazoxanide may have some benefit.72 Microsporidia are also difficult to treat and have variable sensitivity to many agents. Albendazole is effective in treating E intestinalis but not very effective in treating E bieneusi.73,74 Uncontrolled studies have shown the following agents to have some benefit in treating microsporidia: atovaquone,75 furazolidone,76 furazolidone-albendazole,77 and thalidomide.78C cayetanensis infection can be treated effectively with TMP-SMX.79
Table 6.
Antimicrobial therapy of persistent infectious diarrhoea
| Enteropathogen | Antimicrobial therapy | Alternative(s) |
| Protozoa | ||
| Giardia intestinalis | Metronidazole 400 mg tds 7–10 days80 | Tinidazole 2 g single dose80 |
| Cryptosporidium parvum | ?Paromomycin 500 mg qds71 | ?Nitazoxanide14 |
| Cyclospora cayetanensis | TMP-SMX 2 tabs bd 7 days79 | |
| Isospora belli | TMP-SMX 2 tabs qds 10 days | |
| Microsporidia | ||
| Encephalitozoon intestinalis | ? Albendazole 400 mg bd 14–28 days73 | ?Furazolidone 100 mg qds 20 days76 |
| Enterocytozoon bieneusi | ? Atovaquone75 | |
| Entamoeba histolytica | See table 5 ▶ | |
| Balantidium coli | See table 5 ▶ | |
| Helminths | ||
| Strongyloides stercoralis | Albendazole 400 mg od 3 days | Thiabendazole 25 mg/kg bd 2–3 days |
| Schistosoma spp | Praziquantel 2–3 doses on day 1 | Ivermectin 100–200 μg/kg od 2 days |
| S mansoni, S haematobium | Praziquantel 40 mg/kg/d | |
| S japonicum | Praziquantel 60 mg/kg/d | |
| Virus | ||
| Cytomegalovirus | Ganciclovir 5 mg/kg bd 14–21 days | Foscarnet 60 mg/kg tds 14–21 days maintenance therapy required |
Acknowledgments
AC-J is supported by the Digestive Disorders Foundation, UK. This review has been modified and updated from an earlier version authored by MJGF in 2001 (see Farthing2).
REFERENCES
- 1.Wheeler JG, Sethi D, Cowden JM, et al. Study of infectious intestinal disease in England: rates in the community, presenting to general practice, and reported to national surveillance. BMJ 1999;318:1046–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Farthing MJG. Infectious diarrhea. In: Irvine EJ, Hunt RH, eds. Evidence-based gastroenterology. Canada: BC Decker, 2001:323–41.
- 3.Farthing MJG. Pathophysiology of infective diarrhoea. Eur J Gastroenterol Hepatol 1993;5:796–807. [Google Scholar]
- 4.Cassuto J, Jodal M, Tuttle R, et al. 5-Hydroxytryptamine and cholera secretion. Physiological and pharmacological studies in cats and rats. Scand J Gastroenterol 1982;17:695–703. [DOI] [PubMed] [Google Scholar]
- 5.Cassuto J, Fahrenburg J, Jodal M, et al. Release of vasoactive intestinal polypeptide from the cat of the small intestine exposed to cholera toxin. Gut 1981;22:958–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beubler E, Horina G. 5-HT2 and 5-HT3 receptor subtypes mediate cholera toxin-induced intestinal fluid secretion in the rat. Gastroenterology 1990;99:83–9. [DOI] [PubMed] [Google Scholar]
- 7.Turvill JL, Farthing MJG. Effect of granisetron on cholera toxin-induced enteric secretion. Lancet 1997;349:1293. [DOI] [PubMed] [Google Scholar]
- 8.Turvill JL, Connor P, Farthing MJG. Neurokinin 1 and 2 receptors mediate cholera toxin secretion in rat jejunum. Gastroenterology 2000;119:1037–44. [DOI] [PubMed] [Google Scholar]
- 9.Banks MR, Casburn-Jones AC, Farthing MJG. Cholera toxin (CT)Escherichia coli heat labile toxin (LT) and heat stable toxin (STa) have an indirect effect on distal intestinal fluid transport in the rat small intestine. Gut 2002;50 (suppl II) :A69. [Google Scholar]
- 10.Nocerino A, Iafusco M, Guandalini S. Cholera toxin-induced small intestinal secretion has a secretory effect on the colon of the rat. Gastroenterology 1995;108:34. [DOI] [PubMed] [Google Scholar]
- 11.Turvill JL, Mourad FH, Farthing MJG. Crucial role for 5-HT in cholera toxin but not Escherichia coli heat-labile enterotoxin-intestinal secretion in rats. Gastroenterology 1998;115:883–90. [DOI] [PubMed] [Google Scholar]
- 12.Lundgren O, Pergrin AT, Person K, et al. Role of the enteric nervous system in the fluid and electrolyte secretion of rotavirus diarrhea. Science 200 287:491–5. [DOI] [PubMed]
- 13.Farthing MJG. Acute diarrhoea: pathophysiology. In: Gracey M, Walker-Smith JA, eds. Diarrhoeal disease. Philadelphia, PA: Vevey/Lippincott-Raven Publishers, 1997;38:55–71. [Google Scholar]
- 14.Farthing MJG. Dehydration and rehydration in children. In: Arnaud MJ, ed. Hydration throughout life. Paris: John Libbey Eurotext, 1998:159–73.
- 15.Nostrant TT, Kumar NB, Appelman HD. Histopathology differentiates acute self-limited colitis from ulcerative colitis. Gastroenterology 1987;92:318–28. [DOI] [PubMed] [Google Scholar]
- 16.Allison MC, Hamilton-Dutoit SJ, Dhillon AP, et al. The value of rectal biopsy in distinguishing self-limiting colitis from early inflammatory bowel disease. QJM 1987;65:985–95. [PubMed] [Google Scholar]
- 17.International Study Group on Reduced-Osmolality ORS Solutions. Multicentre evaluation of reduced-osmolality oral rehydration salts solution. Lancet 1995;346:282–5. [PubMed] [Google Scholar]
- 18.Thillainayagam AV, Hunt JB, Farthing MJG. Enhancing clinical efficacy of oral rehydration therapy: is low osmolality the key? Gastroenterology 1998;114:197–210. [DOI] [PubMed] [Google Scholar]
- 19.Gore SM, Fontaine O, Pierce NF. Impact or rice-based oral rehydration solution on stool output and duration of diarrhoea: meta-analysis of 13 clinical trials. BMJ 1992;304:287–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ramakrishna BS, Venkataraman S, Srinivasan P, et al. Amylase resistant starch plus oral rehydration solution for cholera. N Engl J Med 2000;342:308–13. [DOI] [PubMed] [Google Scholar]
- 21.Bowie MD, Hill ID, Mann MD. Loperamide for treatment of acute diarrhoea in infants and young children. A double-blind placebo-controlled trial. S Afr med J 1995;85:885–7. [PubMed] [Google Scholar]
- 22.Kaplan MA, Prior MJ, McKonly KI, et al. A multicentre randomised controlled trial of a liquid loperamide product versus placebo in the treatment of acute diarrhoea in children. Clin Pediatr 1999;38:579–91. [DOI] [PubMed] [Google Scholar]
- 23.Ericsson CD, Nicholls-Vasquez I, DuPont HL, et al. Optimal dosing of trimethoprim-sulfamethoxazole when used with loperamide to treat traveler’s diarrhoea. Antimicrob Agents Chemother 1992;36:2821–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Murphy GS, Bodhidatta L, Echeverria P, et al. Ciprofloxacin and loperamide in the treatment of bacillary dysentery. Ann Intern Med 1993;118:582–6. [DOI] [PubMed] [Google Scholar]
- 25.Taylor DN, Sanchez JL, Candler W, et al. Treatment of traveler’s diarrhoea: ciprofloxacin plus loperamide compared with ciprofloxacin alone. A placebo randomized control trial. Ann Intern Med 1991;114:731–9. [DOI] [PubMed] [Google Scholar]
- 26.Powell DW, Field M. Pharmacological approaches to treatment of secretory diarrhoea. In: Field M, Fordtran JS, Schultz SG, eds. Secretory diarrhoea. Bethesda, MD: American Physiological Society, 1980:187–209.
- 27.Farthing MJG, Casburn-Jones A, Banks MR. Getting control of intestinal secretion: thoughts for 2003. Dig Liv Dis 2003;35:378–85. [DOI] [PubMed] [Google Scholar]
- 28.Silberschmidt G, Schick MT, Steffen R, et al. Treatment of travellers’ diarrhoea: zaldaride compared with loperamide and placebo. Eur J Gastroenterol Hepatol 1995;7:871–5. [PubMed] [Google Scholar]
- 29.Okhuysen PC, DuPont HL, Ericsson CD, et al. Zaldaride maleate (a new calmodulin antagonist) versus loperamide in the treatment of traveler’s diarrhoea: a randomised, placebo-controlled trial. Clin Infect Dis 1995;21:341–4. [DOI] [PubMed] [Google Scholar]
- 30.Farthing MJG. Novel targets for the control of secretory diarrhoea. Gut 2002;50 (suppl III) :iii15–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Turvill JL, Farthing MJG. Enkephalins and enkephalinase inhibitors in intestinal fluid and electrolyte transport. Eur J Gastroenterol Hepatol 1997;9:877–80. [DOI] [PubMed] [Google Scholar]
- 32.Farthing MJG. Enkephalinase inhibition: a rationale approach to anti-secretory therapy for acute diarrhoea. Aliment Pharmacol Ther 1999;13 (suppl 6) :1–2. [PubMed] [Google Scholar]
- 33.Salazar-Lindo E, Santisteban-Ponce J, Chea-Woo E, et al. Racecadotril in the treatment of acute watery diarrhea in children. N Engl J Med 2000;343:463–7. [DOI] [PubMed] [Google Scholar]
- 34.Cezard JP, Duhamel JF, Meyer M, et al. Efficacy and tolerability of racecadotril in acute diarrhea in children. Gastroenterology 2001;120:799–805. [DOI] [PubMed] [Google Scholar]
- 35.Ma T, Thiagarajah JR, Yang H, et al. Thiazilinone CFTR inhibitor identified by high throughput screening blocks cholera toxin-induced intestinal fluid secretion. J Clin Invest 2002;110:1651–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.DiCesare D, DuPont HL, Mathewson JJ, et al. A double-blind, randomized, placebo-controlled study of SP 303 (Provir) in the symptomatic treatment of acute diarrhea among travelers to Jamaica and Mexico. Am J Gastroenterol 2002;97:2585–8. [DOI] [PubMed] [Google Scholar]
- 37.Dupont HL, Ericcson C. Prevention and treatment of traveler’s diarrhea. N Engl J Med 1993;328:1821–7. [DOI] [PubMed] [Google Scholar]
- 38.Gorbach SL. Bismuth therapy in gastro-intestinal diseases. Gastroenterology 1990;99:863–75. [DOI] [PubMed] [Google Scholar]
- 39.Ericsson CD, Johnson PC, DuPont HL, et al. Ciprofloxacin or trimethoprim-sulfamethoxazole as initial therapy for traveler’s diarrhea. A placebo-controlled randomized trial. Ann Intern Med 1987;106:216–20. [DOI] [PubMed] [Google Scholar]
- 40.Mattila L, Peltola H, Siitonen A, et al. Short-term treatment of traveler’s diarrhea with norfloxacin: a double-blind, placebo-controlled study during two sessions. Clin Infect Dis 1993;17:779–82. [DOI] [PubMed] [Google Scholar]
- 41.Salam I, Katelaris P, Leigh-Smith S, et al. A randomised placebo-controlled trial of single dose ciprofloxacin in treatment of travellers’ diarrhoea. Lancet 1994;344:1537–9. [DOI] [PubMed] [Google Scholar]
- 42.DuPont HL, Jiang Z-D, Ericsson CD, et al. Rifaximin versus ciprofloxacin for the treatment of traveler’s diarrhea: A randomised, double-blind clinical trial. Clin Infect Dis 2001;33:1807–15. [DOI] [PubMed] [Google Scholar]
- 43.Dutta D, Bhattacharya SK, Bhattacharya MK, et al. Efficacy of norfloxacin and doxycycline for treatment of Vibrio cholerae 0139 infection. J Antimicrob Chemother 1996;37:575–81. [DOI] [PubMed] [Google Scholar]
- 44.Khan WA, Bennish ML, Seas C, et al. Randomised controlled comparison of single-dose ciprofloxacin and doxycyline for cholera caused by Vibrio cholerae 01 or 0139. Lancet 1996;348:296–300. [DOI] [PubMed] [Google Scholar]
- 45.Usubutun S, Agalar C, Diri C, et al. Single dose ciprofloxacin in cholera. Rue J Emerg Med 1997;4:145–9. [PubMed] [Google Scholar]
- 46.Gorbach SL. The discovery of Lactobacillus GG. Nutr Today 1996;31:2S–4. [Google Scholar]
- 47.Guandalini S, Kirjavainen PV, Zikri MA, et al. Lactobacillus GG administered in oral rehydration solution to children with acute diarrhoea: a multicenter European trial. J Pediatr Gastroenterol Nutr 2000;30:54–60. [DOI] [PubMed] [Google Scholar]
- 48.Huang JS, Bousvaros A, Lee JW, et al. Efficacy of probiotic use in acute diarrhea in children: A meta-analysis. Dig Dis Sci 2002;47:2625–34. [DOI] [PubMed] [Google Scholar]
- 49.D’Souza AL, Rajkumar C, Cooke J, et al. Probiotics in prevention of antibiotic associated diarrhoea: Meta-analysis. BMJ 2002;324:1361–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tauxe RV, Puhr ND, Wells JG, et al. Antimicrobial resistance of Shigella isolates in the USA: the importance of international travellers. J Infect Dis 1990;162:1107–11. [DOI] [PubMed] [Google Scholar]
- 51.Bennish ML, Salam MA, Haider R, et al. Therapy for shigellosis. II. Randomized double-blind comparison of ciprofloxacin and ampicillin. J Infect Dis 1990;162:711–16. [DOI] [PubMed] [Google Scholar]
- 52.Khan WA, Seas C, Dhar U, et al. Treatment of shigellosis: V. Comparison of azithromycin and ciprofloxacin. A double-blind, randomised, controlled trial. Ann Intern Med 1997;126:697–703. [DOI] [PubMed] [Google Scholar]
- 53.Bassily S, Hyams KG, El-Masry NA, et al. Short-course norfloxacin and trimethoprim-sulfamethoxazole treatment of shigellosis and salmonellosis in Egypt. Am J Trop Med Hyg 1994;51:219–23. [DOI] [PubMed] [Google Scholar]
- 54.Gotuzzo E, Oberhelman RA, Maguina C, et al. Comparison of single-dose treatment with norfloxacin and standard 5-day treatment with trimethoprim-sulfamethoxazole for acute shigellosis in adults. Antimicrob Agents Chemother 1989;33:1101–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bennish ML, Salam MA, Khan WA, et al. Treatment of shigellosis III. Comparison of one or two-dose ciprofloxacin with standard 5 day therapy. A randomised, blinded trial. Ann Intern Med 1992;117:727–34. [DOI] [PubMed] [Google Scholar]
- 56.Anders BJ, Lauer BA, Paisley JW, et al. Double-blind placebo controlled trial of erythromycin fro treatment of Campylobacter enteritis. Lancet 1982;1:131–2. [DOI] [PubMed] [Google Scholar]
- 57.Mandal BK, Ellis ME, Dunbar EM, et al. Double-blind placebo-controlled trial of erythromycin in the treatment of clinical campylobacter infection. J Antimicrob Chemother 1984;13:619–23. [DOI] [PubMed] [Google Scholar]
- 58.Salazar-Lindo E, Sack RB, Chea-Woo E, et al. Early treatment with erythromycin of Campylobacter jejuni-associated dysentery in children. J Pediatr 1986;109:355–60. [DOI] [PubMed] [Google Scholar]
- 59.Williams MD, Schorling JB, Barrett LJ, et al. Early treatment of Campylobacter jejuni enteritis. Antimicrob Agents Chemother 1989;33:248–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gayraud M, Scavizzi MR, Mollaret HJH, et al. Antibiotic treatment of Yersinia enterocolitica septicaemia: a retrospective review of 43 cases. Clin Infect Dis 1993;17:405–10. [DOI] [PubMed] [Google Scholar]
- 61.Crowe M, Ashford K, Isaphani P. Clinical features and antibiotic treatment of septic arthritis and osteomyelitis due to Yersinia enterocolitica. J Med Microbiol 1996;45:302–9. [DOI] [PubMed] [Google Scholar]
- 62.Teasley DG, Gerding DN, Olson MM, et al. Prospective randomised trial of metronidazole versus vancomycin for Clostridium difficile-associated diarrhoea and colitis. Lancet 1983;2:1043–6. [DOI] [PubMed] [Google Scholar]
- 63.Wilcox MH, Howe R. Diarrhoea caused by Clostridium difficile: response time for treatment with metronidazole and vancomycin. J Antimicrob Chemother 1995;35:673–9. [DOI] [PubMed] [Google Scholar]
- 64.Young GP, Ward PB, Bayley N, et al. Antibiotic-associated colitis due to Clostridium difficile: double-blind comparison of vancomycin and bacitracin. Gastroenterology 1985;89:1038–45. [DOI] [PubMed] [Google Scholar]
- 65.Wenisch C, Parschalk B, Hasenhundl M, et al. Comparison of vancomycin, teicoplanin, metronidazole and fusidic acid for the treatment of Clostridium difficile-associated diarrhea. Clin Infect Dis 1996;22:813–18. [DOI] [PubMed] [Google Scholar]
- 66.Kelly MP, Farthing MJG. Infections of the gastrointestinal tract. In: O’Grady F, Lambert HP, Finch RG, et al. eds. Antibiotic and chemotherapy, 7th edn. London: Churchill Livingstone, 1997:708–20.
- 67.Garcia-Laverde A, de Bonilla L. Clinical trials with metronidazole in human balantidiasis. Am J Trop Med Hyg 1975;24:781–3. [DOI] [PubMed] [Google Scholar]
- 68.Prouix F, Turgeon JPJ, Delage G, et al. Randomized, controlled trial of antibiotic therapy for Escherichia coli O157-H7 enteritis. J Pediatr 1992;121:299–303. [DOI] [PubMed] [Google Scholar]
- 69.Carter AO, Borczyk AA, Carlson JA, et al. A severe outbreak of Escherichia coli O157-H7-associated haemorrhagic colitis in a nursing home. N Engl J Med 1987;317:496–500. [DOI] [PubMed] [Google Scholar]
- 70.Pavia AT, Nichols CR, Green DP, et al. Hemolytic-uremic syndrome during an outbreak of Escherichia coli O157-H7 infections in institutions for mentally retarded persons: clinical and epidemiologic observations. J Pediatr 1990;116:544–51. [DOI] [PubMed] [Google Scholar]
- 71.Bissuel F, Cotte L, Rabodonirina M, et al. Paromomycin: an effective treatment for cryptosporidial diarrhea in patients with AIDS. Clin Infect Dis 1994;18:447–9. [DOI] [PubMed] [Google Scholar]
- 72.Farthing MJG. Clinical aspects of human cryptosporidiosis. Contrib Microbiol 2000;6:50–74. [DOI] [PubMed] [Google Scholar]
- 73.Molina JM, Chastang C, Goguel J, et al. Albendazole for treatment and prophylaxis of microsporidiosis due to Encephalitozoon intestinalis in patients with AIDS: a randomised double-blind trial. J Infect Dis 1998;177:1373–7. [DOI] [PubMed] [Google Scholar]
- 74.Leder K, Ryan N, Spelman D, et al. Microsporidial disease in HIV-infected patients: a report of 42 patients and review of the literature. Scand J Infect Dis 1998;30:331–8. [DOI] [PubMed] [Google Scholar]
- 75.Anwar-Bruni DM, Hogan SE, Schwartz DA, et al. Atovaquone is effective treatment for the symptoms of gastrointestinal microsporidiosis in HIV-1 infected patients. AIDS 1996;10:619–23. [DOI] [PubMed] [Google Scholar]
- 76.Dionisio D, Sterrantino G, Meli M, et al. Use of furazolidone for the treatment of microsporidiosis due to Enterocytozoon bieneusi in patients with AIDS. Recent Prog Med 1995;86:394–7. [PubMed] [Google Scholar]
- 77.Dionisio D, Manneschi LI, Di Lollo S, et al. Persistent damage to Enterocytozoon bieneusi with persistent symptomatic relief after combined furazolidone and albendazole in AIDS patients. J Clin Pathol 1998;51:731–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sharpstone D, Rowbottom A, Francis N, et al. Thalidomide: a novel therapy for microsporidiosis. Gastroenterology 1997;112:1823–9. [DOI] [PubMed] [Google Scholar]
- 79.Hoge CW, Shlim DR, Ghimire M, et al. Placebo-controlled trial of co-trimoxazole for Cyclospora infections among travellers and foreign residents in Nepal. Lancet 1995;345:691–3. [DOI] [PubMed] [Google Scholar]
- 80.Vesy CJ, Peterson WL. Review article: the management of giardiasis. Aliment Pharmacol Ther 1999;13:843–50. [DOI] [PubMed] [Google Scholar]